The Impact of Uremic Toxicity Induced Inflammatory Response on the Cardiovascular Burden in Chronic Kidney Disease
Abstract
:1. Introduction
2. Results
2.1. Correlations between eGFR and Uremic Toxins, Inflammatory Biomarkers and BUCVR
2.2. Correlations between Plasma Levels Uremic Toxins, Inflammatory Biomarkers and BUCVR
2.3. Tissue Expression of Inflammatory Biomarkers and BUCVR in Human Renal Arteries
2.4. Comparisons between Plasma Levels Uremic Toxins, Inflammatory Biomarkers, Vascular Inflammation, BUCVR in Patients with and without Carotid Artery Plaques
2.5. Survival Analysis According to eGFR, Plasma Levels Uremic Toxins, Inflammatory Biomarkers and BUCVR
3. Discussion
4. Materials and Methods
4.1. Subjects
4.1.1. Pre-Dialysis CKD Patients
4.1.2. CKD Transplant Recipients
4.2. Material
4.2.1. Blood Sampling
4.2.2. Renal Arteries Samples
4.2.3. Clinical and Biochemical Characteristics of the Patients (Cohort 1)
4.3. Measurement of Plasma Levels of Uremic Toxins
4.4. Measurement of Plasma Levels of Inflammatory Biomarkers (IL-6, hsCRP, MCP-1, sICAM-1, sVCAM-1 and sFas)
4.5. Biomarkers of the Uremic Cardiovascular Response (BUCVR)-sCD36, Fractalkine and sRAGE Plasma Levels
4.6. Histology and Immunohistochemistry (IHC) of Arteries from CKD Patients and Healthy Controls
4.7. Non-Invasive Evaluation of Atherosclerotic CVD
4.8. Cardiovascular Events during the Follow Up
4.9. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Go, A.S.; Chertow, G.M.; Fan, D.; McCulloch, C.E.; Hsu, C.Y. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Engl. J. Med. 2004, 351, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Lekawanvijit, S.; Krum, H. Cardiorenal syndrome: Acute kidney injury secondary to cardiovascular disease and role of protein-bound uraemic toxins. J. Physiol. 2014, 592, 3969–3983. [Google Scholar] [CrossRef] [PubMed]
- Jourde-Chiche, N.; Dou, L.; Cerini, C.; Dignat-George, F.; Brunet, P. Vascular incompetence in dialysis patients--protein-bound uremic toxins and endothelial dysfunction. Semin. Dial. 2011, 24, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Stenvinkel, P.; Ketteler, M.; Johnson, R.J.; Lindholm, B.; Pecoits-Filho, R.; Riella, M.; Heimbürger, O.; Cederholm, T.; Girndt, M. IL-10, IL-6, and TNF-alpha: Central factors in the altered cytokine network of uremia--the good, the bad, and the ugly. Kidney Int. 2005, 67, 1216–1233. [Google Scholar] [CrossRef] [PubMed]
- Borges, N.A.; Barros, A.F.; Nakao, L.S.; Dolenga, C.J.; Fouque, D.; Mafra, D. Protein-Bound Uremic Toxins from Gut Microbiota and Inflammatory Markers in Chronic Kidney Disease. J. Ren. Nutr. 2016, 26, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Yoshida, M. Protein-bound uremic toxins: New culprits of cardiovascular events in chronic kidney disease patients. Toxins 2014, 6, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Moradi, H.; Sica, D.A.; Kalantar-Zadeh, K. Cardiovascular burden associated with uremic toxins in patients with chronic kidney disease. Am. J. Nephrol. 2013, 38, 136–148. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Campbell, K.L.; Johnson, D.W.; Stanton, T.; Vesey, D.A.; Coombes, J.S.; Weston, K.S.; Hawley, C.M.; McWhinney, B.C.; Ungererf, J.P.J.; et al. Protein-bound uremic toxins, inflammation and oxidative stress: A cross-sectional study in stage 3–4 chronic kidney disease. Arch. Med. Res. 2014, 45, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Kaminski, T.W.; Pawlak, K.; Karbowska, M.; Mysliwiec, M.; Pawlak, D. Indoxyl sulfate - the uremic toxin linking hemostatic system disturbances with the prevalence of cardiovascular disease in patients with chronic kidney disease. BMC Nephrol. 2017, 18, 35. [Google Scholar] [CrossRef] [PubMed]
- Manabe, I. Chronic inflammation links cardiovascular, metabolic and renal diseases. Circ. J. 2011, 75, 2739–2748. [Google Scholar] [CrossRef] [PubMed]
- Barreto, F.C.; Barreto, D.V.; Liabeuf, S.; Meert, N.; Glorieux, G.; Temmar, M.; Choukroun, G.; Vanholder, R.; Massy, Z.A. and on behalf of the European Uremic Toxin Work Group (EUTox). Serum indoxyl sulfate is associated with vascular disease and mortality in chronic kidney disease patients. Clin. J. Am. Soc. Nephrol. 2009, 4, 1551–1558. [Google Scholar] [CrossRef] [PubMed]
- Liabeuf, S.; Barreto, D.V.; Barreto, F.C.; Meert, N.; Glorieux, G.; Schepers, E.; Temmar, M.; Choukroun, G.; Vanholder, R.; Massy, Z.A.; et al. Free p-cresylsulphate is a predictor of mortality in patients at different stages of chronic kidney disease. Nephrol. Dial. Transpl. 2010, 25, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.J.; Liu, H.L.; Pan, C.F.; Chuang, C.K.; Jayakumar, T.; Wang, T.J.; Chen, H.H.; Wu, C.J. Indoxyl sulfate predicts cardiovascular disease and renal function deterioration in advanced chronic kidney disease. Arch. Med. Res. 2012, 43, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Pecoits-Filho, R.; Heimburger, O.; Barany, P.; Suliman, M.; Fehrman-Ekholm, I.; Lindholm, B.; Stenvinkel, P. Associations between circulating inflammatory markers and residual renal function in CRF patients. Am. J. Kidney Dis. 2003, 41, 1212–1218. [Google Scholar] [CrossRef]
- Stinghen, A.E.; Goncalves, S.M.; Martines, E.G.; Nakao, L.S.; Riella, M.C.; Aita, C.A.; Pecoits-Filho, R. Increased plasma and endothelial cell expression of chemokines and adhesion molecules in chronic kidney disease. Nephron Clin. Pract. 2009, 111, c117–c126. [Google Scholar] [CrossRef] [PubMed]
- Dou, L.; Sallee, M.; Cerini, C.; Poitevin, S.; Gondouin, B.; Jourde-Chiche, N.; Fallague, K.; Brunet, P.; Calaf, R.; Dussol, B.; et al. The cardiovascular effect of the uremic solute indole-3 acetic acid. J. Am. Soc. Nephrol. 2015, 26, 876–887. [Google Scholar] [CrossRef] [PubMed]
- Dalboni, M.A.; Sardenberg, C.; Andreoli, M.C.; Watanabe, R.; Canziani, M.E.; Santos, B.F.; Liangos, O.; Jaber, B.L.; Draibe, S.; Cendoroglo, M. Soluble Fas: A novel marker of inflammation in uremia. Artif. Organs 2003, 27, 687–691. [Google Scholar] [CrossRef] [PubMed]
- El-Agroudy, A.E.; El-Baz, A. Soluble Fas: A useful marker of inflammation and cardiovascular diseases in uremic patients. Clin. Exp. Nephrol. 2010, 14, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Yoo, T.H.; Hwang, Y.; Lee, G.H.; Kim, B.; Jang, J.; Yu, H.T.; Kim, M.C.; Cho, J.Y.; Lee, C.J.; et al. Indoxyl sulfate (IS)-mediated immune dysfunction provokes endothelial damage in patients with end-stage renal disease (ESRD). Sci. Rep. 2017, 7, 3057. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chmielewski, M.; Bragfors-Helin, A.C.; Stenvinkel, P.; Lindholm, B.; Anderstam, B. Serum soluble CD36, assessed by a novel monoclonal antibody-based sandwich ELISA, predicts cardiovascular mortality in dialysis patients. Clin. Chim. Acta 2010, 411, 2079–2082. [Google Scholar] [CrossRef] [PubMed]
- Huh, H.Y.; Lo, S.K.; Yesner, L.M.; Silverstein, R.L. CD36 induction on human monocytes upon adhesion to tumor necrosis factor-activated endothelial cells. J. Biol. Chem. 1995, 270, 6267–6271. [Google Scholar] [CrossRef] [PubMed]
- Lekawanvijit, S.; Kompa, A.R.; Krum, H. Protein-bound uremic toxins: A long overlooked culprit in cardiorenal syndrome. Am. J. Physiol. Ren. Physiol. 2016, 311, F52–F62. [Google Scholar] [CrossRef] [PubMed]
- Kashiyama, K.; Sonoda, S.; Muraoka, Y.; Suzuki, Y.; Kamezaki, F.; Tsuda, Y.; Araki, M.; Tamura, M.; Takeuchi, M.; Abe, H.; et al. Coronary plaque progression of non-culprit lesions after culprit percutaneous coronary intervention in patients with moderate to advanced chronic kidney disease: Intravascular ultrasound and integrated backscatter intravascular ultrasound study. Int. J. Cardiovasc. Imaging 2015, 31, 935–945. [Google Scholar] [CrossRef] [PubMed]
- Ross, R. Atherosclerosis-an inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Jofre, R.; Rodriguez-Benitez, P.; Lopez-Gomez, J.M.; Perez-Garcia, R. Inflammatory syndrome in patients on hemodialysis. J. Am. Soc. Nephrol. 2006, 17, S274–S280. [Google Scholar] [CrossRef] [PubMed]
- Vanholder, R.; De Smet, R. Pathophysiologic effects of uremic retention solutes. J. Am. Soc. Nephrol. 1999, 10, 1815–1823. [Google Scholar] [PubMed]
- Yadav, A.K.; Lal, A.; Jha, V. Association of circulating fractalkine (CX3CL1) and CX3CR1 (+) CD4 (+) T cells with common carotid artery intima-media thickness in patients with chronic kidney disease. J. Atheroscler. Thromb. 2011, 18, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Jiang, D. Fractalkine/CX3CR1 and atherosclerosis. Clin. Chim. Acta 2011, 412, 1180–1186. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.D.; Bursill, C.; Guzik, T.J.; Sadowski, J.; Channon, K.M.; Greaves, D.R. Smooth muscle cells in human atherosclerotic plaques express the fractalkine receptor CX3CR1 and undergo chemotaxis to the CX3C chemokine fractalkine (CX3CL1). Circulation 2003, 108, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.J.; Wu, V.; Wu, P.C.; Wu, C.J. Meta-Analysis of the Associations of p-Cresyl Sulfate (PCS) and Indoxyl Sulfate (IS) with Cardiovascular Events and All-Cause Mortality in Patients with Chronic Renal Failure. PLoS ONE 2015, 10, e0132589. [Google Scholar] [CrossRef] [PubMed]
- Richter, B.; Koller, L.; Hohensinner, P.J.; Rychli, K.; Zorn, G.; Goliasch, G.; Berger, R.; Mörtl, D.; Maurer, G.; Huber, K.; et al. Fractalkine is an independent predictor of mortality in patients with advanced heart failure. Thromb. Haemost. 2012, 6, 1220–1227. [Google Scholar]
- Leurs, P.; Lindholm, B. The AGE-RAGE pathway and its relation to cardiovascular disease in patients with chronic kidney disease. Arch. Med. Res. 2013, 44, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Selvin, E.; Halushka, M.K.; Rawlings, A.M.; Hoogeveen, R.C.; Ballantyne, C.M.; Coresh, J.; Astor, B.C. sRAGE and risk of diabetes, cardiovascular disease, and death. Diabetes 2013, 62, 2116–2121. [Google Scholar] [CrossRef] [PubMed]
- Rebholz, C.M.; Astor, B.C.; Grams, M.E.; Halushka, M.K.; Lazo, M.; Hoogeveen, R.C.; Ballantyne, C.M.; Coresh, J.; Selvin, E. Association of plasma levels of soluble receptor for advanced glycation end products and risk of kidney disease: The Atherosclerosis Risk in Communities study. Nephrol. Dial. Transpl. 2015, 30, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Loomis, S.J.; Chen, Y.; Sacks, D.B.; Christenson, E.S.; Christenson, R.H.; Rebholz, C.M.; Selvin, E. Cross-sectional Analysis of AGE-CML, sRAGE, and esRAGE with Diabetes and Cardiometabolic Risk Factors in a Community-Based Cohort. Clin. Chem. 2017, 63, 980–989. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goncalves, S.; Pecoits-Filho, R.; Perreto, S.; Barberato, S.H.; Stinghen, A.E.; Lima, E.G.; Fuerbringer, S.; Sauthier, S.M.; Riella, M.C. Associations between renal function, volume status and endotoxaemia in chronic kidney disease patients. Nephrol. Dial. Transpl. 2006, 21, 2788–2794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guilgen, G.; Werneck, M.L.; de Noronha, L.; Martins, A.P.; Varela, A.M.; Nakao, L.S.; Pecoits-Filho, R. Increased calcification and protein nitration in arteries of chronic kidney disease patients. Blood Purif. 2011, 32, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F.; Feldman, H.I., 3rd; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Levin, A.; Stevens, P.E.; Bilous, R.W.; Coresh, J.; De Francisco, A.L.M.; De Jong, P.E.; Griffith, K.E.; Hemmelgarn, B.R.; Iseki, K.; Lamb, E.J.; et al. KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. Suppl. 2013, 3, 1–150. [Google Scholar]
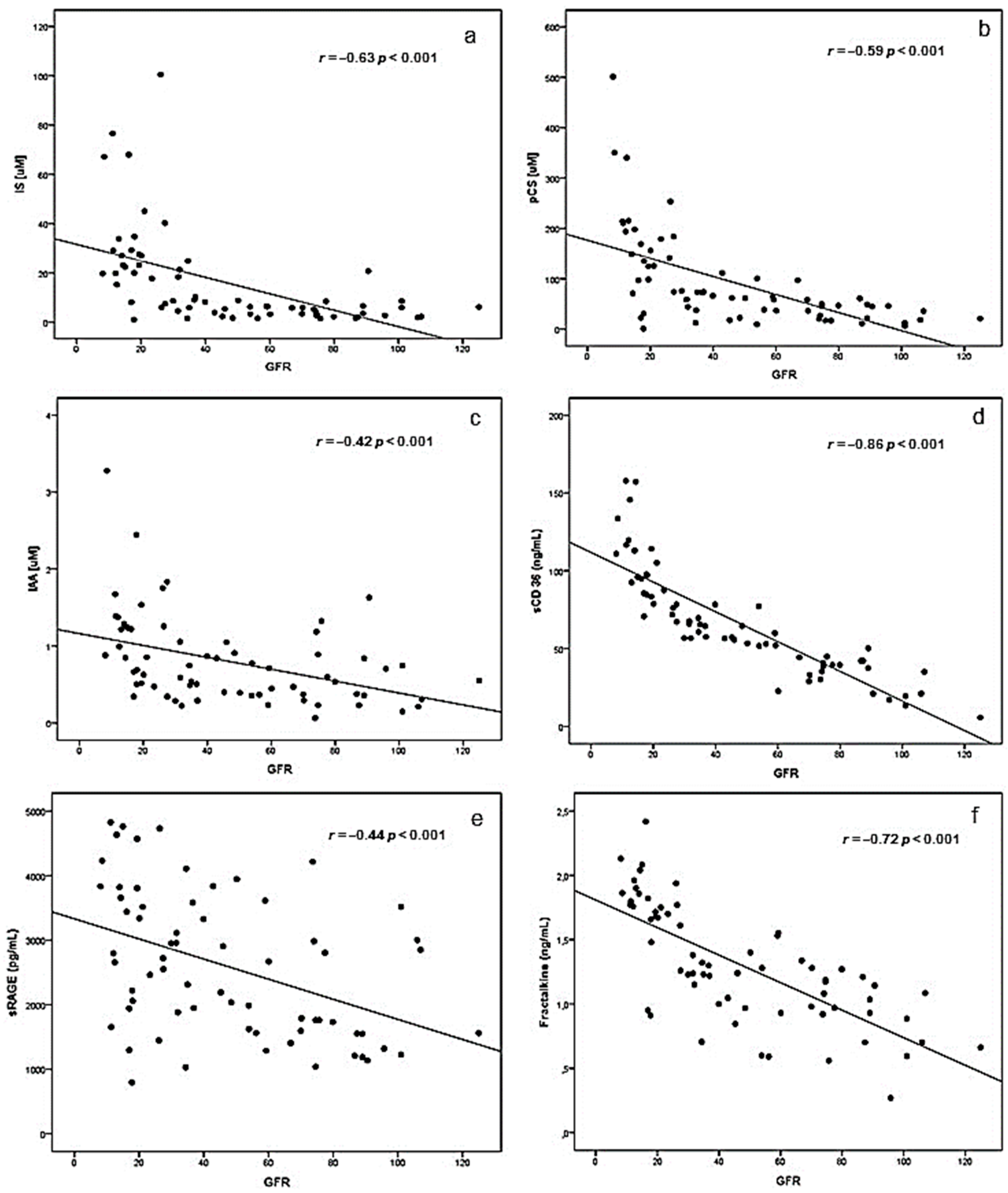
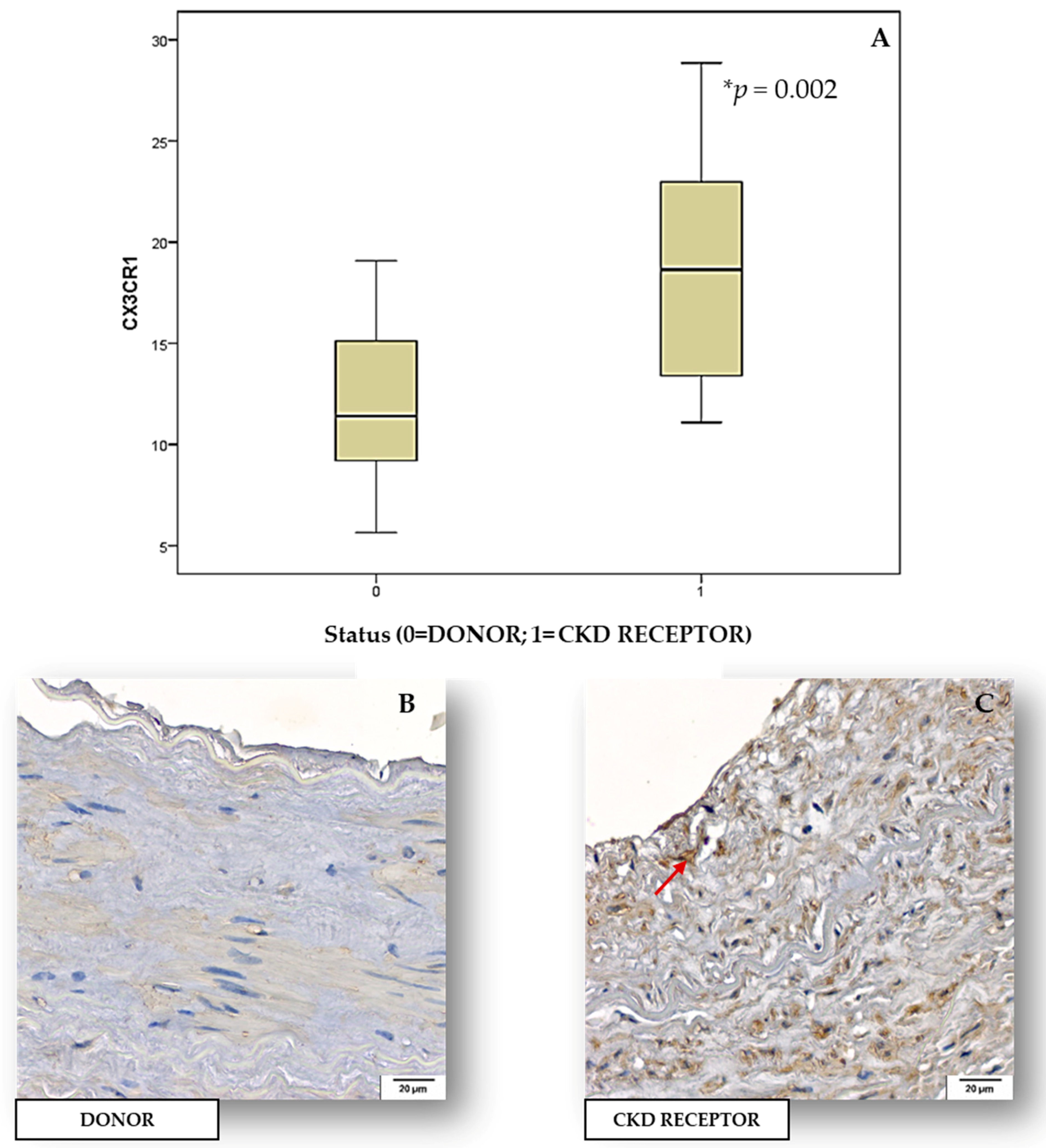
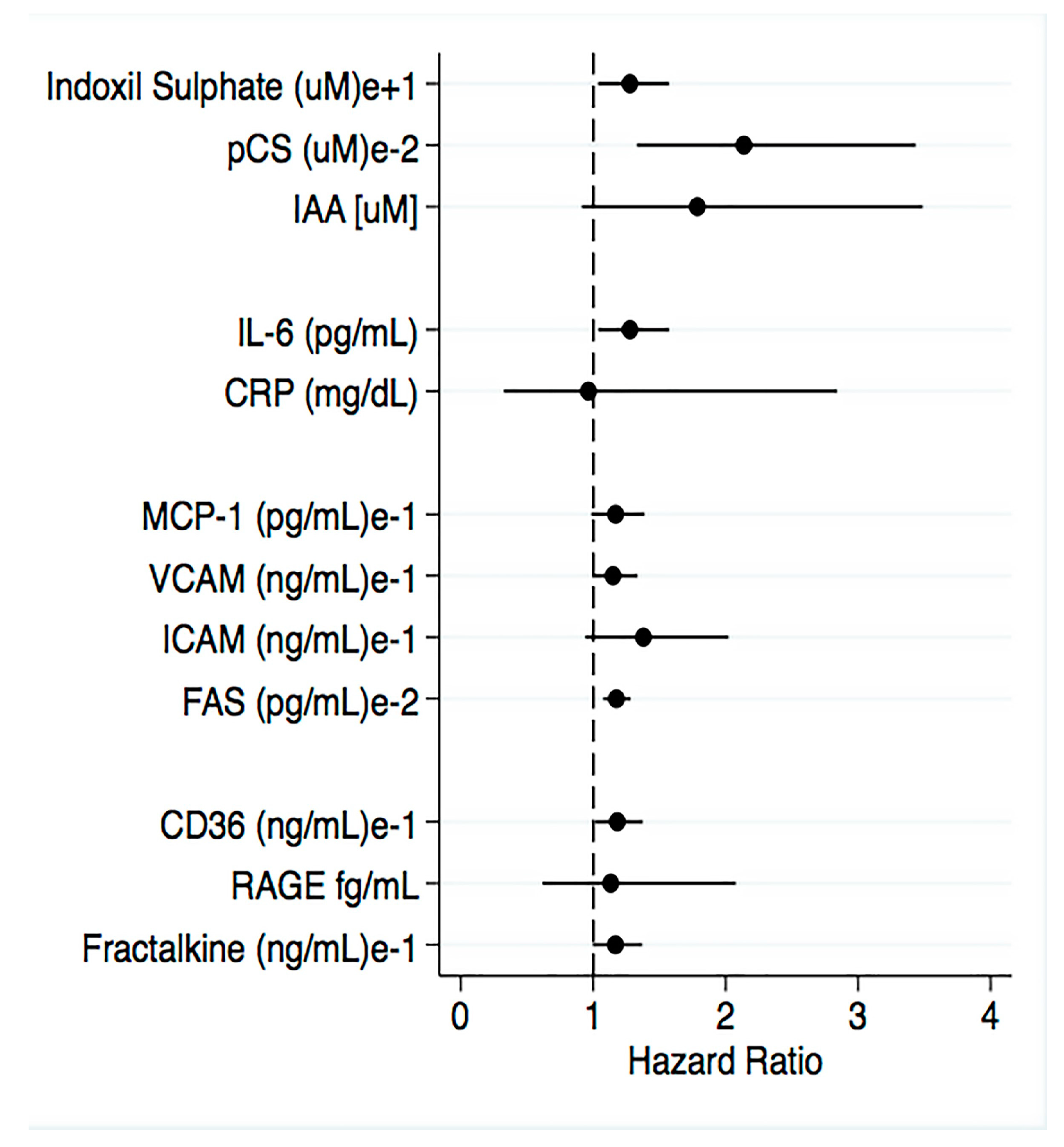

| Analyzed Parameters | |||||
|---|---|---|---|---|---|
| Patients, n | 67 | ||||
| Traditional risk factors | CKD 1 (n = 7) | CKD 2 (n = 15) | CKD 3 (n = 20) | CKD 4 (n = 16) | CKD 5 (n = 9) |
| Mean age ± SD, years | 45 ± 16.7 | 54 ± 11.2 | 61 ± 11.2 | 56 ± 14.9 | 61 ± 8.7 |
| Gender, % females | 6 | 12 | 21 | 10 | 8 |
| Race, % caucasians | 9 | 17 | 28 | 22 | 14 |
| Mean BMI ± SD | 29.6 ± 5.3 | 28.2 ± 3.7 | 28.4 ± 4.0 | 28.4 ± 4.5 | 28 ± 6.1 |
| Diabetes, % | 3 | 9 | 12 | 9 | 3 |
| Hypertension, % | 3 | 12 | 19 | 15 | 8 |
| CVD, % | 9 | 13 | 15 | 10 | 6 |
| Dyslipidemia, % | 5 | 6 | 6 | 5 | 5 |
| Smoking, % | 2 | 3 | 3 | 2 | 3 |
| LVH, % | - | 6 | 6 | 9 | 3 |
| Plaques, % | 3 | 5 | 8 | 9 | 6 |
| Primary Kidney Disease | |||||
| Diabetic nephropathy, % | 3 | 9 | 9 | 5 | 5 |
| Glomeruloesclerosis hypertensive, % | - | 5 | 10 | 9 | 5 |
| Chronic glomerulonephritis, % | 6 | 2 | 2 | 5 | - |
| Others and unknown, % | 2 | 3 | 3 | 3 | 2 |
| Laboratory Parameters | |||||
| GFR (CKD-EPI), mL/min Median (range) | 101.0 90.6–125 | 74.6 60.1–89 | 41.4 30–59.3 | 19.4 15–27.5 | 12.3 8.1–14.4 |
| Proteinuria, mg/24 h | 222.1 | 277.0 | 1091.0 | 879.0 | 3232.0 |
| Albumin, mg/dL | 4.2 | 4.0 | 3.8 | 3.8 | 3.8 |
| Glucose, mg/dL | 85.5 | 92.2 | 106.4 | 122.8 | 107.8 |
| HDL cholesterol, mg/dL | 47.6 | 41.0 | 44.4 | 38.2 | 39.7 |
| LDL cholesterol, mg/dL | 112.0 | 100 | 122 | 109.6 | 135.4 |
| Triglycerides, mg/dL | 175.1 | 217.2 | 168.2 | 202.3 | 191.1 |
| Calcium, mg/dL | 9.2 | 9.3 | 8.9 | 9.0 | 9.2 |
| Phosphorus, mg/dL | 3.3 | 3.7 | 3.7 | 4.3 | 5.0 |
| Hemoglobin, g/dL | 14.6 | 14.4 | 13.9 | 12.0 | 12.3 |
| Analyzed Parameters | ||
|---|---|---|
| Traditional risk factors | Control group (n = 12) | CKD group (n = 14) |
| Mean age ± SD, years | 41.6 ± 10.1 | 37.1 ± 13.1 |
| Gender, % females | 50 | 43 |
| Mean BMI ± SD | 24.7 ± 3.5 | 24.3 ± 3.2 |
| Hypertension, % | 0 | 100 |
| Primary Kidney Disease | ||
| Hypertensive nephrosclerosis, % | - | 43 |
| Chronic glomerulonephritis, % | - | 43 |
| Others and unknown, % | - | 14 |
| Laboratory Parameters | ||
| Creatinine, mg/dL | 0.8 ± 0.2 | 8.6 ± 3.2 |
| Glucose, mg/dL | 93 ± 17.2 | 112.0 ± 28.1 |
| HDL cholesterol, mg/dL | 50.1 ± 10.7 | 45.4 ± 17.4 |
| LDL cholesterol, mg/dL | 112 ± 28.2 | 114 ± 30.3 |
| Calcium, mg/dL | 9.1 ± 0.5 | 9.5 ± 0.9 |
| Phosphorus, mg/dL | 4.01 ± 1.0 | 5.8 ± 1.9 |
| Hemoglobin, g/dL | 13.5 ± 1.9 | 10.6 ± 1.5 |
| Analyzed Parameters | Mean ± SD | Median (Range) |
|---|---|---|
| Uremic Toxins | ||
| IS, uM | 15.8 ±19.4 | 7.6 (1.1–100.5) |
| pCS, uM | 91.5 ± 92.2 | 60.7 (0.9–501.0) |
| IAA, uM | 0.80 ± 0.57 | 0.66 (0.06–3.28) |
| Inflammatory Biomarkers | ||
| IL-6, pg/mL | 4.91 ± 3.37 | 3.69 (0.67–11.0) |
| hsCRP, mg/L | 5.18 ± 6.66 | 2.8 (0.30–39.9) |
| MCP-1, pg/mL | 105.9 ± 31 | 102.9 (54.4 0–229.0) |
| sVCAM, ng/mL | 806 ± 392 | 689 (378–1849) |
| sICAM, ng/mL | 81 ± 14.5 | 80.8 (39.6–156.8) |
| sFas pg/mL | 1339 ± 659 | 1253 (306–4181) |
| BUCVR | ||
| sCD36, ng/mL | 66.6 ± 34.4 | 60.9 (5.8–157.9) |
| sRAGE, pg/mL | 2594 ± 1115 | 2552 (795–4827) |
| Fractalkine, ng/mL | 1.30 ± 0.46 | 1.24 (0.27–2.42) |
| IS | pCS | IAA | ||||
|---|---|---|---|---|---|---|
| r | p | r | p | r | p | |
| IL-6, pg/mL | 0.13 | 0.29 | 0.236 | 0.055 | 0.078 | 0.53 |
| hsCRP, mg/L | 0.10 | 0.42 | 0.024 | 0.84 | 0.020 | 0.87 |
| MCP-1, pg/mL | 0.38 | 0.001 | 0.42 | <0.001 | 0.29 | 0.015 |
| sVCAM, ng/mL | 0.29 | 0.019 | 0.29 | 0.017 | 0.27 | 0.025 |
| sICAM, ng/mL | 0.047 | 0.71 | 0.12 | 0.34 | 0.087 | 0.48 |
| sFas, pg/mL | 0.41 | 0.001 | 0.46 | <0.001 | 0.19 | 0.13 |
| IS | pCS | IAA | ||||
|---|---|---|---|---|---|---|
| r | p | r | p | r | p | |
| sCD36, ng/mL | 0.62 | <0.001 | 0.55 | <0.001 | 0.52 | <0.001 |
| sRAGE, pg/mL | 0.48 | <0.001 | 0.48 | <0.001 | 0.20 | 0.108 |
| Fractalkine, ng/mL | 0.77 | <0.001 | 0.77 | <0.001 | 0.41 | <0.001 |
| MCP-1 | sICAM | sCD36 | sRAGE | Fractalkine | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| r | p | r | p | r | p | r | p | r | p | |
| IL-6, pg/mL | 0.36 | 0.003 | NS | NS | NS | NS | ||||
| hsCRP, mg/L | NS | 0.37 | 0.002 | NS | NS | NS | ||||
| MCP-1, pg/mL | - | NS | 0.30 | 0.015 | 0.28 | 0.022 | 0.38 | 0.002 | ||
| sVCAM, ng/mL | 0.41 | 0.001 | NS | 0.33 | 0.006 | NS | NS | |||
| sICAM, ng/mL | NS | - | NS | NS | NS | |||||
| sFas, pg/mL | NS | NS | 0.47 | <0.001 | 0.39 | 0.001 | 0.43 | <0.001 | ||
| sCD36, ng/mL | 0.30 | 0.015 | NS | - | 0.42 | <0.001 | 0.73 | <0.001 | ||
| sRAGE, pg/mL | 0.28 | 0.022 | NS | 0.42 | <0.001 | - | 0.50 | <0.001 | ||
| Fractalkine, ng/mL | 0.38 | 0.002 | NS | 0.73 | <0.001 | 0.50 | <0.001 | - | ||
| PLAQUES | ||||
|---|---|---|---|---|
| No (n = 44) | Yes (n = 20) | p | ||
| UT | IS, uM | 16.6 ± 21.3 | 14.9 ± 16.0 | 0.957 |
| pCS, uM | 74.9 ± 66.1 | 113 ± 96.5 | 0.160 | |
| IAA, uM | 0.75 ± 0.47 | 0.92 ± 0.77 | 0.378 | |
| Inflammatory Biomarkers | IL-6, pg/mL | 4.4 ± 3.1 | 5.9 ± 3.6 | 0.106 |
| hsCRP, mg/L | 5.1 ± 7.5 | 5.3 ± 4.6 | 0.921 | |
| MCP-1, pg/mL | 102.9 ± 27.9 | 113.4 ± 36.3 | 0.212 | |
| sVCAM-1, ng/mL | 772.7 ± 341.1 | 884.1 ± 480.4 | 0.293 | |
| sICAM-1, ng/mL | 81.0 ± 9.5 | 82.2 ± 22.3 | 0.762 | |
| sFas, pg/mL | 1418.8 ± 694.8 | 1224.0 ± 584.7 | 0.289 | |
| BUCVR | sCD 36, ng/mL | 61.0 ± 30.2 | 79.1 ± 40.1 | 0.049 |
| sRAGE, pg/mL | 2661 ± 1108 | 2508 ± 1129 | 0.611 | |
| Fractalkine, ng/mL | 1.25 ± 0.45 | 1.40 ± 0.45 | 0.223 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Claro, L.M.; Moreno-Amaral, A.N.; Gadotti, A.C.; Dolenga, C.J.; Nakao, L.S.; Azevedo, M.L.V.; De Noronha, L.; Olandoski, M.; De Moraes, T.P.; Stinghen, A.E.M.; et al. The Impact of Uremic Toxicity Induced Inflammatory Response on the Cardiovascular Burden in Chronic Kidney Disease. Toxins 2018, 10, 384. https://doi.org/10.3390/toxins10100384
Claro LM, Moreno-Amaral AN, Gadotti AC, Dolenga CJ, Nakao LS, Azevedo MLV, De Noronha L, Olandoski M, De Moraes TP, Stinghen AEM, et al. The Impact of Uremic Toxicity Induced Inflammatory Response on the Cardiovascular Burden in Chronic Kidney Disease. Toxins. 2018; 10(10):384. https://doi.org/10.3390/toxins10100384
Chicago/Turabian StyleClaro, Ligia Maria, Andrea N. Moreno-Amaral, Ana Carolina Gadotti, Carla J. Dolenga, Lia S. Nakao, Marina L.V. Azevedo, Lucia De Noronha, Marcia Olandoski, Thyago P. De Moraes, Andréa E. M. Stinghen, and et al. 2018. "The Impact of Uremic Toxicity Induced Inflammatory Response on the Cardiovascular Burden in Chronic Kidney Disease" Toxins 10, no. 10: 384. https://doi.org/10.3390/toxins10100384






