Clostridium perfringens Iota-Toxin: Structure and Function
Abstract
:1. Introduction
| Molecular weight | Iota a (Ia) 47,605 Da (413 residues) |
| Iota b (Ib) 74,147 Da (664 residues) | |
| Biological activity | Lethality, Dermonecrosis, Cytotoxicity |
| Enzymatic activity of Ia | NAD+-glycohydrolase (NADase) |
| ADP-ribosyltransferase (ARTase) |
| Toxin | Enzymatic component | Binding component |
|---|---|---|
| C. perfringens Iota-toxin | Ia | Ib |
| B. anthracis Toxin | Lethal factor | Protective antigen |
| Edema factor | ||
| C. botulinum C2toxin | C2I | C2II |
| C. spiroforme Iota-like toxin | Sa | Sb |
2. Characteristics of Iota-Toxin



3. Structure and Function
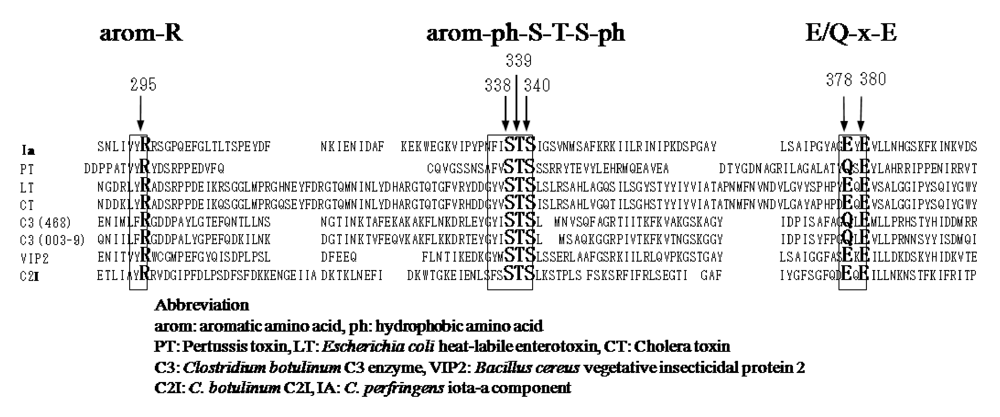
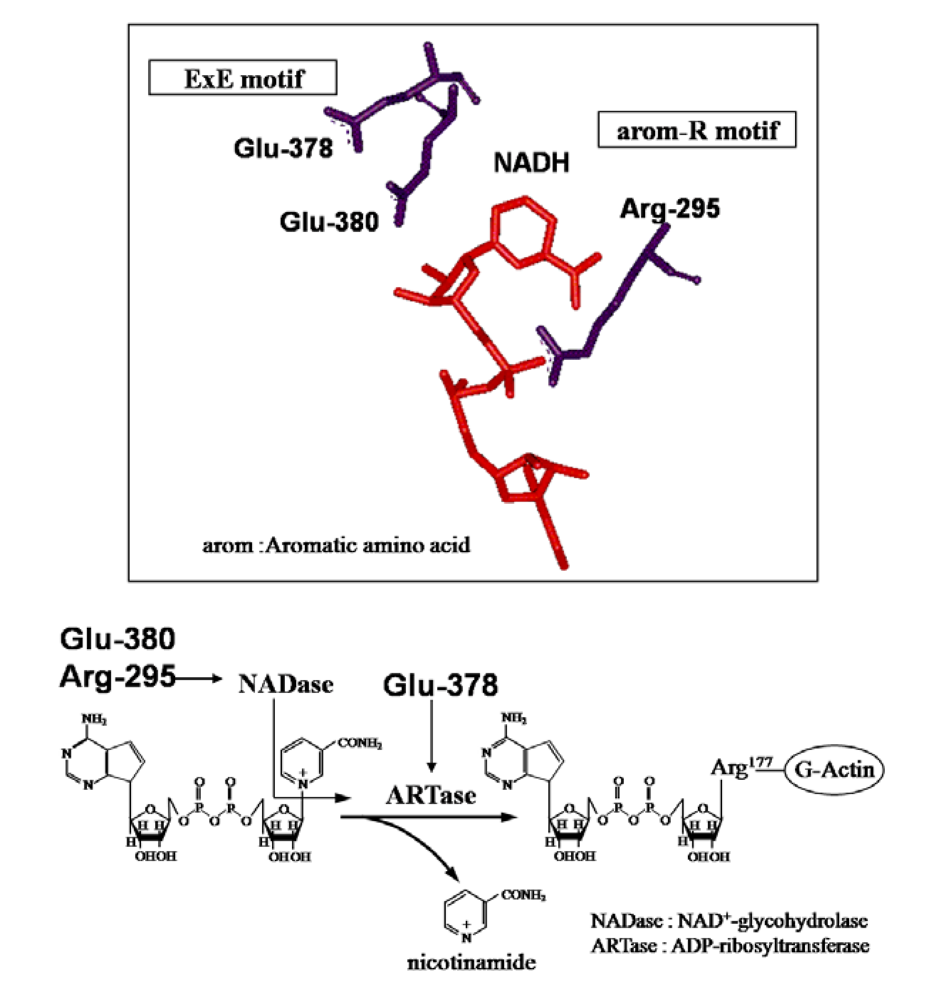


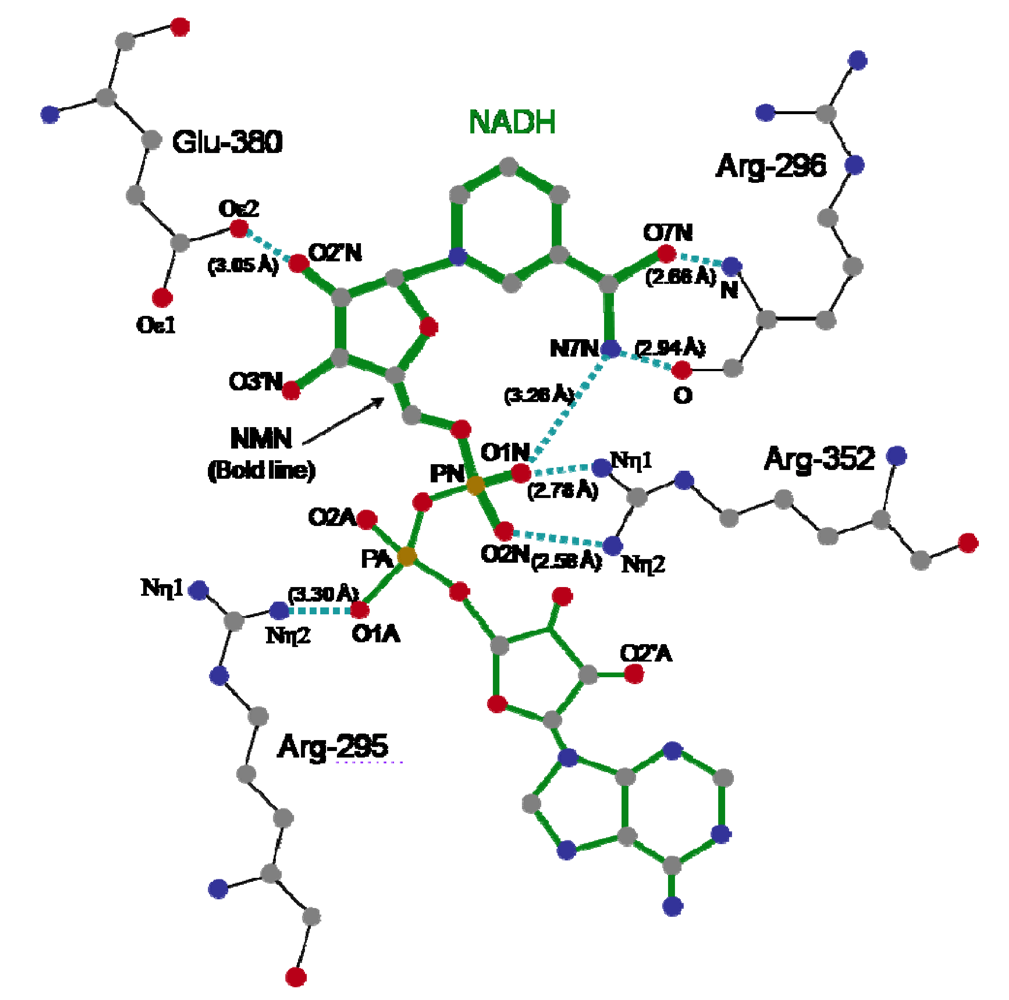


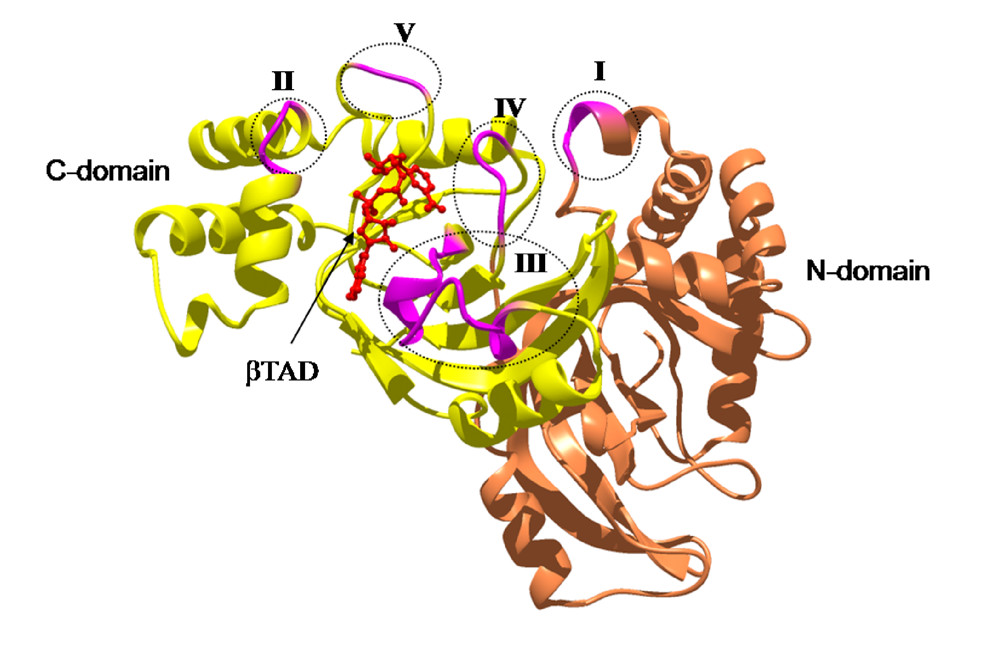
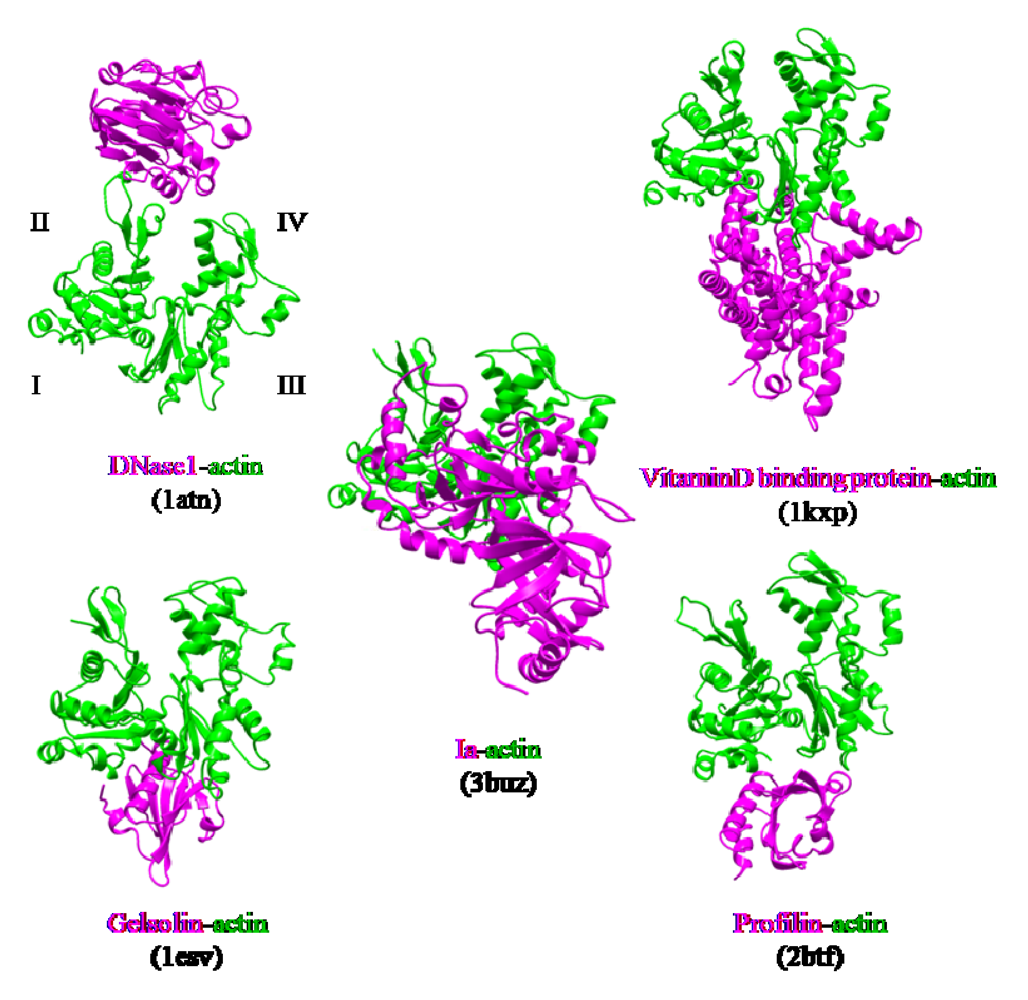
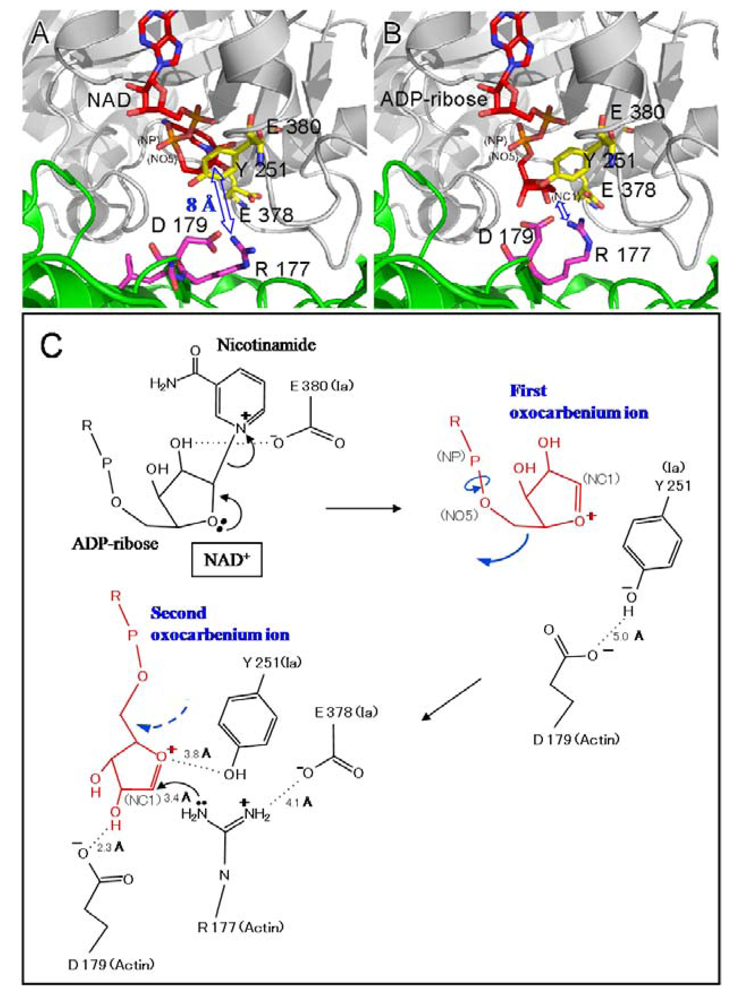
4. Mode of Action
5. Conclusions

References
- McDonel, J.L. Pharmacology of Bacterial Toxins; Pergamon Press: New York, NY, USA, 1986; pp. 477–517. [Google Scholar]
- Sakurai, J. Toxins of Clostridium perfringens. Rev. Med. Microbiol. 1995, 6, 175–185. [Google Scholar]
- Sakurai, J.; Nagahama, M.; Ochi, S. Major toxins of Clostridium perfringens. J. Toxicol.-Toxin Rev. 1997, 16, 195–214. [Google Scholar]
- Songer, J.G. Clostridial enteric diseases of domestic animals. Clin. Microbiol. Rev. 1996, 9, 216–234. [Google Scholar] [PubMed]
- Barth, H.; Aktories, K.; Popoff, M.R.; Stiles, B.G. Binary bacterial toxins: Biochemistry, biology, and applications of common Clostridium and Bacillus proteins. Microbiol. Mol. Biol. Rev. 2004, 68, 373–402. [Google Scholar] [PubMed]
- Aktories, K.; Wegner, A. ADP-ribosylation of actin by clostridial toxins. J. Cell Biol. 1989, 109, 1385–1387. [Google Scholar] [PubMed]
- Gibert, M.; Petit, L.; Raffestin, S.; Okabe, A.; Popoff, M.R. Clostridium perfringens iota-toxin requires activation of both binding and enzymatic components for cytopathic activity. Infect. Immun. 2000, 68, 3848–3853. [Google Scholar] [PubMed]
- Bosworth, T.J. On a new type of toxin produced by Clostridium welchii. J. Comp. Pathol. 1943, 53, 245–255. [Google Scholar]
- Stiles, B.G.; Wilkins, T.D. Purification and characterization of Clostridium perfringens iota toxin: Dependence on two nonlinked proteins for biological activity. Infect. Immun. 1986, 54, 683–688. [Google Scholar] [PubMed]
- Perelle, S.; Gibert, M.; Boquet, P.; Popoff, M.R. Characterization of Clostridium perfringens iota-toxin genes and expression in Escherichia coli. Infect. Immun. 1993, 61, 5147–5156. [Google Scholar] [PubMed]
- Sakurai, J.; Kobayashi, K. Lethal and dermonecrotic activities of Clostridium perfringens iota toxin: Biological activities induced by cooperation of two nonlinked components. Microbiol. Immunol. 1995, 39, 249–253. [Google Scholar] [PubMed]
- Krueger, K.M.; Barbieri, J.T. The family of bacterial ADP-ribosylating exotoxins. Clin. Microbiol. Rev. 1995, 8, 34–47. [Google Scholar] [PubMed]
- Holbourn, K.P.; Shone, C.C.; Acharya, K.R. A family of killer toxins. Exploring the mechanism of ADP-ribosylating toxins. FEBS J. 2006, 273, 4579–4593. [Google Scholar] [CrossRef] [PubMed]
- Gill, D.M.; Meren, R. ADP-ribosylation of membrane proteins catalyzed by cholera toxin: Basis of the activation of adenylate cyclase. Proc. Natl. Acad. Sci. USA. 1978, 75, 3050–3054. [Google Scholar] [CrossRef]
- Katada, T.; Ui, M. Direct modification of the membrane adenylate cyclase system by islet-activating protein due to ADP-ribosylation of a membrane protein. Proc. Natl. Acad. Sci. USA 1982, 79, 3129–3133. [Google Scholar]
- Moss, J.; Garrison, S.; Oppenheimer, N.J.; Richardson, S.H. NAD-dependent ADP-ribosylation of arginine and proteins by Escherichia coli heat-labile enterotoxin. J. Biol. Chem. 1979, 254, 6270–6272. [Google Scholar] [PubMed]
- van Ness, B.G.; Howard, J.B.; Bodley, J.W. ADP-ribosylation of elongation factor 2 by diphtheria toxin. Isolation and properties of the novel ribosyl-amino acid and its hydrolysis products. J. Biol. Chem. 1980, 255, 10717–10720. [Google Scholar] [PubMed]
- Deng, Q.; Barbieri, J.T. Molecular mechanisms of the cytotoxicity of ADP-ribosylating toxins. Ann. Rev. Microbiol. 2008, 62, 271–288. [Google Scholar] [CrossRef]
- Aktories, K.; Weller, U.; Chhatwal, G.S. Clostridium botulinum type C produces a novel ADP-ribosyltransferase distinct from botulinum C2 toxin. FEBS Lett. 1987, 212, 109–113. [Google Scholar] [PubMed]
- Aktories, K.; Bärmann, M.; Ohishi, I.; Tsuyama, S.; Jakobs, K.H.; Habermann, E. Botulinum C2 toxin ADP-ribosylates actin. Nature 1986, 322, 390–392. [Google Scholar] [CrossRef] [PubMed]
- Popoff, M.R.; Rubin, E.J.; Gill, D.M.; Boquet, P. Actin-specific ADP-ribosyltransferase produced by a Clostridium difficile strain. Infect. Immun. 1988, 56, 2299–2306. [Google Scholar] [PubMed]
- Popoff, M.R.; Boquet, P. Clostridium spiroforme toxin is a binary toxin which ADP-ribosylates cellular actin. Biochem. Biophys. Res. Commun. 1988, 152, 1361–1368. [Google Scholar] [PubMed]
- Richard, J.F.; Mainguy, G.; Gibert, M.; Marvaud, J.C.; Stiles, B.G.; Popoff, M.R. Transcytosis of iota-toxin across polarized CaCo-2 cells. Mol. Microbiol. 2002, 43, 907–917. [Google Scholar] [PubMed]
- Hilger, H.; Pust, S.; von Figura, G.; Kaiser, E.; Stiles, B.G.; Popoff, M.R.; Barth, H. The long-lived nature of Clostridium perfringens iota toxin in mammalian cells induces delayed apoptosis. Infect. Immun. 2009, 77, 5593–5601. [Google Scholar] [PubMed]
- Domenighini, M.; Rappuoli, R. Three conserved consensus sequences identify the NAD-binding site of ADP-ribosylating enzymes,expressed by eukaryotes,bacteria and T-even bacteriophages. Mol. Microbiol. 1996, 21, 667–674. [Google Scholar] [PubMed]
- Carpusca, I.; Jank, T.; Aktories, K. Bacillus sphaericus mosquitocidal toxin (MTX) and pierisin: The enigmatic offspring from the family of ADP-ribosyltransferases. Mol. Microbiol. 2006, 62, 621–630. [Google Scholar] [PubMed]
- Nagahama, M.; Sakaguchi, Y.; Kobayashi, K.; Ochi, S.; Sakurai, J. Characterization of the enzymatic component of Clostridium perfringens iota-toxin. J. Bacteriol. 2000, 182, 2096–2103. [Google Scholar] [PubMed]
- Perelle, S.; Domenighini, M.; Popoff, M.R. Evidence that Arg-295, Glu-378, and Glu-380 are active-site residues of the ADP-ribosyltransferase activity of iota toxin. FEBS Lett. 1996, 395, 191–194. [Google Scholar] [CrossRef] [PubMed]
- Barth, H.; Preiss, J.C.; Hofmann, F.; Aktories, K. Characterization of the catalytic site of the ADP-ribosyltransferase Clostridium botulinum C2 toxin by site-directed mutagenesis. J. Biol. Chem. 1998, 273, 29506–29511. [Google Scholar] [PubMed]
- Domenighini, M.; Montecucco, C.; Ripka, W.C.; Rappuoli, R. Computer modeling of the NAD binding site of ADP-ribosylating toxins: Active-site structure and mechanism of NAD binding. Mol. Microbiol. 1991, 5, 23–31. [Google Scholar] [PubMed]
- Radke, J.; Pederson, K.J.; Barbieri, J.T. Pseudomonas aeruginosa exoenzyme S is a biglutamic acid ADP-ribosyltransferase. Infect. Immun. 1999, 67, 1508–1510. [Google Scholar] [PubMed]
- Damme, J.; Jung, M.; Hofmann, F.; Just, I.; Vandekerckhove, J.; Aktories, K. Analysis of the catalytic site of the actin ADP-ribosylating Clostridium perfringens iota toxin. FEBS Lett. 1996, 380, 291–295. [Google Scholar] [PubMed]
- Cieplak, W.; Mead, D.J.; Messer, R.J.; Grant, C.C. Site-directed mutagenic alteration of potential active-site residues of the A subunit of Escherichia coli heat-labile enterotoxin. Evidence for a catalytic role for glutamic acid 112. J. Biol. Chem. 1995, 270, 30545–30550. [Google Scholar] [PubMed]
- Hara, N.; Tsuchiya, M.; Shimoyama, M. Glutamic acid 207 in rodent T-cell RT6 antigens is essential for arginine-specific ADP-ribosylation. J. Biol. Chem. 1996, 271, 29552–29555. [Google Scholar] [PubMed]
- Aktories, K.; Jung, M.; Böhmer, J.; Fritz, G.; Vandekerckhove, J.; Just, I. Studies on the active-site structure of C3-like exoenzymes: Involvement of glutamic acid in catalysis of ADP-ribosylation. Biochimie. 1995, 77, 326–332. [Google Scholar] [PubMed]
- Petosa, C.; Collier, R.J.; Klimpel, K.R.; Leppla, S.H.; Liddington, R.C. Crystal structure of the anthrax toxin protective antigen. Nature 1997, 385, 833–838. [Google Scholar] [PubMed]
- Schleberger, C.; Hochmann, H.; Barth, H.; Aktories, K.; Schulz, G.E. Structure and action of the binary C2 toxin from Clostridium botulinum. J. Mol. Biol. 2006, 364, 705–715. [Google Scholar] [PubMed]
- Gupta, P.K.; Chandra, H.; Gaur, R.; Kurupati, R.K.; Chowdhury, S.; Tandon, V.; Sigh, Y.; Maithal, K. Conformational fluctuations in anthrax protective antigen: A possible role of calcium in the folding pathway of the protein. FEBS Lett. 2003, 554, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Gao-Sheridan, S.; Zhang, S.; Collier, R.J. Exchange characteristics of calcium ions bound to anthrax protective antigen. Biochem. Biophys. Res. Commun. 2002, 300, 61–64. [Google Scholar]
- Leppla, S.A. The Comprehensive Sourcebook of Bacterial Protein Toxins, 2nd ed; Academic Press: San Diego, CA, USA, 1991; pp. 243–261. [Google Scholar]
- Nagahama, M.; Nagayasu, K.; Kobayashi, K.; Sakurai, J. Binding component of Clostridium perfringens iota-toxin induces endocytosis in vero cells. Infect. Immun. 2002, 70, 1909–1914. [Google Scholar] [PubMed]
- Kobayashi, K.; Nagahama, M.; Ohkubo, N.; Kojima, T.; Shirai, H.; Iwamoto, S.; Oda, M.; Sakurai, J. Role of Ca2+-binding motif in cytotoxicity induced by Clostridium perfringens iota-toxin. Microb. Pathog. 2008, 44, 265–270. [Google Scholar] [PubMed]
- Nagahama, M.; Yamaguchi, A.; Hagiyama, T.; Ohkubo, N.; Kobayashi, K.; Sakurai, J. Binding and internalization of Clostridium perfringens iota-toxin in lipid rafts. Infect. Immun. 2004, 72, 3267–3275. [Google Scholar] [PubMed]
- Tsuge, H.; Nagahama, M.; Nishimura, T.; Sakaguchi, Y.; Katunuma, N.; Sakurai, J. Crystallization and preliminary X-ray studies of the Ia component of Clostridium perfringens iota toxin complexed with NADPH. J. Struct. Biol. 1999, 126, 175–177. [Google Scholar] [PubMed]
- Tsuge, H.; Nagahama, M.; Nishimura, H.; Hisatsune, J.; Sakaguchi, Y.; Itogawa, Y.; Katunuma, N.; Sakurai, J. Crystal structure and site-directed mutagenesis of enzymatic components from Clostridium perfringens iota-toxin. J. Mol. Biol. 2003, 325, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Craig, J.A.; Putnam, C.D.; Carozzi, N.B.; Tainer, J.A. Evolution and mechanism from structures of an ADP-ribosylating toxin and NAD complex. Nat. Struct. Biol. 1999, 6, 932–936. [Google Scholar] [PubMed]
- Schleberger, C.; Hochmann, H.; Barth, H.; Aktories, K.; Schulz, G.E. Structure and action of the binary C2 toxin from Clostridium botulinum. J. Mol. Biol. 2006, 384, 705–715. [Google Scholar]
- Sakurai, J.; Nagahama, M.; Hisatsune, J.; Katunuma, N.; Tsuge, H. Clostridium perfringens iota-toxin, ADP-ribosyltransferase: Structure and mechanism of action. Adv. Enzyme Regul. 2003, 43, 361–377. [Google Scholar] [PubMed]
- Menetrey, J.; Flatau, G.; Stura, E.A.; Charbonnier, J.B.; Gas, F.; Teulon, J.M.; Le Du, M.H.; Boquet, P.; Menez, A. NAD binding induces conformational changes in Rho ADP-ribosylating Clostridium botulinum C3 exoenzyme. J. Biol. Chem. 2002, 277, 30950–30957. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Arvai, A.S.; Clancy, S.B.; Tainer, J.A. Crystal structure and novel recognition motif of rho ADP-ribosylating C3 exoenzyme from Clostridium botulinum: Structural insights for recognition specificity and catalysis. J. Mol. Biol. 2001, 305, 95–107. [Google Scholar] [PubMed]
- Tsuge, H.; Nagahama, M.; Oda, M.; Iwamoto, S.; Utsunomiya, H.; Marquez, V.E.; Katunuma, N.; Nishizawa, M.; Sakurai, J. Structural basis of actin recognition and arginine ADP-ribosylation by Clostridium perfringens iota-toxin. Proc. Natl. Acad. Sci. USA. 2008, 105, 7399–7404. [Google Scholar]
- McLaughlin, P.J.; Gooch, J.T.; Mannherz, H.G.; Weeds, A.G. Structure of gelsolin segment 1-actin complex and the mechanism of filament severing. Nature 1993, 364, 685–692. [Google Scholar] [PubMed]
- Otterbein, L.R.; Cosio, C.; Graceffa, P.; Dominguez, R. Crystal structures of the vitamin D-binding protein and its complex with actin: Structural basis of the actin-scavenger system. Proc. Natl.Acad. Sci. USA 2002, 99, 8003–8008. [Google Scholar]
- Kabsch, W.; Mannherz, H.G.; Suck, D.; Pai, E.F.; Holmes, K.C. Atomic structure of the actin:DNase I complex. Nature 1990, 347, 37–44. [Google Scholar] [PubMed]
- Schutt, C.E.; Myslik, J.C.; Rozycki, M.D.; Goonesekere, N.C.; Lindberg, U. The structure of crystalline profilin-beta-actin. Nature 1993, 365, 810–816. [Google Scholar] [PubMed]
- Singh, Y.; Klimpel, K.R.; Arora, N.; Sharma, M.; Leppla, S.H. The chymotrypsin-sensitive site, FFD315, in anthrax toxin protective antigen is required for translocation of lethal factor. J. Biol. Chem. 1994, 269, 29039–29046. [Google Scholar] [PubMed]
- Milne, J.C.; Furlong, D.; Hanna, P.C.; Wall, J.S.; Collier, R.J. Anthrax protective antigen forms oligomers during intoxication of mammalian cells. J. Biol. Chem. 1994, 269, 20607–20612. [Google Scholar] [PubMed]
- Barth, H.; Blocker, D.; Behlke, J.; BergsmafSchutter, W.; Brisson, A.; Benz, R.; Aktories, K. Cellular uptake of Clostridium botulinum C2 toxin requires oligomerization and acidification. J. Biol. Chem. 2000, 275, 16478–16483. [Google Scholar] [PubMed]
- Considine, R.V.; Simpson, L.L. Cellular and molecular actions of binary toxins possessing ADP-ribosyltransferase activity. Toxicon 1991, 29, 913–936. [Google Scholar] [PubMed]
- Knapp, O.; Benz, R.; Gibert, M.; Marvaud, J.C.; Popoff, M.R. Interaction of Clostridium perfringens iota-toxin with lipid bilayer membranes. Demonstration of channel formation by the activated binding component Ib and channel block by the enzyme component Ia. J. Biol. Chem. 2002, 277, 6143–6152. [Google Scholar] [PubMed]
- Blocker, D.; Behlke, J.; Aktories, K.; Barth, H. Cellular uptake of Clostridium perfringens binary iota-toxin. Infect. Immun. 2001, 69, 2980–2987. [Google Scholar] [PubMed]
- Nabi, I.R.; Le, P.U. Caveolae/raft-dependent endocytosis. J. Cell Biol. 2003, 161, 673–677. [Google Scholar] [PubMed]
- Nagahama, M.; Hayashi, S.; Morimitsu, S.; Sakurai, J. Biological activities and pore formation of Clostridium perfringens beta toxin in HL 60 cells. J. Biol. Chem. 2003, 278, 36934–36941. [Google Scholar] [PubMed]
- Nagahama, M.; Hagiyama, T.; Kojima, T.; Aoyanagi, K.; Takahashi, C.; Oda, M.; Sakaguchi, Y.; Oguma, K.; Sakurai, J. Binding and internalization of Clostridium botulinum C2 toxin. Infect. Immun. 2009, 77, 5139–5148. [Google Scholar] [PubMed]
- Duncan, M.J.; Shin, J.S.; Abraham, S.N. Microbial entry through caveolae: Variations on a theme. Cell Microbiol. 2002, 4, 783–791. [Google Scholar] [PubMed]
- Chazal, N.; Gerlier, D. Virus entry, assembly, budding, and membrane rafts. Microbiol. Mol. Biol. Rev. 2003, 67, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Marvaud, J.C.; Smith, T.; Hale, M.L.; Popoff, M.R.; Smith, L.A.; Stiles, B.G. Clostridium perfringens iota-toxin: Mapping of receptor binding and Ia docking domains on Ib. Infect. Immun. 2001, 69, 2435–2441. [Google Scholar] [PubMed]
- Schraw, W.; Li, Y.; McClain, M.S.; van der Goot, F.G.; Cover, T.L. Association of Helicobacter pylori vacuolating toxin (VacA) with lipid rafts. J. Biol. Chem. 2002, 277, 34642–34650. [Google Scholar] [PubMed]
- Gibert, M.; Marvaud, J.C.; Pereira, Y.; Hale, M.L.; Stiles, B.G.; Boquet, P.; Lamaze, C.; Popoff, M.R. Differential requirement for the translocation of clostridial binary toxins: Iota toxin requires a membrane potential gradient. FEBS Lett. 2007, 581, 1287–1296. [Google Scholar] [PubMed]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sakurai, J.; Nagahama, M.; Oda, M.; Tsuge, H.; Kobayashi, K. Clostridium perfringens Iota-Toxin: Structure and Function. Toxins 2009, 1, 208-228. https://doi.org/10.3390/toxins1020208
Sakurai J, Nagahama M, Oda M, Tsuge H, Kobayashi K. Clostridium perfringens Iota-Toxin: Structure and Function. Toxins. 2009; 1(2):208-228. https://doi.org/10.3390/toxins1020208
Chicago/Turabian StyleSakurai, Jun, Masahiro Nagahama, Masataka Oda, Hideaki Tsuge, and Keiko Kobayashi. 2009. "Clostridium perfringens Iota-Toxin: Structure and Function" Toxins 1, no. 2: 208-228. https://doi.org/10.3390/toxins1020208
APA StyleSakurai, J., Nagahama, M., Oda, M., Tsuge, H., & Kobayashi, K. (2009). Clostridium perfringens Iota-Toxin: Structure and Function. Toxins, 1(2), 208-228. https://doi.org/10.3390/toxins1020208




