Western Dietary Pattern Is Associated with Irritable Bowel Syndrome in the French NutriNet Cohort
Abstract
:1. Introduction
2. Materials and Methods
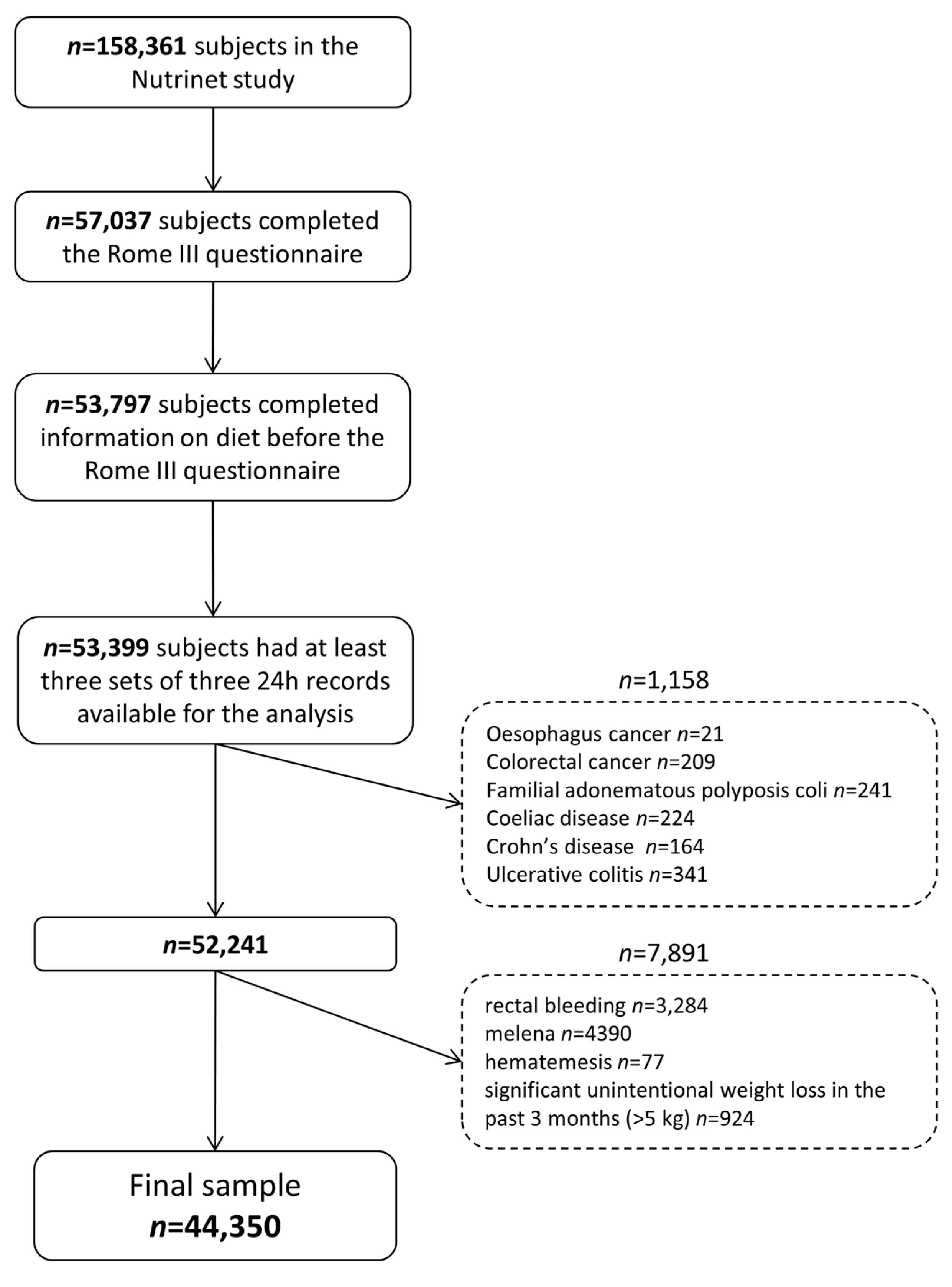
2.1. Population
2.2. Ethics
2.3. Data Collection
2.3.1. Irritable Bowel Syndrome
2.3.2. Dietary Data
2.3.3. Covariates
2.4. Statistical Analyses
3. Results
3.1. Population
3.2. Dietary Patterns
3.3. Characteristics of the Dietary Patterns in the Studied Population
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Longstreth, G.F.; Thompson, W.G.; Chey, W.D.; Houghton, L.A.; Mearin, F.; Spiller, R.C. Functional Bowel Disorders. Gastroenterology 2006, 130, 1480–1491. [Google Scholar] [CrossRef] [PubMed]
- Drossman, D.A.; Hasler, W.L. Rome IV-Functional GI Disorders: Disorders of Gut-Brain Interaction. Gastroenterology 2016, 150, 1257–1261. [Google Scholar] [CrossRef] [PubMed]
- Lovell, R.M.; Ford, A.C. Global prevalence of and risk factors for irritable bowel syndrome: A meta-analysis. Clin. Gastroenterol. Hepatol. 2012, 10, 712–721.e4. [Google Scholar] [CrossRef] [PubMed]
- Dapoigny, M.; Bellanger, J.; Bonaz, B.; Bruley des Varannes, S.; Bueno, L.; Coffin, B.; Ducrotté, P.; Flourié, B.; Lémann, M.; Lepicard, A.; et al. Irritable bowel syndrome in France: A common, debilitating and costly disorder. Eur. J. Gastroenterol. Hepatol. 2004, 16, 995–1001. [Google Scholar] [CrossRef] [PubMed]
- Monsbakken, K.W.; Vandvik, P.O.; Farup, P.G. Perceived food intolerance in subjects with irritable bowel syndrome—Etiology, prevalence and consequences. Eur. J. Clin. Nutr. 2006, 60, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Boettcher, E.; Crowe, S.E. Dietary Proteins and Functional Gastrointestinal Disorders. Am. J. Gastroenterol. 2013, 108, 728–736. [Google Scholar] [CrossRef] [PubMed]
- Farre, R.; Tack, J. Food and Symptom Generation in Functional Gastrointestinal Disorders: Physiological Aspects. Am. J. Gastroenterol. 2013, 108, 698–706. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, S.J.; Lomer, M.C.E.; Gibson, P.R. Short-Chain Carbohydrates and Functional Gastrointestinal Disorders. Am. J. Gastroenterol. 2013, 108, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.R.; Varney, J.; Malakar, S.; Muir, J.G. Food components and irritable bowel syndrome. Gastroenterology 2015, 148, 1158–1174.e4. [Google Scholar] [CrossRef] [PubMed]
- Simren, M.; Mansson, A.; Langkilde, A.M.; Svedlund, J.; Abrahamsson, H.; Bengtsson, U.; Bjornsson, E.S. Food-related gastrointestinal symptoms in the irritable bowel syndrome. Digestion 2001, 63, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Saito, Y.A.; Locke, G.R.; Weaver, A.L.; Zinsmeister, A.R.; Talley, N.J. Diet and functional gastrointestinal disorders: A population-based case-control study. Am. J. Gastroenterol. 2005, 100, 2743–2748. [Google Scholar] [CrossRef] [PubMed]
- Gupta, D.; Ghoshal, U.C.; Misra, A.; Misra, A.; Choudhuri, G.; Singh, K. Lactose intolerance in patients with irritable bowel syndrome from northern India: A case-control study. J. Gastroenterol. Hepatol. 2007, 22, 2261–2265. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, S.J.; Parker, F.C.; Muir, J.G.; Gibson, P.R. Dietary Triggers of Abdominal Symptoms in Patients with Irritable Bowel Syndrome: Randomized placebo-controlled evidence. Clin. Gastroenterol. Hepatol. 2008, 6, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Bijkerk, C.J.; de Wit, N.J.; Muris, J.W.M.; Whorwell, P.J.; Knottnerus, J.A.; Hoes, A.W. Soluble or insoluble fibre in irritable bowel syndrome in primary care? Randomised placebo controlled trial. BMJ 2009, 339, b3154. [Google Scholar] [CrossRef] [PubMed]
- Bohn, L.; Storsrud, S.; Tornblom, H.; Bengtsson, U.; Simren, M. Self-Reported Food-Related Gastrointestinal Symptoms in IBS Are Common and Associated With More Severe Symptoms and Reduced Quality of Life. Am. J. Gastroenterol. 2013, 108, 634–641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eswaran, S.L.; Chey, W.D.; Han-Markey, T.; Ball, S.; Jackson, K. A Randomized Controlled Trial Comparing the Low FODMAP Diet vs. Modified NICE Guidelines in US Adults with IBS-D. Am. J. Gastroenterol. 2016, 111, 1824–1832. [Google Scholar] [CrossRef] [PubMed]
- Newby, P.K.; Tucker, K.L. Empirically Derived Eating Patterns Using Factor or Cluster Analysis: A review. Nutr. Rev. 2004, 62, 177–203. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.R., Jr.; Gross, M.D.; Steffen, L.; Steffes, M.W.; Yu, X.; Svetkey, L.P.; Appel, L.J.; Vollmer, W.M.; Bray, G.A.; Moore, T.; et al. The Effects of Dietary Patterns on Urinary Albumin Excretion: Results of the dietary approaches to stop hypertension (DASH) trial. Am. J. Kidney Dis. 2009, 53, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Kant, A.K. Dietary patterns and health outcomes. J. Am. Diet. Assoc. 2004, 104, 615–635. [Google Scholar] [CrossRef] [PubMed]
- Khayyatzadeh, S.S.; Esmaillzadeh, A.; Saneei, P.; Keshteli, A.H.; Adibi, P. Dietary patterns and prevalence of irritable bowel syndrome in Iranian adults. Neurogastroenterol. Motil. 2016, 28, 1921–1933. [Google Scholar] [CrossRef] [PubMed]
- Hercberg, S.; Castetbon, K.; Czernichow, S.; Malon, A.; Mejean, C.; Kesse, E.; Touvier, M.; Galan, P. The Nutrinet-Santé Study: A web-based prospective study on the relationship between nutrition and health and determinants of dietary patterns and nutritional status. BMC Public Health 2010, 10, 242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rome Foundation. Guidelines—Rome III diagnostic criteria for functional gastrointestinal disorders. J. Gastrointest. Liver Dis. 2006, 15, 307–312. [Google Scholar]
- Drossman, D.A. The functional gastrointestinal disorders and the Rome III process. Gastroenterology 2006, 130, 1377–1390. [Google Scholar] [CrossRef] [PubMed]
- Hercberg, S.; Deheeger, M.; Preziosi, P.; Collectif. Portions Alimentaires; Manuel Photos Pour L’estimation des Quantités; Polytechnica: Paris, France, 2012. [Google Scholar]
- Etude Nutrinet Santé. Table de Composition des Aliments; Economica: Paris, France, 2013. [Google Scholar]
- Touvier, M.; Kesse-Guyot, E.; Méjean, C.; Pollet, C.; Malon, A.; Castetbon, K.; Hercberg, S. Comparison between an interactive web-based self-administered 24 h dietary record and an interview by a dietitian for large-scale epidemiological studies. Br. J. Nutr. 2011, 105, 1055–1064. [Google Scholar] [CrossRef] [PubMed]
- Lassale, C.; Castetbon, K.; Lapote, F.; Camilleri, G.M.; Deschamps, V.; Vernay, M.; Faure, P.; Hercberg, S.; Galan, P.; Kesse-Guyot, E. Validation of a Web-based, self-administered, non-consecutive-day dietary record tool against urinary biomarkers. Br. J. Nutr. 2015, 113, 953–962. [Google Scholar] [CrossRef] [PubMed]
- Lassale, C.; Castetbon, K.; Laporte, F.; Deschamps, V.; Vernay, M.; Camilleri, G.M.; Faure, P.; Hercberg, S.; Galan, P.; Kesse-Guyot, E. Correlations between fruit, vegetables, fish, vitamins, and fatty acids estimated by web-based nonconsecutive dietary records and respective biomarkers of nutritional status. J. Acad. Nutr. Diet. 2016, 116, 427–438.e5. [Google Scholar] [CrossRef] [PubMed]
- Consumption Unit. Available online: https://www.insee.fr/fr/metadonnees/definition/c1802 (accessed on 31 January 2017).
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Assmann, K.E.; Lassale, C.; Andreeva, V.A.; Jeandel, C.; Hercberg, S.; Galan, P.; Kesse-Guyot, E. A healthy dietary pattern at midlife, combined with a regulated energy intake, is related to increased odds for healthy aging. J. Nutr. 2015, 145, 2139–2145. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, H.F. The varimax criterion for analytic rotation in factor analysis. Psychometrika 1958, 23, 187–200. [Google Scholar] [CrossRef]
- O’Rourke, N.O.; Hatcher, L.H. A Step-by-Step Approach to Using SAS for Factor Analysis and Structural Equation Modeling, 2nd ed.; SAS Institute: Cary, NC, USA, 2013. [Google Scholar]
- Schafer, J.L.; Olsen, M.K. Multiple imputation for multivariate missing-data problems: A data analyst’s perspective. Multivar. Behav. Res. 1998, 33, 545–571. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.S.; Yajima, M.; Gelman, A.E.; Hill, J. Multiple imputation with diagnostics (mi) in R: Opening windows into the black box. J. Stat. Softw. 2011, 45, 1–31. [Google Scholar] [CrossRef]
- Analytics, Business Intelligence and Data Management. Available online: https://www.sas.com/en_us/home.html (accessed on 4 September 2017).
- Simren, M.; Agerforz, P.; Bjornsson, E.S.; Abrahamsson, H. Nutrient-dependent enhancement of rectal sensitivity in irritable bowel syndrome (IBS). Neurogastroenterol. Motil. 2007, 19, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Feinle-Bisset, C.; Azpiroz, F. Dietary lipids and functional gastrointestinal disorders. Am. J. Gastroenterol. 2013, 108, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Simrén, M.; Abrahamsson, H.; Björnsson, E.S. Lipid-Induced Colonic Hypersensitivity in the Irritable Bowel Syndrome: The Role of Bowel Habit, Sex, and Psychologic Factors. Clin. Gastroenterol. Hepatol. 2007, 5, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Ostgaard, H.; Hausken, T.; Gundersen, D.; El-Salhy, M. Diet and effects of diet management on quality of life and symptoms in patients with irritable bowel syndrome. Mol. Med. Rep. 2012, 5, 1382–1390. [Google Scholar] [CrossRef] [PubMed]
- Faresjo, A.; Johansson, S.; Faresjo, T.; Roos, S.; Hallert, C. Sex differences in dietary coping with gastrointestinal symptoms. Eur. J. Gastroenterol. Hepatol. 2010, 22, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Reding, K.W.; Cain, K.C.; Jarrett, M.E.; Eugenio, M.D.; Heitkemper, M.M. Relationship between patterns of alcohol consumption and gastrointestinal symptoms among patients with irritable bowel syndrome. Am. J. Gastroenterol. 2013, 108, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Hayes, P.; Corish, C.; O’Mahony, E.; Quigley, E.M.M. A dietary survey of patients with irritable bowel syndrome. J. Hum. Nutr. Diet. 2014, 27, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Holmes, B.A.; Habi-Rachedi, F.; Trotin, B.; Paineau, D.; Guyonnet, D.; Rondeau, P.; Flourié, B.; Whelan, K. Dietary patterns, digestive symptoms, and health-related quality of life in women reporting minor digestive symptoms. Nutrition 2017, 35, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Popkin, B.M.; Gordon-Larsen, P. The nutrition transition: Worldwide obesity dynamics and their determinants. Int. J. Obes. 2004, 28, S2–S9. [Google Scholar] [CrossRef] [PubMed]
- Stuckler, D.; McKee, M.; Ebrahim, S.; Basu, S. Manufacturing epidemics: The role of global producers in increased consumption of unhealthy commodities including processed foods, alcohol, and tobacco. PLoS Med. 2012, 9, e1001235. [Google Scholar] [CrossRef] [PubMed]
- Larochette, B.; Sanchez-Gonzalez, J. Cinquante ans de Consommation Alimentaire: Une Croissance Modérée, Mais de Profonds Changements; Report Number 1568; Coll; Insee Première: Insee, France, 2015. [Google Scholar]
- WHO/FAO. Diet, Nutrition and the Prevention of Chronic Diseases; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Shetty, P. Nutrition transition and its health outcomes. Indian J. Pediatr. 2013, 80, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A. La Transition Nutritionnelle. Available online: http://www.nutriaa.com/docs/proc_univ_2003_107.pdf (accessed on 12 July 2017).
- Combris, P. Que mange-t-on en France et dans les pays industrialisés? In A Table? Peut-On Encore Bien Manger? Editions de l’Aube: Paris, France, 2003; Volume 1, pp. 29–45. [Google Scholar]
- Moubarac, J.C.; Martins, A.P.B.; Claro, R.M.; Levy, R.B.; Cannon, G.; Monteiro, C.A. Consumption of ultra-processed foods and likely impact on human health. Evidence from Canada. Public Health Nutr. 2013, 16, 2240–2248. [Google Scholar] [CrossRef] [PubMed]
- Moayyedi, P.; Quigley, E.M.M.; Lacy, B.E.; Lembo, A.J.; Saito, Y.A.; Schiller, L.R.; Soffer, E.E.; Spiegel, B.M.R.; Ford, A.C. The effect of fiber supplementation on irritable bowel syndrome: A systematic review and meta-analysis. Am. J. Gastroenterol. 2014, 109, 1367–1374. [Google Scholar] [CrossRef] [PubMed]
- Shau, J.P.; Chen, P.H.; Chan, C.F.; Hsu, Y.C.; Wu, T.C.; James, F.E.; Pan, W.H. Fast foods—Are they a risk factor for functional gastrointestinal disorders? Asia Pac. J. Clin. Nutr. 2016, 25, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Heitkemper, M.M. Gender differences in irritable bowel syndrome. Gastroenterology 2002, 123, 1686–1701. [Google Scholar] [CrossRef] [PubMed]
- Heitkemper, M.; Jarrett, M. Irritable bowel syndrome: Does gender matter? J. Psychosom. Res. 2008, 64, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Harris, L.A.; Umar, S.B.; Baffy, N.; Heitkemper, M.M. Irritable bowel syndrome and female patients. Gastroenterol. Clin. N. Am. 2016, 45, 179–204. [Google Scholar] [CrossRef] [PubMed]
- Houghton, L.A.; Heitkemper, M.; Crowell, M.D.; Emmanuel, A.; Halpert, A.; McRoberts, J.A.; Toner, B. Age, gender, and women’s health and the patient. Gastroenterology 2016, 150, 1332–1343e4. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.S.; Irving, P.M.; Shepherd, S.J.; Muir, J.G.; Gibson, P.R. Comparison of the prevalence of fructose and lactose malabsorption across chronic intestinal disorders. Aliment. Pharmacol. Ther. 2009, 30, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.S.; Gearry, R.B.; Muir, J.G.; Irving, P.M.; Rose, R.; Rosella, O.; Haines, M.L.; Shepherd, S.J.; Gibson, P.R. Dietary poorly absorbed, short-chain carbohydrates increase delivery of water and fermentable substrates to the proximal colon. Aliment. Pharmacol. Ther. 2010. [Google Scholar] [CrossRef] [PubMed]
- Muir, J.G.; Shepherd, S.J.; Rosella, O.; Rose, R.; Barrett, J.S.; Gibson, P.R. Fructan and free fructose content of common Australian vegetables and fruit. J. Agric. Food Chem. 2007, 55, 6619–6627. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.S.; Gibson, P.R. Fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs) and nonallergic food intolerance: FODMAPs or food chemicals? Ther. Adv. Gastroenterol. 2012, 5, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Tseng, M.; DeVellis, R.F. Fundamental dietary patterns and their correlates among US whites. J. Am. Diet. Assoc. 2001, 101, 929. [Google Scholar] [CrossRef]
- Hu, F.B.; Rimm, E.B.; Stampfer, M.J.; Ascherio, A.; Spiegelman, D.; Willett, W.C. Prospective study of major dietary patterns and risk of coronary heart disease in men. Am. J. Clin. Nutr. 2000, 72, 912–921. [Google Scholar] [PubMed]
- Fransen, H.P.; Beulens, J.W.J.; May, A.M.; Struijk, E.A.; Boer, J.M.A.; de Wit, G.A.; Onland-Moret, N.C.; van der Schouw, Y.T.; Bueno-de-Mesquita, H.B.; Hoekstra, J.; et al. Dietary patterns in relation to quality-adjusted life years in the EPIC-NL cohort. Prev. Med. 2015, 77, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Guinot, C.; Latreille, J.; Malvy, D.; Preziosi, P.; Galan, P.; Hercberg, S.; Tenenhaus, M. Use of multiple correspondence analysis and cluster analysis to study dietary behaviour: Food consumption questionnaire in the SU.VI.MAX. cohort. Eur. J. Epidemiol. 2001, 17, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Bertrais, S.; Péneau, S.; Estaquio, C.; Dauchet, L.; Vergnaud, A.C.; Czernichow, S.; Galan, P.; Hercberg, S.; Bellisle, F. Dietary patterns and their sociodemographic and behavioural correlates in French middle-aged adults from the SU.VI.MAX cohort. Eur. J. Clin. Nutr. 2008, 63, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Andreeva, V.A.; Jeandel, C.; Ferry, M.; Hercberg, S.; Galan, P. A healthy dietary pattern at midlife is associated with subsequent cognitive performance. J. Nutr. 2012, 142, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Julia, C.; Meunier, N.; Touvier, M.; Ahluwalia, N.; Sapin, V.; Papet, I.; Cano, N.; Hercberg, S.; Galan, P.; Kesse-Guyot, E. Dietary patterns and risk of elevated C-reactive protein concentrations 12 years later. Br. J. Nutr. 2013, 110, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Bedard, A.; Garcia-Aymerich, J.; Sanchez, M.; Le Moual, N.; Clavel-Chapelon, F.; Boutron-Ruault, M.C.; Maccario, J.; Varraso, R. Confirmatory factor analysis compared with principal component analysis to derive dietary patterns: A longitudinal study in adult women. J. Nutr. 2015, 145, 1559–1568. [Google Scholar] [CrossRef] [PubMed]
| Characteristics of Participants | Non Cases Participants | Participants with IBS | p Value * | ||
|---|---|---|---|---|---|
| n = 41,927 (94.5%) | n = 2423 (5.5%) | ||||
| n | % | n | % | ||
| Gender | |||||
| Men | 9183 | 21.9 | 460 | 19.0 | <0.001 |
| Women | 32,744 | 78.1 | 1963 | 81.0 | |
| Age (mean ± SD) | 49.4 | ±14.3 | 56.0 | ±12.0 | <0.0001 |
| Educational level | |||||
| No diploma or primary school | 1218 | 2.91 | 86 | 3.5 | <0.01 |
| Secondary | 13,776 | 32.86 | 863 | 35.6 | |
| High education level | 26,933 | 64.24 | 1474 | 60.8 | |
| BMI | |||||
| BMI < 25 | 29,022 | 70.04 | 1639 | 67.8 | 0.07 |
| BMI 25–30 | 8949 | 21.60 | 556 | 23.0 | |
| BMI ≥ 30 | 3465 | 8.36 | 221 | 9.1 | |
| Marital status | |||||
| Single | 11,146 | 26.58 | 648 | 26.7 | 0.86 |
| Cohabiting | 30,781 | 73.42 | 1775 | 73.3 | |
| Smoking status | |||||
| Non smoker | 21,743 | 51.86 | 1163 | 48.0 | <0.0001 |
| Former smoker | 14,657 | 34.96 | 1000 | 41.3 | |
| Current smoker | 5527 | 13.18 | 260 | 10.7 | |
| Monthly income level | |||||
| Less than 1200 euros per c.u. | 5804 | 15.50 | 276 | 12.7 | <0.0001 |
| From 1200 to 2300 euros per c.u. | 16,371 | 43.71 | 905 | 42.7 | |
| More than 2300 euros per c.u. | 15,277 | 40.79 | 992 | 45.7 | |
| Physical activity level | |||||
| High | 12,714 | 34.78 | 768 | 35.7 | 0.58 |
| Moderate | 15,699 | 42.94 | 922 | 42.8 | |
| Low | 8146 | 22.28 | 462 | 21.5 | |
| Time between inclusion and IBS questionnaire (years) | 3.2 | ±0.99 | 3.2 | ±0.97 | 0.12 |
| Time between dietary records and IBS questionnaire (years) | 1.0 | ±1.1 | 0.88 | ±1.02 | <0.0001 |
| DP “Healthy” | DP “Western” | DP “Traditional” | |
|---|---|---|---|
| Meat, ham | −0.31 | 0.24 | |
| Processed meat | 0.31 | 0.28 | |
| Fish and seafood | 0.22 | −0.13 | |
| Vegetable fat | 0.24 | −0.15 | 0.27 |
| Animal fat | 0.37 | ||
| Dairy products | −0.56 | ||
| Dried fruits | 0.49 | ||
| Potatoes and tubers | 0.41 | ||
| Fruits and vegetable juices | 0.25 | ||
| Starches | −0.16 | 0.18 | |
| Whole grains | 0.60 | −0.12 | |
| Breakfast cereals | 0.34 | −0.14 | |
| Cakes, cookies, pastries and desserts | 0.53 | ||
| Salty snacks | 0.46 | ||
| Organ meat | 0.16 | ||
| Poultry | |||
| Milk | −0.27 | ||
| Fruits | 0.42 | −0.37 | 0.17 |
| Vegetables | 0.46 | −0.39 | 0.25 |
| Sauces | 0.26 | 0.21 | |
| Cheese | 0.26 | 0.45 | |
| Bread | −0.34 | 0.62 | |
| Sugared cereals | −0.24 | ||
| Confectionery | 0.12 | 0.40 | |
| Soft non sugared beverages | 0.41 | 0.14 | |
| Sweetened beverages and sodas | −0.11 | 0.35 | −0.11 |
| Alcoholic beverages | 0.30 | 0.36 | |
| Legumes | 0.34 | ||
| Eggs | −0.14 |
| Characteristics of Participants | Healthy | p Value * | Western | p Value * | Traditional | p Value * | |||
|---|---|---|---|---|---|---|---|---|---|
| Q1 (lowest) | Q5 (highest) | Q1 (lowest) | Q5 (highest) | Q1 (lowest) | Q5 (highest) | ||||
| Gender | |||||||||
| Men | 21.7 | 21.7 | 21.7 | 21.7 | 21.7 | 21.7 | |||
| Women | 78.3 | 78.3 | 78.3 | 78.3 | 78.3 | 78.3 | |||
| Age (SD) | 45.9 (14.7) | 52.8 (13.1) | <0.0001 | 56.2 (12.4) | 41.5 (13.4) | <0.0001 | 45.4 (14.8) | 53.1 (13.1) | <0.0001 |
| Educational level | |||||||||
| No diploma or primary school | 3.7 | 2.5 | <0.0001 | 4.5 | 1.9 | <0.0001 | 2.6 | 3.2 | 0.02 |
| Secondary | 39.6 | 28.9 | 38.6 | 27.1 | 32.6 | 33.8 | |||
| High education level | 57.7 | 68.6 | 57.0 | 71.0 | 64.7 | 63.0 | |||
| BMI | |||||||||
| BMI < 25 | 65.7 | 76.2 | <0.0001 | 63.0 | 74.0 | <0.0001 | 74.8 | 66.1 | <0.0001 |
| BMI 25–30 | 22.8 | 17.7 | 25.3 | 17.9 | 18.1 | 23.6 | |||
| BMI ≥ 30 | 11.5 | 6.1 | 11.1 | 8.1 | 7.1 | 10.3 | |||
| Marital status | |||||||||
| Single | 26.8 | 29.7 | <0.0001 | 27.3 | 30.0 | <0.0001 | 34.3 | 22.2 | <0.0001 |
| Cohabiting | 73.2 | 70.3 | 72.7 | 70.0 | 65.6 | 77.8 | |||
| Smoking status | |||||||||
| Non smoker | 54.8 | 50.1 | <0.0001 | 50.8 | 52.3 | <0.0001 | 55.9 | 47.9 | <0.0001 |
| Former smoker | 29.6 | 39.9 | 40.7 | 29.3 | 31.4 | 38.6 | |||
| Current smoker | 15.6 | 10.0 | 8.5 | 18.4 | 12.7 | 13.5 | |||
| Monthly income level | |||||||||
| Less than 1200 euros per c.u. | 21.3 | 12.1 | <0.0001 | 13.1 | 18.9 | <0.0001 | 19.3 | 13.2 | <0.0001 |
| From 1200 to 2300 euros per c.u. | 46.6 | 41.4 | 43.7 | 44.4 | 43.4 | 43.2 | |||
| More than 2300 euros per c.u. | 32.1 | 46.4 | 43.2 | 36.7 | 37.3 | 43.3 | |||
| Physical activity | |||||||||
| High | 31.8 | 40.1 | <0.0001 | 41.9 | 30.1 | <0.0001 | 33.7 | 37.1 | <0.0001 |
| Moderate | 41.3 | 43.3 | 39.3 | 43.5 | 43.8 | 41.3 | |||
| Low | 26.9 | 16.6 | 18.8 | 26.4 | 22.5 | 21.6 | |||
| Dietary Profile | Model | Number of Cases | Q1 | Q2 | Q3 | Q4 | Q5 | p for Trend | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | OR | 95% CI | OR | 95% CI | OR | 95% CI | |||||
| Healthy | Men | n * | 76 | 89 | 97 | 108 | 90 | |||||
| Model 1 | Ref. | 1.18 | 0.86–1.62 | 1.29 | 0.95–1.76 | 1.44 | 1.07–1.95 | 1.17 | 0.86–1.61 | 0.14 | ||
| Model 2 | Ref. | 1.13 | 0.82–1.55 | 1.19 | 0.87–1.63 | 1.34 | 0.98–1.83 | 1.13 | 0.81–1.57 | 0.24 | ||
| Women | n * | 292 | 385 | 394 | 416 | 476 | ||||||
| Model 1 | Ref. | 1.17 | 1.00–1.37 | 1.10 | 0.94–1.29 | 1.10 | 0.94–1.29 | 1.19 | 1.02–1.39 | 0.12 | ||
| Model 2 | Ref. | 1.13 | 0.97–1.33 | 1.08 | 1.92–1.26 | 1.09 | 0.93–1.28 | 1.19 | 1.02–1.40 | 0.10 | ||
| Western | Men | n * | 67 | 89 | 88 | 107 | 109 | |||||
| Model 1 | Ref. | 1.33 | 0.96–1.84 | 1.30 | 0.94–1.81 | 1.58 | 1.14–2.17 | 1.56 | 1.14–2.17 | <0.01 | ||
| Model 2 | Ref. | 1.31 | 0.95–1.81 | 1.27 | 0.92–1.77 | 1.52 | 1.10–2.11 | 1.52 | 1.08–2.16 | 0.01 | ||
| Women | n * | 442 | 469 | 400 | 359 | 293 | ||||||
| Model 1 | Ref. | 1.2 | 1.04–1.37 | 1.16 | 1.00–1.33 | 1.22 | 1.05–1.42 | 1.28 | 1.08–1.52 | <0.01 | ||
| Model 2 | Ref. | 1.21 | 1.05–1.39 | 1.18 | 1.02–1.36 | 1.26 | 1.08–1.46 | 1.36 | 1.14–1.62 | <0.001 | ||
| Traditional | Men | n * | 85 | 99 | 83 | 93 | 100 | |||||
| Model 1 | Ref. | 1.18 | 0.87–1.59 | 0.98 | 0.71–1.34 | 1.08 | 0.79–1.61 | 1.13 | 0.79–1.61 | 0.72 | ||
| Model 2 | Ref. | 1.19 | 0.88–1.61 | 1.01 | 0.73–1.40 | 1.14 | 0.82–1.59 | 1.25 | 0.86–1.82 | 0.37 | ||
| Women | n * | 309 | 347 | 391 | 431 | 485 | ||||||
| Model 1 | Ref. | 0.99 | 0.85–1.17 | 1.06 | 0.91–1.24 | 1.12 | 0.95–1.31 | 1.19 | 1.00–1.41 | 0.02 | ||
| Model 2 | Ref. | 1.01 | 0.86–1.19 | 1.10 | 0.93–1.29 | 1.18 | 1.00–1.39 | 1.29 | 1.08–1.54 | <0.01 | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buscail, C.; Sabate, J.-M.; Bouchoucha, M.; Kesse-Guyot, E.; Hercberg, S.; Benamouzig, R.; Julia, C. Western Dietary Pattern Is Associated with Irritable Bowel Syndrome in the French NutriNet Cohort. Nutrients 2017, 9, 986. https://doi.org/10.3390/nu9090986
Buscail C, Sabate J-M, Bouchoucha M, Kesse-Guyot E, Hercberg S, Benamouzig R, Julia C. Western Dietary Pattern Is Associated with Irritable Bowel Syndrome in the French NutriNet Cohort. Nutrients. 2017; 9(9):986. https://doi.org/10.3390/nu9090986
Chicago/Turabian StyleBuscail, Camille, Jean-Marc Sabate, Michel Bouchoucha, Emmanuelle Kesse-Guyot, Serge Hercberg, Robert Benamouzig, and Chantal Julia. 2017. "Western Dietary Pattern Is Associated with Irritable Bowel Syndrome in the French NutriNet Cohort" Nutrients 9, no. 9: 986. https://doi.org/10.3390/nu9090986
APA StyleBuscail, C., Sabate, J.-M., Bouchoucha, M., Kesse-Guyot, E., Hercberg, S., Benamouzig, R., & Julia, C. (2017). Western Dietary Pattern Is Associated with Irritable Bowel Syndrome in the French NutriNet Cohort. Nutrients, 9(9), 986. https://doi.org/10.3390/nu9090986






