Probiotics Prevent Late-Onset Sepsis in Human Milk-Fed, Very Low Birth Weight Preterm Infants: Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Data Extraction and Meta-Analysis
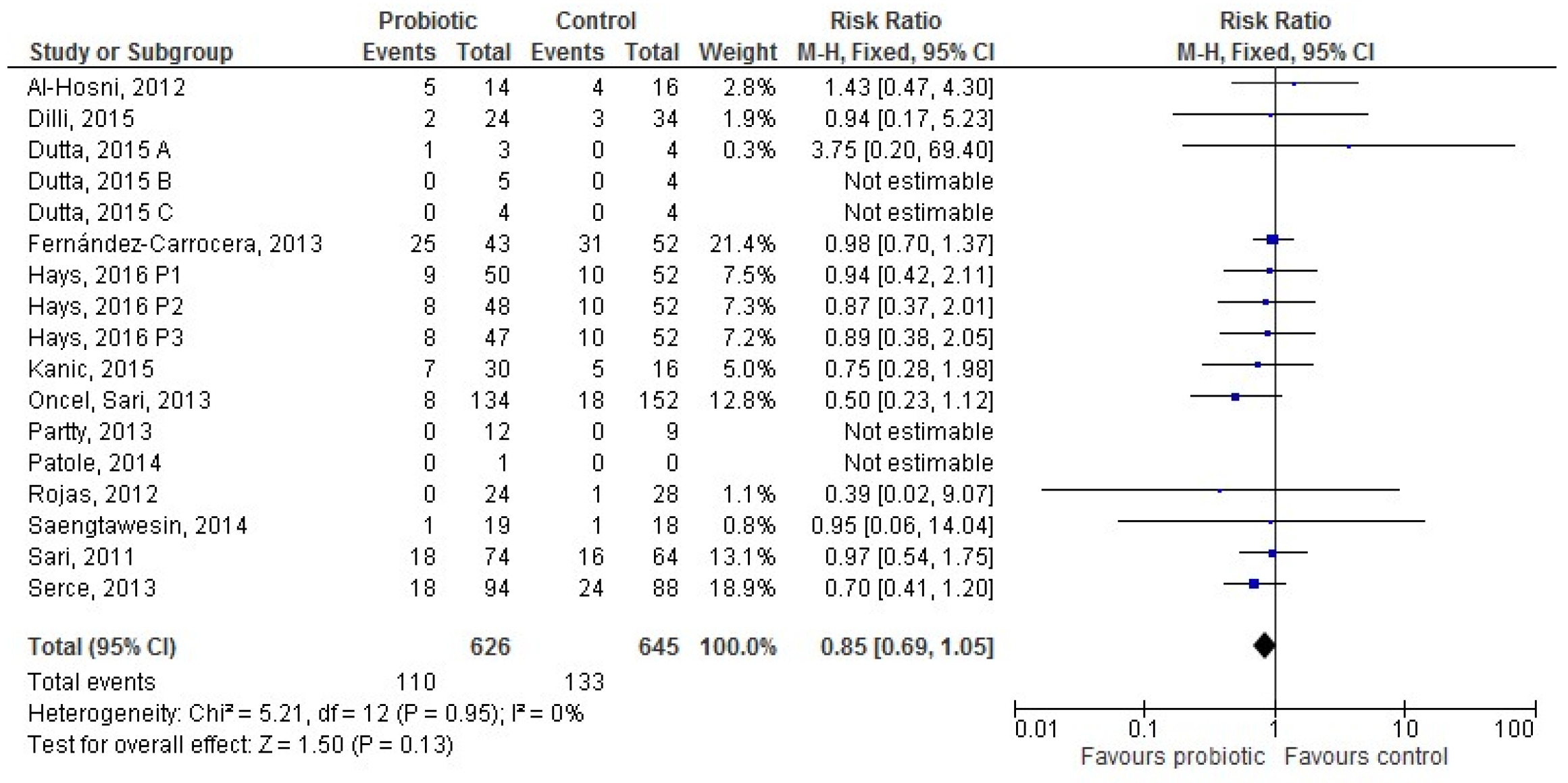
3. Results
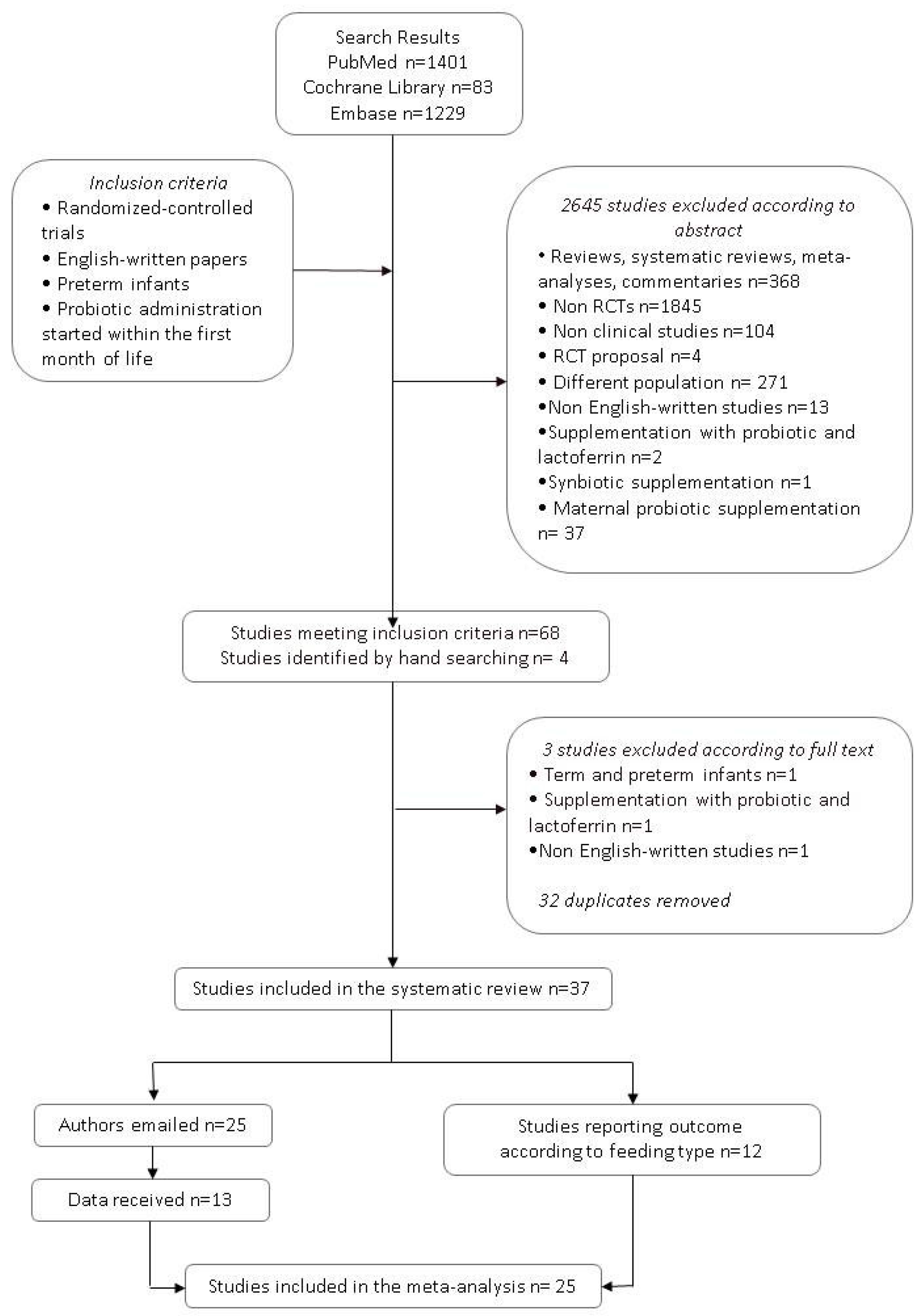
3.1. Literature Search
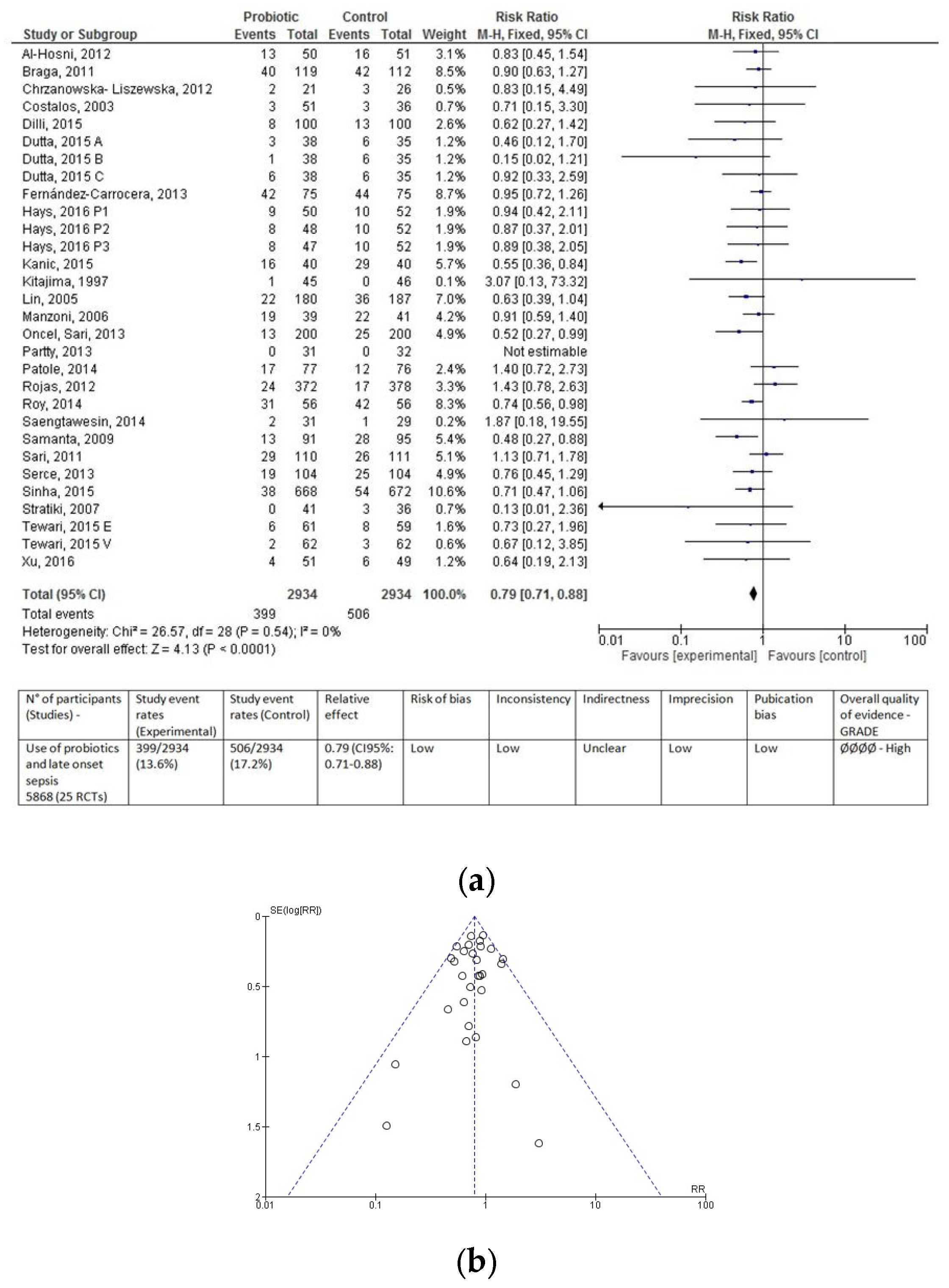
3.2. Probiotic and LOS: Overall Population
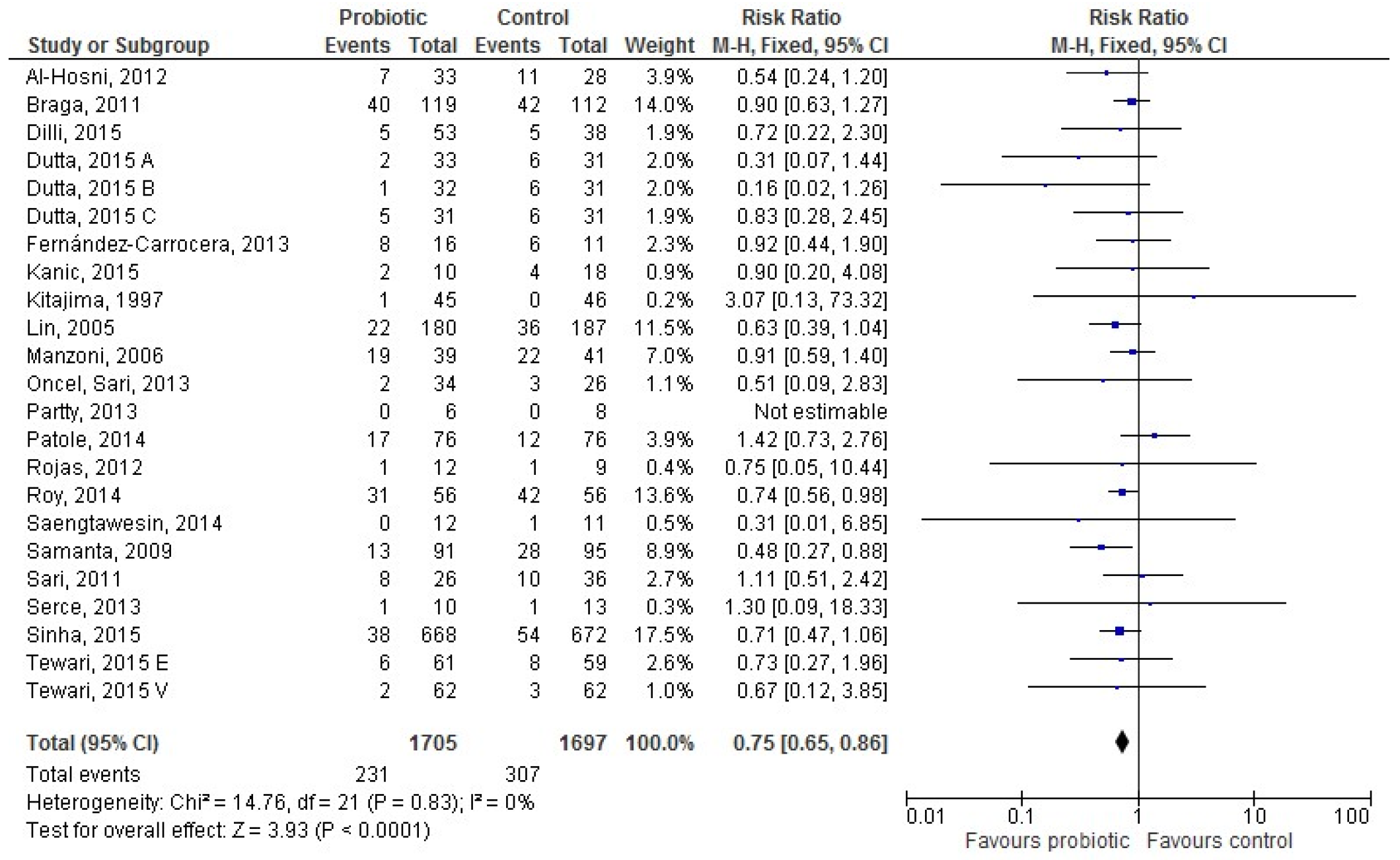
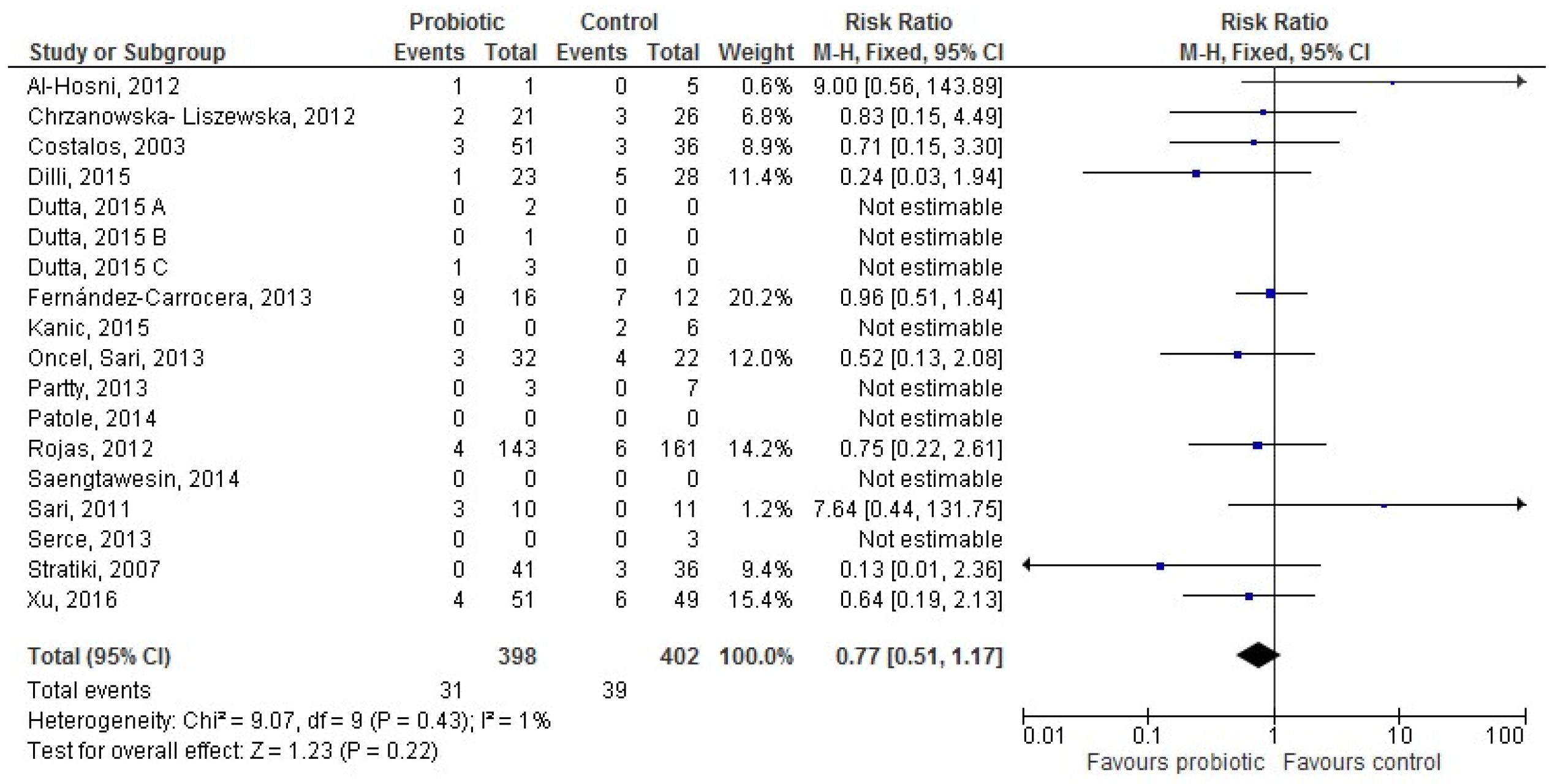
3.3. Probiotic and LOS According to Type of Feeding
3.3.1. Population Characteristics: VLBW and Extremely Low Birth Weight (ELBW) Infants
3.3.2. Probiotic Characteristics
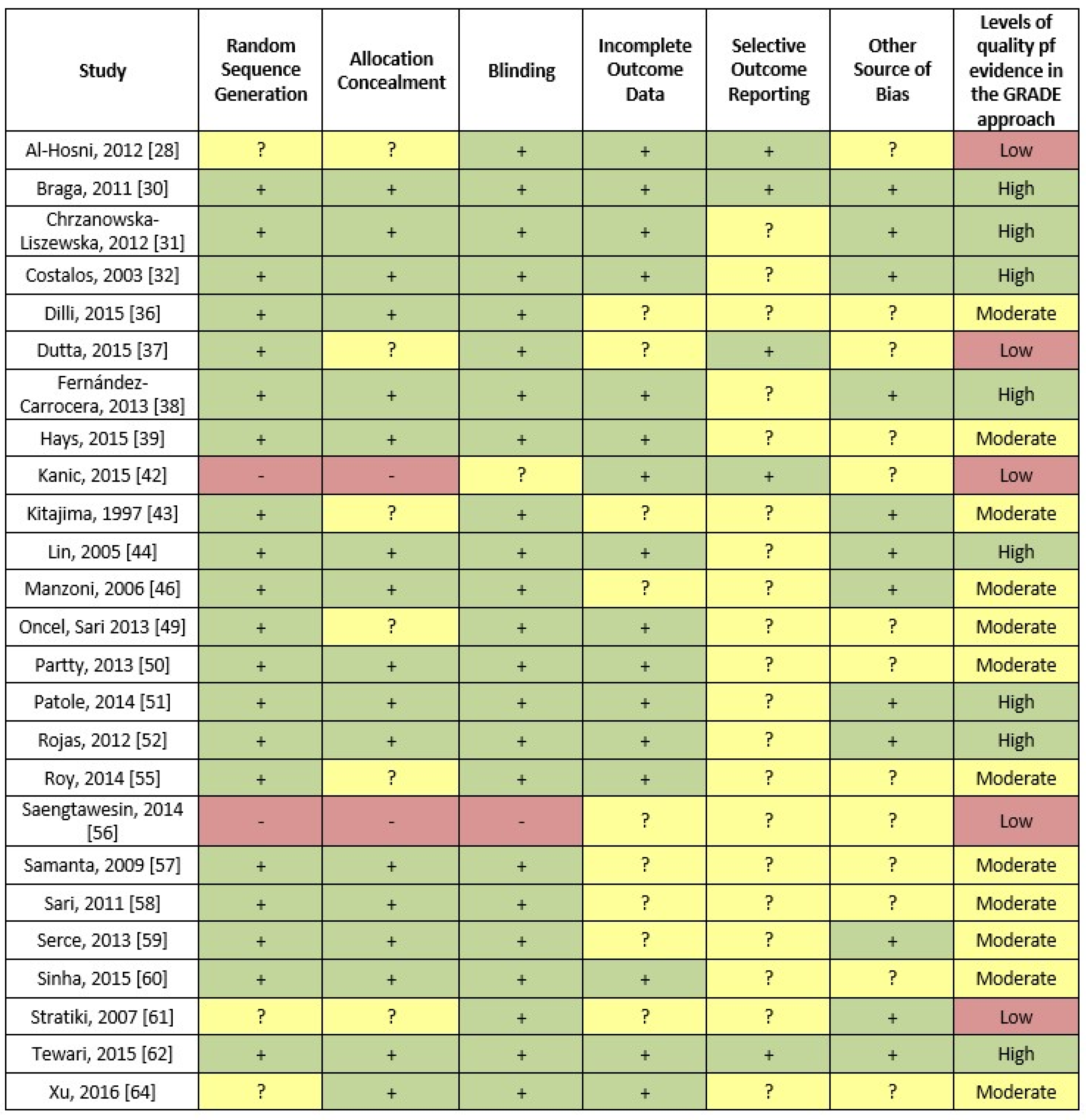
3.4. Methodological Study Quality
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Liu, L.; Johnson, H.L.; Cousens, S.; Perin, J.; Scott, S.; Lawn, J.E.; Rudan, I.; Campbell, H.; Cibulskis, R.; Li, M.; et al. Global, regional, and national causes of child mortality: An updated systematic analysis for 2010 with time trends since 2000. Lancet 2012, 379, 2151–2161. [Google Scholar] [CrossRef]
- Dong, Y.; Speer, C.P. Late-onset neonatal sepsis: Recent developments. Arch. Dis. Child. Fetal Neonatal Ed. 2015, 100, F257–F263. [Google Scholar] [CrossRef] [PubMed]
- Stoll, B.J.; Hansen, N.; Fanaroff, A.A.; Wright, L.L.; Carlo, W.A.; Ehrenkranz, R.A.; Lemons, J.A.; Donovan, E.F.; Stark, A.R.; Tyson, J.E.; et al. Late-onset sepsis in very low birth weight neonates: The experience of the NICHD Neonatal Research Network. Pediatrics 2002, 110, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Stoll, B.J.; Hansen, N.I.; Adams-Chapman, I.; Fanaroff, A.A.; Hintz, S.R.; Vohr, B.R.; Higgins, R.D.; National Institute of Child Health and Human Development Neonatal Research Network. Neurodevelopmental and growth impairment among extremely low-birth-weight infants with neonatal infection. JAMA 2004, 292, 2357–2365. [Google Scholar] [CrossRef] [PubMed]
- Collado, M.C.; Cernada, M.; Neu, J.; Pérez-Martínez, G.; Gormaz, M.; Vento, M. Factors influencing gastrointestinal tract and microbiota immune interaction in preterm infants. Pediatr. Res. 2015, 77, 726–731. [Google Scholar] [CrossRef] [PubMed]
- Berrington, J.E.; Stewart, C.J.; Embleton, N.D.; Cummings, S.P. Gut microbiota in preterm infants: Assessment and relevance to health and disease. Arch. Dis. Child. Fetal Neonatal Ed. 2013, 98, F286–F290. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.J.; Marrs, E.C.L.; Magorrian, S.; Nelson, A.; Lanyon, C.; Perry, J.D.; Embleton, N.D.; Cummings, S.P.; Berrington, J.E. The preterm gut microbiota: Changes associated with necrotizing enterocolitis and infection. Acta Paediatr. 2012, 101, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Mai, V.; Torrazza, R.M.; Ukhanova, M.; Wang, X.; Sun, Y.; Li, N.; Shuster, J.; Sharma, R.; Hudak, M.L.; Neu, J. Distortions in development of intestinal microbiota associated with late onset sepsis in preterm infants. PLoS ONE 2013, 8, e52876. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E.; Guarner, F.; Guerrant, R.; Holt, P.R.; Quigley, E.M.; Sartor, R.B.; Sherman, P.M.; Mayer, E.A. An update on the use and investigation of probiotics in health and disease. Gut 2013, 62, 787–796. [Google Scholar] [CrossRef] [PubMed]
- Slattery, J.; Macfabe, D.F.; Frye, R.E. The significance of the enteric microbiome on the development of childhood disease: A review of prebiotic and probiotic therapies in disorders of childhood. Clin. Med. Insights Pediatr. 2016, 91–107. [Google Scholar] [CrossRef]
- AlFaleh, K.; Anabrees, J. Probiotics for prevention of necrotizing enterocolitis in preterm infants. Cochrane Database Syst. Rev. 2014, 9, 584–671. [Google Scholar] [CrossRef]
- Aceti, A.; Gori, D.; Barone, G.; Callegari, M.L.; Di Mauro, A.; Fantini, M.P.; Indrio, F.; Maggio, L.; Meneghin, F.; Morelli, L.; et al. Probiotics for prevention of necrotizing enterocolitis in preterm infants: Systematic review and meta-analysis. Ital. J. Pediatr. 2015, 41, 89. [Google Scholar] [CrossRef] [PubMed]
- Aceti, A.; Gori, D.; Barone, G.; Callegari, M.L.; Fantini, M.P.; Indrio, F.; Maggio, L.; Meneghin, F.; Morelli, L.; Zuccotti, G.; et al. Probiotics and time to achieve full enteral feeding in human milk-fed and formula-fed preterm infants: Systematic review and meta-analysis. Nutrients 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Athalie-Jape, G.; Deshpande, G.; Rao, S.; Patole, S. Benefits of probiotics on enteral nutrition in preterm neonates-a systematic review. J. Paediatr. Child Health 2014, 50, 15. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.C.; Athalye-Jape, G.K.; Deshpande, G.C.; Simmer, K.N.; Patole, S.K. Probiotic Supplementation and Late-Onset Sepsis in Preterm Infants: A Meta-analysis. Pediatrics 2016, 137, e20153684. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.-Q.; Hu, H.-J.; Liu, C.-Y.; Shakya, S.; Li, Z.-Y. Probiotics for Preventing Late-Onset Sepsis in Preterm Neonates: A PRISMA-Compliant Systematic Review and Meta-Analysis of Randomized Controlled Trials. Medicine 2016, 95, e2581. [Google Scholar] [CrossRef] [PubMed]
- Carlisle, E.M.; Morowitz, M.J. The intestinal microbiome and necrotizing enterocolitis. Curr. Opin. Pediatr. 2013, 25, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, A.R.; Barile, D.; Underwood, M.A.; Mills, D.A. The impact of the milk glycobiome on the neonate gut microbiota. Annu. Rev. Anim. Biosci. 2015, 3, 419–445. [Google Scholar] [CrossRef] [PubMed]
- Repa, A.; Thanhaeuser, M.; Endress, D.; Weber, M.; Kreissl, A.; Binder, C.; Berger, A.; Haiden, N. Probiotics (Lactobacillus acidophilus and Bifidobacterium bifidum) prevent NEC in VLBW infants fed breast milk but not formula. Pediatr. Res. 2015, 77, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Ward, R.E.; Ninonuevo, M.; Mills, D.A.; Lebrilla, C.B.; German, J.B. In vitro fermentation of breast milk oligosaccharides by bifidobacterium infantis and Lactobacillus gasseri. Appl. Environ. Microbiol. 2006, 72, 4497–4499. [Google Scholar] [CrossRef] [PubMed]
- Underwood, M.A.; German, J.B.; Lebrilla, C.B.; Mills, D.A. Bifidobacterium longum subspecies infantis: Champion colonizer of the infant gut. Pediatr. Res. 2014, 77, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.E.; Kunz, R.; Falck-Ytter, Y.; Alonso-Coello, P.; Schünemann, H.J. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008, 336, 924–926. [Google Scholar] [CrossRef] [PubMed]
- Awad, H.; Mokhtar, G.; Imam, S.S.; Gad, G.I.; Hafez, H.; Aboushady, N. Comparison between killed and living probiotic usage versus placebo for the prevention of necrotizing enterocolitis and sepsis in neonates. Pakistan J. Biol. Sci. 2010, 13, 253–262. [Google Scholar] [CrossRef]
- Manzoni, P.; Rinaldi, M.; Cattani, S.; Pugni, L.; Romeo, M.G.; Messner, H. Bovine Lactoferin Supplementation for Prevention of Late-Onset Sepsis in Very Low-Birth-Weight Neonates. J. Am. Med. Assoc. 2009, 302, 1421–1428. [Google Scholar] [CrossRef] [PubMed]
- Ren, B. Preventive effect of Bifidobacterium tetravaccine tablets in premature infants with necrotizing enterocolitis. J. Pediatr. Pharm. 2010, 16, 24–25. [Google Scholar]
- Al-Hosni, M.; Duenas, M.; Hawk, M.; Stewart, L.A.; Borghese, R.A.; Cahoon, M.; Atwood, L.; Howard, D.; Ferrelli, K.; Soll, R. Probiotics-supplemented feeding in extremely low-birth-weight infants. J. Perinatol. 2012, 32, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Bin-Nun, A.; Bromiker, R.; Wilschanski, M.; Kaplan, M.; Rudensky, B.; Caplan, M.; Hammerman, C. Oral probiotics prevent necrotizing enterocolitis in very low birth weight neonates. J. Pediatr. 2005, 147, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Braga, T.D.; da Silva, G.A.; de Lira, P.I.; de Carvalho Lima, M. Efficacy of bifidobacterium breve and Lactobacillus casei oral supplementation on necrotizing enterocolitis in very-low-birth-weight preterm infants: A double-blind, randomized, controlled trial. Am. J. Clin. Nutr. 2011, 93, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Chrzanowska-Liszewska, D.; Seliga-Siwecka, J.; Kornacka, M.K. The effect of Lactobacillus rhamnosus GG supplemented enteral feeding on the microbiotic flora of preterm infants-double blinded randomized control trial. Early Hum. Dev. 2012, 88, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Costalos, C.; Skouteri, V.; Gounaris, A.; Sevastiadou, S.; Triandafilidou, A.; Ekonomidou, C.; Kontaxaki, F.; Petrochilou, V. Enteral feeding of premature infants with Saccharomyces boulardii. Early Hum. Dev. 2003, 74, 89–96. [Google Scholar] [CrossRef]
- Costeloe, K.; Hardy, P.; Juszczak, E.; Wilks, M.; Millar, M.R. Bifidobacterium breve BBG-001 in very preterm infants: A randomised controlled phase 3 trial. Lancet 2016, 387, 649–660. [Google Scholar] [CrossRef]
- Dani, C.; Biadaioli, R.; Bertini, G.; Martelli, E.; Rubaltelli, F.F. Probiotics feeding in prevention of urinary tract and necrotizing enterocolitis in preterm infants. Biol. Neonate 2002, 82, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Demirel, G.; Erdeve, O.; Celik, I.H.; Dilmen, U. Saccharomyces boulardii for prevention of necrotizing enterocolitis in preterm infants: A randomized, controlled study. Acta Paediatr. 2013, 102, 560–565. [Google Scholar] [CrossRef] [PubMed]
- Dilli, D.; Aydin, B.; Fettah, N.D.; Özyazıcı, E.; Beken, S.; Zenciroğlu, A.; Okumuş, N.; Özyurt, B.M.; İpek, M.Ş.; Akdağ, A.; et al. The propre-save study: Effects of probiotics and prebiotics alone or combined on necrotizing enterocolitis in very low birth weight infants. J. Pediatr. 2015, 166, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; Ray, P.; Narang, A. Comparison of stool colonization in premature infants by three dose regimes of a probiotic combination: A randomized controlled trial. Am. J. Perinatol. 2015, 32, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Carrocera, L.A.; Solis-Herrera, A.; Cabanillas-Ayón, M.; Gallardo-Sarmiento, R.B.; García-Pérez, C.S.; Montaño-Rodríguez, R.; Echániz-Aviles, M.O.L.; Fernandez-Carrocera, L.A.; Cabanillas-Ayon, M.; Gallardo-Sarmiento, R.B.; et al. double-blind, randomised clinical assay to evaluate the efficacy of probiotics in preterm newborns weighing less than 1500 g in the prevention of necrotising enterocolitis. Arch. Dis. Child. Fetal Neonatal Ed. 2013, 98, F5. [Google Scholar] [CrossRef] [PubMed]
- Hays, S.; Jacquot, A.; Gauthier, H.; Kempf, C.; Beissel, A.; Pidoux, O.; Jumas-Bilak, E.; Decullier, E.; Lachambre, E.; Beck, L.; et al. Probiotics and growth in preterm infants: A randomized controlled trial, PREMAPRO study. Clin. Nutr. 2016, 35, 802–811. [Google Scholar] [CrossRef] [PubMed]
- Hikaru, U.; Koichi, S.; Yayoi, S.; Hiromichi, S.; Hiroaki, S.; Yoshikazu, O.; Seigo, S.; Nagata, S.; Toshiaki, S.; Yamashiro, Y. Bifidobacteria prevents preterm infants from developing infection and sepsis. Int. J. Probiotics Prebiotics 2010, 5, 33–36. [Google Scholar]
- Jacobs, A.S.E.; Tobin, J.M. Probiotic effects on late-onset sepsis in very preterm infants: A randomized controlled trial. Pediatrics 2013, 132, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Kanic, Z.; Micetic Turk, D.; Burja, S.; Kanic, V.; Dinevski, D. Influence of a combination of probiotics on bacterial infections in very low birthweight newborns. Wien. Klin. Wochenschr. 2015, 127, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, H.; Sumida, Y.; Tanaka, R.; Yuki, N.; Takayama, H.; Fujimura, M. Early administration of Bifidobacterium breve to preterm infants: Randomised controlled trial. Arch. Dis. Child. Fetal Neonatal Ed. 1997, 76, F101–F107. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-C.; Su, B.-H.; Chen, A.-C.; Lin, T.-W.; Tsai, C.-H.; Yeh, T.-F.; Oh, W. Oral probiotics reduce the incidence and severity of necrotizing enterocolitis in very low birth weight infants. Pediatrics 2005, 115, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-C.; Hsu, C.-H.; Chen, H.-L.; Chung, M.-Y.; Hsu, J.-F.; Lien, R.-I.; Tsao, L.-Y.; Chen, C.-H.; Su, B.-H. Oral probiotics prevent necrotizing enterocolitis in very low birth weight preterm infants: A multicenter, randomized, controlled trial. Pediatrics 2008, 122, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Manzoni, P.; Mostert, M.; Leonessa, M.L.; Priolo, C.; Farina, D.; Monetti, C.; Latino, M.A.; Gomirato, G. Oral supplementation with Lactobacillus casei subspecies rhamnosus prevents enteric colonization by Candida species in preterm neonates: A randomized study. Clin. Infect. Dis. 2006, 42, 1735–1742. [Google Scholar] [CrossRef] [PubMed]
- Mihatsch, W.A.; Vossbeck, S.; Eikmanns, B.; Hoegel, J.; Pohlandt, F. Effect of bifidobacterium lactis on the incidence of nosocomial infections in very-low-birth-weight infants: A randomized controlled trial. Neonatology 2010, 98, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Millar, M.R.R.; Bacon, C.; Smith, S.L.L.; Walker, V.; Hall, M.A.A. Enteral feeding of premature infants with Lactobacillus GG. Arch. Dis. Child. 1993, 69, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Oncel, M.Y.; Sari, F.N.; Arayici, S.; Guzoglu, N.; Erdeve, O.; Uras, N.; Oguz, S.S.; Dilmen, U. Lactobacillus Reuteri for the prevention of necrotising enterocolitis in very low birthweight infants: A randomised controlled trial. Arch. Dis. Child. Fetal Neonatal Ed. 2014, 99, F110–F115. [Google Scholar] [CrossRef] [PubMed]
- Pärtty, A.; Luoto, R.; Kalliomäki, M.; Salminen, S.; Isolauri, E. Effects of early prebiotic and probiotic supplementation on development of gut microbiota and fussing and crying in preterm infants: A randomized, double-blind, placebo-controlled trial. J. Pediatr. 2013, 163. [Google Scholar] [CrossRef] [PubMed]
- Patole, S.; Keil, A.D.; Chang, A.; Nathan, E.; Doherty, D.; Simmer, K.; Esvaran, M.; Conway, P. Effect of Bifidobacterium breve M-16V supplementation on fecal bifidobacteria in preterm neonates-a randomised double blind placebo controlled trial. PLoS ONE 2014, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Rojas, M.A.; Lozano, J.M.; Rojas, M.X.; Rodriguez, V.A.; Rondon, M.A.; Bastidas, J.A.; Perez, L.A.; Rojas, C.; Ovalle, O.; Garcia-Harker, J.E.; et al. Prophylactic probiotics to prevent death and nosocomial infection in preterm infants. Pediatrics 2012, 130, e1113–e1120. [Google Scholar] [CrossRef] [PubMed]
- Romeo, M.G.; Romeo, D.M.; Trovato, L.; Oliveri, S.; Palermo, F.; Cota, F.; Betta, P. Role of probiotics in the prevention of the enteric colonization by Candida in preterm newborns: Incidence of late-onset sepsis and neurological outcome. J. Perinatol. 2011, 31, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Rougé, C.; Piloquet, H.; Butel, M.-J.; Berger, B.; Rochat, F.; Ferraris, L.; Des Robert, C.; Legrand, A.; de la Cochetiere, M.-F.; N’Guyen, J.-M.; et al. Oral supplementation with probiotics in very-low-birth-weight preterm infants: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2009, 89, 1828–1835. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Chaudhuri, J.; Sarkar, D.; Ghosh, P.; Swapna, C. Role of enteric supplementation of probiotic on late-onset sepsisi by candida species in preterm low-boirth weight neonates: A randomized double blind, placebo-controlled trial. N. Am. J. Med. Sci. 2014, 6, 50–57. [Google Scholar] [CrossRef]
- Saengtawesin, V.; Tangpolkaiwalsak, R.; Kanjanapattankul, W. Effect of oral probiotics supplementation in the prevention of necrotizing enterocolitis among very low birth weight preterm infants. J. Med. Assoc. Thail. 2014, 97, S20–S25. [Google Scholar] [PubMed]
- Samanta, M.; Sarkar, M.; Ghosh, P.; Ghosh, J.; Sinha, M.; Chatterjee, S. Prophylactic probiotics for prevention of necrotizing enterocolitis in very low birth weight newborns. J. Trop. Pediatr. 2008, 55, 128–131. [Google Scholar] [CrossRef] [PubMed]
- Sari, F.N.; Dizdar, E.A.; Oguz, S.; Erdeve, O.; Uras, N.; Dilmen, U. Oral probiotics: Lactobacillus sporogenes for prevention of necrotizing enterocolitis in very low-birth weight infants: A randomized, controlled trial. Eur. J. Clin. Nutr. 2011, 65, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Serce, O.; Benzer, D.; Gursoy, T.; Karatekin, G.; Ovali, F. Efficacy of saccharomyces boulardii on necrotizing enterocolitis or sepsis in very low birth weight infants: A randomised controlled trial. Early Hum. Dev. 2013, 89, 1033–1036. [Google Scholar] [CrossRef] [PubMed]
- Sinha, A.; Gupta, S.S.; Chellani, H.; Maliye, C.; Kumari, V.; Arya, S.; Garg, B.S.; Gaur, S.D.; Gaind, R.; Deotale, V.; et al. Role of probiotics VSL#3 in prevention of suspected sepsis in low birthweight infants in India: A randomised controlled trial. BMJ Open 2015, 5, e006564. [Google Scholar] [CrossRef] [PubMed]
- Stratiki, Z.; Costalos, C.; Sevastiadou, S.; Kastanidou, O.; Skouroliakou, M.; Giakoumatou, A.; Petrohilou, V. The effect of a bifidobacter supplemented bovine milk on intestinal permeability of preterm infants. Early Hum. Dev. 2007, 83, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Tewari, V.V.; Dubey, S.K.; Gupta, G. Bacillus clausii for prevention of late-onset sepsis in preterm infants: A randomized controlled trial. J. Trop. Pediatr. 2015, 61, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Totsu, S.; Yamasaki, C.; Terahara, M.; Uchiyama, A.; Kusuda, S. Bifidobacterium and enteral feeding in preterm infants: Cluster-randomized trial. Pediatr. Int. 2014, 56, 714–719. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Wang, Y.; Wang, Y.; Fu, J.; Sun, M.; Mao, Z.; Vandenplas, Y. A double-blinded randomized trial on growth and feeding tolerance with Saccharomyces boulardii CNCM I-745 in formula-fed preterm infants. J. Pediatr. (Rio. J.) 2016, 92, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Drago, L.; de Vecchi, E. Should Lactobacillus sporogenes and Bacillus coagulans have a future? J. Chemother. 2009, 21, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Maffei, D.; Schanler, R.J. Human milk is the feeding strategy to prevent necrotizing enterocolitis! Semin. Perinatol. 2017, 41, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Schanler, R.J.; Shulman, R.J.; Lau, C. Feeding strategies for premature infants: Beneficial outcomes of feeding fortified human milk versus preterm formula. Pediatrics 1999, 103, 1150–1157. [Google Scholar] [CrossRef] [PubMed]
- Corvaglia, L.; Fantini, M.P.; Aceti, A.; Gibertoni, D.; Rucci, P.; Baronciani, D.; Faldella, G. Predictors of full enteral feeding achievement in very low birth weight infants. PLoS ONE 2014, 9, e92235. [Google Scholar] [CrossRef] [PubMed]
- Embleton, N.D.; Zalewski, S.; Berrington, J.E. Probiotics for prevention of necrotizing enterocolitis and sepsis in preterm infants. Curr. Opin. Infect Dis. 2016, 29, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Eidelman, A.I. Breastfeeding and the use of human milk: An analysis of the American Academy of Pediatrics 2012 Breastfeeding Policy Statement. Breastfeed. Med. 2012, 129, e827–e841. [Google Scholar] [CrossRef] [PubMed]
- Underwood, M.A. Human milk for premature infant. Pediatr. Clin. N. Am. 2013, 60, 189–207. [Google Scholar] [CrossRef] [PubMed]
- Peila, C.; Moro, G.; Bertino, E.; Cavallarin, L.; Giribaldi, M.; Giuliani, F.; Cresi, F.; Coscia, A. The effect of holder pasteurization on nutrients and biologically-active components in donor human milk: A review. Nutrients 2016, 8, 477. [Google Scholar] [CrossRef] [PubMed]
- Dang, D.; Zhou, W.; Lun, Z.J.; Mu, X.; Wang, D.X.; Wu, H. Meta-analysis of probiotics and/or prebiotics for the prevention of eczema. J. Int. Med. Res. 2013, 41, 1426–1436. [Google Scholar] [CrossRef] [PubMed]
- Bertelli, C.; Pillonel, T.; Torregrossa, A.; Prod’hom, G.; Fischer, C.J.; Greub, G.; Giannoni, E. Bifidobacterium longum bacteremia in preterm infants receiving probiotics. Clin. Infect. Dis. 2015, 60, 924–927. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Study Details | Study Population | Intervention Specie | Placebo |
|---|---|---|---|---|
| Dose (D) | ||||
| Start of Treatment (S) | ||||
| End of Treatment (E) | ||||
| Al-Hosni, 2012 [28] | P | Preterm infants with BW 501–1000g, appropriate for gestational age, and ≤ 14 days of age at time of feeding initiation | Lactobacillus rhamnosus GG Bifidobacterium Infantis | Extra milk |
| DB | D: 0.5 × 109 CFU each probiotic, OD | |||
| R | S: first enteral feeding | |||
| C | E: discharge or until 34 w postmenstrual age | |||
| Multic. | ||||
| Bin-Nun, 2005 [29] | P | Preterm infants with BW < 1500g, who began enteral feeding on a weekday | Bifidobacterium infantis Streptococcus thermophileus Bifidobacterium bifidus | HM or FM |
| B | D: 0.35 × 109 CFU each probiotic, OD | |||
| R | S: Start of enteral feeding | |||
| C | E: 36 w postconceptual age | |||
| Braga, 2011 [30] | P | Inborn infants with BW 750–1499 g | Lactobacillus casei Bifidobacterium breve | Extra HM |
| DB | D: 3.5 × 107 CFU to 3.5 × 109 CFU OD | |||
| R | S: Day 2 | |||
| C | E: Day 30, NEC diagnosis, discharge, death whichever occurred first | |||
| Chrzanowska-Liszewska, 2012 [31] | P | Preterm infants with GA< 32 w and BW> 1000g, who started enteral formula feeding before enrollment | Lactobacillus rhamnosus | MDX |
| DB | D: 6 × 109 CFU, OD | |||
| R | S: Day 0–3 of life | |||
| C | E: Day 42 of supplementation | |||
| Costalos, 2003 [32] | P | GA 28–32 w No major GI problem Not receiving antibiotics Not receiving breast milk | Saccharomyces boulardii | MDX |
| R | D: 1×109 CFU BD | |||
| C | S: Non-specified | |||
| Median duration of probiotic supplementation: 30 days | ||||
| Costeloe, 2016 [33] | P | Preterm infants with GA 23–30 w No severe malformation or any GI tract Malformation | Bifidobacterium breve BBG-001 | Corn starch |
| B | D: 8.3–8.8 log10 | |||
| R | S: 43.9 h (median age) | |||
| C | E: 36 weeks’ postmenstrual age or discharge | |||
| Multic. | ||||
| Dani, 2002 [34] | P | Infants with GA< 33 w or BW < 1500 g | Lactobacillus rhamnosus GG | MDX |
| DB | D: 6×109 CFU OD | |||
| R | S: First feed | |||
| C | E: Discharge | |||
| Multic. | ||||
| Demirel, 2013 [35] | P | Preterm infants with GA≤ 32 w and BW≤ 1500 g, who survived to feed enterally | Saccharomyces boulardii | None |
| B | D: 5 × 109 CFU OD | |||
| R | S: First feed | |||
| C | E: Discharge | |||
| Dilli, 2015 [36] | P | Preterm infants with GA< 32 w and BW< 1500 g, born at or transferred to the NICU within the first week of life and fed enterally before inclusion | Bifidobacterium lactis | MDX powder |
| DB | D: 5 × 109 CFU | |||
| R | S: Beyond d7 after birth | |||
| C | E: Death or discharge (max 8 weeks) | |||
| Multic | ||||
| Dutta, 2015 [37] | P | Preterm infants with GA 27–33 w, < 96 h of age, tolerating milk ≥ 15 mL/kg/day No GI/life-threatening malformations No NEC/sepsis | Lactobacillus acidophilus Lactobacillus rhamnosus Bifidobacterium longum Saccharomyces boulardii | Potato starch, MDX, magnesium stearate. |
| B | Total D: 1010 CFU (high dose) or 109 CFU (low dose), BD | |||
| R | S: Age< 96 h | |||
| C | E: Day 14 (short course) or day 21 (long course) | |||
| Fernandez-Carrocera, 2013 [38] | P | Preterm infants with BW< 1500g | Lactobacillus acidophilus 1 CFU/g Lactobacillus rhamnosus 4.4 × 108 CFU/g Lactobacillus casei 1 × 109 CFU/g Lactobacillus plantarum 1.76 × 108 CFU/g Bifidobacterium infantis 2.76 × 107 CFU/g Streptococcus thermophilus 6.6 × 105 CFU/g | None |
| DB | Total D: 1g powder OD | |||
| R | S: Start of enteral feeding | |||
| C | E: Non-specified | |||
| Hays, 2015 [39] | P | Preterm infants with GA 25–31 w and BW 700–1600, AGA, admitted to hospital within day 7 of life, Who initiated enteral feeding before day 5. Infants with NEC ≥ IB were excluded No severe malformation No severe clinical or surgical condition | Bifidobacterium lactis only Bifidobacterium longum only Bifidobacterium lactis + Bifidobacterium longum | MDX |
| DB | D: 109 CFU each strain, OD | |||
| R | S: Non-specified | |||
| C | Duration: 4 weeks if GA≥ 29 w , 6 weeks if GA≤ 28 w or until feeding interruption for more than 72 h | |||
| Multic. | ||||
| Hikaru, 2012 [ 40] | P | Extremely preterm infants and VLBW infants No major gastrointestinal tract surgery or multiple anomalies | Bifidobacterium breve | None |
| R | D: 109 CFU, OD | |||
| C | S: Day of birth | |||
| E: Discharge | ||||
| Jacobs, 2013 [41] | P | Preterm infants with GA< 32 w and BW< 1500 g | Bifidobacterium infantis BB-02 300 CFU × 106 Streptococcus thermophilus Th-4 350 CFU × 106 Bifidobacterium lactis BB-12 350 CFU × 106 | MDX powder |
| DB | Total D: 1 × 109 CFU × 1.5 g maltodextrin powder OD | |||
| R | S: enteral feed ≥ 1 mL every 4 h | |||
| C | E: discharge or term corrected age | |||
| Multic. | ||||
| Kanic, 2015 [42] | P | Preterm infants with GA< 33 w and BW< 1500 g | Lactobacillus acidophilus Enterococus faecium Bifidobacterium infantis | None |
| R | Total D: 0.6 × 107 CFU, BD | |||
| C | S: Start of enteral feeding | |||
| E: Discharge | ||||
| Kitajima, 1997 [43] | P | Preterm infants with BW< 1500 g | Bifidobacterium breve YIT4010 | Distilled water |
| R | D: 0.5 × 109 CFU OD | |||
| C | S: Within 24 h of life | |||
| Duration of probiotic supplementation: 28 days | ||||
| Lin, 2005 [44] | P | Infants with BW< 1500 g, who started to feed enterally and survived beyond day 7 | Lactobacillus acidophilus | None |
| Bifidobacterium infantis | ||||
| B | D: ≥ 106 CFU each probiotic (= 125 mg/kg), BD | |||
| R | S. Start of enteral feeding | |||
| C | E: Discharge | |||
| Lin, 2008 [45] | P | Preterm infants with GA< 34 w and BW< 1500 g, who survived to feed enterally | Lactobacillus acidophilus NCDO 1746 Bifidobacterium bifidum NCDO 1453 | None |
| B | D: 1 × 109 CFU each probiotic (= 125 mg/kg), BD | |||
| R | S: Day 2 of age | |||
| C | Duration: 6 weeks | |||
| Multic. | ||||
| Manzoni, 2006 [46] | P | Infants with BW< 1500 g, ≥ 3 days of life, who started enteral feeding with HM | Lactobacillus rhamnosus LGG | None |
| DB | D: 6 × 109 CFU/day | |||
| R | S: Day 3 of life | |||
| C | E: End of the 6th week or discharge | |||
| Mihatsch, 2010 [47] | P | Preterm infants with GA< 30 w and BW≤ 1500 g | Bifidobacterium lactis BB12 | Indistinguishable powder |
| R | D: 2 × 109 CFU/kg 6 times a day | |||
| C | S: Start of enteral feeding | |||
| E: Non-specified | ||||
| Millar, 1993 [48] | P | Preterm infants with GA≤ 33 w | Lactobacillus GG | None |
| DB | D: 108 CFU, BD | |||
| R | S: Start of enteral feed | |||
| Duration: 14 days | ||||
| Oncel, Sari, 2013 [49] | P | Preterm infants with GA≤ 32 w and BW≤ 1500 g, who survived to feed enterally | Lactobacillus reuteri DSM 17938 | Oil base |
| DB | D: 1 × 108 CFU OD | |||
| R | S: First feed | |||
| C | E: Death or discharge | |||
| Partty, 2013 [50] | P | Preterm infants with GA 32–36 w and BW> 1500 g | Lactobacillus rhamnosus GG | Microcrystal line cellulose and dextrose anhydrate |
| DB | D: 1 × 109 CFU | |||
| R | S: Day 1 | |||
| C | E: OD until day 30, BD until day 60 | |||
| Patole, 2014 [51] | P | Preterm infants with GA< 33 w and BW< 1500 g | Bifidobacterium breve M16-V | Dextrin |
| DB | D: 3 × 109 CFU OD (1.5 × 109 CFU OD for newborns≤ 27 w until they reached 50 ml/kg/day enteral feeds) | |||
| R | S: Start of enteral feed | |||
| C | E: Corrected age of 37 w | |||
| Rojas, 2012 [52] | P | Preterm infants with BW≤ 2000 g, hemodynamically stable, ≤ 48 h of age (regardless start of enteral feeding) | Lactobacillus reuteri DSM 17938 | Oil base |
| DB | D: 1 × 108 CFU OD | |||
| R | S: Age≤ 48 h | |||
| C | E: Death or discharge | |||
| Multic. | ||||
| Romeo, 2011 [53] | P | Preterm infants with GA< 37 w and BW< 2500g, who reached stable enteral feeding within 72 h of life | Lactobacillus reuteri ATCC 55730, 1 × 108 CFU OD | None |
| R | Lactobacillus rhamnosus ATCC 53103, 6 × 109 CFU OD | |||
| C | S: Within 72 h of life | |||
| E: After 6 w or at discharge | ||||
| Rougé, 2009 [54] | P | Preterm infants with GA< 32 w and BW≤ 1500 g, ≤2 w of age, without any disease other than those linked to prematurity, who started enteral feeding before inclusion | Bifidobacterium longum BB536 Lactobacillus rhamnosus GG BB536-LGG | MDX |
| DB | Total D: 1 × 108 CFU/day | |||
| R | S: Start of enteral feeding | |||
| C | E: Discharge | |||
| Bic. | ||||
| Roy, 2014 [55] | P | Preterm infants with GA< 37 w and BW< 2500 g | Lactobacillus acidophilus 1.25 × 109 CFU Bifidobacterium longum 0.1250 × 109 CFU Bifidobacterium bifidum 0.125 × 109 CFU Bifidobacterium lactis 1 × 109 CFU | Sterile water |
| R | Total D: 0.5 g powder, BD | |||
| DB | S: Within 72 h of life | |||
| C | E: After 6 w or at discharge | |||
| Saengtawesin, 2014 [56] | P | Preterm infants with GA≤ 34 w and BW≤ 1500g | Lactobacillus acidophilus 1 × 109 CFU Bifidobacterium bifidum 1 × 109 CFU | None |
| R | Total D: 125 mg/kg BD | |||
| C | S: Start of enteral feeding | |||
| E: End of 6th w of supplementation or discharge | ||||
| Samanta, 2009 [57] | P | Preterm infants with GA< 32 w and BW< 1500g, Who started to feed enterally and survived beyond 48 h of life | Bifidobacterium infantis Bifidobacterium bifidum Bifidobacterium longum Lactobacillus acidophilus | None |
| DB | D: 2.5 × 109 CFU each probiotic, BD | |||
| R | S: Non specified | |||
| C | E: Discharge | |||
| Sari, 2011 [58] | P | Preterm infants with GA< 33 w and BW< 1500 g, Who survived to feed enterally | Lactobacillus sporogenes | None |
| B | D: 0.35 × 109 CFU, OD | |||
| R | S: Start of enteral feeding | |||
| C | E: Discharge | |||
| Serce, 2013 [59] | P | Preterm infants with GA≤ 32 w and GA≤ 1500g, who survived to feed enterally | Saccharomyces boulardii | Distilled water |
| DB | D: 0.5 × 109 CFU, BD | |||
| R | S: Start of enteral feeding | |||
| C | E: Discharge | |||
| Sinha, 2015 [60] | P | Preterm infants with GA≥ 34 w and BW 1500–2500 g | Streptococcus thermophiles Bifidobacterium breve Bifidobacterium longum Bifidobacterium infantis Lactobacillus acidophilus Lactobacillus plantarum Lactobacillus paracasei Lactobacillus delbrueckii spp bulgaricus | MDX |
| DB | Total D: 10 × 109 CFU per day | |||
| R | S: Day 3 of life | |||
| C | Duration: 30 days | |||
| Bic. | ||||
| Stratiki, 2007 [61] | P | Preterm infants with GA 27–37 w, formula fed | Bifidobacterium lactis | None |
| B | D: 2 × 107 CFU/ g milk powder | |||
| R | S: Start of enteral feeding | |||
| C | E: Discharge | |||
| Tewari, 2015 [62] | P | Preterm infants with GA< 34 w | Bacillus clausii | Sterile water |
| DB | D: 2.9 × 109 spores | |||
| R | S: D5 in asymptomatic, d10 in symptomatic infants | |||
| C | E: 6 w of life, discharge, death, LOS diagnosis, whichever occurred first | |||
| Totsu, 2014 [63] | P | Infants with BW< 1500 g | Bifidobacterium bifidum | Dextrin |
| DB | D: 2.5 × 109 CFU, divided in two doses | |||
| CLR | S: Within 48 h after birth | |||
| C | E: Body weight 2000 g | |||
| Multic. | ||||
| Xu, 2016 [64] | P | Preterm infants with GA> 30 and BW 1500–2500 g, formula fed | Saccharomyces boulardii | None |
| B | D: 109 CFU/Kg , BD | |||
| R | S: Start of enteral feeding | |||
| C | E: 28th day of life or discharge |
| Authors, Year | Study Summary | Reason for Exclusion |
|---|---|---|
| Awad, 2000 [25] | Living vs. killed Lactobacillus acidophilus vs. placebo given to neonates admitted to the study NICU | Term and preterm infants included |
| Manzoni, 2009 [26] | Bovine Lactoferrin (BLF) alone or BLF plus Lactobacillus rhamnosus GG given to VLBW neonates | Supplementation with probiotic and lactoferrin |
| Ren B, 2010 [27] | Bacillus clausii and Clostridium (butyricum) San Chang Le Kang given to preterm infants (exact gestational age unclear) | Non English-written study |
| Author, Year | Previous LOS Rate | Number of Subjects | LOS in Probiotic Group | LOS in Control Group |
|---|---|---|---|---|
| Al-Hosni, 2012 [28] | Not stated | 50 probiotic 51 control | 13/50 | 16/51 |
| Bin-Nun, 2005 [29] | Not stated | 72 probiotic 73 control | 31/72 | 24/73 |
| Braga, 2011 [30] | Not stated | 119 probiotic 112 placebo | 40/119 | 42/112 |
| Chrzanowska-Liszewska, 2012 [31] | Not stated | 21 probiotic 26 placebo | 2/21 | 3/26 |
| Costalos, 2003 [32] | Not Stated | 51 probiotic 36 placebo | 3/51 | 3/36 |
| Costeloe, 2015 [33] | 15% | 650 probiotic 660 placebo | 73/650 | 77/660 |
| Dani, 2002 [34] | Not stated | 295 probiotic 290 placebo | 14/295 | 12/290 |
| Demirel, 2013 [35] | Not stated | 135 probiotic 136 control | 20/135 | 21/136 |
| Dilli, 2015 [36] | Not stated | 100 probiotic 100 placebo | 8/100 | 13/100 |
| Dutta, 2015 A [37] | Not stated | 38 probiotic 35 placebo | 3/38 | 6/35 |
| Dutta, 2015 B [37] | Not stated | 38 probiotic 35 placebo | 1/38 | 6/35 |
| Dutta, 2015 C [37] | Not stated | 38 probiotic 35 placebo | 6/38 | 6/35 |
| Fernández-Carrocera, 2013 [38] | Not stated | 75 probiotic 75 placebo | 42/75 | 44/75 |
| Hays, 2015 P1 [39] | Not stated | 50 probiotic 52 placebo | 9/50 | 10/52 |
| Hays, 2015 P2 [39] | Not stated | 48 probiotic 52 placebo | 8/48 | 10/52 |
| Hays, 2015 P3 [39] | Not stated | 47 probiotic 52 placebo | 8/47 | 10/52 |
| Hikaru, 2012 [40] | Not stated | 108 probiotic 100 control | 10/108 | 22/100 |
| Jacobs, 2013 [41] | 23% | 548 probiotic 551 placebo | 72/548 | 89/551 |
| Kanic, 2015 [42] | Not stated | 40 probiotic 40 control | 16/40 | 29/40 |
| Kitajima, 1997 [43] | Not stated | 45 probiotic 46 placebo | 1/45 | 0/46 |
| Lin, 2005 [44] | Not stated | 180 probiotic 187 control | 22/180 | 36/187 |
| Lin, 2008 [45] | Not stated | 217 placebo 217 control | 40/217 | 24/217 |
| Manzoni, 2006 [46] | Not stated | 39 probiotic 41 control | 19/39 | 22/41 |
| Mihatsch, 2010 [47] | 40% | 91 probiotic 89 placebo | 28/91 | 29/89 |
| Millar, 1993 [48] | Not stated | 10 probiotic 10 control | 0/10 | 0/10 |
| Oncel, Sari 2013 [49] | Not stated | 200 probiotic 200 placebo | 13/200 | 25/200 |
| Partty, 2013 [50] | Not stated | 31 probiotic 32 placebo | 0/31 | 0/32 |
| Patole, 2014 [51] | Not stated | 77 probiotic 76 placebo | 17/77 | 12/76 |
| Rojas, 2012 [52] | 28% | 372 probiotic 378 placebo | 24/372 | 17/378 |
| Romeo, 2011 A [53] | Not stated | 83 probiotic 83 control | 1/83 | 9/83 |
| Romeo, 2011 B [53] | Not stated | 83 probiotic 83 control | 2/83 | 9/83 |
| Rougé, 2009 [54] | Not stated | 45 probiotic 49 placebo | 15/45 | 13/49 |
| Roy, 2014 [55] | 33% | 56 probiotic 56 placebo | 31/56 | 42/56 |
| Saengtawesin, 2014 [56] | Not stated | 31 probiotic 29 control | 2/31 | 1/29 |
| Samanta, 2009 [57] | Not stated | 91 probiotic 95 control | 13/91 | 28/95 |
| Sari, 2011 [58] | Not stated | 110 probiotic 111 control | 29/110 | 26/111 |
| Serce, 2013 [59] | 19% | 104 probiotic 104 placebo | 19/104 | 25/104 |
| Sinha, 2015 [60] | 17% | 668 probiotic 672 placebo | 38/668 | 54/672 |
| Stratiki, 2007 [61] | Not stated | 41 probiotic 36 control | 0/41 | 3/36 |
| Tewari, 2015 E [62] | 21% | 61 probiotic 59 placebo | 6/61 | 8/59 |
| Tewari, 2015 V [62] | 21% | 62 probiotic 62 placebo | 2/62 | 3/62 |
| Totsu, 2014 [63] | Not stated | 153 probiotic 130 placebo | 6/153 | 10/130 |
| Xu, 2016 [64] | Not stated | 51 probiotic 49 control | 4/51 | 6/49 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aceti, A.; Maggio, L.; Beghetti, I.; Gori, D.; Barone, G.; Callegari, M.L.; Fantini, M.P.; Indrio, F.; Meneghin, F.; Morelli, L.; et al. Probiotics Prevent Late-Onset Sepsis in Human Milk-Fed, Very Low Birth Weight Preterm Infants: Systematic Review and Meta-Analysis. Nutrients 2017, 9, 904. https://doi.org/10.3390/nu9080904
Aceti A, Maggio L, Beghetti I, Gori D, Barone G, Callegari ML, Fantini MP, Indrio F, Meneghin F, Morelli L, et al. Probiotics Prevent Late-Onset Sepsis in Human Milk-Fed, Very Low Birth Weight Preterm Infants: Systematic Review and Meta-Analysis. Nutrients. 2017; 9(8):904. https://doi.org/10.3390/nu9080904
Chicago/Turabian StyleAceti, Arianna, Luca Maggio, Isadora Beghetti, Davide Gori, Giovanni Barone, Maria Luisa Callegari, Maria Pia Fantini, Flavia Indrio, Fabio Meneghin, Lorenzo Morelli, and et al. 2017. "Probiotics Prevent Late-Onset Sepsis in Human Milk-Fed, Very Low Birth Weight Preterm Infants: Systematic Review and Meta-Analysis" Nutrients 9, no. 8: 904. https://doi.org/10.3390/nu9080904
APA StyleAceti, A., Maggio, L., Beghetti, I., Gori, D., Barone, G., Callegari, M. L., Fantini, M. P., Indrio, F., Meneghin, F., Morelli, L., Zuccotti, G., Corvaglia, L., & On behalf of the Italian Society of Neonatology. (2017). Probiotics Prevent Late-Onset Sepsis in Human Milk-Fed, Very Low Birth Weight Preterm Infants: Systematic Review and Meta-Analysis. Nutrients, 9(8), 904. https://doi.org/10.3390/nu9080904











