The Effect of a Standardized Ginger Extract on Chemotherapy-Induced Nausea-Related Quality of Life in Patients Undergoing Moderately or Highly Emetogenic Chemotherapy: A Double Blind, Randomized, Placebo Controlled Trial
Abstract
1. Introduction
2. Methods
2.1. Sample and Recruitment
2.2. Intervention
2.3. Procedure
2.4. Outcome Measures
2.4.1. Primary Outcome
2.4.2. Secondary Outcomes
2.5. Adverse Events
2.6. Statistical Analysis
3. Results
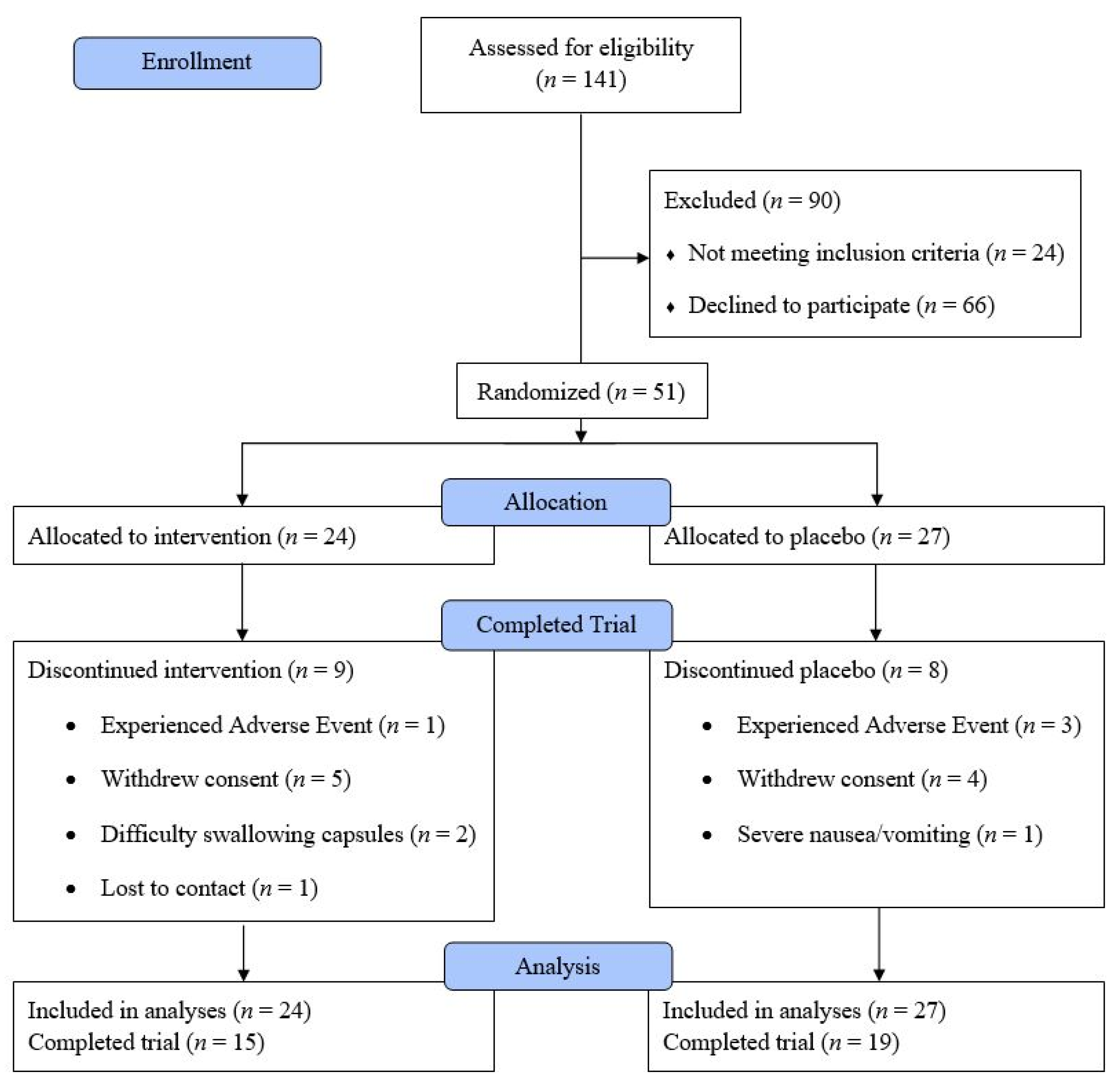
3.1. Patient Demographics and Adherence
3.2. CINV-Related Quality of Life
3.3. Nausea and Vomiting Symptoms
3.4. Fatigue, Nutrition Status, and Cancer-Related Quality of Life
3.5. Participant Blinding and Adherence
3.6. Effect of Prognostic Factors on CINV-Related Outcomes
3.7. Adverse Events
3.8. Incomplete Questionnaires
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hsieh, R.K.; Chan, A.; Kim, H.K.; Yu, S.; Kim, J.G.; Lee, M.A.; Dalen, J.; Jung, H.; Liu, Y.P.; Burke, T.A.; et al. Baseline patient characteristics, incidence of cinv, and physician perception of cinv incidence following moderately and highly emetogenic chemotherapy in asia pacific countries. Support. Care Cancer 2015, 23, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Davidson, W.; Teleni, L.; Muller, J.; Ferguson, M.; McCarthy, A.; Vick, J.; Isenring, E. Malnutrition and chemotherapy-induced nausea and vomiting: Implications for practice. Oncol. Nurs. Forum 2012, 39, E340–E345. [Google Scholar] [CrossRef] [PubMed]
- Bloechl-Daum, B.; Deuson, R.R.; Mavros, P.; Hansen, M.; Herrstedt, J. Delayed nausea and vomiting continue to reduce patients’ quality of life after highly and moderately emetogenic chemotherapy despite antiemetic treatment. J. Clin. Oncol. 2006, 24, 4472–4478. [Google Scholar] [CrossRef] [PubMed]
- Neymark, N.; Crott, R. Impact of emesis on clinical and economic outcomes of cancer therapy with highly emetogenic chemotherapy regimens: A retrospective analysis of three clinical trials. Support. Care Cancer 2005, 13, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Molassiotis, A.; Yung, H.P.; Yam, B.M.; Chan, F.Y.; Mok, T.S. The effectiveness of progressive muscle relaxation training in managing chemotherapy-induced nausea and vomiting in chinese breast cancer patients: A randomised controlled trial. Support. Care Cancer 2002, 10, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, M.; Brito-Dellan, N.; Davis, M.; Leach, M.; Lagman, R. Olanzapine as an antiemetic in refractory nausea and vomiting in advanced cancer. J. Pain Symptom Manag. 2003, 25, 578–582. [Google Scholar] [CrossRef]
- Marx, W.M.; Teleni, L.; McCarthy, A.L.; Vitetta, L.; McKavanagh, D.; Thomson, D.; Isenring, E. Ginger (Zingiber officinale) and chemotherapy-induced nausea and vomiting: A systematic literature review. Nutr. Rev. 2013, 71, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Marx, W.; Ried, K.; McCarthy, A.L.; Vitetta, L.; Sali, A.; McKavanagh, D.; Isenring, E. Ginger—Mechanism of action in chemotherapy-induced nausea and vomiting: A review. Crit. Rev. Food Sci. Nutr. 2015. [Google Scholar] [CrossRef] [PubMed]
- Marx, W.; Kiss, N.; Isenring, L. Is ginger beneficial for nausea and vomiting? An update of the literature. Curr. Opin. Support. Palliat. Care 2015, 9, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Chaiyakunapruk, N.; Kitikannakorn, N.; Nathisuwan, S.; Leeprakobboon, K.; Leelasettagool, C. The efficacy of ginger for the prevention of postoperative nausea and vomiting: A meta-analysis. Am. J. Obstet. Gynecol. 2006, 194, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Viljoen, E.; Visser, J.; Koen, N.; Musekiwa, A. A systematic review and meta-analysis of the effect and safety of ginger in the treatment of pregnancy-associated nausea and vomiting. Nutr. J. 2014, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- Marx, W.; McCarthy, A.; Ried, K.; Vitetta, L.; McKavanagh, D.; Thomson, D.; Sali, A.; Isenring, L. Can ginger ameliorate chemotherapy-induced nausea? Protocol of a randomized double blind, placebo-controlled trial. BMC Complement. Altern. Med. 2014, 14, 134. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.; Seo, W. Systematic review and meta-analysis of the correlates of cancer-related fatigue. Worldviews Evid Based Nurs. 2011, 8, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Zick, S.M.; Ruffin, M.T.; Lee, J.; Normolle, D.P.; Siden, R.; Alrawi, S.; Brenner, D.E. Phase II trial of encapsulated ginger as a treatment for chemotherapy-induced nausea and vomiting. Support. Care Cancer 2009, 17, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Schag, C.C.; Heinrich, R.L.; Ganz, P.A. Karnofsky performance status revisited: Reliability, validity, and guidelines. J. Clin. Oncol. 1984, 2, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Roila, F.; Hesketh, P.J.; Herrstedt, J. Prevention of chemotherapy- and radiotherapy-induced emesis: Results of the 2004 perugia international antiemetic consensus conference. Ann. Oncol. 2006, 17, 20–28. [Google Scholar] [PubMed]
- Lindley, C.M.; Hirsch, J.D.; O'Neill, C.V.; Transau, M.C.; Gilbert, C.S.; Osterhaus, J.T. Quality of life consequences of chemotherapy-induced emesis. Qual. Life Res. 1992, 1, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.R.; Pearson, J.D.; Cai, B.; Elmer, M.; Horgan, K.; Lindley, C. Assessing the impact of chemotherapy-induced nausea and vomiting on patients’ daily lives: A modified version of the functional living index-emesis (flie) with 5-day recall. Support. Care Cancer 2003, 11, 522–527. [Google Scholar] [PubMed]
- Rhodes, V.A.; McDaniel, R.W. The index of nausea, vomiting, and retching: A new format of the lndex of nausea and vomiting. Oncol. Nurs. Forum 1999, 26, 889–894. [Google Scholar] [PubMed]
- National Comprehensive Cancer Network (NCCN). NCCN Practice Guidelines in Oncology™ (v.1.2015): Antiemesis. Available online: http://www.nccn.org/professionals/physician_gls/pdf/antiemesis.pdf (accessed on 18 December 2016).
- Isenring, E.; Bauer, J.; Capra, S. The scored patient-generated subjective global assessment (PG-SGA) and its association with quality of life in ambulatory patients receiving radiotherapy. Eur. J. Clin. Nutr. 2003, 57, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Cella, D.; Hahn, E.A.; Dineen, K. Meaningful change in cancer-specific quality of life scores: Differences between improvement and worsening. Qual. Life Res. 2002, 11, 207–221. [Google Scholar] [CrossRef] [PubMed]
- Yellen, S.B.; Cella, D.F.; Webster, K.; Blendowski, C.; Kaplan, E. Measuring fatigue and other anemia-related symptoms with the functional assessment of cancer therapy (FACT) measurement system. J. Pain Symptom Manag. 1997, 13, 63–74. [Google Scholar] [CrossRef]
- Cella, D.; Eton, D.T.; Lai, J.S.; Peterman, A.H.; Merkel, D.E. Combining anchor and distribution-based methods to derive minimal clinically important differences on the functional assessment of cancer therapy (FACT) anemia and fatigue scales. J. Pain Symptom Manag. 2002, 24, 547–561. [Google Scholar] [CrossRef]
- Van Belle, S.; Paridaens, R.; Evers, G.; Kerger, J.; Bron, D.; Foubert, J.; Ponnet, G.; Vander Steichel, D.; Heremans, C.; Rosillon, D. Comparison of proposed diagnostic criteria with FACT-F and VAS for cancer-related fatigue: Proposal for use as a screening tool. Support. Care Cancer 2005, 13, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Webster, K.; Cella, D.; Yost, K. The functional assessment of chronic illness therapy (FACIT) measurement system: Properties, applications, and interpretation. Health Qual. Life Outcomes 2003, 1, 79. [Google Scholar] [CrossRef] [PubMed]
- Hickok, J.T.; Roscoe, J.A.; Morrow, G.R. The role of patients’ expectations in the development of anticipatory nausea related to chemotherapy for cancer. J. Pain Symptom Manag. 2001, 22, 843–850. [Google Scholar] [CrossRef]
- Pirri, C.; Katris, P.; Trotter, J.; Bayliss, E.; Bennett, R.; Drummond, P. Risk factors at pretreatment predicting treatment-induced nausea and vomiting in australian cancer patients: A prospective, longitudinal, observational study. Support. Care Cancer 2011, 19, 1549–1563. [Google Scholar] [CrossRef] [PubMed]
- Chang, V.T.; Hwang, S.S.; Feuerman, M. Validation of the edmonton symptom assessment scale. Cancer 2000, 88, 2164–2171. [Google Scholar] [CrossRef]
- Roila, F.; Herrstedt, J.; Aapro, M.; Gralla, R.J.; Einhorn, L.H.; Ballatori, E.; Bria, E.; Clark-Snow, R.A.; Espersen, B.T.; Feyer, P.; et al. Guideline update for mascc and esmo in the prevention of chemotherapy- and radiotherapy-induced nausea and vomiting: Results of the perugia consensus conference. Ann. Oncol. 2010, 21, v232–v243. [Google Scholar] [CrossRef] [PubMed]
- Fahimi, F.; Khodadad, K.; Amini, S.; Naghibi, F.; Salamzadeh, J.; Baniasadi, S. Evaluating the effect of zingiber officinalis on nausea and vomiting in patients receiving cisplatin based regimens. Iran. J. Pharm. Res. 2011, 10, 379–384. [Google Scholar] [PubMed]
- Cohen, L.; de Moor, C.A.; Eisenberg, P.; Ming, E.E.; Hu, H. Chemotherapy-induced nausea and vomiting: Incidence and impact on patient quality of life at community oncology settings. Support. Care Cancer 2007, 15, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Sontakke, S.; Thawani, V.; Naik, M.S. Ginger as an antiemetic in nausea and vomiting induced by chemotherapy: A randomized, cross-over, double blind study. Indian J. Pharmacol. 2003, 35, 32–36. [Google Scholar]
- Manusirivithaya, S.; Sripramote, M.; Tangjitgamol, S.; Sheanakul, C.; Leelahakorn, S.; Thavaramara, T.; Tangcharoenpanich, K. Antiemetic effect of ginger in gynecologic oncology patients receiving cisplatin. Int. J. Gynecol. Cancer 2004, 14, 1063–1069. [Google Scholar] [CrossRef] [PubMed]
- Sprangers, M.A.G.; Schwartz, C.E. Integrating response shift into health-related quality of life research: A theoretical model. Soc. Sci. Med. 1999, 48, 1507–1515. [Google Scholar] [CrossRef]
- Konmun, J.; Danwilai, K.; Ngamphaiboon, N.; Sripanidkulchai, B.; Sookprasert, A.; Subongkot, S. A phase ii randomized double-blind placebo-controlled study of 6-gingerol as an anti-emetic in solid tumor patients receiving moderately to highly emetogenic chemotherapy. Med. Oncol. (Northwood, London, England) 2017, 34, 69. [Google Scholar] [CrossRef] [PubMed]
- Marx, W.; McKavanagh, D.; McCarthy, A.L.; Bird, R.; Ried, K.; Chan, A.; Isenring, L. The effect of ginger (Zingiber officinale) on platelet aggregation: A systematic literature review. PLoS ONE 2015, 10, e0141119. [Google Scholar]
- Petrella, T.; Clemons, M.; Joy, A.; Young, S.; Callaghan, W.; Dranitsaris, G. Identifying patients at high risk for nausea and vomiting after chemotherapy: The development of a practical validated prediction tool. J. Support. Oncol. 2009, 7, 9–16. [Google Scholar]
- Yu, Y.; Zick, S.; Li, X.; Zou, P.; Wright, B.; Sun, D. Examination of the pharmacokinetics of active ingredients of ginger in humans. AAPS J. 2011, 13, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Zick, S.M.; Djuric, Z.; Ruffin, M.T.; Litzinger, A.J.; Normolle, D.P.; Alrawi, S.; Feng, M.R.; Brenner, D.E. Pharmacokinetics of 6-gingerol, 8-gingerol, 10-gingerol, and 6-shogaol and conjugate metabolites in healthy human subjects. Cancer Epidemiol. Biomark. Prev. 2008, 17, 1930–1936. [Google Scholar] [CrossRef] [PubMed]
| Total | Intervention Group | Control Group | |
|---|---|---|---|
| n | 51 | 24 | 27 |
| Age (mean ± SD, years) | 58 ± 12 | 57 ± 14 | 59 ± 11 |
| Gender (n, % female) | 32 (63) | 16 (66) | 16 (59) |
| Race (n, % Caucasian) | 42 (82) | 18 (75) | 24 (88) |
| Primary diagnoses (n) | |||
| Breast | 13 | 7 | 6 |
| Colon | 19 | 8 | 11 |
| Lymphoma | 11 | 5 | 6 |
| Other | 8 | 4 | 4 |
| Chemotherapy Emetogenicity (n) | |||
| HEC | 8.0 | 4.0 | 4.0 |
| MEC | 43.0 | 20.0 | 23.0 |
| Receiving aprepitant (n) | 18.0 | 7.0 | 11.0 |
| Cycle 1 | Cycle 2 | Cycle 3 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total | Placebo | Intervention | p Value | Total | Placebo | Intervention | p Value | Total | Placebo | Intervention | p Value | |
| CINV-QoL | 123 (103, 126) | 111 (99, 126) | 124.5 (113.2, 126) | 0.043 * | 124 (108, 126) | 117 (109, 126) | 124 (108, 126) | 0.916 | 122 (107, 126) | 120 (111, 126) | 123.5 (107, 126) | 0.931 |
| Nausea-related QoL | 60 (51, 63) | 54 (46, 63) | 61.5 (56.2, 63) | 0.029 * | 61 (49, 63) | 56 (49, 63) | 61 (52.2, 63) | 0.494 | 56 (49, 63) | 56 (49, 63) | 56.5 (46.7, 63) | 0.931 |
| Vomiting-related QoL | 63 (51, 63) | 63 (51, 63) | 63 (54, 63) | 0.237 | 63 (51.9, 63) | 63 (52, 63) | 63 (54.7, 63) | 0.663 | 63 (51, 63) | 63 (54, 63) | 59 (50.2, 63) | 0.414 |
| Global cancer-related QoL | 78.1 ± 19.5 | 71.9 ± 18.3 | 85.1 ± 18.9 | 0.015 * | 71.1 ± 14.5 | 67.6 ± 10.2 | 74.9 ± 17.7 | 0.077 | 79.1 ± 14.9 | 75 ± 13.8 | 83.6 ± 15 | 0.040 * |
| Fatigue | 36.7 ± 12.8 | 32.2 ± 10.8 | 41.8 ± 13 | 0.006 * | 36.1 ± 9.4 | 34.5 ± 7.9 | 37.7 ± 10.8 | 0.23 | 39.1 ± 9.2 | 36.1 ± 7.2 | 42.4 ± 10.2 | 0.013 * |
| Nutrition status at start of cycle (n, well nourished) | 44 | 22 | 22 | 0.371 | 38 | 19 | 19 | 0.500 | 37 | 19 | 18 | 0.622 |
| Cycle 1 | Cycle 2 | Cycle 3 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total | Placebo | Intervention | p Value | Total | Placebo | Intervention | p Value | Total | Placebo | Intervention | p Value | |
| Anticipatory CINV score | 8 (8, 9) | 8 (8, 8) | 8 (8, 8) | 0.44 | 8 (8, 9) | 8 (8, 9) | 8 (8, 9) | 0.61 | 8 (8, 8) | 8 (8, 9) | 8 ( 8,8) | 0.76 |
| Acute CINV score | 9 (9, 11) | 9 (9, 11) | 10 (9, 10.7) | 0.84 | 9 (9, 10) | 9 (8, 10) | 9 (9, 9) | 0.94 | 9 (9, 11) | 9 (9, 11) | 10 (9, 11) | 0.12 |
| Vomiting score | 3 (3, 3) | 3 (3, 3) | 3 (3, 3) | 0.41 | 3 (3, 3) | 3 (3, 3) | 3 (3, 3) | 0.99 | 3 (3, 3) | 3 (3, 3) | 3 (3, 3) | 0.17 |
| Nausea score | 3 (3, 5) | 3 (3, 5) | 3.6 (3, 5) | 0.46 | 3 (3, 4) | 3 (3, 4) | 3 (3, 3) | 0.63 | 3 (3, 5) | 3 (3, 5) | 3 (3, 5) | 0.79 |
| Retching score | 2 (2, 2) | 2 (2, 2) | 2 (2, 2) | 0.78 | 2 (2, 2) | 2 (2, 2) | 2 (2, 2) | 0.99 | 2 (2, 2) | 2 (2, 2) | 2 (2, 2) | 0.46 |
| Delayed CINV score | 32 (27, 34) | 32 (28, 34) | 31 (26, 34.7) | 0.74 | 30 (28, 31) | 30 (29, 31) | 29 (28, 31) | 0.26 | 29 (27, 30) | 29 (27, 30) | 29 (27, 30) | 0.86 |
| Vomiting score | 9 (9, 11) | 9 (9, 12) | 9 (9, 9.7) | 0.74 | 9 (9, 10) | 9 (9, 10) | 9 (9, 10) | 0.95 | 9 (9, 9) | 9 (9, 10) | 9 (9, 9) | 0.69 |
| Nausea score | 13 (9, 19) | 15 (9, 20) | 11 (9, 17) | 0.27 | 13 (9, 15) | 12 (9, 16) | 14.5 (9, 15) | 0.54 | 12 (9, 16) | 12 (9, 16) | 12 (9, 16.5) | 0.42 |
| Retching score | 6 (6, 6) | 6 (6, 6) | 6 (6, 6.05) | 0.28 | 6 (6, 6) | 6 (6, 6) | 6 (6, 6.7) | 0.56 | 6 (6, 6) | 6 (6, 6) | 6 (6, 6) | 0.41 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marx, W.; McCarthy, A.L.; Ried, K.; McKavanagh, D.; Vitetta, L.; Sali, A.; Lohning, A.; Isenring, E. The Effect of a Standardized Ginger Extract on Chemotherapy-Induced Nausea-Related Quality of Life in Patients Undergoing Moderately or Highly Emetogenic Chemotherapy: A Double Blind, Randomized, Placebo Controlled Trial. Nutrients 2017, 9, 867. https://doi.org/10.3390/nu9080867
Marx W, McCarthy AL, Ried K, McKavanagh D, Vitetta L, Sali A, Lohning A, Isenring E. The Effect of a Standardized Ginger Extract on Chemotherapy-Induced Nausea-Related Quality of Life in Patients Undergoing Moderately or Highly Emetogenic Chemotherapy: A Double Blind, Randomized, Placebo Controlled Trial. Nutrients. 2017; 9(8):867. https://doi.org/10.3390/nu9080867
Chicago/Turabian StyleMarx, Wolfgang, Alexandra L. McCarthy, Karin Ried, Dan McKavanagh, Luis Vitetta, Avni Sali, Anna Lohning, and Elisabeth Isenring. 2017. "The Effect of a Standardized Ginger Extract on Chemotherapy-Induced Nausea-Related Quality of Life in Patients Undergoing Moderately or Highly Emetogenic Chemotherapy: A Double Blind, Randomized, Placebo Controlled Trial" Nutrients 9, no. 8: 867. https://doi.org/10.3390/nu9080867
APA StyleMarx, W., McCarthy, A. L., Ried, K., McKavanagh, D., Vitetta, L., Sali, A., Lohning, A., & Isenring, E. (2017). The Effect of a Standardized Ginger Extract on Chemotherapy-Induced Nausea-Related Quality of Life in Patients Undergoing Moderately or Highly Emetogenic Chemotherapy: A Double Blind, Randomized, Placebo Controlled Trial. Nutrients, 9(8), 867. https://doi.org/10.3390/nu9080867





