Food Processing: The Influence of the Maillard Reaction on Immunogenicity and Allergenicity of Food Proteins
Abstract
:1. Food Processing
2. Thermal Processing Induces Conformational Changes in Food Proteins
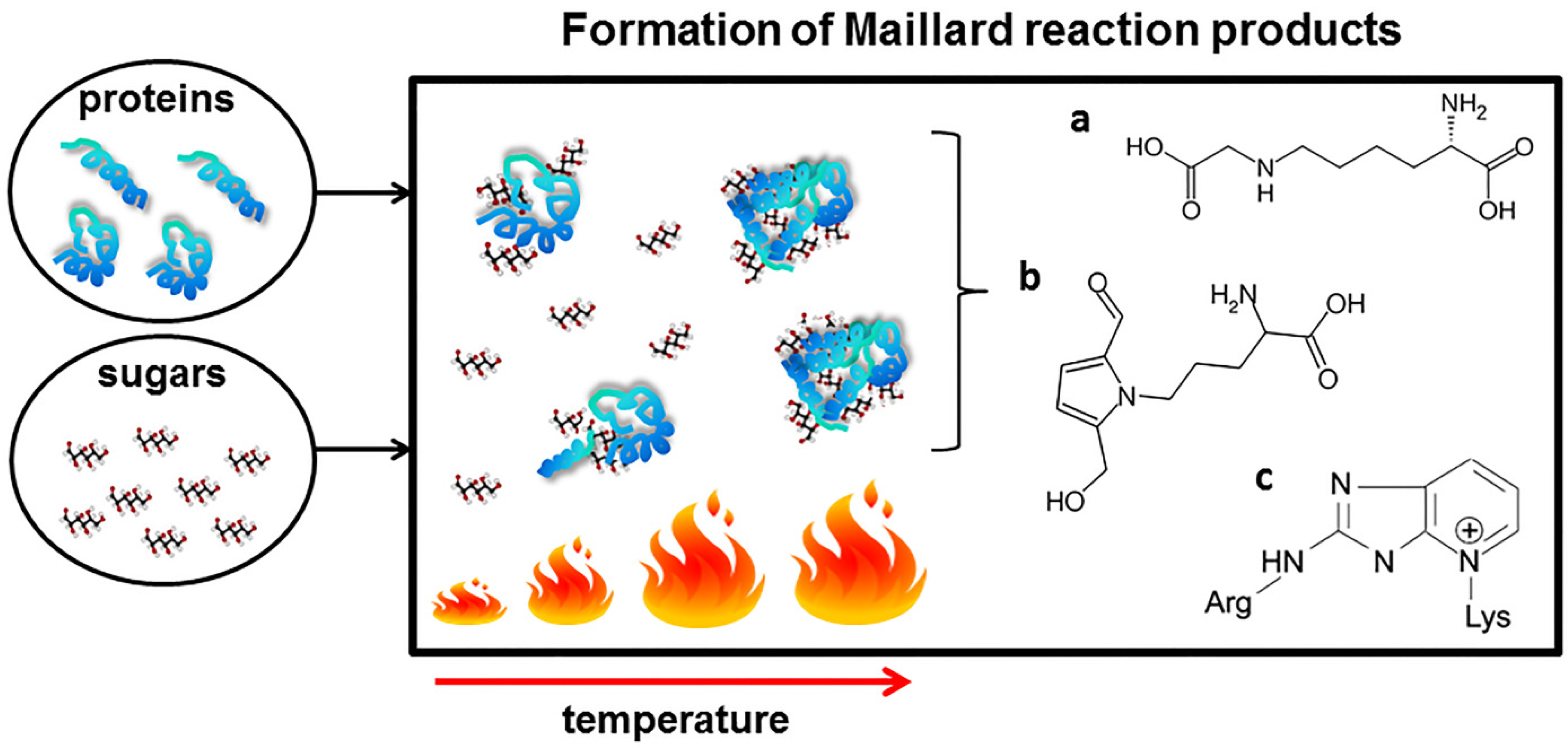
3. The Maillard Reaction in Food Processing
4. Influence of Maillard Reaction of Digestibility of Proteins
5. Absorption of MRPs in Intestine
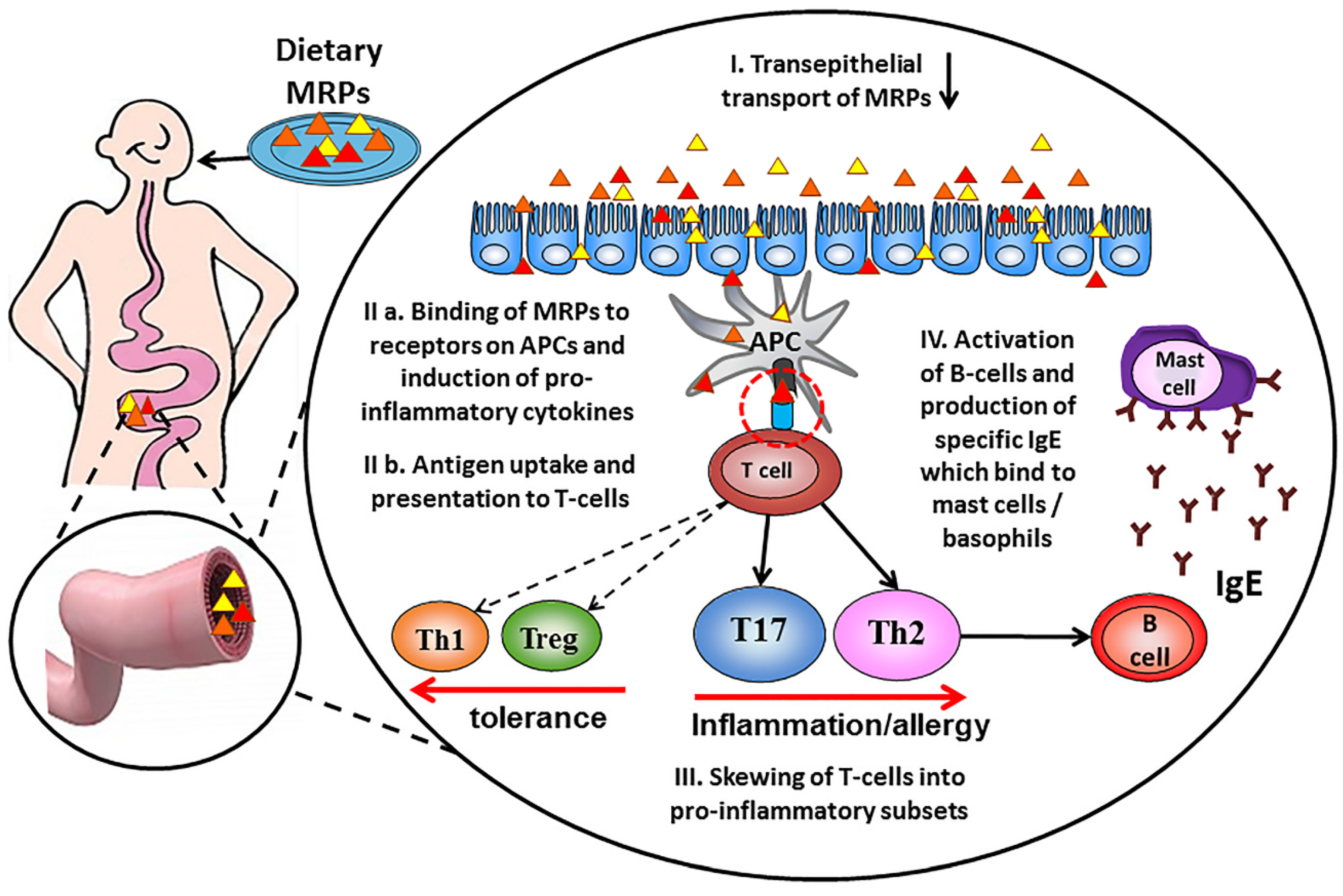
6. Influence of Maillard Reaction on Immunogenicity of Proteins
6.1. MR-Modified Proteins and Allergic Sensitization
6.2. Interaction of MR-Modified Proteins with Receptors Present on APCs
6.3. Influence of MR-Modified Proteins on T-Cell Activation and Polarization
6.4. Role of Agglomeration in Immunogenicity of MR-Modified Proteins
6.5. Influence of MR-Modified Proteins on B-Cells Switching and the Production of Antigen Specific IgG and IgE
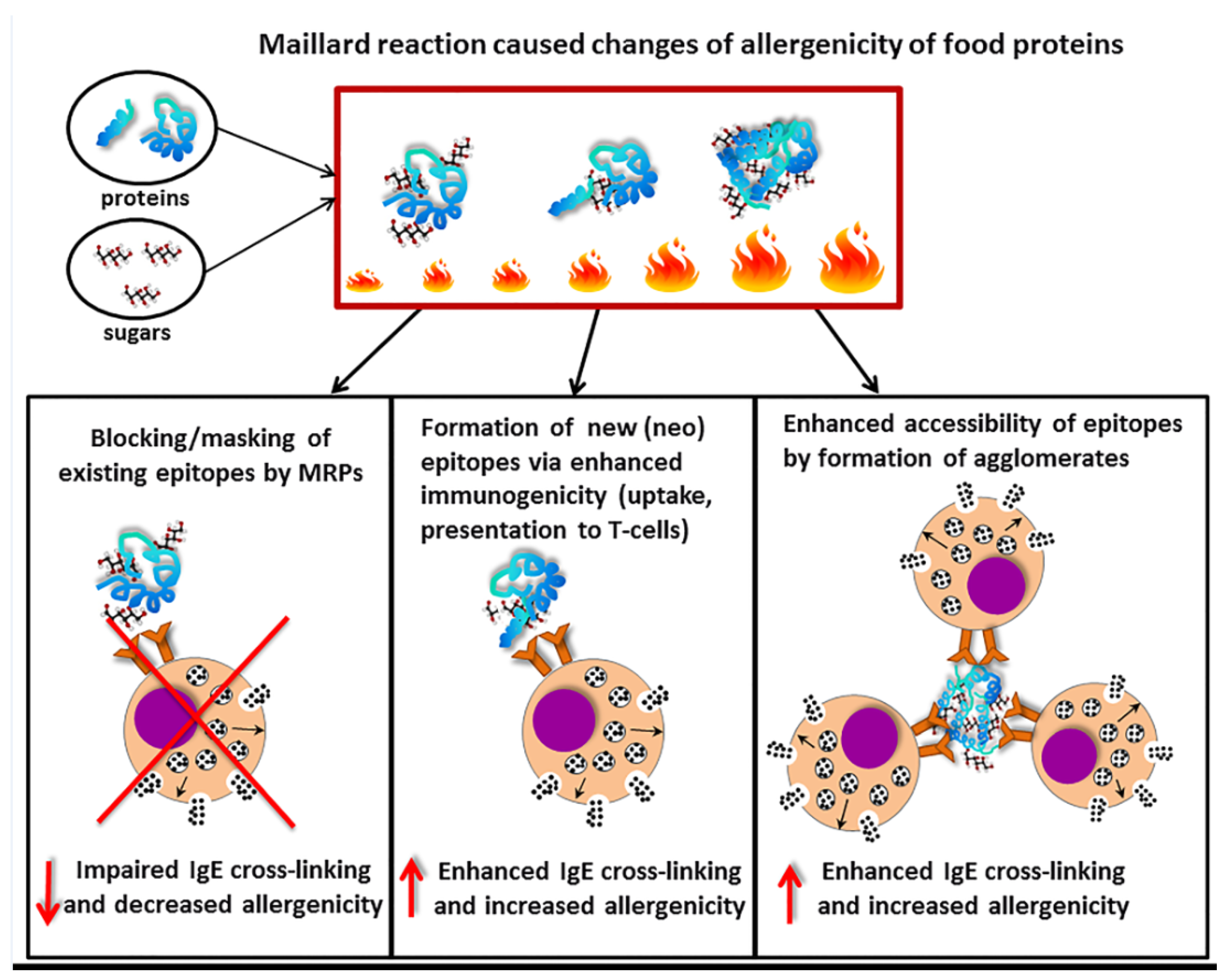
7. Influence of Maillard Reaction on Recognition of Food Allergens by Specific IgE
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pereira, R.N.; Vicente, A.A. Environmental impact of novel thermal and non-thermal technologies in food processing. Food Res. Int. 2010, 43, 1936–1943. [Google Scholar] [CrossRef] [Green Version]
- Ling, B.; Tang, J.; Kong, F.; Mitcham, E.J.; Wang, S. Kinetics of food quality changes during thermal processing: A review. Food Bioprocess Technol. 2015, 8, 343–358. [Google Scholar] [CrossRef]
- Sevenich, R.; Bark, F.; Kleinstueck, E.; Crews, C.; Pye, C.; Hradecky, J.; Reineke, K.; Lavilla, M.; Martinez-de-Maranon, I.; Briand, J.C.; et al. The impact of high pressure thermal sterilization on the microbiological stability and formation of food processing contaminants in selected fish systems and baby food puree at pilot scale. Food Control 2015, 50, 539–547. [Google Scholar] [CrossRef]
- Peng, P.; Song, H.; Zhang, T.; Addy, M.; Zhang, Y.; Cheng, Y.; Hatzenbeller, R.; Zhu, X.; Liu, S.; Liu, Y.; et al. Concentrated high intensity electric field (chief) system for non-thermal pasteurization of liquid foods: Modeling and simulation of fluid mechanics, electric analysis, and heat transfer. Comput. Chem. Eng. 2017, 97, 183–193. [Google Scholar] [CrossRef]
- Halpin, R.M.; Duffy, L.; Cregenzán-Alberti, O.; Lyng, J.G.; Noci, F. The effect of non-thermal processing technologies on microbial inactivation: An investigation into sub-lethal injury of Escherichia coli and Pseudomonas fluorescens. Food Control 2014, 41, 106–115. [Google Scholar] [CrossRef]
- Roberts, P.B. Food irradiation is safe: Half a century of studies. Radiat. Phys. Chem. 2014, 105, 78–82. [Google Scholar] [CrossRef]
- Thirumdas, R.; Sarangapani, C.; Annapure, U.S. Cold plasma: A novel non-thermal technology for food processing. Food Biophys. 2015, 10, 1–11. [Google Scholar] [CrossRef]
- Davis, P.J.; Williams, S.C. Protein modification by thermal processing. Allergy 1998, 53, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Tong, P.; Gao, J.; Chen, H.; Li, X.; Zhang, Y.; Jian, S.; Wichers, H.; Wu, Z.; Yang, A.; Liu, F. Effect of heat treatment on the potential allergenicity and conformational structure of egg allergen ovotransferrin. Food Chem. 2012, 131, 603–610. [Google Scholar] [CrossRef]
- Murayama, K.; Tomida, M. Heat-induced secondary structure and conformation change of bovine serum albumin investigated by fourier transform infrared spectroscopy. Biochemistry 2004, 43, 11526–11532. [Google Scholar] [CrossRef] [PubMed]
- Somoza, V.; Wenzel, E.; Weiss, C.; Clawin-Radecker, I.; Grubel, N.; Erbersdobler, H.F. Dose-dependent utilisation of casein-linked lysinoalanine, N(epsilon)-fructoselysine and N(epsilon)-carboxymethyllysine in rats. Mol. Nutr. Food Res. 2006, 50, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Van Boekel, M.A. Kinetic aspects of the maillard reaction: A critical review. Mol. Nutr. Food Res. 2001, 45, 150–159. [Google Scholar]
- Hellwig, M.; Henle, T. Baking, ageing, diabetes: A short history of the maillard reaction. Angew. Chem. Int. Ed. 2014, 53, 10316–10329. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, F.C.; Coimbra, J.S.; de Oliveira, E.B.; Zuniga, A.D.; Rojas, E.E. Food protein-polysaccharide conjugates obtained via the maillard reaction: A review. Crit. Rev. Food Sci. Nutr. 2016, 56, 1108–1125. [Google Scholar] [CrossRef] [PubMed]
- Henle, T. Protein-bound advanced glycation endproducts (ages) as bioactive amino acid derivatives in foods. Amino Acids 2005, 29, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Somoza, V. Five years of research on health risks and benefits of maillard reaction products: An update. Mol. Nutr. Food Res. 2005, 49, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ru, Q.; Ding, Y. Glycation a promising method for food protein modification: Physicochemical properties and structure, a review. Food Res. Int. 2012, 49, 170–183. [Google Scholar] [CrossRef]
- Ames, J.M. Applications of the maillard reaction in the food industry. Food Chem. 1998, 62, 431–439. [Google Scholar] [CrossRef]
- Teodorowicz, M.; Jansen, A.P.H.; Roovers, M.H.W.M.; Ruinemans-Koerts, J.; Wichers, H.J.; Savelkoul, H.F.J. Maillard-type neoallergens present in processed soy extract may cause an allergic reaction in soy allergic patients. Clin. Trans. Allergy 2015, 5, 21. [Google Scholar] [CrossRef]
- Heilmann, M.; Wellner, A.; Gadermaier, G.; Ilchmann, A.; Briza, P.; Krause, M.; Nagai, R.; Burgdorf, S.; Scheurer, S.; Vieths, S.; et al. Ovalbumin modified with pyrraline, a maillard reaction product, shows enhanced T-cell immunogenicity. J. Biol. Chem. 2014, 289, 7919–7928. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, A.E.; Hillson, W.R.; Noti, M.; Gartlan, K.H.; Johnson, S.; Thomas, B.; Artis, D.; Sattentau, Q.J. Dry roasting enhances peanut-induced allergic sensitization across mucosal and cutaneous routes in mice. J. Allergy Clin. Immunol. 2014, 134, 1453–1456. [Google Scholar] [CrossRef] [PubMed]
- Iwan, M.; Vissers, Y.M.; Fiedorowicz, E.; Kostyra, H.; Kostyra, E.; Savelkoul, H.F.; Wichers, H.J. Impact of maillard reaction on immunoreactivity and allergenicity of the hazelnut allergen cor a 11. J.Agric. Food Chem. 2011, 59, 7163–7171. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, K.; Schweimer, K.; Reese, G.; Randow, S.; Suhr, M.; Becker, W.M.; Vieths, S.; Rosch, P. Structure and stability of 2s albumin-type peanut allergens: Implications for the severity of peanut allergic reactions. Biochem. J. 2006, 395, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Suhr, M.; Wicklein, D.; Lepp, U.; Becker, W.M. Isolation and characterization of natural ara h 6: Evidence for a further peanut allergen with putative clinical relevance based on resistance to pepsin digestion and heat. Mol. Nutr. Food Res. 2004, 48, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Apostolovic, D.; Stanic-Vucinic, D.; de Jongh, H.H.; de Jong, G.A.; Mihailovic, J.; Radosavljevic, J.; Radibratovic, M.; Nordlee, J.A.; Baumert, J.L.; Milcic, M.; et al. Conformational stability of digestion-resistant peptides of peanut conglutins reveals the molecular basis of their allergenicity. Sci. Rep. 2016, 6, 29249. [Google Scholar] [CrossRef] [PubMed]
- Bogh, K.L.; Madsen, C.B. Food allergens: Is there a correlation between stability to digestion and allergenicity? Crit. Rev. Food Sci. Nutr. 2016, 56, 1545–1567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bannon, G.; Fu, T.J.; Kimber, I.; Hinton, D.M. Protein digestibility and relevance to allergenicity. Environ. Health Perspect. 2003, 111, 1122–1124. [Google Scholar] [CrossRef] [PubMed]
- Peram, M.R.; Loveday, S.M.; Ye, A.; Singh, H. In vitro gastric digestion of heat-induced aggregates of β-lactoglobulin. J. Dairy Sci. 2013, 96, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Corzo-Martínez, M.; Soria, A.C.; Belloque, J.; Villamiel, M.; Moreno, F.J. Effect of glycation on the gastrointestinal digestibility and immunoreactivity of bovine β-lactoglobulin. Int. Dairy J. 2010, 20, 742–752. [Google Scholar] [CrossRef] [Green Version]
- Corzo-Martinez, M.; Moreno, F.J.; Olano, A.; Villamiel, M. Role of pyridoxamine in the formation of the amadori/heyns compounds and aggregates during the glycation of beta-lactoglobulin with galactose and tagatose. J. Agric. Food Chem. 2010, 58, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Shahab, U.; Baig, M.H.; Khan, M.S.; Khan, M.S.; Srivastava, A.K.; Saeed, M.; Moinuddin. Inhibitory effect of metformin and pyridoxamine in the formation of early, intermediate and advanced glycation end-products. PLoS ONE 2013, 8, e72128. [Google Scholar] [CrossRef] [PubMed]
- Dominika, Ś.; Arjan, N.; Karyn, R.P.; Henryk, K. The study on the impact of glycated pea proteins on human intestinal bacteria. Int. J. Food Microbiol. 2011, 145, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Teodorowicz, M.; Fiedorowicz, E.; Kostyra, H.; Wichers, H.; Kostyra, E. Effect of maillard reaction on biochemical properties of peanut 7s globulin (ara h 1) and its interaction with a human colon cancer cell line (caco-2). Eur. J. Nutr. 2013, 52, 1927–1938. [Google Scholar] [CrossRef] [PubMed]
- Luz Sanz, M.; Corzo-Martinez, M.; Rastall, R.A.; Olano, A.; Moreno, F.J. Characterization and in vitro digestibility of bovine beta-lactoglobulin glycated with galactooligosaccharides. J. Agric. Food Chem. 2007, 55, 7916–7925. [Google Scholar] [CrossRef] [PubMed]
- Katayama, S.; Shima, J.; Saeki, H. Solubility improvement of shellfish muscle proteins by reaction with glucose and its soluble state in low-ionic-strength medium. J. Agric. Food Chem. 2002, 50, 4327–4332. [Google Scholar] [CrossRef] [PubMed]
- Sato, R.; Sawabe, T.; Kishimura, H.; Hayashi, K.; Saeki, H. Preparation of neoglycoprotein from carp myofibrillar protein and alginate oligosaccharide: Improved solubility in low ionic strength medium. J. Agric. Food Chem. 2000, 48, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Yeboah, F.K.; Alli, I.; Yaylayan, V.A.; Yasuo, K.; Chowdhury, S.F.; Purisima, E.O. Effect of limited solid-state glycation on the conformation of lysozyme by esi-msms peptide mapping and molecular modeling. Bioconj. Chem. 2004, 15, 27–34. [Google Scholar] [CrossRef] [PubMed]
- De Jongh, H.H.J.; Taylor, S.L.; Koppelman, S.J. Controlling the aggregation propensity and thereby digestibility of allergens by maillardation as illustrated for cod fish parvalbumin. J. Biosci. Bioeng. 2011, 111, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Teodorowicz, M.; Wichers, H.J.; van Boekel, M.A.; Hettinga, K.A. Generation of soluble advanced glycation end products receptor (srage)-binding ligands during extensive heat treatment of whey protein/lactose mixtures is dependent on glycation and aggregation. J Agric. Food Chem. 2016, 64, 6477–6486. [Google Scholar] [CrossRef] [PubMed]
- Seiquer, I.; Diaz-Alguacil, J.; Delgado-Andrade, C.; Lopez-Frias, M.; Munoz Hoyos, A.; Galdo, G.; Navarro, M.P. Diets rich in maillard reaction products affect protein digestibility in adolescent males aged 11–14 y. Am. J. Clin. Nutr. 2006, 83, 1082–1088. [Google Scholar] [PubMed]
- Hellwig, M.; Bunzel, D.; Huch, M.; Franz, C.M.; Kulling, S.E.; Henle, T. Stability of individual maillard reaction products in the presence of the human colonic microbiota. J. Agric. Food Chem. 2015, 63, 6723–6730. [Google Scholar] [CrossRef] [PubMed]
- Hellwig, M.; Henle, T. Release of pyrraline in absorbable peptides during simulated digestion of casein glycated by 3-deoxyglucosone. Eur. Food Res. Technol. 2013, 237, 47–55. [Google Scholar] [CrossRef]
- Koschinsky, T.; He, C.-J.; Mitsuhashi, T.; Bucala, R.; Liu, C.; Buenting, C.; Heitmann, K.; Vlassara, H. Orally absorbed reactive glycation products (glycotoxins): An environmental risk factor in diabetic nephropathy. Proc. Natl. Acad. Sci. USA 1997, 94, 6474–6479. [Google Scholar] [CrossRef] [PubMed]
- Uribarri, J.; Cai, W.; Sandu, O.; Peppa, M.; Goldberg, T.; Vlassara, H. Diet-derived advanced glycation end products are major contributors to the body’s age pool and induce inflammation in healthy subjects. Ann. N. Y. Acad. Sci. 2005, 1043, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Andrade, C.; Tessier, F.J.; Niquet-Leridon, C.; Seiquer, I.; Pilar Navarro, M. Study of the urinary and faecal excretion of nepsilon-carboxymethyllysine in young human volunteers. Amino Acids 2012, 43, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Foerster, A.; Henle, T. Glycation in food and metabolic transit of dietary ages (advanced glycation end-products): Studies on the urinary excretion of pyrraline. Biochem. Soc. Trans. 2003, 31, 1383–1385. [Google Scholar] [CrossRef] [PubMed]
- Forster, A.; Kuhne, Y.; Henle, T. Studies on absorption and elimination of dietary maillard reaction products. Ann. N. Y. Acad. Sci. 2005, 1043, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Semba, R.D.; Ang, A.; Talegawkar, S.; Crasto, C.; Dalal, M.; Jardack, P.; Traber, M.G.; Ferrucci, L.; Arab, L. Dietary intake associated with serum versus urinary carboxymethyl-lysine, a major advanced glycation end product, in adults: The energetics study. Eur. J. Clin. Nutr. 2012, 66, 3–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hellwig, M.; Matthes, R.; Peto, A.; Löbner, J.; Henle, T. N-ε-fructosyllysine and n-ε-carboxymethyllysine, but not lysinoalanine, are available for absorption after simulated gastrointestinal digestion. Amino Acids 2014, 46, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Ames, J.M. Evidence against dietary advanced glycation endproducts being a risk to human health. Mol. Nutr. Food Res. 2007, 51, 1085–1090. [Google Scholar] [CrossRef] [PubMed]
- Roth-Walter, F.; Berin, M.C.; Arnaboldi, P.; Escalante, C.R.; Dahan, S.; Rauch, J.; Jensen-Jarolim, E.; Mayer, L. Pasteurization of milk proteins promotes allergic sensitization by enhancing uptake through peyer’s patches. Allergy 2008, 63, 882–890. [Google Scholar] [CrossRef] [PubMed]
- Stojadinovic, M.; Pieters, R.; Smit, J.; Velickovic, T.C. Cross-linking of beta-lactoglobulin enhances allergic sensitization through changes in cellular uptake and processing. Toxicol. Sci. 2014, 140, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Tuohy, K.M.; Hinton, D.J.; Davies, S.J.; Crabbe, M.J.; Gibson, G.R.; Ames, J.M. Metabolism of maillard reaction products by the human gut microbiota—implications for health. Mol. Nutr. Food Res. 2006, 50, 847–857. [Google Scholar] [CrossRef] [PubMed]
- Helou, C.; Marier, D.; Jacolot, P.; Abdennebi-Najar, L.; Niquet-Leridon, C.; Tessier, F.J.; Gadonna-Widehem, P. Microorganisms and maillard reaction products: A review of the literature and recent findings. Amino Acids 2014, 46, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Seiquer, I.; Rubio, L.A.; Peinado, M.J.; Delgado-Andrade, C.; Navarro, M.P. Maillard reaction products modulate gut microbiota composition in adolescents. Mol. Nutr. Food Res. 2014, 58, 1552–1560. [Google Scholar] [CrossRef] [PubMed]
- Teodorowicz, M.; Świątecka, D.; Savelkoul, H.; Wichers, H.; Kostyra, E.C. Hydrolysates of glycated and heat-treated peanut 7s globulin (ara h 1) modulate human gut microbial proliferation, survival and adhesion. J. Appl. Microbiol. 2014, 116, 424–434. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.F.; Ramasamy, R.; Schmidt, A.M. Mechanisms of disease: Advanced glycation end-products and their receptor in inflammation and diabetes complications. Nat. Rev. Endocrinol. 2008, 4, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.E.; Prasad, C.; Vijayagopal, P.; Juma, S.; Imrhan, V. Advanced glycation end products, inflammation, and chronic metabolic diseases: Links in a chain? Crit. Rev. Food Sci. Nutr. 2016, 56, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.C.; Zarrin, A.A. The production and regulation of ige by the immune system. Nat. Rev. Immunol. 2014, 14, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Guilliams, M.; Bruhns, P.; Saeys, Y.; Hammad, H.; Lambrecht, B.N. The function of Fc[gamma] receptors in dendritic cells and macrophages. Nat. Rev. Immunol. 2014, 14, 94–108. [Google Scholar] [CrossRef] [PubMed]
- Bertheloot, D.; Naumovski, A.L.; Langhoff, P.; Horvath, G.L.; Jin, T.; Xiao, T.S.; Garbi, N.; Agrawal, S.; Kolbeck, R.; Latz, E. Rage enhances tlr responses through binding and internalization of rna. J. Immunol. 2016, 197, 4118–4126. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.M.; Vianna, M.; Gerlach, M.; Brett, J.; Ryan, J.; Kao, J.; Esposito, C.; Hegarty, H.; Hurley, W.; Clauss, M.; et al. Isolation and characterization of two binding proteins for advanced glycosylation end products from bovine lung which are present on the endothelial cell surface. J. Biol. Chem. 1992, 267, 14987–14997. [Google Scholar] [PubMed]
- Maldonado, S.; Dai, J.; Singh, S.; Mwangi, D.; Rivera, A.; Fitzgerald-Bocarsly, P. Human pdcs express the c-type lectin receptor dectin-1 and uptake and kill Aspergillus fumigatus spores in vitro (mpf4p.734). J. Immunol. 2015, 194, 136.10. [Google Scholar]
- Zhu, L.L.; Zhao, X.Q.; Jiang, C.; You, Y.; Chen, X.P.; Jiang, Y.Y.; Jia, X.M.; Lin, X. C-type lectin receptors dectin-3 and dectin-2 form a heterodimeric pattern-recognition receptor for host defense against fungal infection. Immunity 2013, 39, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Sprokholt, J.K.; Overmars, R.J.; Geijtenbeek, T.B.H. Dc-sign in infection and immunity. In C-type Lectin Receptors in Immunity; Yamasaki, S., Ed.; Springer: Tokyo, Japan, 2016; pp. 129–150. [Google Scholar]
- Lakshminarayan, R.; Wunder, C.; Becken, U.; Howes, M.T.; Benzing, C.; Arumugam, S.; Sales, S.; Ariotti, N.; Chambon, V.; Lamaze, C.; et al. Galectin-3 drives glycosphingolipid-dependent biogenesis of clathrin-independent carriers. Nat. Cell Biol. 2014, 16, 592–603. [Google Scholar] [CrossRef] [PubMed]
- Azad, A.K.; Rajaram, M.V.S.; Schlesinger, L.S. Exploitation of the macrophage mannose receptor (cd206) in infectious disease diagnostics and therapeutics. J. Cytol. Mol. Biol. 2014, 1, 1000003. [Google Scholar] [PubMed]
- Gosselin, E.J.; Wardwell, K.; Gosselin, D.R.; Alter, N.; Fisher, J.L.; Guyre, P.M. Enhanced antigen presentation using human fc gamma receptor (monocyte/macrophage)-specific immunogens. J. Immunol. 1992, 149, 3477–3481. [Google Scholar] [PubMed]
- Van der Heijden, F.L.; Joost van Neerven, R.J.; van Katwijk, M.; Bos, J.D.; Kapsenberg, M.L. Serum-ige-facilitated allergen presentation in atopic disease. J. Immunol. 1993, 150, 3643. [Google Scholar] [PubMed]
- Buttari, B.; Profumo, E.; Capozzi, A.; Facchiano, F.; Saso, L.; Sorice, M.; Rigano, R. Advanced glycation end products of human beta(2) glycoprotein i modulate the maturation and function of dcs. Blood 2011, 117, 6152–6161. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.; Jia, Q.; Liang, C.; Luo, Y.; Huang, D.; Sun, A.; Wang, K.; Zou, Y.; Chen, H. Advanced glycosylation end products might promote atherosclerosis through inducing the immune maturation of dendritic cells. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 2157–2163. [Google Scholar] [CrossRef] [PubMed]
- Ilchmann, A.; Burgdorf, S.; Scheurer, S.; Waibler, Z.; Nagai, R.; Wellner, A.; Yamamoto, Y.; Yamamoto, H.; Henle, T.; Kurts, C.; et al. Glycation of a food allergen by the maillard reaction enhances its t-cell immunogenicity: Role of macrophage scavenger receptor class a type i and ii. J. Allergy Clin. Immunol. 2010, 125, 175–183.e111. [Google Scholar] [CrossRef] [PubMed]
- Ott, C.; Jacobs, K.; Haucke, E.; Navarrete Santos, A.; Grune, T.; Simm, A. Role of advanced glycation end products in cellular signaling. Redox Biol. 2014, 2, 411–429. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.M.; Mitsuhashi, T.; Wojciechowicz, D.; Shimizu, N.; Li, J.; Stitt, A.; He, C.; Banerjee, D.; Vlassara, H. Molecular identity and cellular distribution of advanced glycation endproduct receptors: Relationship of p60 to ost-48 and p90 to 80k-h membrane proteins. Proc. Natl. Acad. Sci. USA 1996, 93, 11047–11052. [Google Scholar] [CrossRef] [PubMed]
- Vlassara, H.; Li, Y.M.; Imani, F.; Wojciechowicz, D.; Yang, Z.; Liu, F.T.; Cerami, A. Identification of galectin-3 as a high-affinity binding protein for advanced glycation end products (age): A new member of the age-receptor complex. Mol. Med. 1995, 1, 634–646. [Google Scholar] [PubMed]
- Ohgami, N.; Nagai, R.; Ikemoto, M.; Arai, H.; Kuniyasu, A.; Horiuchi, S.; Nakayama, H. Cd36, a member of the class b scavenger receptor family, as a receptor for advanced glycation end products. J. Biol. Chem. 2001, 276, 3195–3202. [Google Scholar] [CrossRef] [PubMed]
- Chatzigeorgiou, A.; Kandaraki, E.; Piperi, C.; Livadas, S.; Papavassiliou, A.G.; Koutsilieris, M.; Papalois, A.; Diamanti-Kandarakis, E. Dietary glycotoxins affect scavenger receptor expression and the hormonal profile of female rats. J. Endocrinol. 2013, 218, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Araki, N.; Higashi, T.; Mori, T.; Shibayama, R.; Kawabe, Y.; Kodama, T.; Takahashi, K.; Shichiri, M.; Horiuchi, S. Macrophage scavenger receptor mediates the endocytic uptake and degradation of advanced glycation end products of the maillard reaction. Eur. J. Biochem. 1995, 230, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, P.; Ghosh, D.; Basu, M.K. Modulation of macrophage mannose receptor affects the uptake of virulent and avirulent leishmania donovani promastigotes. J. Parasitol. 2001, 87, 1023–1027. [Google Scholar] [CrossRef]
- Hilmenyuk, T.; Bellinghausen, I.; Heydenreich, B.; Ilchmann, A.; Toda, M.; Grabbe, S.; Saloga, J. Effects of glycation of the model food allergen ovalbumin on antigen uptake and presentation by human dendritic cells. Immunology 2010, 129, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Royer, P.J.; Emara, M.; Yang, C.; Al-Ghouleh, A.; Tighe, P.; Jones, N.; Sewell, H.F.; Shakib, F.; Martinez-Pomares, L.; Ghaemmaghami, A.M. The mannose receptor mediates the uptake of diverse native allergens by dendritic cells and determines allergen-induced t cell polarization through modulation of ido activity. J. Immunol. 2010, 185, 1522–1531. [Google Scholar] [CrossRef] [PubMed]
- Mueller, G.A.; Maleki, S.J.; Johnson, K.; Hurlburt, B.K.; Cheng, H.; Ruan, S.; Nesbit, J.B.; Pomés, A.; Edwards, L.L.; Schorzman, A.; et al. Identification of maillard reaction products on peanut allergens that influence binding to the receptor for advanced glycation end products. Allergy 2013, 68, 1546–1554. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Teodorowicz, M.; van Boekel, M.A.; Wichers, H.J.; Hettinga, K.A. The decrease in the igg-binding capacity of intensively dry heated whey proteins is associated with intense maillard reaction, structural changes of the proteins and formation of rage-ligands. Food Funct. 2016, 7, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Holik, A.K.; Rohm, B.; Somoza, M.M.; Somoza, V. N(epsilon)-carboxymethyllysine (cml), a maillard reaction product, stimulates serotonin release and activates the receptor for advanced glycation end products (rage) in sh-sy5y cells. Food Funct. 2013, 4, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Neeper, M.; Schmidt, A.M.; Brett, J.; Yan, S.D.; Wang, F.; Pan, Y.C.; Elliston, K.; Stern, D.; Shaw, A. Cloning and expression of a cell surface receptor for advanced glycosylation end products of proteins. J. Biol. Chem. 1992, 267, 14998–15004. [Google Scholar] [PubMed]
- Bastos, D.H.M.; Gugliucci, A. Contemporary and controversial aspects of the maillard reaction products. Curr. Opin. Food Sci. 2015, 1, 13–20. [Google Scholar] [CrossRef]
- Devangelio, E.; Santilli, F.; Formoso, G.; Ferroni, P.; Bucciarelli, L.; Michetti, N.; Clissa, C.; Ciabattoni, G.; Consoli, A.; Davì, G. Soluble rage in type 2 diabetes: Association with oxidative stress. Free. Radic. Biol. Med. 2007, 43, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Raucci, A.; Cugusi, S.; Antonelli, A.; Barabino, S.M.; Monti, L.; Bierhaus, A.; Reiss, K.; Saftig, P.; Bianchi, M.E. A soluble form of the receptor for advanced glycation endproducts (rage) is produced by proteolytic cleavage of the membrane-bound form by the sheddase a disintegrin and metalloprotease 10 (adam10). FASEB J. 2008, 22, 3716–3727. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, S.-I.; Nakamura, N.; Suematsu, M.; Kaseda, K.; Matsui, T. Advanced glycation end products: A molecular target for vascular complications in diabetes. Mol. Med. 2015, 21, S32–S40. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, S.; Nakamura, K.; Matsui, T.; Ueda, S.; Fukami, K.; Okuda, S. Agents that block advanced glycation end product (age)-rage (receptor for ages)-oxidative stress system: A novel therapeutic strategy for diabetic vascular complications. Exp. Opin. Investig. Drugs 2008, 17, 983–996. [Google Scholar] [CrossRef] [PubMed]
- Gugliucci, A.; Menini, T. The axis age-rage-soluble rage and oxidative stress in chronic kidney disease. In Oxidative Stress and Inflammation in Non-Communicable Diseases—Molecular Mechanisms and Perspectives in Therapeutics; Camps, J., Ed.; Springer International Publishing: Cham, Germany, 2014; pp. 191–208. [Google Scholar]
- Reddy, V.P.; Beyaz, A. Inhibitors of the maillard reaction and age breakers as therapeutics for multiple diseases. Drug Discov. Today 2006, 11, 646–654. [Google Scholar] [CrossRef] [PubMed]
- Maillard-Lefebvre, H.; Boulanger, E.; Daroux, M.; Gaxatte, C.; Hudson, B.I.; Lambert, M. Soluble receptor for advanced glycation end products: A new biomarker in diagnosis and prognosis of chronic inflammatory diseases. Rheumatology 2009, 48, 1190–1196. [Google Scholar] [CrossRef] [PubMed]
- Zill, H.; Bek, S.; Hofmann, T.; Huber, J.; Frank, O.; Lindenmeier, M.; Weigle, B.; Erbersdobler, H.F.; Scheidler, S.; Busch, A.E.; et al. Rage-mediated mapk activation by food-derived age and non-age products. Biochem. Biophys. Res. Commun. 2003, 300, 311–315. [Google Scholar] [CrossRef]
- Hou, F.F.; Ren, H.; Owen, W.F., Jr.; Guo, Z.J.; Chen, P.Y.; Schmidt, A.M.; Miyata, T.; Zhang, X. Enhanced expression of receptor for advanced glycation end products in chronic kidney disease. J. Am. Soc. Nephrol. JASN 2004, 15, 1889–1896. [Google Scholar] [CrossRef] [PubMed]
- Kislinger, T.; Fu, C.; Huber, B.; Qu, W.; Taguchi, A.; Du Yan, S.; Hofmann, M.; Yan, S.F.; Pischetsrieder, M.; Stern, D.; et al. N(epsilon)-(carboxymethyl)lysine adducts of proteins are ligands for receptor for advanced glycation end products that activate cell signaling pathways and modulate gene expression. J. Biol. Chem. 1999, 274, 31740–31749. [Google Scholar] [CrossRef] [PubMed]
- Buetler, T.M.; Leclerc, E.; Baumeyer, A.; Latado, H.; Newell, J.; Adolfsson, O.; Parisod, V.; Richoz, J.; Maurer, S.; Foata, F.; et al. Nε-carboxymethyllysine-modified proteins are unable to bind to rage and activate an inflammatory response. Mol. Nutr. Food Res. 2008, 52, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Buetler, T.M.; Latado, H.; Leclerc, E.; Weigle, B.; Baumeyer, A.; Heizmann, C.W.; Scholz, G. Glycolaldehyde-modified β-lactoglobulin ages are unable to stimulate inflammatory signaling pathways in rage-expressing human cell lines. Mol. Nutr. Food Res. 2011, 55, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Muscat, S.; Pelka, J.; Hegele, J.; Weigle, B.; Münch, G.; Pischetsrieder, M. Coffee and maillard products activate nf-κb in macrophages via H2O2 production. Mol. Nutr. Food Res. 2007, 51, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Cellmer, T.; Bratko, D.; Prausnitz, J.M.; Blanch, H.W. Protein aggregation in silico. Trends Biotechnol. 2007, 25, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-J.; Wang, P.-W.; Yang, I.H.; Wu, C.-L.; Chuang, J.-H. Amyloid-beta mediates the receptor of advanced glycation end product-induced pro-inflammatory response via toll-like receptor 4 signaling pathway in retinal ganglion cell line rgc-5. Int. J. Biochem. Cell Biol. 2015, 64, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sadigh-Eteghad, S.; Sabermarouf, B.; Majdi, A.; Talebi, M.; Farhoudi, M.; Mahmoudi, J. Amyloid-beta: A crucial factor in Alzheimer’s disease. Med. Princ. Pract. 2015, 24, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Cianferoni, A.; Spergel, J.M. Food allergy: Review, classification and diagnosis. Allergol. Int. 2009, 58, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Rahaman, T.; Vasiljevic, T.; Ramchandran, L. Conformational changes of beta-lactoglobulin induced by shear, heat, and ph-effects on antigenicity. J. Dairy Sci. 2015, 98, 4255–4265. [Google Scholar] [CrossRef] [PubMed]
- Scheurer, S.; Lauer, I.; Foetisch, K.; San Miguel Moncin, M.; Retzek, M.; Hartz, C.; Enrique, E.; Lidholm, J.; Cistero-Bahima, A.; Vieths, S. Strong allergenicity of pru av 3, the lipid transfer protein from cherry, is related to high stability against thermal processing and digestion. J. Allergy Clin. Immunol. 2004, 114, 900–907. [Google Scholar] [CrossRef] [PubMed]
- Gruber, P.; Vieths, S.; Wangorsch, A.; Nerkamp, J.; Hofmann, T. Maillard reaction and enzymatic browning Aaffect the allergenicity of pru av 1, the major allergen from cherry (Prunus avium). J. Agric. Food Chem. 2004, 52, 4002–4007. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Saiz, R.; Belloque, J.; Molina, E.; Lopez-Fandino, R. Human immunoglobulin e (ige) binding to heated and glycated ovalbumin and ovomucoid before and after in vitro digestion. J. Agric. Food Chem. 2011, 59, 10044–10051. [Google Scholar] [CrossRef] [PubMed]
- Taheri-Kafrani, A.; Gaudin, J.C.; Rabesona, H.; Nioi, C.; Agarwal, D.; Drouet, M.; Chobert, J.M.; Bordbar, A.K.; Haertle, T. Effects of heating and glycation of beta-lactoglobulin on its recognition by ige of sera from cow milk allergy patients. J. Agric. Food Chem. 2009, 57, 4974–4982. [Google Scholar] [CrossRef] [PubMed]
- Vissers, Y.M.; Blanc, F.; Skov, P.S.; Johnson, P.E.; Rigby, N.M.; Przybylski-Nicaise, L.; Bernard, H.; Wal, J.-M.; Ballmer-Weber, B.; Zuidmeer-Jongejan, L.; et al. Effect of heating and glycation on the allergenicity of 2s albumins (ara h 2/6) from peanut. PLoS ONE 2011, 6, e23998. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vissers, Y.M.; Iwan, M.; Adel-Patient, K.; Stahl Skov, P.; Rigby, N.M.; Johnson, P.E.; Mandrup Muller, P.; Przybylski-Nicaise, L.; Schaap, M.; Ruinemans-Koerts, J.; et al. Effect of roasting on the allergenicity of major peanut allergens ara h 1 and ara h 2/6: The necessity of degranulation assays. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2011, 41, 1631–1642. [Google Scholar] [CrossRef] [PubMed]
- Cucu, T.; De Meulenaer, B.; Bridts, C.; Devreese, B.; Ebo, D. Impact of thermal processing and the maillard reaction on the basophil activation of hazelnut allergic patients. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2012, 50, 1722–1728. [Google Scholar] [CrossRef] [PubMed]
- Maleki, S.J.; Chung, S.-Y.; Champagne, E.T.; Raufman, J.-P. The effects of roasting on the allergenic properties of peanut proteins. J. Allergy Clin. Immunol. 2000, 106, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Gruber, P.; Becker, W.M.; Hofmann, T. Influence of the maillard reaction on the allergenicity of rara h 2, a recombinant major allergen from peanut (Arachis hypogaea), its major epitopes, and peanut agglutinin. J. Agric. Food Chem. 2005, 53, 2289–2296. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, A.; Watanabe, K.; Ojima, T.; Ahn, D.H.; Saeki, H. Effect of maillard reaction on allergenicity of scallop tropomyosin. J. Agric. Food Chem. 2005, 53, 7559–7564. [Google Scholar] [CrossRef] [PubMed]
- Toda, M.; Heilmann, M.; Ilchmann, A.; Vieths, S. The maillard reaction and food allergies: Is there a link? Clin. Chem. Lab. Med. CCLM/FESCC 2014, 52, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Gupta, K.; Sharma, A.; Das, M.; Ansari, I.A.; Dwivedi, P.D. Maillard reaction in food allergy: Pros and cons. Crit. Rev. Food Sci. Nutr. 2016, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Vojdani, A. Detection of ige, igg, iga and igm antibodies against raw and processed food antigens. Nutr. Metab. 2009, 6, 22. [Google Scholar] [CrossRef] [PubMed]
- Sampson, H.A.; Ho, D.G. Relationship between food-specific ige concentrations and the risk of positive food challenges in children and adolescents. J. Allergy Clin. Immunol. 1997, 100, 444–451. [Google Scholar] [CrossRef]
- Huby, R.D.; Dearman, R.J.; Kimber, I. Why are some proteins allergens? Toxicol. Sci. 2000, 55, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Aalberse, R.C. Structural biology of allergens. J. Allergy Clin. Immunol. 2000, 106, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Bredehorst, R.; David, K. What establishes a protein as an allergen? J. Chromatogr. B Biomed. Sci. Appl. 2001, 756, 33–40. [Google Scholar] [CrossRef]
- James, J.A.; Harley, J.B. B-cell epitope spreading in autoimmunity. Immunol. Rev. 1998, 164, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Dall’Antonia, F.; Pavkov-Keller, T.; Zangger, K.; Keller, W. Structure of allergens and structure based epitope predictions. Methods 2014, 66, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Pomes, A.; Chruszcz, M.; Gustchina, A.; Wlodawer, A. Interfaces between allergen structure and diagnosis: Know your epitopes. Curr. Allergy Asthma Rep. 2015, 15, 506. [Google Scholar] [CrossRef] [PubMed]
- Handlogten, M.W.; Kiziltepe, T.; Serezani, A.P.; Kaplan, M.H.; Bilgicer, B. Inhibition of weak-affinity epitope-ige interactions prevents mast cell degranulation. Nat. Chem. Biol. 2013, 9, 789–795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baenziger, J.; Kornfeld, S.; Kochwa, S. Structure of the carbohydrate units of ige immunoglobulin. I. Over-all composition, glycopeptide isolation, and structure of the high mannose oligosaccharide unit. J. Biol. Chem. 1974, 249, 1889–1896. [Google Scholar] [PubMed]
- Gould, H.J.; Sutton, B.J. Ige in allergy and asthma today. Nat. Rev. Immunol. 2008, 8, 205–217. [Google Scholar] [CrossRef] [PubMed]
- El-Khouly, F.; Lewis, S.A.; Pons, L.; Burks, A.W.; Hourihane, J.O.B. Igg and ige avidity characteristics of peanut allergic individuals. Pediatr. Allergy Immunol. 2007, 18, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Lin, J.; Bardina, L.; Goldis, M.; Nowak-Węgrzyn, A.; Shreffler, W.G.; Sampson, H.A. Correlation of ige/igg4 milk epitopes and affinity of milk-specific ige antibodies with different phenotypes of clinical milk allergy. J. Allergy Clin. Immunol. 2010, 125, 695–702.e6. [Google Scholar] [CrossRef] [PubMed]
- Gould, H.J.; Sutton, B.J.; Beavil, A.J.; Beavil, R.L.; McCloskey, N.; Coker, H.A.; Fear, D.; Smurthwaite, L. The biology of ige and the basis of allergic disease. Ann. Rev. Immunol. 2003, 21, 579–628. [Google Scholar] [CrossRef] [PubMed]
- Knol, E.F. Requirements for effective ige cross-linking on mast cells and basophils. Mol. Nutr. Food Res. 2006, 50, 620–624. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teodorowicz, M.; Van Neerven, J.; Savelkoul, H. Food Processing: The Influence of the Maillard Reaction on Immunogenicity and Allergenicity of Food Proteins. Nutrients 2017, 9, 835. https://doi.org/10.3390/nu9080835
Teodorowicz M, Van Neerven J, Savelkoul H. Food Processing: The Influence of the Maillard Reaction on Immunogenicity and Allergenicity of Food Proteins. Nutrients. 2017; 9(8):835. https://doi.org/10.3390/nu9080835
Chicago/Turabian StyleTeodorowicz, Malgorzata, Joost Van Neerven, and Huub Savelkoul. 2017. "Food Processing: The Influence of the Maillard Reaction on Immunogenicity and Allergenicity of Food Proteins" Nutrients 9, no. 8: 835. https://doi.org/10.3390/nu9080835




