Energy and Protein in Critically Ill Patients with AKI: A Prospective, Multicenter Observational Study Using Indirect Calorimetry and Protein Catabolic Rate
Abstract
1. Introduction
- (a)
- to evaluate the validity of conventional predictive formulas and equations for the calculation of energy expenditure and protein needs, by using IC and the nPCR as gold standards;
- (b)
- to compare prescribed and actually received nutrients with estimated and measured needs.
2. Materials and Methods
2.1. Patients
2.2. Data Collection and Calculations
2.3. Indirect Calorimetry Instrumentation and Measurements
2.4. Artificial Nutrition
2.5. Reference Body Weight
2.6. Statistical Analysis
3. Results
3.1. Nutrition
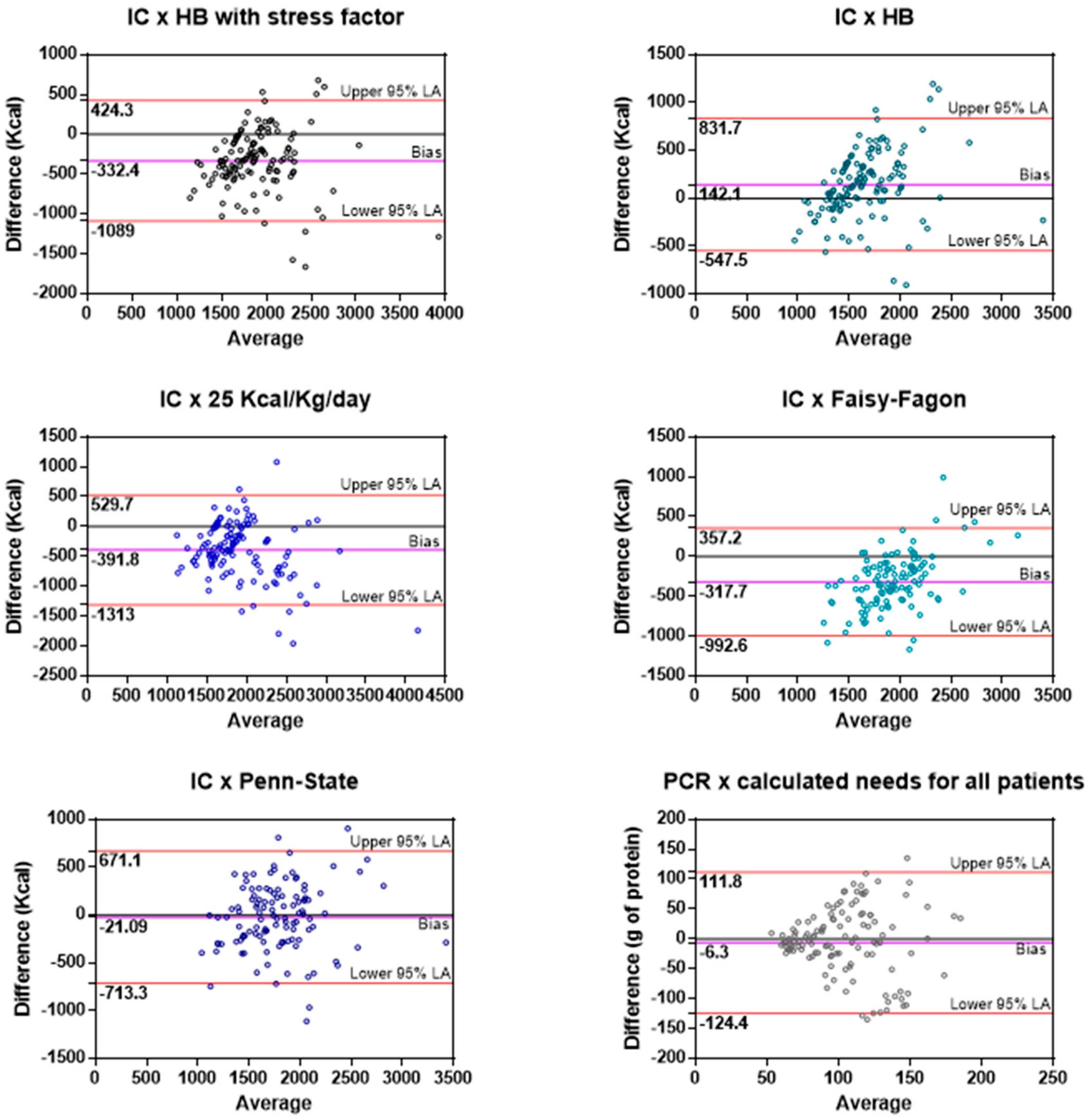
3.1.1. Indirect Calorimetry and Predictive Equations
3.1.2. Protein Catabolic Rate and Recommended Formulas
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, P.K.; Burdmann, E.A.; Mehta, R.L. Acute kidney injury: Global health alert. Kidney Int. 2013, 83, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Lameire, N.H.; Bagga, A.; Cruz, D.; de Maeseneer, J.; Endre, Z.; Kellum, J.A.; Liu, K.D.; Mehta, R.L.; Pannu, N.; Biesen, W.V.; et al. Acute kidney injury: An increasing global concern. Lancet 2013, 382, 170–179. [Google Scholar] [CrossRef]
- Susantitaphong, T.; Cruz, D.N.; Cerda, J.; Abulfaraj, M.; Alqahtani, F.; Koulouridis, I.; Jaber, B.L. World incidence of AKI: A meta-analysis. Clin. J. Am. Soc. Nephrol. 2013, 8, 1482–1493. [Google Scholar] [CrossRef] [PubMed]
- Uchino, S.; Bellomo, R.; Morimatsu, H.; Morgera, S.; Schetz, M.; Tan, I.; Bouman, C.; Macedo, E.; Gibney, N.; Tolwani, A.; et al. Continuous renal replacement therapy: A worldwide practice survey. The beginning and ending supportive therapy for the kidney (B.E.S.T. kidney) investigators. Intensive Care Med. 2007, 33, 1563–1570. [Google Scholar] [CrossRef] [PubMed]
- Fiaccadori, E.; Lombardi, M.; Leonardi, S.; Rotelli, C.F.; Tortorella, G.; Borghetti, A. Prevalence and clinical outcome associated with preexisting malnutrition in acute renal failure: A prospective cohort study. J. Am. Soc. Nephrol. 1999, 10, 581–593. [Google Scholar] [PubMed]
- Fiaccadori, E.; Regolisti, G.; Maggiore, U. Specialized nutritional support interventions in critically ill patients on renal replacement therapy. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Fiaccadori, E.; Sabatino, A.; Morabito, S.; Bozzoli, L.; Donadio, C.; Maggiore, U.; Regolisti, G. Hyper/Hypoglycemia and acute kidney injury in critically ill patients. Clin. Nutr. 2016, 35, 317–321. [Google Scholar] [CrossRef] [PubMed]
- Bellomo, R.; Cass, A.; Cole, L.; Finfer, S.; Gallagher, M.; Lee, J.; Lo, S.; McArthur, C.; McGuinness, S.; Myburgh, J.; et al. Calorie intake and patient outcomes in severe acute kidney injury: Findings from the randomized evaluation of normal vs. augmented level of replacement therapy (RENAL) study trial. Crit. Care 2014, 18, R45. [Google Scholar] [CrossRef] [PubMed]
- Oshima, T.; Berger, M.M.; de Waele, E.; Guttormsen, A.B.; Heidegger, C.-P.; Hiesmayr, M.; Singer, P.; Wernerman, J.; Pichard, C. Indirect calorimetry in nutritional therapy A position paper by the ICALIC study group. Clin. Nutr. 2017, 36, 651–662. [Google Scholar] [CrossRef] [PubMed]
- Fiaccadori, E.; Maggiore, U.; Rotelli, C.; Giacosa, R.; Picetti, E.; Parenti, E.; Meschi, T.; Borghi, L.; Tagliavini, D.; Cabassi, A. Effects of energy intakes on nitrogen balance in patients with acute renal failure: A pilot study. Nephrol. Dial. Transpl. 2005, 20, 1976–1980. [Google Scholar] [CrossRef] [PubMed]
- Kross, E.K.; Sena, M.; Schmidt, K.; Stapleton, R.D. A comparison of predictive equations of energy expenditure and measured energy expenditure in critically ill patients. J. Crit. Care 2012, 27, e5–e12. [Google Scholar] [CrossRef] [PubMed]
- Faisy, C.; Guerot, E.; Diehl, J.L.; Labrousse, J.; Fagon, J.Y. Assessment of resting energy expenditure in mechanically ventilated patients. Am. J. Clin. Nutr. 2003, 78, 241–249. [Google Scholar] [PubMed]
- Frankenfield, D.; Hise, M.; Malone, A.; Russell, M.; Gradwell, E.; Compher, C. Prediction of resting metabolic rate in critically ill adult patients: Results of a systematic review of the evidence. J. Am. Diet Assoc. 2007, 107, 1552–1561. [Google Scholar] [CrossRef] [PubMed]
- Frankenfield, D.C.; Coleman, A.; Alam, S.; Cooney, R.N. Analysis of estimation methods for resting metabolic rate in critically ill adults. JPEN J. Parenter. Enter. Nutr. 2009, 33, 27–36. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, A.; Hildebrandt, L. Comparison of formulaic equations to determine energy expenditure in the critically ill patient. Nutrition 2003, 19, 233–239. [Google Scholar] [CrossRef]
- De Góes, C.R.; Berbel-Bufarah, M.N.; Sanches, A.C.; Xavier, P.S.; Balbi, A.L.; Ponce, S. Poor agreement between predictive equations of energy expenditure and measured energy expenditure in critically ill acute kidney injury patients. Ann. Nutr. Metab. 2016, 68, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Chima, C.S.; Meyer, L.; Hummel, A.C.; Bosworth, C.; Heyka, R.; Paganini, E.P.; Werynski, A. Protein catabolic rate in patients with acute renal failure on continuous arteriovenous hemofiltration and total parenteral nutrition. J. Am. Soc. Nephrol. 1993, 3, 1516–1521. [Google Scholar] [PubMed]
- Macias, W.L.; Alaka, K.J.; Murphy, M.H.; Miller, M.E.; Clark, W.R.; Mueller, B.A. Impact of the nutritional regimen on protein catabolism and nitrogen balance in patients with acute renal failure. JPEN J. Parenter. Enter. Nutr. 1996, 20, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Leblanc, M.; Garred, L.J.; Cardinal, J.; Pichette, V.; Nolin, L.; Ouimet, D.; Geadah, D. Catabolism in critical illness: Estimation from urea nitrogen appearance and creatinine production during continuous renal replacement therapy. Am. J. Kidney Dis. 1998, 32, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Marshall, M.R.; Golper, T.A.; Shaver, M.J.; Alam, M.G.; Chatoth, D.K. Urea kinetics during sustained low-efficiency dialysis in critically ill patients requiring renal replacement therapy. Am. J. Kidney Dis. 2002, 39, 556–570. [Google Scholar] [CrossRef] [PubMed]
- Kritmetapak, K.; Peerapornratana, S.; Srisawat, N.; Somlaw, N.; Lakananurak, N.; Dissayabutra, T.; Phonork, C.; Leelahavanichkul, A.; Tiranathanagul, K.; Susantithapong, P.; et al. The impact pf macro-and micronutrients on predicting outcomes of critically ill patients requiring continuous renal replacement therapy. PLoS ONE 2016, 11, e0156634. [Google Scholar] [CrossRef] [PubMed]
- Khwaja, A. KDIGO Clinical practice guidelines for acute kidney injury. Kidney Int. 2012, 2, 1–138. [Google Scholar] [CrossRef] [PubMed]
- Cano, N.; Fiaccadori, E.; Tesinsky, P.; Toigo, G.; Druml, W.; Kuhlmann, M.; Mann, H.; Hörl, W.H. ESPEN guidelines on enteral nutrition: Adult renal failure. Clin. Nutr. 2006, 25, 295–310. [Google Scholar] [CrossRef] [PubMed]
- Cano, N.; Aparicio, M.; Brunori, G.; Carrero, J.J.; Cianciaruso, B.; Fiaccadori, E.; Lindholm, B.; Teplan, V.; Fouque, D.; Guarnieri, G. ESPEN guidelines on parenteral nutrition: Adult renal failure. Clin. Nutr. 2009, 28, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.A.; Benedict, F.G. A biometric study of human basal metabolism. Proc. Natl. Acad. Sci. USA 1918, 4, 370–373. [Google Scholar] [CrossRef] [PubMed]
- Garred, L.J. Dialysate-based kinetic modeling. Adv. Ren. Replace. Ther. 1995, 2, 305–318. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Boullata, J.; Williams, J.; Cottrell, F.; Hudson, L.; Compher, C. Accurate determination of energy needs in hospitalized patients. J. Am. Diet Assoc. 2007, 107, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Reid, C.L. Poor agreement between continuous measurements of energy expenditure and routinely used prediction equations in intensive care unit patients. Clin. Nutr. 2007, 26, 649–657. [Google Scholar] [CrossRef] [PubMed]
- Robins, K.; Stankorb, S.M.; Salgueiro, M. Energy expenditure in acute posttraumatic amputation: Comparison of four methods for assessment. Nutr. Clin. Pract. 2013, 28, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Long, C.L.; Schaffel, N.; Geiger, J.W.; Schiller, W.R.; Blackmore, W.S. Metabolic response to injury and illness: Estimation of energy and protein needs from indirect calorimetry and nitrogen balance. JPEN J. Parenter. Enter. Nutr. 1979, 3, 452–456. [Google Scholar] [CrossRef] [PubMed]
- Casati, A.; Colombo, S.; Leggieri, C.; Muttini, S.; Capocasa, T.; Gallioli, G. Measured versus calculated energy expenditure in pressure support ventilated ICU patients. Minerva Anestesiol. 1996, 62, 165–170. [Google Scholar] [PubMed]
- O’Leary-Kelley, C.M.; Puntillo, K.A.; Barr, J.; Stotts, N.; Douglas, M.K. Nutritional adequacy in patients receiving mechanical ventilation who are fed enterally. Am. J. Crit. Care 2005, 14, 222–231. [Google Scholar] [PubMed]
- Hickmann, C.E.; Roeseler, J.; Castanares-Zapatero, D.; Herrera, E.I.; Mongodin, A.; Laterre, P.F. Energy expenditure in the critically ill performing early physical therapy. Intensive Care Med. 2014, 40, 548–555. [Google Scholar] [CrossRef] [PubMed]
- De Waele, E.; Spapen, H.; Honoré, P.M.; Mattens, S.; van Gorp, V.; Diltoer, M.; Huyghens, L. Introducing a new generation indirect calorimeter for estimating energy requirements in adult intensive care unit patients: Feasibility, practical considerations, and comparison with a mathematical equation. J. Crit. Care 2013, 28, e1–e6. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.N.; Heuberger, R.A. Predictive equations for energy needs for the critically ill. Respir. Care 2009, 54, 509–521. [Google Scholar] [PubMed]
- Kreymann, K.G.; Berger, M.M.; Deutz, N.E.; Hiesmayr, M.; Jolliet, P.; Kazandjiev, G.; Nitenberg, G.; van den Berghe, G.; Wernerman, J.; Ebner, C.; et al. ESPEN guidelines on enteral nutrition: Intensive care. Clin. Nutr. 2006, 25, 210–223. [Google Scholar] [CrossRef] [PubMed]
- Segadilha, N.L.; Rocha, E.E.; Tanaka, L.M.; Gomes, K.L.; Espinoza, R.E.; Peres, W.A. Energy expenditure in critically ill elderly patients: Indirect calorimetry vs. predictive equations. JPEN J. Parenter. Enter. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Picolo, M.F.; Lago, A.F.; Menegueti, M.G.; Nicolini, E.A.; Basile-Filho, A.; Nunes, A.A.; Martins-Filho, O.A.; Auxiliadora-Martins, M. Harris-Benedict equation and resting energy expenditure estimates in critically ill ventilator patients. Am. J. Crit. Care 2016, 25, e21–e29. [Google Scholar] [CrossRef] [PubMed]
- Zusman, O.; Theilla, M.; Cohen, J.; Kagan, I.; Bendavid, I.; Singer, P. Resting energy expenditure, calorie and protein consumption in critically ill patients: A retrospective cohort study. Crit. Care 2016, 20, 367. [Google Scholar] [CrossRef] [PubMed]
- Allingstrup, M.J.; Esmailzadeh, N.; Wilkens Knudsen, A.; Espersen, K.; Hartvig Jensen, T.; Wiis, J.; Perner, A.; Kondrup, J. Provision of protein and energy in relation to measured requirements in intensive care patients. Clin. Nutr. 2012, 31, 462–468. [Google Scholar] [CrossRef] [PubMed]
- Van Strack, S.R.J.; Weijs, P.J.; Koopmans, R.H.; Sauerwein, H.P.; Beishuizen, A.; Girbes, A.R. Optimal nutrition during the period of mechanical ventilation decreases mortality in critically ill, long-term acute female patients: A prospective observational cohort study. Crit. Care 2009, 13, R132. [Google Scholar] [CrossRef] [PubMed]
- Singer, P.; Hiesmayr, M.; Biolo, G.; Felbinger, T.W.; Berger, M.M.; Goeters, C.; Kondrup, J.; Wunder, C.; Pichard, C. Pragmatic approach to nutrition in the ICU: Expert opinion regarding which calorie protein target. Clin. Nutr. 2014, 33, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Bufarah, M.N.B.; de Góes, C.R.; Oliveira, M.C.; Ponce, D.; Balbi, A.L. Estimating catabolism: A possible tool for nutritional monitoring of patients with acute kidney injury. J. Ren. Nutr. 2017, 27, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Fiaccadori, E.; Regolisti, G.; Cademartiri, C.; Cabassi, A.; Picetti, E.; Barbagallo, M.; Gherli, T.; Castellano, G.; Morabito, S.; Maggiore, U. Efficacy and safety of a citrate-based protocol for sustained low-efficiency dialysis in AKI using standard dialysis equipment. Clin. J. Am. Soc. Nephrol. 2013, 8, 1670–1678. [Google Scholar] [CrossRef] [PubMed]
- Umber, A.; Wolley, M.J.; Golper, T.A.; Shaver, M.J.; Marshall, M.R. Amino acid losses during sustained low efficiency dialysis in critically ill patients with acute kidney injury. Clin. Nephrol. 2014, 81, 93–99. [Google Scholar] [CrossRef] [PubMed]
| At ICU Admission | |
|---|---|
| Age (SD) | 67 (15) |
| Male sex (%) | 31/42 (72) |
| Body weight at admission (kg) (SD) | 83 (25.9) |
| BMI (SD) | 29 (9.4) |
| APACHE II (SD) | 22.4 (7.0) |
| SOFA score (median (range)) | |
| - Circulatory | 3.0 (0–4) |
| - Coagulation | 1.0 (0–3) |
| - Liver | 0.0 (0–3) |
| - Renal | 2.0 (0–4) |
| - Neurologic | 0.0 (0–4) |
| - Respiratory | 3.0 (0–4) |
| - Total | 9.1 (3.2) |
| Source of admission (%) | |
| - Other hospital | 1/42 (2) |
| - Medical ward | 11/42 (26) |
| - Surgical ward | 15/42 (36) |
| - Other ICUs | 7/42 (17) |
| - Emergency room | 8/42 (19) |
| ICU admission main diagnosis (%) | |
| - Trauma | 3/42 (7) |
| - Septic Shock | 12/42 (29) |
| - Respiratory | 7/42 (17) |
| - Oncological | 1/42 (2.25) |
| - Cardiac | 1/42 (2.25) |
| - Intoxication | 1/42 (2.25) |
| - Renal | 5/42 (12) |
| - Vascular | 6/42 (14) |
| - Gastrointestinal | 5/42 (12) |
| - Other | 1/42 (2.25) |
| Admission type (%) | |
| - Elective surgery | 3/42 (7) |
| - Urgent surgery | 19/42 (45) |
| - Medical | 20/42 (48) |
| Chronic comorbidities (%) | |
| - Hypertension | 31/42 (74) |
| - Diabetes | 16/42 (38) |
| - COPD | 6/42 (14) |
| - CAD | 6/42 (14) |
| - CHF | 7/42 (17) |
| - Peripheral vascular disease | 6/42 (14) |
| - Immunocompromised | 4/42 (10) |
| - Liver disease | 5/42 (12) |
| - Malignancy | 4/42 (10) |
| - CKD (no dialysis) | 7/42 (17) |
| Acute comorbidities % (at admission or up to 24 h from admission) | |
| - Severe sepsis/septic shock | 16/42 (38) |
| - IMV | 31/42 (71) |
| - NIMV | 1/42 (2) |
| - Shock | 8/42 (19) |
| - AKI | 31/42 (71) |
| - Oliguria | 18/42 (40) |
| - RRT | 6/42 (14) |
| - ARDS | 11/42 (26) |
| - Vasoactive drugs | 20/42 (48) |
| - Major bleeding (≥3 units blood needed) | 7/42 (17) |
| AKI characteristics | |
| AKI at admission (%) | 22/42 (52) |
| Time to AKI from ICU admission (days) (median (range)) | 1.0 (0–26) |
| sCr mg/dL at admission (median (range)) | 1.7 (0.4–17.6) |
| BUN at admission mg/dL (SD) | 44 (36) |
| sCr mg/dL at AKI diagnosis (median (range)) | 2.1 (0.8–17.6) |
| BUN at AKI diagnosis mg/dL (SD) | 52 (34) |
| Urinary output at AKI diagnosis (mL) (median (range)) | 1160 (0–4860) |
| APACHE II at AKI diagnosis (SD) | 22.7 (6.1) |
| sCr mg/dL at RRT start (median (range)) | 4.4 (1.1–17.6) |
| BUN at RRT start diagnosis mg/dL (SD) | 93 (52) |
| Urinary output at RRT start (mL) (median (range)) | 375 (0–4330) |
| APACHE II at RRT start (SD) | 25.8 (5.2) |
| Patients on RRT (%) | 19/42 (45) |
| Number of RRT sessions in the last 24 h before IC | 38/130 |
| Type of RRT (%) | |
| - SLED | 32/38 (84) |
| - HD | 4/38 (11) |
| - HDF | 1/38 (2.5) |
| - CVVH | 1/38 (2.5) |
| Duration RRT days (median (range)) | 9 (1–86) |
| Still on RRT at discharge/death | 6/19 (31.6) |
| Clinical Variable | |
|---|---|
| Maximum Temperature on the Last 24 h (°C) (Mean (SD)) | 37 (0.9) |
| Vasoactive drugs (%) | 43/130 (33) |
| - Norepinephrine | 43/43 (100) |
| - Epinephrine | 0/43 |
| - Dopamine | 0/43 |
| - Dobutamine | 2/43 (5) |
| Other drugs (%) | 74/130 (57) |
| - Barbiturates | 1/74 (1) |
| - Propofol | 6/74 (8) |
| - Benzodiazepines | 17/74 (23) |
| - Neuromuscular blockers | 2/74 (3) |
| - Morphine/Fentanyl | 69/74 (93) |
| Insulin (%) | 103/130 (79%) |
| Insulin IU in the last 24 h (mean (SD)) | 55 (36) |
| Blood glucose at the time of measurement (mean (SD)) | 138 (40) |
| Variables | |
|---|---|
| Total Days of Nutrition | 654 |
| EN only (%) | 294/654 (45) |
| PN only (%) | 204/649 (31) |
| EN + PN (%) | 156/649 (24) |
| Prescribed energy and protein needs (n = 654) | |
| - kcal/day | 1551 (644) |
| - Protein total g/day | 70.5 (38.2) |
| Delivered energy and protein needs (n = 654) | |
| - kcal/day | 1408 (651) * |
| - Protein total g/day | 63.4 (35.3) * |
| Propofol (%) | 32/649 (5) |
| Energy received through somministration of propofol—kcal | 139 (114) |
| Prokinetic drugs (%) | 10/42 (24) |
| - Domperidone (%) | 8/10 (80) |
| - Metoclopramide (%) | 4/10 (40) |
| All Patients (n IC = 130; n PCR = 123) | On MV (n IC = 113) | All on RRT (n IC = 38; n PCR = 39) | RRT-MV (n IC = 31) | No RRT (n IC = 92; n PCR = 84) | No RRT+ MV (n IC = 83) | |
|---|---|---|---|---|---|---|
| IC | 1724 (431) | 1771 (431) | 1730 (445) | 1798 (469) | 1722 (428) | 1762 (420) |
| Equations | ||||||
| - Harris-Benedict with stress factor | 2057 (436) a | NA | 2002 (277) a | NA | 2079 (286) c | NA |
| - Harris-Benedict | 1582 (335) a | NA | 1540 (213) c | NA | 1599 (374) a | NA |
| - 25 kcal/kg | 2116 (560) a | NA | 2174 (463) a | NA | 2092 (596) a | NA |
| - Faisy-Fagon | NA | 2089 (280) a | NA | 2119 (180) a | NA | 2079 (307) a |
| - Penn State | NA | 1793 (392) | NA | 1866 (271) | NA | 1796 (427) |
| Prescribed | 1575 (672) a | 1618 (682) b | 1593 (561) | 1659 (552) | 1568 (716) b | 1603 (724) b |
| Received | 1439 (680) b | 1475 (690) a | 1443 (568) c | 1470 (580) c | 1438 (724) c | 1476 (727) c |
| ANOVA | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
| PCR g/24 h | 102.1 (38.7) | NA | 89.5 (29.3) | NA | 107.9 (41.1) | NA |
| nPCR (g/kg) * | 1.2 (1.09, 0.4–2.7) | NA | 1.0 (0.8, 0.4–2.3) | NA | 1.3 (1.2, 0.6–2.7) | NA |
| Calculated protein need * | 108.3 (46.2) | NA | 154.0 (51.8) d | NA | 87.1 (25.7) d | NA |
| Prescribed protein * | 76.5 (37.3) d | NA | 83.7 (30.1) | NA | 73.2 (40.0) d | NA |
| Received protein * | 69.2 (35.3) d | NA | 76.7 (31.0) e | NA | 66.2 (37.0) d | NA |
| ANOVA | <0.0001 | NA | <0.0001 | NA | <0.0001 | NA |
| Equation | % of Estimates 90–110% of IC | % of Estimates <90% of IC (Underfeeding) | % of Estimates >110% of IC (Overfeeding) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| All Patients | RRT | No RRT | All Patients | RRT | No RRT | All Patients | RRT | No RRT | |
| HB + 30% SF | 26 | 18 | 29 | 5 | 8 | 3 | 9 | 74 | 67 |
| HB | 38 | 42 | 36 | 46 | 47 | 46 | 16 | 11 | 18 |
| 25 kcal/kg/day | 28 | 18 | 32 | 5 | 3 | 7 | 67 | 79 | 62 |
| Faisy-Fagon | 31 | 31 | 31 | 5 | 7 | 5 | 64 | 62 | 64 |
| Penn-State | 40 | 38 | 40 | 27 | 24 | 27 | 34 | 38 | 32 |
| All Patients (n = 81) | On MV (n = 72) | |
|---|---|---|
| IC | 1735 (413) | 1791 (399) |
| Equations | ||
| Harris-Benedict with stress factor | 1995 (462) a | NA |
| Harris-Benedict | 1535 (356) a | NA |
| 25 kcal/kg | 2031 (576) a | NA |
| Faisy-Fagon | NA | 2081 (298) a |
| Penn State | NA | 1767 (427) |
| ANOVA | <0.0001 | <0.0001 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabatino, A.; Theilla, M.; Hellerman, M.; Singer, P.; Maggiore, U.; Barbagallo, M.; Regolisti, G.; Fiaccadori, E. Energy and Protein in Critically Ill Patients with AKI: A Prospective, Multicenter Observational Study Using Indirect Calorimetry and Protein Catabolic Rate. Nutrients 2017, 9, 802. https://doi.org/10.3390/nu9080802
Sabatino A, Theilla M, Hellerman M, Singer P, Maggiore U, Barbagallo M, Regolisti G, Fiaccadori E. Energy and Protein in Critically Ill Patients with AKI: A Prospective, Multicenter Observational Study Using Indirect Calorimetry and Protein Catabolic Rate. Nutrients. 2017; 9(8):802. https://doi.org/10.3390/nu9080802
Chicago/Turabian StyleSabatino, Alice, Miriam Theilla, Moran Hellerman, Pierre Singer, Umberto Maggiore, Maria Barbagallo, Giuseppe Regolisti, and Enrico Fiaccadori. 2017. "Energy and Protein in Critically Ill Patients with AKI: A Prospective, Multicenter Observational Study Using Indirect Calorimetry and Protein Catabolic Rate" Nutrients 9, no. 8: 802. https://doi.org/10.3390/nu9080802
APA StyleSabatino, A., Theilla, M., Hellerman, M., Singer, P., Maggiore, U., Barbagallo, M., Regolisti, G., & Fiaccadori, E. (2017). Energy and Protein in Critically Ill Patients with AKI: A Prospective, Multicenter Observational Study Using Indirect Calorimetry and Protein Catabolic Rate. Nutrients, 9(8), 802. https://doi.org/10.3390/nu9080802





