Intra-Amniotic Administration (Gallus gallus) of Cicer arietinum and Lens culinaris Prebiotics Extracts and Duck Egg White Peptides Affects Calcium Status and Intestinal Functionality
Abstract
:1. Introduction
2. Materials and Methods
2.1. Extraction of Prebiotics
2.2. Preparation of Desalted Duck Egg White Peptides (DPs)
2.3. Animals and Design
2.4. Serum Bone Alkaline Phosphatase and Liver and Bone Mineral Analysis
2.5. Isolation of Total RNA from Chicken Duodenum
2.6. Real Time Polymerase Chain Reaction (RT-PCR)
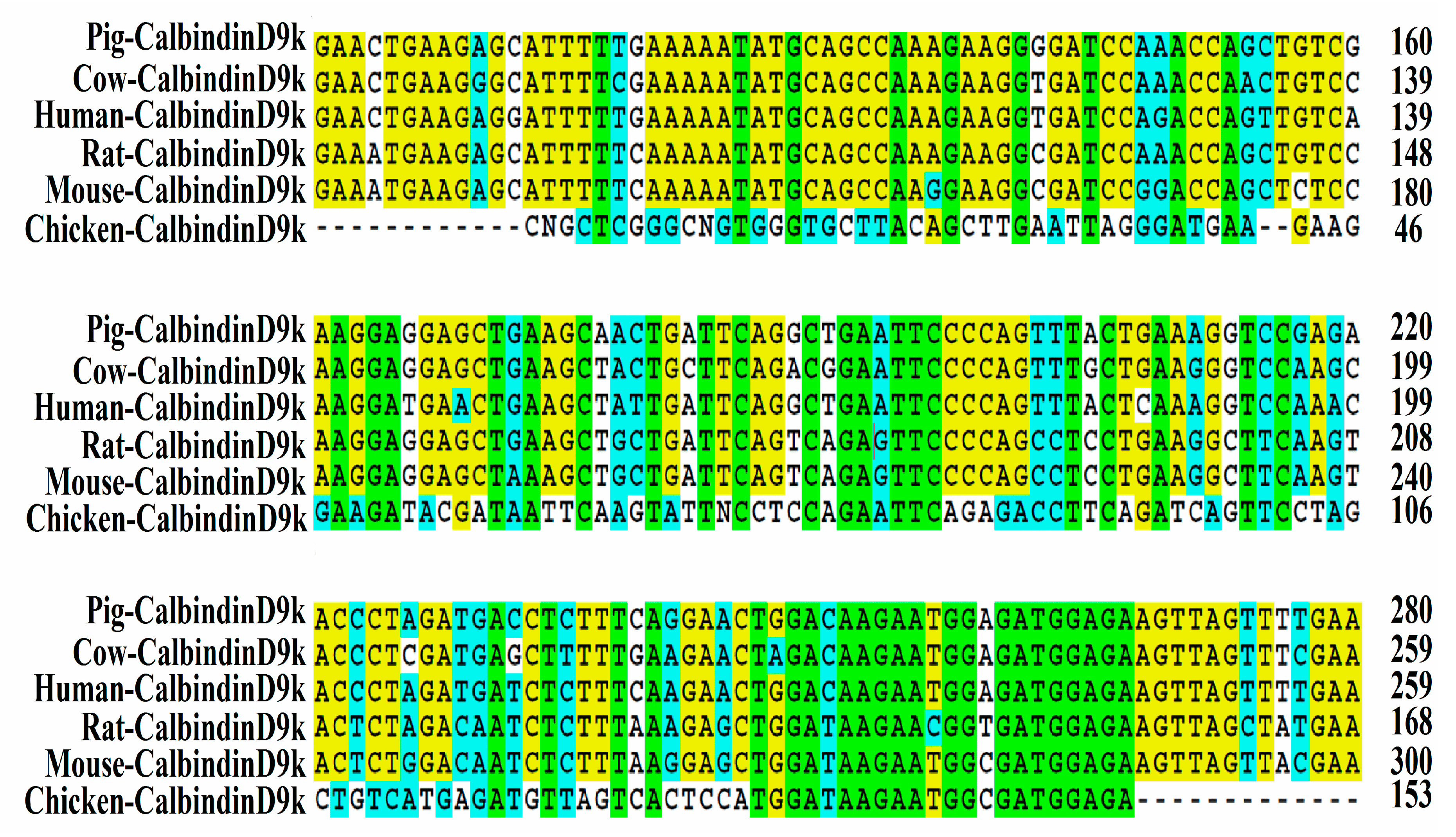
2.7. Isolation of Chicken Intestinal CalbindinD9k Gene Fragment
2.8. Primer Design
2.9. Real-Time qPCR Design
2.10. Collection of Microbial Samples and Intestinal Contents DNA Isolation
2.11. Primers Design and PCR Amplification of Bacterial 16S rDNA
2.12. Morphological Examination
2.13. Statistical Analysis
3. Results
3.1. Isolation and Sequencing of Partial Chicken Intestinal CalbindinD9k cDNA
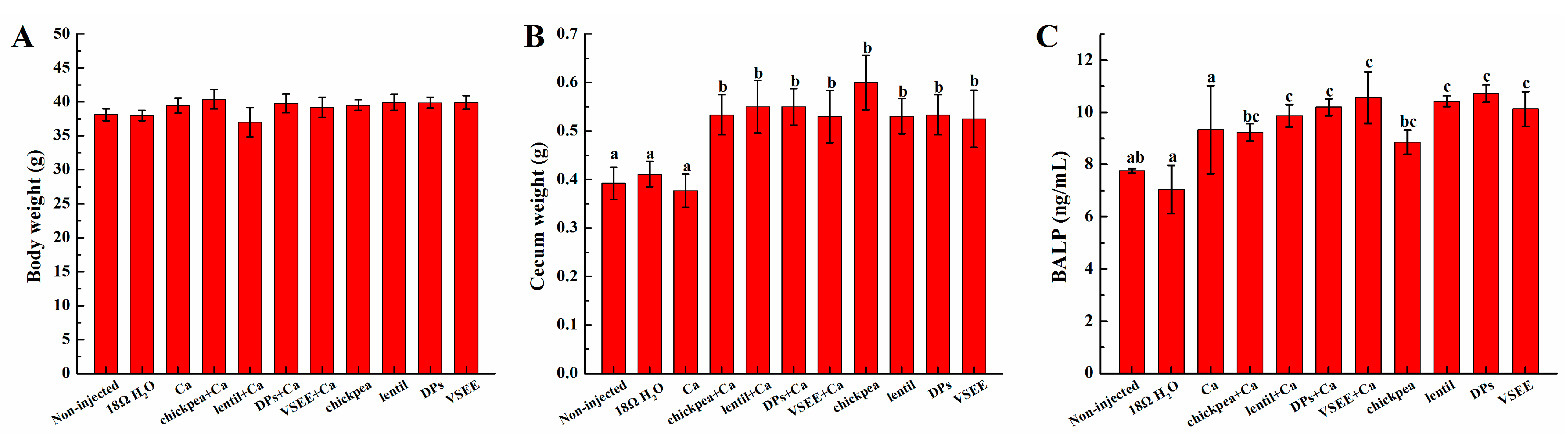
3.2. Body and Cecum Weight
3.3. Serum Bone Alkaline Phosphate (BALP) Content
3.4. Liver and Bone Mineral Content
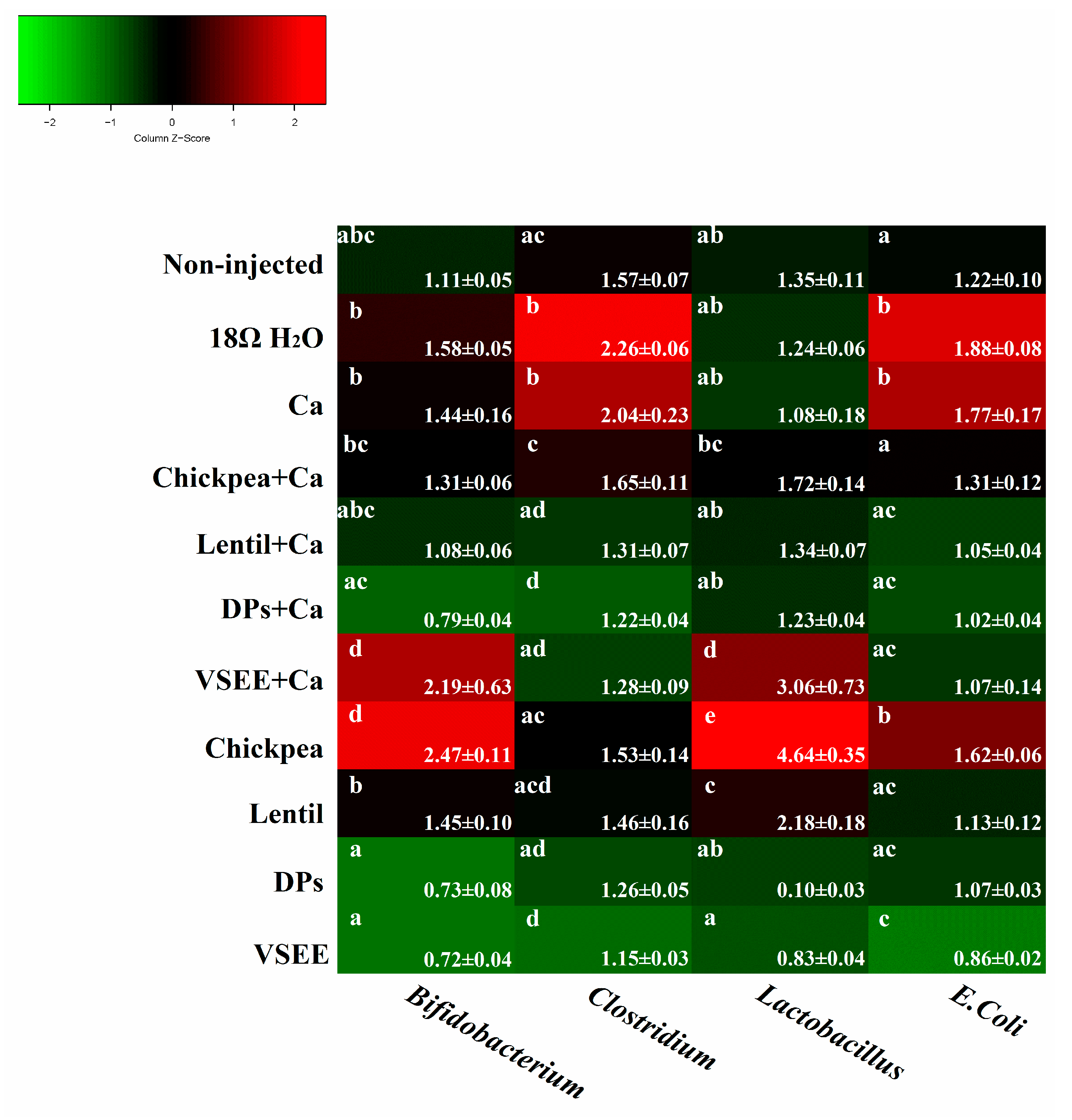
3.5. Intestinal Content Bacterial Genera- and Species-Level Analysis
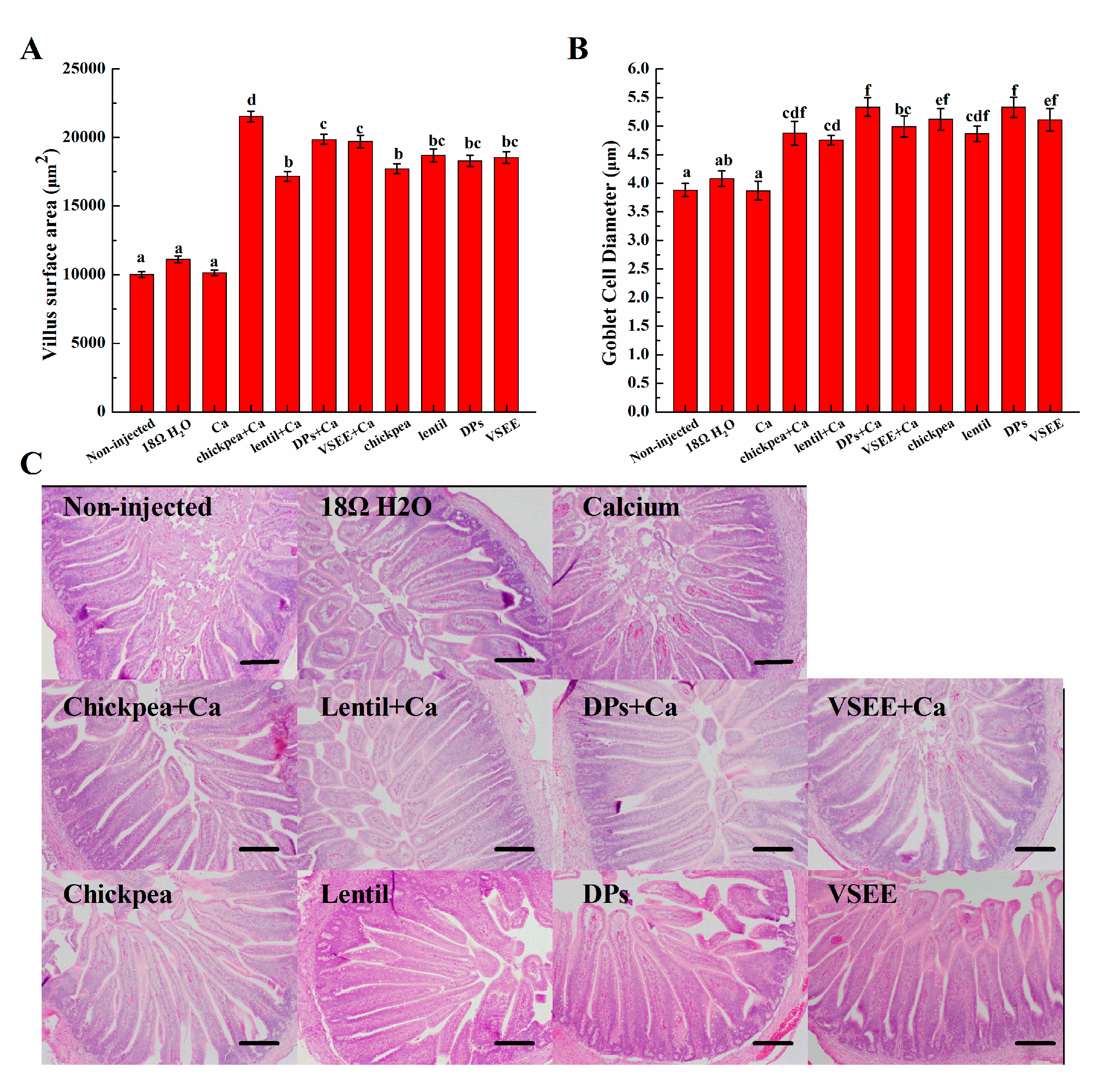
3.6. Morphometric Measurements
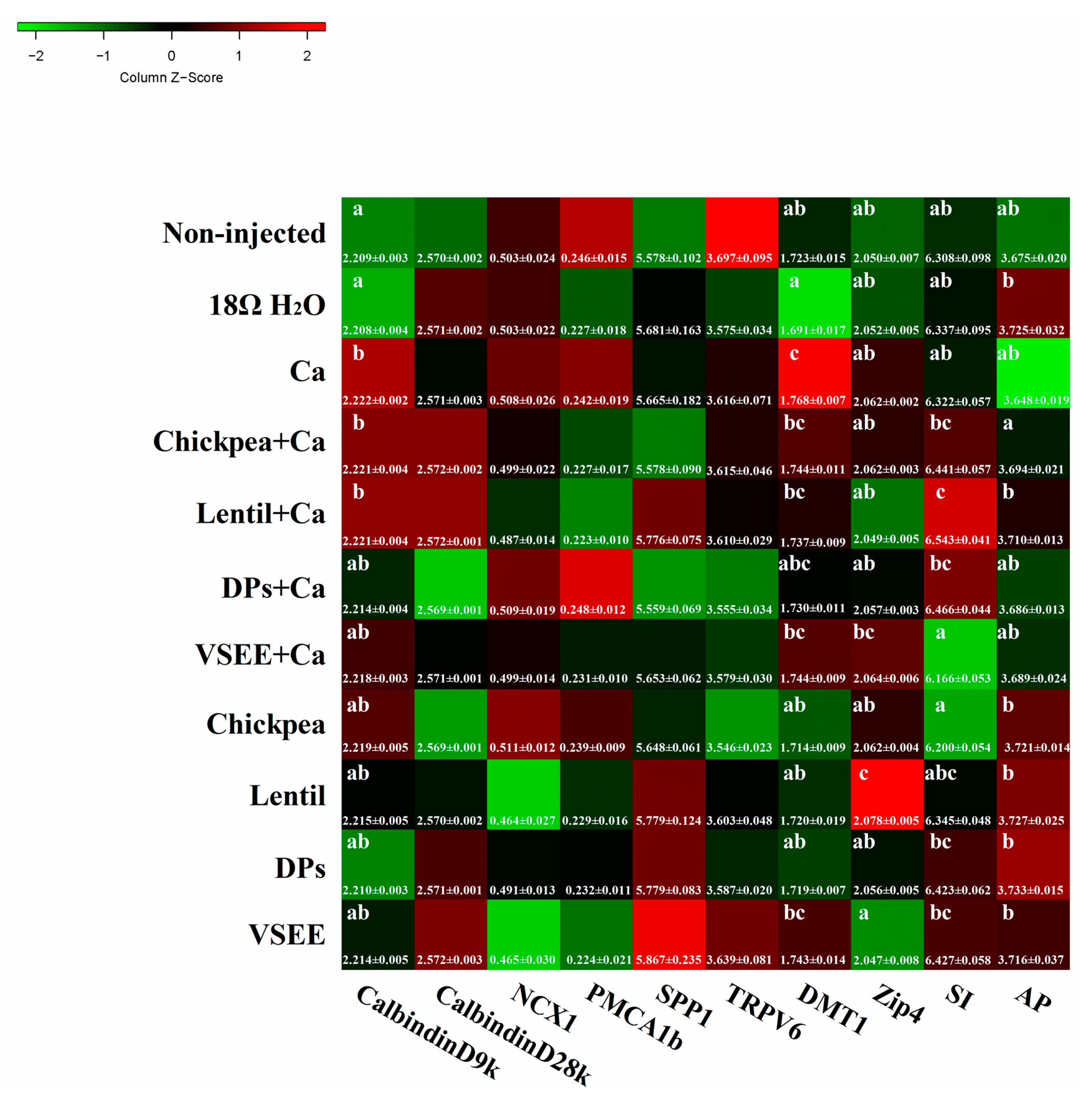
3.7. Calcium Metabolism Genes
3.8. BBM Functional Genes
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| Ca | Calcium |
| Fe | Iron |
| Mg | Magnesium |
| Zn | Zinc |
| DPs | Desalted duck egg white peptides |
| VSEE | Val-Ser-Glu-Glu |
| CPPs | Casein phosphopeptides |
| TRPV6 | Transient receptor potential vanilloid 6 |
| PMCA1b | Plasma membrane Ca2+ ATPase |
| SPP1 | Secreted Phosphoprotein 1 |
| NCX1 | Sodium calcium exchanger 1 |
| DMT1 | Divalent metal transporter 1 |
| AP | Amino peptidase |
| SI | Sucrose isomaltase |
| BBM | Brush border membrane |
References
- Bailey, R.L.; Dodd, K.W.; Goldman, J.A.; Gahche, J.J.; Dwyer, J.T.; Moshfegh, A.J.; Sempos, C.T.; Picciano, M.F. Estimation of total usual calcium and vitamin D intakes in the United States. J. Nutr. 2010, 140, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Bryk, G.; Coronel, M.Z.; Pellegrini, G.; Mandalunis, P.; Rio, M.E.; de Portela, M.L.P.M.; Zeni, S.N. Effect of a combination GOS/FOS® prebiotic mixture and interaction with calcium intake on mineral absorption and bone parameters in growing rats. Eur. J. Nutr. 2015, 54, 913–923. [Google Scholar] [CrossRef] [PubMed]
- Whisner, C.M.; Martin, B.R.; Schoterman, M.H.; Nakatsu, C.H.; McCabe, L.D.; McCabe, G.P.; Wastney, M.E.; Van Den Heuvel, E.G.; Weaver, C.M. Galacto-oligosaccharides increase calcium absorption and gut bifidobacteria in young girls: A double-blind cross-over trial. Br. J. Nutr. 2013, 110, 1292–1303. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.; Jung, C.; Choi, H.; Kim, C.; Ha, H. Effectiveness of phosvitin peptides on enhancing bioavailability of calcium and its accumulation in bones. Food Chem. 2005, 93, 577–583. [Google Scholar] [CrossRef]
- Sun, N.; Wu, H.; Du, M.; Tang, Y.; Liu, H.; Fu, Y.; Zhu, B. Food protein-derived calcium chelating peptides: A review. Trends Food Sci. Technol. 2016, 58, 140–148. [Google Scholar] [CrossRef]
- García-Vieyra, M.I.; Del Real, A.; López, M.G. Agave fructans: Their effect on mineral absorption and bone mineral content. J. Med. Food 2014, 17, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Holloway, L.; Moynihan, S.; Abrams, S.A.; Kent, K.; Hsu, A.R.; Friedlander, A.L. Effects of oligofructose-enriched inulin on intestinal absorption of calcium and magnesium and bone turnover markers in postmenopausal women. Br. J. Nutr. 2007, 97, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.-R.; Wang, L.; Wang, R.; Chen, Z.-X. Calcium-binding capacity of wheat germ protein hydrolysate and characterization of peptide-calcium complex. J. Agric. Food Chem. 2013, 61, 7537–7544. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.M.W.; de Souza, R.; Kendall, C.W.C.; Emam, A.; Jenkins, D.J.A. Colonic health: Fermentation and short chain fatty acids. J. Clin. Gastroenterol. 2006, 40, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Tuohy, K.M.; Rouzaud, G.C.M.; Bruck, W.M.; Gibson, G.R. Modulation of the human gut microflora towards improved health using prebiotics—Assessment of efficacy. Curr. Pharm. Des. 2005, 11, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M.B.; Cumps, J.; Devogelaer, J. Dietary chicory inulin increases whole-body bone mineral density in growing male rats. J. Nutr. 2002, 132, 3599–3602. [Google Scholar] [PubMed]
- Rodrigues, F.C.; Barbosa Castro, A.S.; Rodrigues, V.C.; Fernandes, S.A.; Filomeno Fontes, E.A.; de Oliveira, T.T.; Duarte Martino, H.S.; de Luces Fortes Ferreira, C.L. Yacon Flour and Bifidobacterium longum Modulate Bone Health in Rats. J. Med. Food 2012, 15, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Whisner, C.M.; Martin, B.R.; Nakatsu, C.H.; McCabe, G.P.; McCabe, L.D.; Peacock, M.; Weaver, C.M. Soluble maize fibre affects short-term calcium absorption in adolescent boys and girls: A randomised controlled trial using dual stable isotopic tracers. Br. J. Nutr. 2014, 112, 446–456. [Google Scholar] [CrossRef] [PubMed]
- Tenorio, M.D.; Espinosa-Martos, I.; Préstamo, G.; Rupérez, P. Soybean whey enhance mineral balance and caecal fermentation in rats. Eur. J. Nutr. 2010, 49, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Parvaneh, K.; Jamaluddin, R.; Karimi, G.; Erfani, R. Effect of probiotics supplementation on bone mineral content and bone mass density. Sci. World J. 2014, 2014, 595962. [Google Scholar] [CrossRef] [PubMed]
- Scholz-Ahrens, K.E.; Ade, P.; Marten, B.; Weber, P.; Timm, W.; Asil, Y.; Glueer, C.-C.; Schrezenmeir, J. Prebiotics, probiotics, and synbiotics affect mineral absorption, bone mineral content, and bone structure. J. Nutr. 2007, 137, 838S–846S. [Google Scholar] [PubMed]
- Perego, S.; Del Favero, E.; De Luca, P.; Dal Piaz, F.; Fiorilli, A.; Ferraretto, A. Calcium bioaccessibility and uptake by human intestinal like cells following in vitro digestion of casein phosphopeptide-calcium aggregates. Food Funct. 2015, 6, 1796–1807. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.-L.; Zhao, L.-N.; Cai, X.; Wang, S.-Y.; Huang, Y.-F.; Hong, J.; Rao, P.-F. Purification and characterisation of a glutamic acid-containing peptide with calcium-binding capacity from whey protein hydrolysate. J. Dairy Res. 2015, 82, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Harnedy, P.A.; O’Keeffe, M.B.; Zhang, L.; Li, B.; Hou, H.; FitzGerald, R.J. Fractionation and identification of Alaska pollock skin collagen-derived mineral chelating peptides. Food Chem. 2015, 173, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Mu, X.; Huang, H.; Nie, R.; Liu, Z.; Zeng, M. Isolation of a calcium-binding peptide from tilapia scale protein hydrolysate and its calcium bioavailability in rats. J. Funct. Foods 2014, 6, 575–584. [Google Scholar] [CrossRef]
- Lv, Y.; Bao, X.; Liu, H.; Ren, J.; Guo, S. Purification and characterization of caclium-binding soybean protein hydrolysates by Ca2+/Fe3+ immobilized metal affinity chromatography (IMAC). Food Chem. 2013, 141, 1645–1650. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Ren, L.; Jiang, J. Purification of a histidine-containing peptide with calcium binding activity from shrimp processing byproducts hydrolysate. Eur. Food Res. Technol. 2011, 232, 281–287. [Google Scholar] [CrossRef]
- Jeon, S.-J.; Lee, J.-H.; Song, K.-B. Isolation of a calcium-binding peptide from chlorella protein hydrolysates. J. Food Sci. Nutr. 2010, 15, 282–286. [Google Scholar] [CrossRef]
- Ferraretto, A.; Signorile, A.; Gravaghi, C.; Fiorilli, A.; Tettamanti, G. Casein phosphopeptides influence calcium uptake by cultured human intestinal HT-29 tumor cells. J. Nutr. 2001, 131, 1655–1661. [Google Scholar] [PubMed]
- Perego, S.; Zabeo, A.; Marasco, E.; Giussani, P.; Fiorilli, A.; Tettamanti, G.; Ferraretto, A. Casein phosphopeptides modulate calcium uptake and apoptosis in Caco2 cells through their interaction with the TRPV6 calcium channel. J. Funct. Foods 2013, 5, 847–857. [Google Scholar] [CrossRef]
- Perego, S.; Cosentino, S.; Fiorilli, A.; Tettamanti, G.; Ferraretto, A. Casein phosphopeptides modulate proliferation and apoptosis in HT-29 cell line through their interaction with voltage-operated L-type calcium channels. J. Nutr. Biochem. 2012, 23, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Tako, E.; Bar, H.; Glahn, R.P. The combined application of the Caco-2 cell bioassay coupled with in vivo (Gallus gallus) feeding trial represents an effective approach to predicting Fe bioavailability in humans. Nutrients 2016, 8, 732. [Google Scholar] [CrossRef] [PubMed]
- Tako, E.; Glahn, R. Intra-amniotic administration and dietary inulin affect the iron status and intestinal functionality of iron-deficient broiler chickens. Poult. Sci. 2012, 91, 1361–1370. [Google Scholar] [CrossRef] [PubMed]
- Yegani, M.; Korver, D.R. Factors affecting intestinal health in poultry. Poult. Sci. 2008, 87, 2052–2063. [Google Scholar] [CrossRef] [PubMed]
- Backhed, F.; Ley, R.E.; Sonnenburg, J.L.; Peterson, D.A.; Gordon, J.I. Host-bacterial mutualism in the human intestine. Science 2005, 307, 1915–1920. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.M.; Choct, M.; Iji, P.A.; Bruerton, K. Trace mineral interactions in broiler chicken diets. Br. Poult. Sci. 2010, 51, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Xi, P.; Junliang, D.; Debing, L.; Guang, Y. Pathology of lymphoid organs in chickens fed a diet deficient in zinc. Avian Pathol. 2004, 33, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Burrell, A.L.; Dozier, W.A.; Davis, A.J.; Compton, M.M.; Freeman, M.E.; Vendrell, P.F.; Ward, T.L. Responses of broilers to dietary zinc concentrations and sources in relation to environmental implications. Br. Poult. Sci. 2004, 45, 255–263. [Google Scholar] [CrossRef] [PubMed]
- De Almeida Costa, G.E.; da Silva Queiroz-Monici, K.; Reis, S.M. P.M.; de Oliveira, A.C. Chemical composition, dietary fibre and resistant starch contents of raw and cooked pea, common bean, chickpea and lentil legumes. Food Chem. 2006, 94, 327–330. [Google Scholar] [CrossRef]
- Rebello, C.J.; Greenway, F.L.; Finley, J.W. Whole grains and pulses: A comparison of the nutritional and health benefits. J. Agric. Food Chem. 2014, 62, 7029–7049. [Google Scholar] [CrossRef] [PubMed]
- Fernando, W.M.; Hill, J.E.; Zello, G.A.; Tyler, R.T.; Dahl, W.J.; Van Kessel, A.G. Diets supplemented with chickpea or its main oligosaccharide component raffinose modify faecal microbial composition in healthy adults. Benef. Microbes 2010, 1, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.R.; Thavarajah, D.; Combs, G.F.; Thavarajah, P. Lentil (Lens culinaris L.): A prebiotic-rich whole food legume. Food Res. Int. 2013, 51, 107–113. [Google Scholar] [CrossRef]
- Zhao, N.; Hu, J.; Hou, T.; Ma, Z.; Wang, C.; He, H. Effects of desalted duck egg white peptides and their products on calcium absorption in rats. J. Funct. Foods 2014, 8, 234–242. [Google Scholar] [CrossRef]
- Hou, T.; Liu, W.; Shi, W.; Ma, Z.; He, H. Desalted duck egg white peptides promote calcium uptake by counteracting the adverse effects of phytic acid. Food Chem. 2017, 219, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Tako, E.; Glahn, R.P.; Knez, M.; Stangoulis, J.C.R. The effect of wheat prebiotics on the gut bacterial population and iron status of iron deficient broiler chickens. Nutr. J. 2014, 13, 58. [Google Scholar] [CrossRef] [PubMed]
- Lieben, L.; Stockmans, I.; Moermans, K.; Carmeliet, G. Maternal hypervitaminosis D reduces fetal bone mass and mineral acquisition and leads to neonatal lethality. Bone 2013, 57, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Pacifici, S.; Song, J.; Zhang, C.; Wang, Q.; Glahn, R.P.; Kolba, N.; Tako, E. Intra amniotic administration of raffinose and stachyose affects the intestinal brush border functionality and alters gut microflora populations. Nutrients 2017, 9, 304. [Google Scholar] [CrossRef] [PubMed]
- Hartono, K.; Reed, S.; Ankrah, N.A.; Glahn, R.P.; Tako, E. Alterations in gut microflora populations and brush border functionality following intra-amniotic daidzein administration. RSC Adv. 2015, 5, 6407–6412. [Google Scholar] [CrossRef]
- Zhu, X.Y.; Zhong, T.; Pandya, Y.; Joerger, R.D. 16S rRNA-based analysis of microbiota from the cecum of broiler chickens. Appl. Environ. Microbiol. 2002, 68, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Van Cromphaut, S.J.; Dewerchin, M.; Hoenderop, J.G.; Stockmans, I.; Van Herck, E.; Kato, S.; Bindels, R.J.; Collen, D.; Carmeliet, P.; Bouillon, R. Duodenal calcium absorption in vitamin D receptor–knockout mice: Functional and molecular aspects. Proc. Natl. Acad. Sci. USA 2001, 98, 13324–13329. [Google Scholar] [CrossRef] [PubMed]
- Kellett, G.L. Alternative perspective on intestinal calcium absorption: Proposed complementary actions of Cav1. 3 and TRPV6. Nutr. Rev. 2011, 69, 347–370. [Google Scholar] [CrossRef] [PubMed]
- Fisher, L.W.; Fedarko, N.S. Six genes expressed in bones and teeth encode the current members of the SIBLING family of proteins. Connect. Tissue Res. 2003, 44, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Kim, J.; Kang, E.-J.; Lee, S.-W.; Park, M.-C.; Park, Y.-B.; Lee, S.-K. Osteopontin might be involved in bone remodelling rather than in inflammation in ankylosing spondylitis. Rheumatology 2008, 47, 1775–1779. [Google Scholar] [CrossRef] [PubMed]
- Tako, E.; Glahn, R.P. White beans provide more bioavailable iron than red beans: Studies in poultry (Gallus gallus) and an in vitro digestion/Caco-2 model. Int. J. Vitam. Nutr. Res. 2010, 80, 416. [Google Scholar] [CrossRef] [PubMed]
- Tako, E.; Rutzke, M.A.; Glahn, R.P. Using the domestic chicken (Gallus gallus) as an in vivo model for iron bioavailability. Poult. Sci. 2010, 89, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Reed, S.; Qin, X.; Ran-Ressler, R.; Brenna, J.T.; Glahn, R.P.; Tako, E. Dietary zinc deficiency affects blood linoleic acid: Dihomo-γ-linolenic acid (LA: DGLA) ratio; a sensitive physiological marker of zinc status in vivo (Gallus gallus). Nutrients 2014, 6, 1164–1180. [Google Scholar] [CrossRef] [PubMed]
- Liao, E.Y.; Luo, X.H.; Wang, W.B.; Wu, X.P.; Zhou, H.D.; Dai, R.C.; Liao, H.J.; Yang, C. Effects of different nylestriol/levonorgestrel dosages on bone metabolism in female Sprague–Dawley rats with retinoic acid-induced osteoporosis. Endocr. Res. 2003, 29, 23–42. [Google Scholar] [CrossRef] [PubMed]
- Wei, M.; Yang, Z.; Li, P.; Zhang, Y.; Sse, W.C. Anti-osteoporosis activity of naringin in the retinoic acid-induced osteoporosis model. Am. J. Chin. Med. 2007, 35, 663–667. [Google Scholar] [CrossRef] [PubMed]
- Kalu, D.N. The ovariectomized rat model of postmenopausal bone loss. Bone Miner. 1991, 15, 175–191. [Google Scholar] [CrossRef]
- Jung, W.-K.; Lee, B.-J.; Kim, S.-K. Fish-bone peptide increases calcium solubility and bioavailability in ovariectomised rats. Br. J. Nutr. 2006, 95, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, Y.; Song, S.; Wang, X.; Qin, Y.; Si, S.; Guo, Y. Combined oral administration of bovine collagen peptides with calcium citrate inhibits bone loss in ovariectomized rats. PLoS ONE 2015, 10, e0135019. [Google Scholar] [CrossRef] [PubMed]
- Cosentino, S.; Donida, B.M.; Marasco, E.; Del Favero, E.; Cantu, L.; Lombardi, G.; Colombini, A.; Iametti, S.; Valaperta, S.; Fiorilli, A.; et al. Calcium ions enclosed in casein phosphopeptide aggregates are directly involved in the mineral uptake by differentiated HT-29 cells. Int. Dairy J. 2010, 20, 770–776. [Google Scholar] [CrossRef]
- Weaver, C.M.; Martin, B.R.; Nakatsu, C.H.; Armstrong, A.P.; Clavijo, A.; McCabe, L.D.; McCabe, G.P.; Duignan, S.; Schoterman, M.H.; van den Heuvel, E.G. Galactooligosaccharides improve mineral absorption and bone properties in growing rats through gut fermentation. J. Agric. Food Chem. 2011, 59, 6501–6510. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.-C.; Wu, J.-B.; Lu, T.-J.; Lin, W.-C. The prebiotic effect of Anoectochilus formosanus and its consequences on bone health. Br. J. Nutr. 2013, 109, 1779–1788. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.W.; Duncan, S.H.; Leitch, E.C.M.; Child, M.W.; Flint, H.J. pH and peptide supply can radically alter bacterial populations and short-chain fatty acid ratios within microbial communities from the human colon. Appl. Environ. Microbiol. 2005, 71, 3692–3700. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M.B.; Van Loo, J.A.; Gibson, G.R. The bifidogenic nature of chicory inulin and its hydrolysis products. J. Nutr. 1998, 128, 11–19. [Google Scholar] [PubMed]
- Hou, T.; Wang, C.; Ma, Z.; Shi, W.; Lui, W.; He, H. Desalted duck egg white peptides: Promotion of calcium uptake and structure characterization. J. Agric. Food Chem. 2015, 63, 8170–8176. [Google Scholar] [CrossRef] [PubMed]
| Analyte | Forward Primer (5′-3′) (Nucleotide Position) | Reverse Primer (5′-3′) | Base Pair | GI Identifier |
|---|---|---|---|---|
| Zip4 | TCTCCTTAGCAGACAATTGAG | GTGACAAACAAGTAGGCGAAAC | 95 | 107050877 |
| DMT-1 | TTGATTCAGAGCCTCCCATTAG | GCGAGGAGTAGGCTTGTATTT | 101 | 206597489 |
| CalBindinD9k | CCTGCAGAAATGAAGAGCATTTT | CTCCATCGCCATTCTTATCCA | 125 | SAMN06269460 |
| CalBindinD28k | CGGAGGAGAATTTCCTGTTGT | CCACTGTGGTCACTGTCATATT | 97 | NM_205513.1 |
| PMCA1b | TGCAGATGCTGTGGGTAAAT | CCATAAGGCTTCCGCAATAGA | 100 | NM_001168002.3 |
| NCX1 | CCTGACGGAGAAATAAGGAAGA | CCCAGGAGAAGACACAGATAAA | 114 | NM_001079473.1 |
| SPP1 | GACACTGACGAGTCTGATGAAG | CTGAAGCCATATGCCACACT | 113 | NM_204535.4 |
| SI | CCAGCAATGCCAGCATATTG | CGGTTTCTCCTTACCACTTCTT | 95 | 2246388 |
| TRPV6 | GCTCCCAGAACCTTCTCTATTT | CCAGGTAATCCTGAGCTCTAATG | 123 | XM_004938142.2 |
| AP | CGTCAGCCAGTTTGACTATGTA | CTCTCAAAGAAGCTGAGGATGG | 138 | 45382360 |
| AKP | CTCATTCCAGCGTACTCTTCTT | GTGTGTAGATCAAAGGGCTACT | 100 | XM_015291489.1 |
| 18S rRNA | GCAAGACGAACTAAAGCGAAAG | TCGGAACTACGACGGTATCT | 100 | 7262899 |
| Group | Bone (mg/kg) | Liver (mg/kg) | ||||||
|---|---|---|---|---|---|---|---|---|
| Ca | Mg | Fe | Zn | Ca | Mg | Fe | Zn | |
| Non-injected | 22,985 ± 962.5 | 370 ± 8.72 | 12.8 ± 0.56 | 31.4 ± 2.08 | 56.4 ± 1.89 | 165.4 ± 4.05 | 55.4 ± 5.89 | 23.0 ± 1.35 |
| 18 MΩ H2O | 21,253 ± 431.6 | 339 ± 5.56 | 12.4 ± 1.12 | 33.9 ± 1.54 | 56.2 ± 1.83 | 157.7 ± 5.80 | 43.6 ± 4.63 | 22.2 ± 1.18 |
| Ca | 25,173 ± 1552.6 | 364 ± 18.30 | 12.1 ± 0.99 | 34.1 ± 0.94 | 51.1 ± 1.96 | 158.0 ± 2.90 | 48.3 ± 3.53 | 22.1 ± 1.66 |
| Chickpea + Ca | 23,271 ± 1172.5 | 345 ± 5.14 | 10.8 ± 0.92 | 35.9 ± 1.72 | 55.7 ± 2.03 | 152.5 ± 3.77 | 52.6 ± 3.81 | 24.2 ± 3.23 |
| Lentil + Ca | 23,823 ± 1123.7 | 397 ± 24.36 | 11.9 ± 0.80 | 33.4 ± 1.32 | 55.7 ± 2.77 | 166.5 ± 4.34 | 51.9 ± 6.94 | 24.5 ± 2.84 |
| DPs + Ca | 25,981 ± 2518.5 | 335 ± 54.94 | 11.0 ± 0.72 | 34.9 ± 2.55 | 55.3 ± 2.73 | 158.1 ± 4.44 | 53.2 ± 7.43 | 24.4 ± 2.36 |
| VSEE + Ca | 22,587 ± 1046.2 | 350 ± 13.96 | 13.6 ± 1.37 | 32.8 ± 0.87 | 64.9 ± 2.74 | 161.6 ± 3.39 | 60.5 ± 4.98 | 25.1 ± 2.90 |
| Chickpea | 24,910 ± 1397.2 | 374 ± 15.14 | 13.7 ± 0.93 | 33.2 ± 3.06 | 63.6 ± 3.68 | 164.9 ± 4.68 | 51.5 ± 5.59 | 20.8 ± 1.76 |
| Lentil | 24,824 ± 1178.7 | 394 ± 8.84 | 13.0 ± 1.48 | 35.9 ± 2.13 | 66.7 ± 2.44 | 164.5 ± 2.75 | 50.6 ± 7.55 | 24.1 ± 1.67 |
| DPs | 24,967 ± 1849.3 | 381 ± 33.63 | 11.2 ± 0.95 | 30.4 ± 0.63 | 60.8 ± 4.99 | 156.2 ± 2.98 | 55.3 ± 5.47 | 26.5 ± 2.38 |
| VSEE | 24,582 ± 655.4 | 368 ± 10.98 | 13.9 ± 0.77 | 30.8 ± 0.75 | 61.8 ± 1.60 | 164.9 ± 3.69 | 60.0 ± 7.26 | 22.8 ± 1.26 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hou, T.; Kolba, N.; Glahn, R.P.; Tako, E. Intra-Amniotic Administration (Gallus gallus) of Cicer arietinum and Lens culinaris Prebiotics Extracts and Duck Egg White Peptides Affects Calcium Status and Intestinal Functionality. Nutrients 2017, 9, 785. https://doi.org/10.3390/nu9070785
Hou T, Kolba N, Glahn RP, Tako E. Intra-Amniotic Administration (Gallus gallus) of Cicer arietinum and Lens culinaris Prebiotics Extracts and Duck Egg White Peptides Affects Calcium Status and Intestinal Functionality. Nutrients. 2017; 9(7):785. https://doi.org/10.3390/nu9070785
Chicago/Turabian StyleHou, Tao, Nikolai Kolba, Raymond P. Glahn, and Elad Tako. 2017. "Intra-Amniotic Administration (Gallus gallus) of Cicer arietinum and Lens culinaris Prebiotics Extracts and Duck Egg White Peptides Affects Calcium Status and Intestinal Functionality" Nutrients 9, no. 7: 785. https://doi.org/10.3390/nu9070785
APA StyleHou, T., Kolba, N., Glahn, R. P., & Tako, E. (2017). Intra-Amniotic Administration (Gallus gallus) of Cicer arietinum and Lens culinaris Prebiotics Extracts and Duck Egg White Peptides Affects Calcium Status and Intestinal Functionality. Nutrients, 9(7), 785. https://doi.org/10.3390/nu9070785






