Associations between Red Meat Intakes and the Micronutrient Intake and Status of UK Females: A Secondary Analysis of the UK National Diet and Nutrition Survey
Abstract
:1. Introduction
2. Materials and Methods
2.1. Nutrients of Interest
2.2. National Diet and Nutrition Survey (NDNS)
2.2.1. Subjects and Study Design
2.2.2. Socioeconomic Status
2.2.3. Micronutrient Data
2.2.4. Response Rates
2.3. Statistical Analyses
3. Results
3.1. Participants
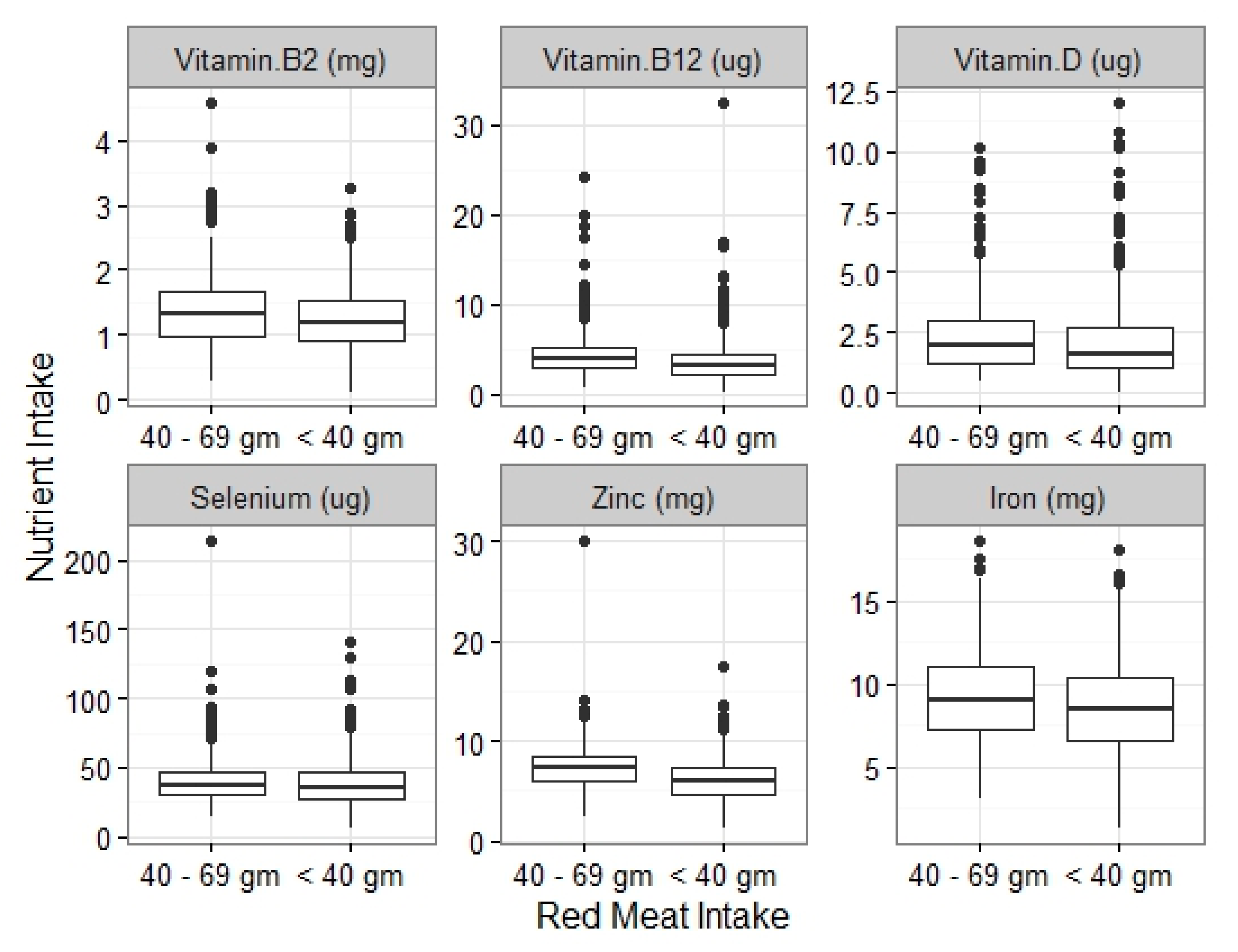
3.2. Micronutrient Intakes
3.3. Regression Modelling
3.4. Micronutrient Status
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- McNeill, S.; van, E.M.E. Red meat in global nutrition. Meat Sci. 2012, 92, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Bates, B.; Cox, L.; Nicholson, S.; Page, P.; Prentice, A.; Steer, T.; Swan, G. National Diet and Nutrition Survey Results from Years 5 and 6 (Combined) of the Rolling Programme (2012/2013–2013/2014); Public Health England: London, UK, 2016. [Google Scholar]
- SACN: Iron and Health. Available online: http://www.fcrn.org.uk/sites/default/files/sacn_iron_and_health_report.pdf (accessed on 18 July 2017).
- McClung, J.P.; Murray-Kolb, L.E. Iron nutrition and premenopausal women: Effects of poor iron status on physical and neuropsychological performance. Annu. Rev. Nutr. 2013, 33, 271–288. [Google Scholar] [CrossRef] [PubMed]
- Coad, J.; Conlon, C. Iron deficiency in women: Assessment, causes and consequences. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.M. The reproductive ecology of iron in women. Am. J. Phys. Anthropol. 2016, 159, S172–S195. [Google Scholar] [CrossRef] [PubMed]
- Greig, A.J.; Patterson, A.J.; Collins, C.E.; Chalmers, K.A. Iron deficiency, cognition, mental health and fatigue in women of childbearing age: A systematic review. J. Nutr. Sci. 2013, 2, e14. [Google Scholar] [CrossRef] [PubMed]
- Maret, W. Zinc biochemistry: From a single zinc enzyme to a key element of life. Adv. Nutr. 2013, 4, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M. Role of nutritional zinc in the prevention of osteoporosis. Mol. Cell. Biochem. 2010, 338, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S. Discovery of human zinc deficiency: Its impact on human health and disease. Adv. Nutr. 2013, 4, 176–190. [Google Scholar] [CrossRef] [PubMed]
- Williams, P. Nutritional composition of red meat. Nutr. Diet. 2007, 64, S113–S119. [Google Scholar] [CrossRef]
- Kennedy, D.O. B Vitamins and the Brain: Mechanisms, Dose and Efficacy—A Review. Nutrients 2016, 8, 68. [Google Scholar] [CrossRef] [PubMed]
- Clarke, M.; Ward, M.; Strain, J.J.; Hoey, L.; Dickey, W.; McNulty, H. B-vitamins and bone in health and disease: The current evidence. Proc. Nutr. Soc. 2014, 73, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Hatfield, D.L.; Tsuji, P.A.; Carlson, B.A.; Gladyshev, V.N. Selenium and selenocysteine: Roles in cancer, health, and development. Trends Biochem. Sci. 2014, 39, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Mistry, H.D.; Broughton, P.F.; Redman, C.W.; Poston, L. Selenium in reproductive health. Am. J. Obstet. Gynecol. 2012, 206, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, M.; von Versen-Hoynck, F. Vitamin D—Roles in women’s reproductive health? Reprod. Biol. Endocrinol. 2011, 9, 146. [Google Scholar] [CrossRef] [PubMed]
- WCRF: Animal Foods. Available online: http://www.wcrf.org/int/research-we-fund/cancer-prevention-recommendations/animal-foods (accessed on 18 July 2017).
- EU. Regulation (EC) No. 1924/2006 of the European parliament and of the council of 20 December 2006 on nutrition and health claims made on foods. Off. J. Eur. Union 2006, L404/409. [Google Scholar]
- Composition of Foods Integrated Dataset (CoFID). Available online: https://www.gov.uk/government/publications/composition-of-foods-integrated-dataset-cofid (accessed on 18 July 2017).
- AHDB. Best Practise Guides: Understanding the Nutrition and Health Claims Regulations on Beef, Lamb and Pork; Agriculture & Horticulture Development Board: Warwickshire, UK, 2017. [Google Scholar]
- Bates, B.; Lennox, A.; Prentice, A.; Bates, C.; Page, P.; Nicholson, S.; Swan, G. National Diet and Nutrition Survey Results from Years 1, 2, 3 and 4 (combined) of the Rolling Programme (2008/2009–2011/2012); Public Health England: London, UK, 2014; pp. 40–52. [Google Scholar]
- Guess, N. Dietary intake in people consuming a reduced-carbohydrate diet in the National Diet and Nutrition Survey. J. Hum. Nutr. Diet. 2017, 30, 360–368. [Google Scholar] [CrossRef] [PubMed]
- WHO. Iron Deficiency Anaemia; Assessment, Prevention, and Control: A Guide for Programme Managers; World Health Organisation: Geneva, Switzerland, 2001. [Google Scholar]
- WHO. Conclusions of a WHO technical consultation on folate and vitamin B12 deficiencies. Food Nutr. Bull. 2008, 29, S238–S244. [Google Scholar]
- DoH. Nutrition and Bone Health with Particular Reference to Calcium and Vitamin D; Department of Health: London, UK, 1998.
- IARC. IARC Monographs Evaluate Consumption of Red Meat and Processed Meat; The International Agency for Research on Cancer: Lyon, France, 2015. [Google Scholar]
- De Smet, S.; Vossen, E. Meat: The balance between nutrition and health. A Review. Meat Sci. 2016, 120, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Johnson, I.T. The cancer risk related to meat and meat products. Br. Med. Bull. 2016, 121, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Eatwell Guide. Available online: https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/528193/Eatwell_guide_colour.pdf (accessed on 16 July 2017).
- Simpson, J.L.; Bailey, L.B.; Pietrzik, K.; Shane, B.; Holzgreve, W. Micronutrients and women of reproductive potential: Required dietary intake and consequences of dietary deficiency or excess. Part II—Vitamin D, vitamin A, iron, zinc, iodine, essential fatty acids. J. Matern. Fetal Neonatal Med. 2011, 24, 1–24. [Google Scholar] [PubMed]
- Fathizadeh, S.; Amani, R.; Haghighizadeh, M.H.; Hormozi, R. Comparison of serum zinc concentrations and body antioxidant status between young women with premenstrual syndrome and normal controls: A case-control study. Int. J. Reprod. Biomed. 2016, 14, 699–704. [Google Scholar]
- Lomagno, K.A.; Hu, F.; Riddell, L.J.; Booth, A.O.; Szymlek-Gay, E.A.; Nowson, C.A.; Byrne, L.K. Increasing iron and zinc in pre-menopausal women and its effects on mood and cognition: A systematic review. Nutrients 2014, 6, 5117–5141. [Google Scholar] [CrossRef] [PubMed]
- Kamudoni, P.; Poole, C.; Davies, S.J. An estimate of the economic burden of vitamin D deficiency in pregnant women in the United Kingdom. Gynecol. Endocrinol. 2016, 32, 592–597. [Google Scholar] [CrossRef] [PubMed]
- IOM. Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2010. [Google Scholar]
- Dragsted, L.O. Biomarkers of meat intake and the application of nutrigenomics. Meat Sci. 2010, 84, 301–307. [Google Scholar] [CrossRef] [PubMed]
| Age | LRNI a | |
|---|---|---|
| 11–18 years (n, %) | 439 (31.7%) | -- |
| 19–64 years (n, %) | 945 (68.3%) | -- |
| Weight (kg) (n = 1301) | 56.4 (56.4–77.8) | -- |
| BMI (kg/m2) (n = 1296) | -- | |
| Less than 25 kg/m2 | 687 (53%) | -- |
| Equal or More than 25 kg/m2 | 609 (47%) | -- |
| SES (IMD Score) | -- | |
| 0.53–8.49 | 245 (21.3%) | -- |
| 8.49–13.79 | 225 (19.6%) | -- |
| 13.79–21.35 | 212 (18.5%) | -- |
| 21.35–34.17 | 230 (20%) | -- |
| 34.17–87.8 | 237 (20.6%) | -- |
| Fe < LRNI (n, %) | 422 (30.5%) | 4.7 mg/day M; 8.0 mg/day F |
| Zn < LRNI (n, %) | 135 (9.8%) | 5.5 mg/day M; 4.0 mg/day F |
| Vit. B1 < LRNI (n, %) | 1 (0.1%) | 0.23 mg/1000 kcal |
| Vit. B2 < LRNI (n, %) | 203 (14.7%) | 0.8 mg/day |
| Vit. B12 < LRNI (n, %) | 23 (1.7%) | 1.0 μg/day |
| Se < LRNI (n, %) | 671 (48.5%) | 40 μg/day |
| K < LRNI (n, %) | 348 (25.1%) | 2000 mg/day |
| Vit. D intake (µg) | 1.98 (1.29–3.11) | -- |
| Hemoglobin (mg/dL) (n = 444) | 13.3 (12.6–13.8) | -- |
| Plasma Ferritin (mg/dL) (n = 450) | 34.86 (19.15–69.23) | -- |
| Plasma Zn (μmol/L) | 13.97 (12.46–15.55) | -- |
| Serum vit. B12 (pmol/L) (n = 446) | 256 (204–312.25) | -- |
| Serum 25-hydroxyvitamin D (nmol/L) (n = 445) | 41.65 (26.68–59.43) | -- |
| Red Meat < 70 (n = 976) | Red Meat ≥ 70 (n = 408) | P Univariate | Adjusted P b | Red Meat < 40 (n = 597) | Red Meat ≥ 40 (n = 787) | P Univariate | Adjusted P b | Red Meat 40–69 g (n = 379) | P Univariate (Compared against < 40) | Adjusted P b | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Demographics. | |||||||||||
| Age | |||||||||||
| 11–18 years | 332 (34%) | 107 (26.2%) | 0.005 | ---- | 210 (35.2%) | 229 (29.1%) | 0.016 | --- | 122 (32.2%) | 0.373 | -- |
| 19–64 years | 664 (66%) | 301 (73.8%) | 387 (64.8%) | 558 (70.9%) | 257 (67.8%) | ||||||
| Weight (kg) | 64.7 (55.7–75.6) | 67 [57.4–81.8] | 0.001 * | --- | 63.9 [55.4–75.5] | 66.2 [57.2–79.1] | 0.007 | --- | 67.6 (15.9) | 0.285 | -- |
| Micronutrient intakes. | |||||||||||
| Fe < LRNI | 340 (34.8%) | 82 (20.1%) | <0.001 * | 0.166 | 225 (37.7%) | 197 (25%) | <0.001 * | 0.357 | 115 (30.3%) | 0.019 | |
| Zn < LRNI | 131 (13.4%) | 4 (1%) | <0.001 * | <0.001 * | 113 (18.9%) | 22 (2.8%) | <0.001 * | <0.001 * | 18 (4.7%) | <0.001 * | <0.001 * |
| Vit. B2 < LRNI | 159 (16.3%) | 44 (10.8%) | 0.008 | 0.829 | 107 (17.9%) | 96 (12.2%) | 0.003 | 0.2 | 107 (17.9%) | 0.083 | |
| Vit. B12 < LRNI | 23 (2.4%) | 0 (0%) | 0.002 | 0.99 | 22 (3.7%) | 1 (0.1%) | <0.001 * | 0.991 | 1 (0.3%) | <0.001 * | 0.99 |
| K < LRNI | 275 (28.2%) | 73 (17.9%) | <0.001 * | 0.592 | 186 (31.2%) | 162 (20.6%) | <0.001 * | 0.156 | 89 (23.5%) | 0.009 | |
| Se < LRNI | 497 (50.9%) | 174 (42.6%) | 0.005 | <0.001 * | 306 (51.3%) | 365 (46.4%) | 0.072 | <0.001 * | 191 (50.4%) | 0.973 | |
| Vit. D intake (µg) | 1.74 (1.1–2.85) | 2.26 (1.63–3.31) | <0.001 * | <0.001 * | 1.6 (1–2.74) | 2.12 (1.47–3.18) | <0.001 * | <0.001 * | 1.98 (1.2–3) | <0.001 * | <0.001 * |
| Blood analytes a. | |||||||||||
| Hemoglobin (g/dL) | 13.2 (12.6–13.8) | 13.3 (12.9–13.9) | 0.193 | --- | 13.2 (12.6–13.8) | 13.3 (12.8–13.9) | 0.296 | --- | 13.3 (12.6–13.8) | 0.361 | -- |
| Ferritin (µg/L) | 31.1 (18.5–61.5) | 45.4 (23–79.6) | 0.014 | --- | 29.3 (16.4–55.9) | 40.3 (20.6–72.6) | 0.065 | --- | 33.2 (19.7–66.6) | 0.109 | -- |
| Zn (µmol/L) | 14.1 (12.5–14.1) | 13.9 (12.5–15.5) | 0.572 | --- | 14.3 (12.6–15.6) | 13.9 (12.2–15.3) | 0.289 | --- | 13.8 (12.5–15.6) | 0.454 | -- |
| Vit. B12 (pmol/L) | 256 (198–314) | 256 (211–303) | 0.776 | --- | 258 (198–311) | 256 (206–316) | 0.876 | --- | 254 (195–327) | 0.907 | -- |
| Vit. D (nmol/L) | 40.2 (25.3–58.4) | 44.5 (29.7–62.6) | 0.075 | --- | 41.1 (27.9–58.4) | 42.1 (26.3–61.4) | 0.844 | --- | 39.2 (22.8–58.3) | 0.386 | -- |
| Iron Intake below LRNI | Zinc Intake below LRNI | |||
|---|---|---|---|---|
| Model R2 Total = 0.518 | Model R2 Total = 0.544 | |||
| Independent Variable | Relative Risk (95% CI) | p | Relative Risk (95% CI) | p |
| Daily Red meat intake (<40 g daily compared to 40–69 g) | ---- | --- | 4.6 (2.28–9.28) | <0.001 * |
| Total fruits and vegetables | 0.992 (0.99–0.994) | <0.001 * | 0.995 (0.993–0.998) | <0.001 * |
| Total white meat intake | --- | --- | --- | --- |
| Total fish intake | --- | --- | --- | --- |
| Age (adults) | 0.472 (0.311–0.716) | <0.001 * | 0.1 (0.052–0.194) | <0.001 * |
| BMI (<25 kg/m2) | ---- | --- | --- | --- |
| QIMD | --- | --- | --- | --- |
| Energy intake (Kcal/day) | 0.996 (0.996–0.997) | <0.001 * | 0.995 (0.994–0.996) | <0.001 * |
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Derbyshire, E. Associations between Red Meat Intakes and the Micronutrient Intake and Status of UK Females: A Secondary Analysis of the UK National Diet and Nutrition Survey. Nutrients 2017, 9, 768. https://doi.org/10.3390/nu9070768
Derbyshire E. Associations between Red Meat Intakes and the Micronutrient Intake and Status of UK Females: A Secondary Analysis of the UK National Diet and Nutrition Survey. Nutrients. 2017; 9(7):768. https://doi.org/10.3390/nu9070768
Chicago/Turabian StyleDerbyshire, Emma. 2017. "Associations between Red Meat Intakes and the Micronutrient Intake and Status of UK Females: A Secondary Analysis of the UK National Diet and Nutrition Survey" Nutrients 9, no. 7: 768. https://doi.org/10.3390/nu9070768
APA StyleDerbyshire, E. (2017). Associations between Red Meat Intakes and the Micronutrient Intake and Status of UK Females: A Secondary Analysis of the UK National Diet and Nutrition Survey. Nutrients, 9(7), 768. https://doi.org/10.3390/nu9070768





