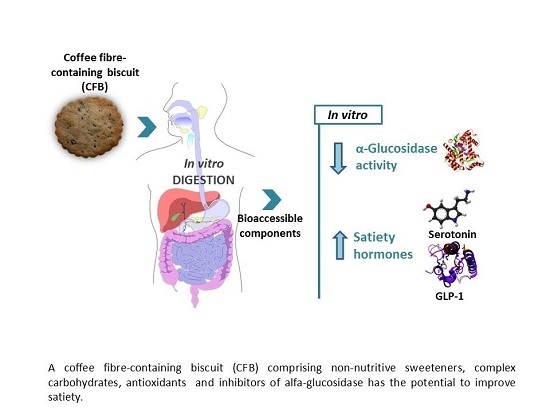
Biscuits with No Added Sugar Containing Stevia, Coffee Fibre and Fructooligosaccharides Modifies α-Glucosidase Activity and the Release of GLP-1 from HuTu-80 Cells and Serotonin from Caco-2 Cells after In Vitro Digestion
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Apparatus and Materials
2.3. Food Ingredients
2.4. Food Samples
2.5. Cell Culture: Caco-2 and HuTu-80
2.6. Bioaccessibility of Food Components
2.6.1. Total Carbohydrates
2.6.2. Galactomannan
2.6.3. Free Sugars
2.6.4. Soluble Proteins and Peptides
2.6.5. Free Amino Groups
2.6.6. Total Phenolic Content
2.6.7. Antioxidant Capacity
2.7. Health-Promoting Properties of Bioaccessible Food Components
2.7.1. Alpha-Glucosidase Inhibition Assay
2.7.2. Assays of Serotonin and GLP-1 Secretion
- Cell viability: Cytotoxic effects of test samples were excluded by performing the colorimetric MTT assay [20]. Differentiated Caco-2 cells were incubated with samples (0.05, 0.5 and 5 mg/mL) diluted in PBS containing ascorbic acid (0.1%) and after a 5-min exposure to cells, samples were removed. HuTu-80 cells were first starved with a serum free medium, glucose and glutamine, for 1 h prior to the incubation with the samples (0.01, 0.05 and 0.5 mg/mL) diluted in the starving-medium. After 90-min of exposure, samples were finally removed from HuTu-80 cells.
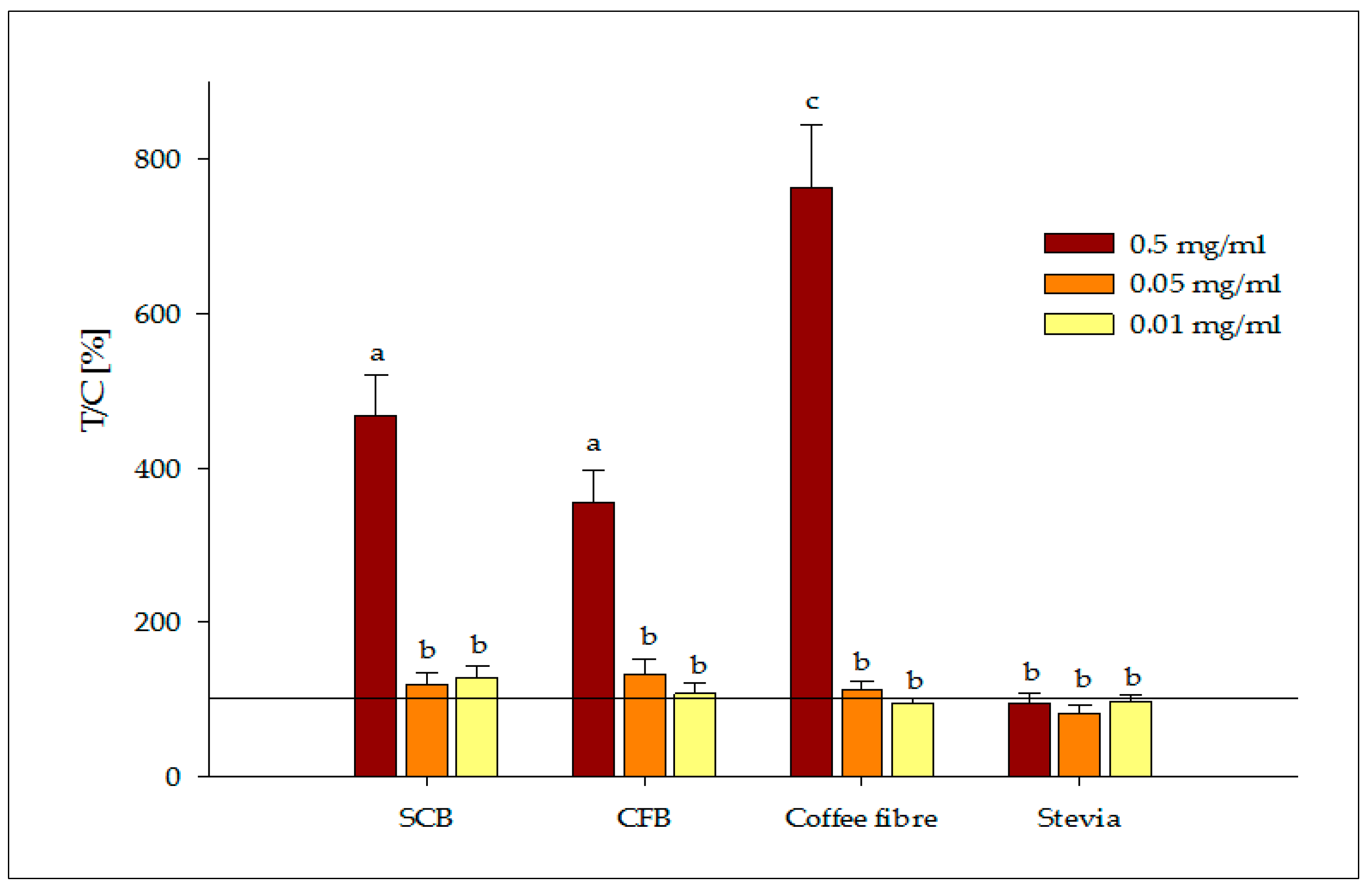
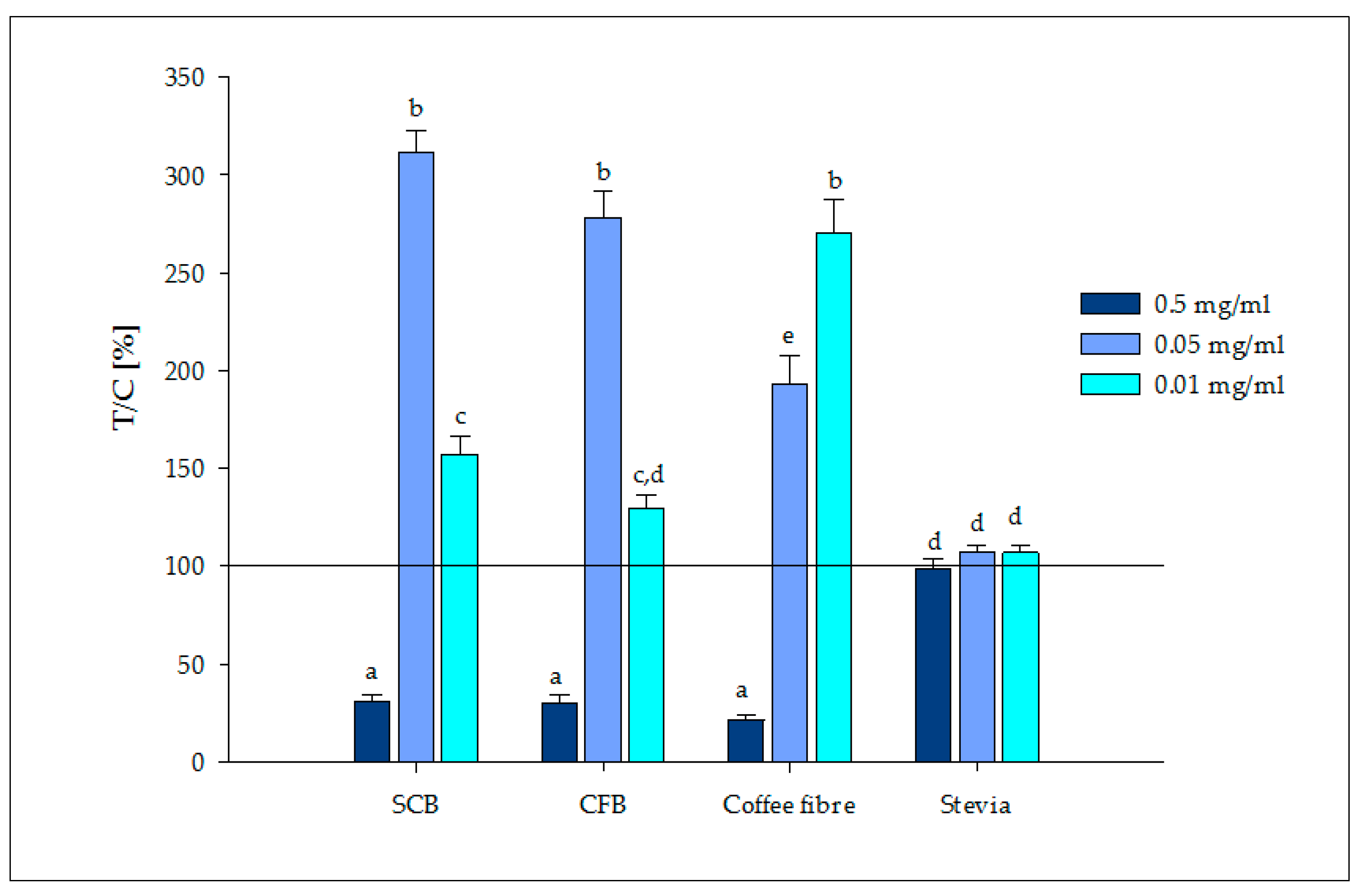
- Stimulation and quantification of serotonin: Caco-2 cells were supplemented with the bioaccessible fractions (150 μL) at three different concentrations (0.5, 0.05, 0.01 mg/mL) in duplicate. Cells were washed with PBS prior to the addition of samples. Then, cells were stimulated for 5 min in darkness in an orbital shaker. Supernatants were removed from cells and frozen until further quantification of the serotonin hormone could be completed. Positive (cinnamaldehyde, 5 mM) and negative (buffer) controls were also tested.
- Stimulation and quantification of GLP-1: HuTu-80 cells were first starved with a serum free medium, glucose and glutamine, for 1 h prior to stimulation with the bioaccessible fractions (500 μL) at 0.01, 0.05 and 0.5 mg/mL in duplicate. Positive (glutamine 40 mM) and negative (medium) controls were also tested. Cells were exposed to samples for 90 min at 37 °C. After stimulation, supernatants were collected and frozen until further quantification of the GLP-1 hormone could be completed.
2.8. Statistical Analysis
3. Results and Discussion
3.1. Bioaccessibility of Food Components
3.2. Health-Promoting Properties of Foods
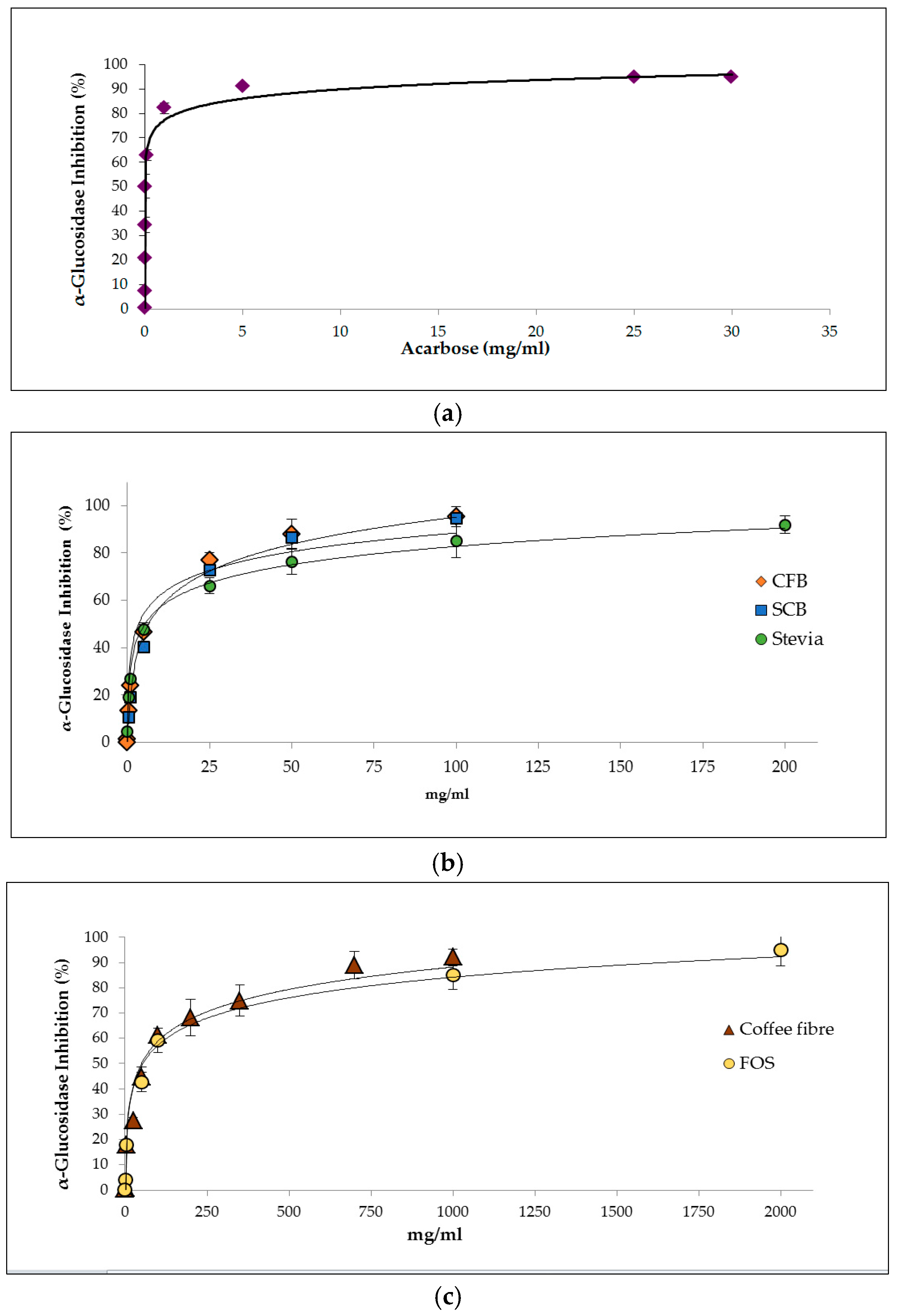
3.2.1. Inhibition of α-Glucosidase Activity
3.2.2. Release of Satiety Hormones
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cornelsen, L.; Carreido, A. Health Related Taxes on Foods and Beverages; Food Research Collaboration: London, UK, 2015; pp. 1–23. [Google Scholar]
- World Health Organization (WHO). Guideline: Sugars Intake for Adults and Children; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Van Kleef, E.; Van Trijp, J.C.M.; Van Den Borne, J.J.G.C.; Zondervan, C. Successful development of satiety enhancing food products: Towards a multidisciplinary agenda of research challenges. Crit. Rev. Food Sci. Nutr. 2012, 52, 611–628. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Saez, N.; Tamargo, A.; Domínguez, I.; Rebollo-Hernanz, M.; Mesías, M.; Morales, F.J.; Martín-Cabrejas, M.A.; del Castillo, M.D. Use of spent coffee grounds as food ingredient in bakery products. Food Chem. 2017, 216, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Kafka, A.; Kang, B.H.; Goundra, R.; Kwon, Y.I.; Apostolidis, E. In vitro evaluation and determination of responsible fraction of coffee beans and dried sugar beet leaves for alpha-glucosidase inhibition. Int. J. Appl. Res. Nat. Prod. 2014, 7, 15–20. [Google Scholar]
- Mathern, J.R.; Raatz, S.K.; Thomas, W.; Slavin, J.L. Effect of fenugreek fiber on satiety, blood glucose and insulin response and energy intake in obese subjects. Phytother. Res. 2009, 23, 1543–1548. [Google Scholar] [CrossRef] [PubMed]
- Hamden, K.; Jaouadi, B.; Carreau, S.; Bejar, S.; Elfeki, A. Inhibitory effect of fenugreek galactomannan on digestive enzymes related to diabetes, hyperlipidemia, and liver-kidney dysfunctions. Biotechnol. Bioprocess. Eng. 2010, 15, 407–413. [Google Scholar] [CrossRef]
- Dreher, M.L. Role of fiber and healthy dietary patterns in body weight regulation and weight loss. Adv. Obes. Weight Manag. Control 2015, 3, 00068. [Google Scholar] [CrossRef]
- De Munter, J.S.L.; Hu, F.B.; Spiegelman, D.; Franz, M.; van Dam, R.M. Whole grain, bran, and germ intake and risk of type 2 diabetes: A prospective cohort study and systematic review. PLoS Med. 2007, 4, e261. [Google Scholar] [CrossRef] [PubMed]
- Majouli, K.; Hlila, M.B.; Hamdi, A.; Flamini, G.; Jannet, H.B.; Kenani, A. Antioxidant activity and α-glucosidase inhibition by essential oils from Hertia cheirifolia (L.). Ind. Crop. Prod. 2016, 82, 23–28. [Google Scholar] [CrossRef]
- Ruiz-Ruiz, J.C.; Moguel-Ordoñez, Y.B.; Matus-Basto, A.J.; Segura-Campos, M.R. Antidiabetic and antioxidant activity of Stevia rebaudiana extracts (Var. Morita) and their incorporation into a potential functional bread. J. Food Sci. Technol. 2015, 52, 7894–7903. [Google Scholar] [CrossRef] [PubMed]
- Van Bloemendaal, L.; IJzerman, R.G.; Ten Kulve, J.S.; Barkhof, F.; Konrad, R.J.; Drent, M.L.; Veltman, D.J.; Diamant, M. GLP-1 receptor activation modulates appetite- and reward-related brain areas in humans. Diabetes 2014, 63, 4186–4196. [Google Scholar] [CrossRef] [PubMed]
- Mawe, G.M.; Hoffman, J.M. Serotonin signaling in the gastrointestinal tract: Functions, dysfunctions, and therapeutic targets. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 473–486. [Google Scholar] [CrossRef] [PubMed]
- Masuko, T.; Minami, A.; Iwasaki, N.; Majima, T.; Nishimura, S.I.; Lee, Y.C. Carbohydrate analysis by a phenol-sulfuric acid method in microplate format. Anal. Biochem. 2005, 339, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Go, K.; Horikawa, Y.; Garcia, R.; Villarreal, F.J. Fluorescent method for detection of cleaved collagens using O-phthaldialdehyde (OPA). J. Biochem. Biophys. Methods 2008, 70, 878–882. [Google Scholar] [CrossRef] [PubMed]
- Contini, M.; Baccelloni, S.; Massantini, R.; Anelli, G. Extraction of natural antioxidants from hazelnut (Corylus avellana L.) shell and skin wastes by long maceration at room temperature. Food Chem. 2008, 110, 659–669. [Google Scholar] [CrossRef]
- Oki, T.; Nagai, S.; Yoshinaga, M.; Nishiba, Y.; Suda, I. Contribution of β-carotene to radical scavenging capacity varies among orange-fleshed sweet potato cultivars. Food Sci. Technol. Res 2006, 12, 156–160. [Google Scholar] [CrossRef]
- Berthelot, K.; Delmotte, F.M. Purification and characterization of an α-glucosidase from Rhizobium sp. (Robinia pseudoacacia L.) strain USDA 4280. Appl. Environ. Microbiol. 1999, 65, 2907–2911. [Google Scholar] [PubMed]
- Geddes, R.; Taylor, J.A. Lysosomal glycogen storage induced by Acarbose, a 1,4-oa-glucosidase inhibitor. Biochem. J 1985, 228, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.V.; Herst, P.M.; Tan, A.S. Tetrazolium dyes as tools in cell biology: New insights into their cellular reduction. Biotechnol. Annu. Rev. 2005, 11, 127–152. [Google Scholar] [PubMed]
- Simões, J.; Nunes, F.M.; Domingues, M.R.; Coimbra, M.A. Extractability and structure of spent coffee ground polysaccharides by roasting pre-treatments. Carbohydr. Polym. 2013, 97, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.R.; Preedy, V.R.; Zibadi, S. Wheat and Rice in Disease Prevention and Health: Benefits, Risks and Mechanisms of Whole Grains in Health Promotion, 1st ed; Watson, R.R., Preedy, V., Zibad, S., Eds.; Elsevier Academic Press: San Diego, CA, USA, 2014. [Google Scholar]
- Butterworth, P.J.; Warren, F.J.; Ellis, P.R. Human α-amylase and starch digestion: An interesting marriage. Starch-Stärke 2011, 63, 395–405. [Google Scholar] [CrossRef]
- Simpson, B.K. Food Biochemistry and Food Processing; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Allen, L.; Prentice, A. Encyclopedia of Human Nutrition, 2nd ed.; Allen, L., Prentice, A., Eds.; Elsevier Academic Press: San Diego, CA, USA, 2005. [Google Scholar]
- Augustin, L.S.A.; Kendall, C.W.C.; Jenkins, D.J.A.; Willett, W.C.; Astrup, A.; Barclay, A.W.; Björck, I.; Brand-Miller, J.C.; Brighenti, F.; Buyken, A.E.; et al. Glycemic index, glycemic load and glycemic response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr. Metab. Cardiovasc. Dis. 2015, 25, 795–815. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, F.; Poli, A. The glycemic index of bread and biscuits is markedly reduced by the addition of a proprietary fiber mixture to the ingredients. Nutr. Metab. Cardiovasc. Dis. 2008, 18, 602–605. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.Z.; Empie, M.W. Fructose metabolism in humans—What isotopic tracer studies tell us. Nutr. Metab. 2012, 9, 89. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Scientific opinion on flavouring group evaluation 310 (FGE. 310): Rebaudioside A from chemical group 30. EFSA J. 2011, 9, 2181. [Google Scholar]
- Brand-Miller, J.; Foster-Powell, K.; Colagiuri, S.; Barclay, A. Low GI Diet Diabetes Handbook; Hachette: New York, NY, USA, 2012. [Google Scholar]
- Fåk, F.; Jakobsdottir, G.; Kulcinskaja, E.; Marungruang, N.; Matziouridou, C.; Nilsson, U.; Stålbrand, H.; Nyman, M. The physico-chemical properties of dietary fibre determine metabolic responses, short-chain fatty acid profiles and gut microbiota composition in rats fed low- and high-fat diets. PLoS ONE 2015, 10, e0127252. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Scientific opinion on the substantiation of a health claim related to non-digestible carbohydrates and a reduction of post-prandial glycaemic responses pursuant to Article 13 (5) of Regulation (EC) No. 1924/2006. EFSA J. 2014. [Google Scholar] [CrossRef]
- Periche, A.; Koutsidis, G.; Escriche, I. Composition of antioxidants and amino acids in Stevia leaf infusions. Plant Foods Hum. Nutr. 2014, 69, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Arraez, S.; Sahuquillo, S.; Capella, J.V.; Ortol, M.D.; Castell, M.L. Influence of healthy sweeteners (tagatose and oligofructose) on the physicochemical characteristics of orange marmalade. J. Texture Stud. 2015, 46, 272–280. [Google Scholar] [CrossRef]
- Cian, R.E.; Vioque, J.; Drago, S.R. Structure-mechanism relationship of antioxidant and ACE I inhibitory peptides from wheat gluten hydrolysate fractionated by pH. Food Res. Int. 2015, 69, 216–223. [Google Scholar] [CrossRef]
- Del Rio, D.; Agnoli, C.; Pellegrini, N.; Krogh, V.; Brighenti, F.; Mazzeo, T.; Masala, G.; Bendinelli, B.; Berrino, F.; Sieri, S.; et al. Total antioxidant capacity of the diet is associated with lower risk of ischemic stroke in a large Italian cohort. J. Nutr. 2011, 141, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Gironés-Vilaplana, A.; Moreno, D.A.; García-Viguera, C. Phytochemistry and biological activity of Spanish Citrus fruits. Food Funct. 2014, 5, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T.; Yoshimoto, C.; Osajima, K.; Oki, T.; Osajima, Y. In vitro survey of α-glucosidase inhibitory food components. Biosci. Biotechnol. Biochem. 1996, 60, 2019–2022. [Google Scholar] [CrossRef] [PubMed]
- Del Castillo, M.D.; Fernandez-Gomez, B.; Ullate, M.; Mesa, M.D. Uso de Productos de la Cascarilla de Café Para la Prevención y Tratamiento de las Patologías que Conforman el Síndrome Metabólico y de Sus Factores de Riesgo. Available online: https://patentscope.wipo.int/search/es/detail.jsf?docId=WO2016097450 (accessed on 1 July 2017).
- Patil, P.; Mandal, S.; Tomar, S.K.; Anand, S. Food protein-derived bioactive peptides in management of type 2 diabetes. Eur. J. Nutr. 2015, 54, 863–880. [Google Scholar] [CrossRef] [PubMed]
- Gostner, J.M.; Schroecksnadel, S.; Jenny, M.; Klein, A.; Ueberall, F.; Schennach, H.; Fuchs, D. Coffee extracts suppress tryptophan breakdown in mitogen-stimulated peripheral blood mononuclear cells. J. Am. Coll. Nutr. 2015, 34, 212–223. [Google Scholar] [CrossRef] [PubMed]
- Ripken, D.; van der Wielen, N.; Wortelboer, H.M.; Meijerink, J.; Witkamp, R.F.; Hendriks, H.F.J. Nutrient-induced glucagon like peptide-1 release is modulated by serotonin. J. Nutr. Biochem. 2016, 32, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Anton, S.D.; Martin, C.K.; Han, H.; Coulon, S.; Cefalu, W.T.; Geiselman, P.; Williamson, D.A. Effects of stevia, aspartame, and sucrose on food intake, satiety, and postprandial glucose and insulin levels. Appetite 2010, 55, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; DiSilvio, B.; Fernstrom, M.H.; Fernstrom, J.D. The chronic ingestion of diets containing different proteins produces marked variations in brain tryptophan levels and serotonin synthesis in the rat. Neurochem. Res. 2011, 36, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lan, N.; Ren, J.; Wu, Y.; Wang, S.; Huang, X.F.; Yu, Y. Orientin improves depression-like behavior and BDNF in chronic stressed mice. Mol. Nutr. Food Res. 2015, 59, 1130–1142. [Google Scholar] [CrossRef] [PubMed]
- Holik, A.-K.; Rohm, B.; Somoza, M.M.; Somoza, V. N(ε)-Carboxymethyllysine (CML), a Maillard reaction product, stimulates serotonin release and activates the receptor for advanced glycation end products (RAGE) in SH-SY5Y cells. Food Funct. 2013, 4, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Serna, E.; Martinez-Saez, N.; Mesias, M.; Morales, F.J.; del Castillo, M.D. Use of coffee silverskin and stevia to improve the formulation of biscuits. Pol. J. Food Nutr. Sci. 2014, 64, 243–251. [Google Scholar] [CrossRef]
- Crane, J.D.; Palanivel, R.; Mottillo, E.P.; Bujak, A.L.; Wang, H.; Ford, R.J.; Collins, A.; Blümer, R.M.; Fullerton, M.D.; Yabut, J.M.; et al. Inhibiting peripheral serotonin synthesis reduces obesity and metabolic dysfunction by promoting brown adipose tissue thermogenesis. Nat. Med. 2014, 21, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.G.; Bloom, S.R. Gut hormones and the regulation of energy homeostasis. Nature 2006, 444, 854–859. [Google Scholar] [CrossRef] [PubMed]
- Hochkogler, C.M.; Lieder, B.; Rust, P.; Berry, D.; Meier, S.M.; Pignitter, M.; Riva, A.; Leitinger, A.; Bruk, A.; Wagner, S.; et al. A 12-week intervention with nonivamide, a TRPV1 agonist, prevents a dietary-induced body fat gain and increases peripheral serotonin in moderately overweight subjects. Mol. Nutr. Food Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Jokura, H.; Watanabe, I.; Umeda, M.; Hase, T.; Shimotoyodome, A. Coffee polyphenol consumption improves postprandial hyperglycemia associated with impaired vascular endothelial function in healthy male adults. Nutr. Res. 2015, 35, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Fujita, Y.; Wideman, R.D.; Speck, M.; Asadi, A.; King, D.S.; Webber, T.D.; Haneda, M.; Kieffer, T.J. Incretin release from gut is acutely enhanced by sugar but not by sweeteners in vivo. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E473–E479. [Google Scholar] [CrossRef] [PubMed]
- Frost, G.; Sleeth, M.L.; Sahuri-Arisoylu, M.; Lizarbe, B.; Cerdan, S.; Brody, L.; Anastasovska, J.; Ghourab, S.; Hankir, M.; Zhang, S.; et al. The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat. Commun. 2014, 5, 3611. [Google Scholar] [CrossRef] [PubMed]
- Hochkogler, C.M.; Rohm, B.; Hojdar, K.; Pignitter, M.; Widder, S.; Ley, J.P.; Krammer, G.E.; Somoza, V. The capsaicin analog nonivamide decreases total energy intake from a standardized breakfast and enhances plasma serotonin levels in moderately overweight men after administered in an oral glucose tolerance test: A randomized, crossover trial. Mol. Nutr. Food Res. 2014, 58, 1282–1290. [Google Scholar] [CrossRef] [PubMed]
- Geraedts, M.C.P.; Troost, F.J.; Fischer, M.A.; Edens, L.; Saris, W.H. Direct induction of CCK and GLP-1 release from murine endocrine cells by intact dietary proteins. Mol. Nutr. Food Res. 2011, 55, 476–484. [Google Scholar] [CrossRef] [PubMed]
| Ingredients (g) | SCB | CFB |
|---|---|---|
| Wheat flour | 56.00 | 59.40 |
| Water | 20.00 | 21.20 |
| Sunflower oil | 7.75 | 8.20 |
| Baking powder | 0.55 | 0.58 |
| Salt | 0.37 | 0.39 |
| Soy lecithin | 0.33 | 0.35 |
| Sucrose | 15.00 | - |
| Stevia | - | 2.10 |
| FOS | - | 3.50 |
| Coffee fibre | - | 4.20 |
| TOTAL dough | 100 | 100 |
| Estimated fibre content (g fibre/100 g biscuit) | 1.90 | 7.50 1 |
| Bioaccessible Compounds | SCB | CFB | Coffee Fibre | Stevia |
|---|---|---|---|---|
| Total Carbohydrates | ||||
| mg glucose eq./g digest | 647.01 ± 70.0 a | 609.34 ± 17.20 a | 113.66 ± 7.57 b | n.d. |
| Galactomannan | ||||
| mg/g digest | ND | 1.60 ± 0.11 a | 19.49 ± 1.19 b | n.d. |
| Sugars | ||||
| mg glucose/g digest | 54.84 ± 9.94 a | 28.78 ± 0.65 b | 0.01 ± 0.00 c | 40.02 ± 0.57 d |
| mg fructose/g digest | 60.91 ± 3.31 a | 39.21 ± 1.48 b | 0.21 ± 0.01 c | 6.20 ± 0.46 d |
| mg mannose/g digest | 0.74 ± 0.11 a | 0.59 ± 0.11 a | 0.29 ± 0.01 b | ND |
| Soluble proteins | ||||
| mg BSA eq./g digest | 4.11 ± 0.09 a | 3.88 ± 0.30 a | 8.75 ± 0.28 b | n.d. |
| Free amino groups | ||||
| mg Nα-acetyl-l-lysine eq./g digest | 7.95 ± 0.19 a | 9.16 ± 0.83 a | 24.0 ± 2.53 b | n.d. |
| Total phenolic content | ||||
| mg CGA eq./g digest | 8.86 ± 0.49 a | 8.98 ± 0.52 a | 15.56 ± 0.95 b | n.d. |
| Samples | IC50 (mg/mL) | mg Acarbose eq./g Digest |
|---|---|---|
| Acarbose | 0.004 ± 0.00 a | - |
| Stevia | 5.53 ± 0.35 b,c | 0.79 ± 0.05 |
| SCB | 6.22 ± 0.33 b | 0.70 ± 0.04 |
| CFB | 3.32 ± 0.12 c | 1.32 ± 0.05 |
| Coffee fibre | 23.9 ± 1.31 d | 0.18 ± 0.01 |
| FOS | 53.4 ± 2.22 e | 0.08 ± 0.00 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinez-Saez, N.; Hochkogler, C.M.; Somoza, V.; Del Castillo, M.D. Biscuits with No Added Sugar Containing Stevia, Coffee Fibre and Fructooligosaccharides Modifies α-Glucosidase Activity and the Release of GLP-1 from HuTu-80 Cells and Serotonin from Caco-2 Cells after In Vitro Digestion. Nutrients 2017, 9, 694. https://doi.org/10.3390/nu9070694
Martinez-Saez N, Hochkogler CM, Somoza V, Del Castillo MD. Biscuits with No Added Sugar Containing Stevia, Coffee Fibre and Fructooligosaccharides Modifies α-Glucosidase Activity and the Release of GLP-1 from HuTu-80 Cells and Serotonin from Caco-2 Cells after In Vitro Digestion. Nutrients. 2017; 9(7):694. https://doi.org/10.3390/nu9070694
Chicago/Turabian StyleMartinez-Saez, Nuria, Christina Maria Hochkogler, Veronika Somoza, and Maria Dolores Del Castillo. 2017. "Biscuits with No Added Sugar Containing Stevia, Coffee Fibre and Fructooligosaccharides Modifies α-Glucosidase Activity and the Release of GLP-1 from HuTu-80 Cells and Serotonin from Caco-2 Cells after In Vitro Digestion" Nutrients 9, no. 7: 694. https://doi.org/10.3390/nu9070694
APA StyleMartinez-Saez, N., Hochkogler, C. M., Somoza, V., & Del Castillo, M. D. (2017). Biscuits with No Added Sugar Containing Stevia, Coffee Fibre and Fructooligosaccharides Modifies α-Glucosidase Activity and the Release of GLP-1 from HuTu-80 Cells and Serotonin from Caco-2 Cells after In Vitro Digestion. Nutrients, 9(7), 694. https://doi.org/10.3390/nu9070694