A Review of Recruitment, Adherence and Drop-Out Rates in Omega-3 Polyunsaturated Fatty Acid Supplementation Trials in Children and Adolescents
Abstract
:1. Introduction
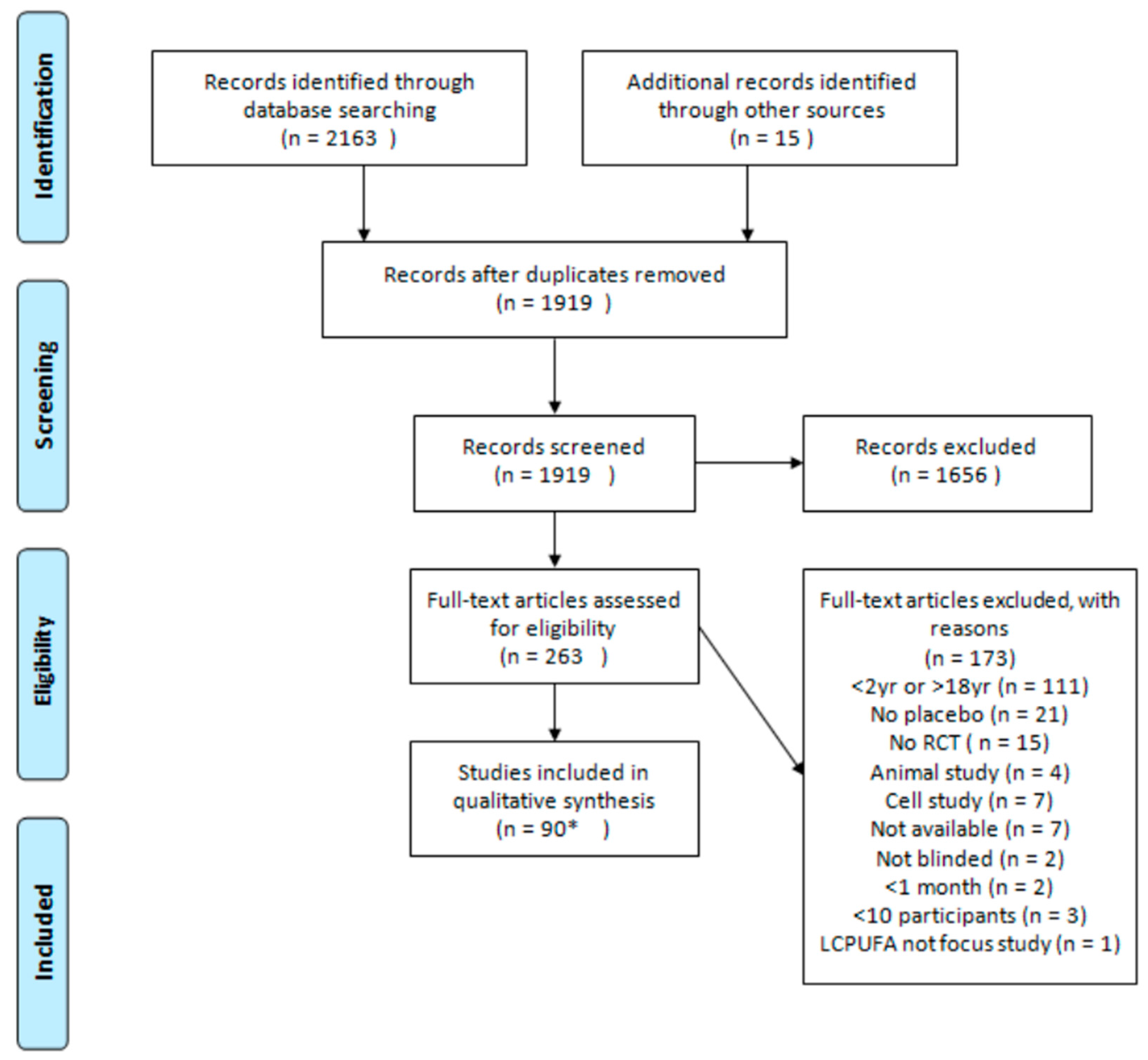
2. Materials and Methods
- Invited: The total number of potential participants invited to participate;
- Responded: The total number of potential participants who responded to the invitation or the number of participants that were screened for participation in the study; and
- Started: The number of participants who were assessed as eligible and began supplementation.
Statistics
3. Results
3.1. Study Characteristics
3.2. Recruitment
3.3. Supplementation
3.4. Adherence
3.5. Drop-Out
4. Discussion
4.1. Recruitment
4.2. Adherence
4.3. Drop-Out
4.4. Strengths and Limitations
5. Conclusions
6. Recommendations
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Deacon, G.; Kettle, C.; Hayes, D.; Dennis, C.; Tucci, J. Omega 3 polyunsaturated fatty acids and the treatment of depression. Crit. Rev. Food Sci. Nutr. 2017, 57, 212–223. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Harris, W.S.; Chung, M.; Lichtenstein, A.H.; Balk, E.M.; Kupelnick, B.; Jordan, H.S.; Lau, J. N-3 fatty acids from fish or fish-oil supplements, but not α-linolenic acid, benefit cardiovascular disease outcomes in primary- and secondary-prevention studies: A systematic review. Am. J. Clin. Nutr. 2006, 84, 5–17. [Google Scholar] [PubMed]
- Joffre, C.; Nadjar, A.; Lebbadi, M.; Calon, F.; Laye, S. N-3 lcpufa improves cognition: The young, the old and the sick. Prostaglandins Leukot. Essent. Fat. Acids 2014, 91, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Akobeng, A. Understanding randomised controlled trials. Arch. Dis. Child 2005, 90, 840–844. [Google Scholar] [CrossRef] [PubMed]
- Robinson, L.; Adair, P.; Coffey, M.; Harris, R.; Burnside, G. Identifying the participant characteristics that predict recruitment and retention of participants to randomised controlled trials involving children: A systematic review. Trials 2016, 17, 294. [Google Scholar] [CrossRef] [PubMed]
- McDonald, A.M.; Knight, R.C.; Campbell, M.K.; Entwistle, V.A.; Grant, A.M.; Cook, J.A.; Elbourne, D.R.; Francis, D.; Garcia, J.; Roberts, I. What influences recruitment to randomised controlled trials? A review of trials funded by two uk funding agencies. Trials 2006, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Watson, J.M.; Torgerson, D.J. Increasing recruitment to randomised trials: A review of randomised controlled trials. BMC Med. Res. Methodol. 2006, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Denhoff, E.R.; Milliren, C.E.; de Ferranti, S.D.; Steltz, S.K.; Osganian, S.K. Factors associated with clinical research recruitment in a pediatric academic medical center—A web-based survey. PLoS ONE 2015, 10, e0140768. [Google Scholar] [CrossRef] [PubMed]
- Steinbeck, K.; Baur, L.; Cowell, C.; Pietrobelli, A. Clinical research in adolescents: Challenges and opportunities using obesity as a model. Int. J. Obes. 2009, 33, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Crutzen, R.; Viechtbauer, W.; Kotz, D.; Spigt, M. No differential attrition was found in randomized controlled trials published in general medical journals: A meta-analysis. J. Clin. Epidemiol. 2013, 66, 948–954. [Google Scholar] [CrossRef] [PubMed]
- Crutzen, R.; Viechtbauer, W.; Spigt, M.; Kotz, D. Differential attrition in health behaviour change trials: A systematic review and meta-analysis. Psychol. Health 2015, 30, 122–134. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, C.E.; Kumaravel, B.; Dumville, J.C.; Torgerson, D.J.; Group, T.A.S. Assessing the impact of attrition in randomized controlled trials. J. Clin. Epidemiol. 2010, 63, 1264–1270. [Google Scholar] [CrossRef] [PubMed]
- Aronson, J.K. Compliance, concordance, adherence. Br. J. Clin. Pharmacol. 2007, 63, 383–384. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.T.; Bussell, J.K. Medication adherence: Who cares? Mayo Clin. Proce. 2011, 86, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Howie, E.K.; Straker, L.M. Rates of attrition, non-compliance and missingness in randomized controlled trials of child physical activity interventions using accelerometers: A brief methodological review. J. Sci. Med. Sport 2016, 19, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Reijen, M.; Vriend, I.; Mechelen, W.; Finch, C.F.; Verhagen, E.A. Compliance with sport injury prevention interventions in randomised controlled trials: A systematic review. Sports Med. 2016, 46, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Fewtrell, M.S.; Kennedy, K.; Singhal, A.; Martin, R.M.; Ness, A.; Hadders-Algra, M.; Koletzko, B.; Lucas, A. How much loss to follow-up is acceptable in long-term randomised trials and prospective studies? Arch. Dis. Child 2008, 93, 458–461. [Google Scholar] [CrossRef] [PubMed]
- Montori, V.M.; Guyatt, G.H. Intention-to-treat principle. CMAJ Can. Med. Assoc. J. 2001, 165, 1339–1341. [Google Scholar]
- Van der Wurff, I.; von Schacky, C.; Berge, K.; Kirschner, P.; de Groot, R. A protocol for a randomised controlled trial investigating the effect of increasing omega-3 index with krill oil supplementation on learning, cognition, behaviour and visual processing in typically developing adolescents. BMJ Open 2016, 6, e011790. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Braegger, C.; Decsi, T.; Kolacek, S.; Mihatsch, W.; Moreno, L.A.; Puntis, J.; Shamir, R.; Szajewska, H.; Turck, D.; et al. Supplementation of n-3 lcpufa to the diet of children older than 2 years: A commentary by the espghan committee on nutrition. J. Pediatr. Gastr. Nutr. 2011, 53, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Transler, C.; Eilander, A.; Mitchell, S.; van de Meer, N. The impact of polyunsaturated fatty acids in reducing child attention deficit and hyperactivity disorders. J. Atten. Disord. 2010, 14, 232–246. [Google Scholar] [CrossRef] [PubMed]
- Vesco, A.T.; Lehmann, J.; Gracious, B.L.; Arnold, L.E.; Young, A.S.; Fristad, M.A. Omega-3 supplementation for psychotic mania and comorbid anxiety in children. J. Child Adolesc. Psychopharmacol. 2015, 25, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Kuratko, C.N.; Barrett, E.C.; Nelson, E.B.; Salem, N. The relationship of docosahexaenoic acid (dha) with learning and behavior in healthy children: A review. Nutrients 2013, 5, 2777–2810. [Google Scholar] [CrossRef] [PubMed]
- Penagini, F.; Dilillo, D.; Borsani, B.; Cococcioni, L.; Galli, E.; Bedogni, G.; Zuin, G.; Zuccotti, G.V. Nutrition in pediatric inflammatory bowel disease: From etiology to treatment. A systematic review. Nutrients 2016, 8, 334. [Google Scholar] [CrossRef] [PubMed]
- Ortega, R.; Rodríguez-Rodríguez, E.; López-Sobaler, A. Effects of omega 3 fatty acids supplementation in behavior and non-neurodegenerative neuropsychiatric disorders. Br. J. Nutr. 2012, 107, S261–S270. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.J.; Ross, M. Fatty acid metabolism in neurodevelopmental disorder: A new perspective on associations between attention-deficit/hyperactivity disorder, dyslexia, dyspraxia and the autistic spectrum. Prostaglandin Leukot. Essent. 2000, 63, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Roux, C. Use of omega-3 for improving behavioural outcomes in autism spectrum disorder in children: A review of the literature. Aust. J. Herb. Med. 2015, 27, 105–111. [Google Scholar]
- Taylor, R.; Connock, M. Effects of Oily Fish/Omega-3 Fatty Acids on Behavioural, Cogitative and Educational Outcomes of Normal School Children: A Systematic Review; Department of Public Health and Epidemiology Report; University of Birmingham: Birmingham, UK, 2007. [Google Scholar]
- Eilander, A.; Hundscheid, D.; Osendarp, S.; Transler, C.; Zock, P. Effects of n-3 long chain polyunsaturated fatty acid supplementation on visual and cognitive development throughout childhood: A review of human studies. Prostaglandin Leukot. Essent. 2007, 76, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Frensham, L.J.; Bryan, J.; Parletta, N. Influences of micronutrient and omega-3 fatty acid supplementation on cognition, learning, and behavior: Methodological considerations and implications for children and adolescents in developed societies. Nutr. Rev. 2012, 70, 594–610. [Google Scholar] [CrossRef] [PubMed]
- Gajos, J.M.; Beaver, K.M. The effect of omega-3 fatty acids on aggression: A meta-analysis. Neurosci. Biobehav. Rev. 2016, 69, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Kidd, P.M. Omega-3 dha and epa for cognition, behavior, and mood: Clinical findings and structural-functional synergies with cell membrane phospholipids. Altern. Med. Rev. 2007, 12, 207. [Google Scholar] [PubMed]
- Königs, A.; Kiliaan, A.J. Critical appraisal of omega-3 fatty acids in attention-deficit/hyperactivity disorder treatment. Neuropsychiatr. Dis. Treat. 2016, 12, 1869. [Google Scholar] [PubMed]
- Bryan, J.; Osendarp, S.; Hughes, D.; Calvaresi, E.; Baghurst, K.; van Klinken, J.-W. Nutrients for cognitive development in school-aged children. Nutr. Rev. 2004, 62, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Clayton, E.H.; Hanstock, T.L.; Garg, M.L.; Hazell, P.L. Long chain omega-3 polyunsaturated fatty acids in the treatment of psychiatric illnesses in children and adolescents. Acta Neuropsychiatr. 2007, 19, 92–103. [Google Scholar] [CrossRef] [PubMed]
- Cooper, R.E.; Tye, C.; Kuntsi, J.; Vassos, E.; Asherson, P. Omega-3 polyunsaturated fatty acid supplementation and cognition: A systematic review and meta-analysis. J. Psychopharmacol. 2015, 29, 753. [Google Scholar] [CrossRef] [PubMed]
- Cooper, R.E.; Tye, C.; Kuntsi, J.; Vassos, E.; Asherson, P. The effect of omega-3 polyunsaturated fatty acid supplementation on emotional dysregulation, oppositional behaviour and conduct problems in adhd: A systematic review and meta-analysis. J. Affect. Disord. 2016, 190, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Bent, S.; Bertoglio, K.; Hendren, R.L. Omega-3 fatty acids for autistic spectrum disorder: A systematic review. J. Autism Dev. Disord. 2009, 39, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Bloch, M.H.; Qawasmi, A. Omega-3 fatty acid supplementation for the treatment of children with attention-deficit/hyperactivity disorder symptomatology: Systematic review and meta-analysis. J. Am. Acad. Child Psychiatry 2011, 50, 991–1000. [Google Scholar] [CrossRef] [PubMed]
- Bonafini, S.; Antoniazzi, F.; Maffeis, C.; Minuz, P.; Fava, C. Beneficial effects of ω-3 pufa in children on cardiovascular risk factors during childhood and adolescence. Prostaglandin Other Lipid Mediat. 2015, 120, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Campbell, A.; Price, J.; Hiatt, W.R. Omega-3 fatty acids for intermittent claudication. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef]
- Gillies, D.; Sinn, J.K.H.; Lad, S.S.; Leach, M.J.; Ross, M.J. Polyunsaturated fatty acids (pufa) for attention deficit hyperactivity disorder (adhd) in children and adolescents. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]
- De Ley, M.; de Vos, R.; Hommes, D.W.; Stokkers, P.C. Fish oil for induction of remission in ulcerative colitis. Cochrane Database Syst. Rev. 2007, 4, CD005986. [Google Scholar]
- Bath-Hextall, F.J.; Jenkinson, C.; Humphreys, R.; Williams, H.C. Dietary supplements for established atopic eczema. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]
- Thien, F.C.K.; De Luca, S.; Woods, R.K.; Abramson, M.J. Dietary marine fatty acids (fish oil) for asthma in adults and children. Cochrane Database Syst. Rev. 2002. [Google Scholar] [CrossRef]
- Pattanittum, P.; Kunyanone, N.; Brown, J.; Sangkomkamhang, U.S.; Barnes, J.; Seyfoddin, V.; Marjoribanks, J. Dietary supplements for dysmenorrhoea. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef]
- Irving, C.B.; Mumby-Croft, R.; Joy, L.A. Polyunsaturated fatty acid supplementation for schizophrenia. Cochrane Database Syst. Rev. 2006. [Google Scholar] [CrossRef]
- Tan, M.L.; Ho, J.J.; Teh, K.H. Polyunsaturated fatty acids (pufas) for children with specific learning disorders. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef]
- Farinotti, M.; Vacchi, L.; Simi, S.; Di Pietrantonj, C.; Brait, L.; Filippini, G. Dietary interventions for multiple sclerosis. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]
- Sarmento Vasconcelos, V.; Macedo, C.R.; de Souza Pedrosa, A.; Pereira Gomes Morais, E.; Porfirio, G.J.; Torloni, M.R. Polyunsaturated fatty acid supplementation for drug-resistant epilepsy. Cochrane Database Syst. Rev. 2016, 8, CD011014. [Google Scholar]
- Dewey, A.; Baughan, C.; Dean, T.P.; Higgins, B.; Johnson, I. Eicosapentaenoic acid (epa, an omega-3 fatty acid from fish oils) for the treatment of cancer cachexia. Cochrane Database Syst. Rev. 2007. [Google Scholar] [CrossRef]
- Oliver, C.; Watson, H. Omega-3 fatty acids for cystic fibrosis. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef]
- Hartweg, J.; Perera, R.; Montori, V.M.; Dinneen, S.F.; Neil, A.H.; Farmer, A.J. Omega-3 polyunsaturated fatty acids (pufa) for type 2 diabetes mellitus. Cochrane Database Syst. Rev. 2008. [Google Scholar] [CrossRef]
- James, S.; Montgomery, P.; Williams, K. Omega-3 fatty acids supplementation for autism spectrum disorders (asd). Cochrane Database Syst. Rev. 2011. [Google Scholar] [CrossRef]
- Lev-Tzion, R.; Griffiths, A.M.; Ledder, O.; Turner, D. Omega 3 fatty acids (fish oil) for maintenance of remission in crohn’s disease. Cochrane Database Syst. Rev. 2014. [Google Scholar] [CrossRef]
- Turner, D.; Steinhart, A.H.; Griffiths, A.M. Omega 3 fatty acids (fish oil) for maintenance of remission in ulcerative colitis. Cochrane Database Syst. Rev. 2007. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The prisma statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Elbarbary, N.S.; Ismail, E.A.R.; Farahat, R.K.; El-Hamamsy, M. Ω-3 fatty acids as an adjuvant therapy ameliorates methotrexate-induced hepatotoxicity in children and adolescents with acute lymphoblastic leukemia: A randomized placebo-controlled study. Nutrition 2016, 32, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Assareh, M.; Davari Ashtiani, R.; Khademi, M.; Jazayeri, S.; Rai, A.; Nikoo, M. Efficacy of polyunsaturated fatty acids (pufa) in the treatment of attention deficit hyperactivity disorder: A randomized, double-blind, placebo-controlled clinical trial. J. Atten. Disord. 2012, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Behdani, F.; Hebrani, P.; Naseraee, A.; Haghighi, M.B.; Akhavanrezayat, A. Does omega-3 supplement enhance the therapeutic results of methylphenidate in attention deficit hyperactivity disorder patients? J. Res. Med. Sci. 2013, 18, 653–658. [Google Scholar] [PubMed]
- Bélanger, S.A.; Vanasse, M.; Spahis, S.; Sylvestre, M.P.; Lippé, S.; L’Heureux, F.; Ghadirian, P.; Vanasse, C.M.; Levy, E. Omega-3 fatty acid treatment of children with attention-deficit hyperactivity disorder: A randomized, double-blind, placebo-controlled study. Paediatr. Child Health 2009, 14, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Bos, D.J.; Oranje, B.; Veerhoek, E.S.; Van Diepen, R.M.; Weusten, J.M.H.; Demmelmair, H.; Koletzko, B.; De Sain-Van Der Velden, M.G.M.; Eilander, A.; Hoeksma, M.; et al. Reduced symptoms of inattention after dietary omega-3 fatty acid supplementation in boys with and without attention deficit/hyperactivity disorder. Neuropsychopharmacology 2015, 40, 2298–2306. [Google Scholar] [CrossRef] [PubMed]
- Dubnov-Raz, G.; Khoury, Z.; Wright, I.; Raz, R.; Berger, I. The effect of alpha-linolenic acid supplementation on adhd symptoms in children: A randomized controlled double-blind study. Front. Hum. Neurosci. 2014, 8, 780. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, P.A.; Birberg-Thornberg, U.; Duchén, K.; Landgren, M.; Malmberg, K.; Pelling, H.; Strandvik, B.; Karlsson, T. Epa supplementation improves teacher-rated behaviour and oppositional symptoms in children with adhd. Acta Paediatr. 2010, 99, 1540–1549. [Google Scholar] [CrossRef] [PubMed]
- Hariri, M.; Djazayery, A.; Djalali, M.; Saedisomeolia, A.; Rahimi, A.; Abdolahian, E. Effect of n-3 supplementation on hyperactivity, oxidative stress and inflammatory mediators in children with attention-deficit-hyperactivity disorder. Malays. J. Nutr. 2012, 18, 329–335. [Google Scholar] [PubMed]
- Hirayama, S.; Hamazaki, T.; Terasawa, K. Effect of docosahexaenoic acid-containing food administration on symptoms of attention-deficit/hyperactivity disorder—A placebo-controlled double-blind study. Eur. J. Clin. Nutr. 2004, 58, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.; Östlund, S.; Fransson, G.; Kadesjö, B.; Gillberg, C. Omega-3/omega-6 fatty acids for attention deficit hyperactivity disorder: A randomized placebo-controlled trial in children and adolescents. J. Atten. Disord. 2009, 12, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.; Mansson, J.E.; Ostlund, S.; Fransson, G.; Areskoug, B.; Hjalmarsson, K.; Landgren, M.; Kadesjo, B.; Gillberg, C. Fatty acids in adhd: Plasma profiles in a placebo-controlled study of omega 3/6 fatty acids in children and adolescents. Atten. Deficit Hyperact. Disord. 2012, 4, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Matsudaira, T.; Gow, R.V.; Kelly, J.; Murphy, C.; Potts, L.; Sumich, A.; Ghebremeskel, K.; Crawford, M.A.; Taylor, E. Biochemical and psychological effects of omega-3/6 supplements in male adolescents with attention-deficit/hyperactivity disorder: A randomized, placebo-controlled, clinical trial. J. Child Adolesc. Psychopharmacol. 2015, 25, 775–782. [Google Scholar] [CrossRef] [PubMed]
- Milte, C.M.; Parletta, N.; Buckley, J.D.; Coates, A.M.; Young, R.M.; Howe, P.R. Increased erythrocyte eicosapentaenoic acid and docosahexaenoic acid are associated with improved attention and behavior in children with adhd in a randomized controlled three-way crossover trial. J. Atten. Disord. 2013, 19, 954–964. [Google Scholar] [CrossRef] [PubMed]
- Milte, C.M.; Parletta, N.; Buckley, J.D.; Coates, A.M.; Young, R.M.; Howe, P.R. Eicosapentaenoic and docosahexaenoic acids, cognition, and behavior in children with attention-deficit/hyperactivity disorder: A randomized controlled trial. Nutrition 2012, 28, 670–677. [Google Scholar] [CrossRef] [PubMed]
- Perera, H.; Jeewandara, K.C.; Seneviratne, S.; Guruge, C. Combined ω3 and ω6 supplementation in children with attention-deficit hyperactivity disorder (adhd) refractory to methylphenidate treatment a double-blind, placebo-controlled study. J. Child Neurol. 2012, 27, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Raz, R.; Carasso, R.L.; Yehuda, S. The influence of short-chain essential fatty acids on children with attention-deficit/hyperactivity disorder: A double-blind placebo-controlled study. J. Child Adolesc. Psychopharmacol. 2009, 19, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Sinn, N.; Bryan, J. Effect of supplementation with polyunsaturated fatty acids and micronutrients on learning and behavior problems associated with child adhd. J. Dev. Behav. Pediatr. 2007, 28, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Sinn, N.; Bryan, J.; Wilson, C. Cognitive effects of polyunsaturated fatty acids in children with attention deficit hyperactivity disorder symptoms: A randomised controlled trial. Prostaglandins Leukot. Essent. Fat. Acids 2008, 78, 311–326. [Google Scholar] [CrossRef] [PubMed]
- Stevens, L.; Zhang, W.; Peck, L.; Kuczek, T.; Grevstad, N.; Mahon, A.; Zentall, S.S.; Arnold, L.E.; Burgess, J.R. Efa supplementation in children with inattention, hyperactivity, and other disruptive behaviors. Lipids 2003, 38, 1007–1021. [Google Scholar] [CrossRef] [PubMed]
- Vaisman, N.; Kaysar, N.; Zaruk-Adasha, Y.; Pelled, D.; Brichon, G.; Zwingelstein, G.; Bodennec, J. Correlation between changes in blood fatty acid composition and visual sustained attention performance in children with inattention: Effect of dietary n-3 fatty acids containing phospholipids. Am. J. Clin. Nutr. 2008, 87, 1170–1180. [Google Scholar] [PubMed]
- Voigt, R.G.; Llorente, A.M.; Jensen, C.L.; Fraley, J.K.; Berretta, M.C.; Heird, W.C. A randomized, double-blind, placebo-controlled trial of docosahexaenoic acid supplementation in children with attention-deficit/hyperactivity disorder. J. Pediatr. 2001, 139, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Widenhorn-Müller, K.; Schwanda, S.; Scholz, E.; Spitzer, M.; Bode, H. Effect of supplementation with long-chain ω-3 polyunsaturated fatty acids on behavior and cognition in children with attention deficit/hyperactivity disorder (adhd): A randomized placebo-controlled intervention trial. Prostaglandin Leukot. Essent. 2014, 91, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Manor, I.; Magen, A.; Keidar, D.; Rosen, S.; Tasker, H.; Cohen, T.; Richter, Y.; Zaaroor-Regev, D.; Manor, Y.; Weizman, A. Safety of phosphatidylserine containing omega3 fatty acids in adhd children: A double-blind placebo-controlled trial followed by an open-label extension. Eur. Psychiatry J. Assoc. Eur. Psychiatr. 2013, 28, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Manor, I.; Magen, A.; Keidar, D.; Rosen, S.; Tasker, H.; Cohen, T.; Richter, Y.; Zaaroor-Regev, D.; Manor, Y.; Weizman, A. The effect of phosphatidylserine containing omega3 fatty-acids on attention-deficit hyperactivity disorder symptoms in children: A double-blind placebo-controlled trial, followed by an open-label extension. Eur. Psychiatry J. Assoc. Eur. Psychiatr. 2012, 27, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.L.; Zhou, T.T.; Ma, L.P.; Yuan, D.J.; Peng, Y.M. Protective effects of dietary supplementation with natural omega-3 polyunsaturated fatty acids on the visual acuity of school-age children with lower iq or attention-deficit hyperactivity disorder. Nutrition 2015, 31, 935–940. [Google Scholar] [CrossRef] [PubMed]
- Kean, J.D.; Sarris, J.; Scholey, A.; Silberstein, R.; Downey, L.A.; Stough, C. Reduced inattention and hyperactivity and improved cognition after marine oil extract (pcso-524®) supplementation in children and adolescents with clinical and subclinical symptoms of attention-deficit hyperactivity disorder (adhd): A randomised, double-blind, placebo-controlled trial. Psychopharmacology 2017, 234, 403–420. [Google Scholar] [PubMed]
- Ross, S.M. Omega-3 fatty acids, part i: The effects of n-3 polyunsaturated fatty acid in the treatment of attention-deficit hyperactivity disorder in children. Holist. Nurs. Pract. 2012, 26, 356–359. [Google Scholar] [CrossRef] [PubMed]
- Raine, A.; Portnoy, J.; Liu, J.; Mahoomed, T.; Hibbeln, J.R. Reduction in behavior problems with omega-3 supplementation in children aged 8–16 years: A randomized, double-blind, placebo-controlled, stratified, parallel-group trial. J. Child Psychol. Psychiatry 2015, 56, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Covar, R.; Gleason, M.; Macomber, B.; Stewart, L.; Szefler, P.; Engelhardt, K.; Murphy, J.; Liu, A.; Wood, S.; DeMichele, S.; et al. Impact of a novel nutritional formula on asthma control and biomarkers of allergic airway inflammation in children. Clin. Exp. Allergy 2010, 40, 1163–1174. [Google Scholar] [CrossRef] [PubMed]
- Hodge, L.; Salome, C.M.; Hughes, J.M.; Liu-Brennan, D.; Rimmer, J.; Allman, M.; Pang, D.; Armour, C.; Woolcock, A.J. Effect of dietary intake of omega-3 and omega-6 fatty acids on severity of asthma in children. Eur. Respir. J. 1998, 11, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-C.; Yang, Y.-H.; Chuang, S.-Y.; Huang, S.-Y.; Pan, W.-H. Reduced medication use and improved pulmonary function with supplements containing vegetable and fruit concentrate, fish oil and probiotics in asthmatic school children: A randomised controlled trial. Br. J. Nutr. 2013, 110, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Nagakura, T.; Matsuda, S.; Shichijyo, K.; Sugimoto, H.; Hata, K. Dietary supplementation with fish oil rich in omega-3 polyunsaturated fatty acids in children with bronchial asthma. Eur. Respir. J. 2000, 16, 861–865. [Google Scholar] [CrossRef] [PubMed]
- Bent, S.; Bertoglio, K.; Ashwood, P.; Bostrom, A.; Hendren, R.L. A pilot randomized controlled trial of omega-3 fatty acids for autism spectrum disorder. J. Autism Dev. Disord. 2011, 41, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Bent, S.; Hendren, R.L.; Zandi, T.; Law, K.; Choi, J.E.; Widjaja, F.; Kalb, L.; Nestle, J.; Law, P. Internet-based, randomized, controlled trial of omega-3 fatty acids for hyperactivity in autism. J. Am. Acad. Child Adolesc. Psychiatry 2014, 53, 658–666. [Google Scholar] [CrossRef] [PubMed]
- Mankad, D.; Dupuis, A.; Smile, S.; Roberts, W.; Brian, J.; Lui, T.; Genore, L.; Zaghloul, D.; Iaboni, A.; Marcon, P.M.A. A randomized, placebo controlled trial of omega-3 fatty acids in the treatment of young children with autism. Mol. Autism 2015, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Voigt, R.G.; Mellon, M.W.; Katusic, S.K.; Weaver, A.L.; Matern, D.; Mellon, B.; Jensen, C.L.; Barbaresi, W.J. Dietary docosahexaenoic acid supplementation in children with autism. J. Pediatr. Gastr. Nutr. 2014, 58, 715–722. [Google Scholar]
- Gracious, B.L.; Chirieac, M.C.; Costescu, S.; Finucane, T.L.; Youngstrom, E.A.; Hibbeln, J.R. Randomized, placebo-controlled trial of flax oil in pediatric bipolar disorder. Bipolar Disord. 2010, 12, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Alicandro, G.; Faelli, N.; Gagliardini, R.; Santini, B.; Magazzù, G.; Biffi, A.; Risé, P.; Galli, C.; Tirelli, A.S.; Loi, S.; et al. A randomized placebo-controlled study on high-dose oral algal docosahexaenoic acid supplementation in children with cystic fibrosis. Prostaglandin Leukot. Essent. 2013, 88, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Romano, C.; Cucchiara, S.; Barabino, A.; Annese, V.; Sferlazzas, C. Usefulness of omega-3 fatty acid supplementation in addition to mesalazine in maintaining remission in pediatric crohn’s disease: A double-blind, randomized, placebo-controlled study. World J. Gastroenterol. 2005, 11, 7118–7121. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.J.; Montgomery, P. The oxford-durham study: A randomized, controlled trial of dietary supplementation with fatty acids in children with developmental coordination disorder. Pediatrics 2005, 115, 1360–1366. [Google Scholar] [CrossRef] [PubMed]
- Kairaluoma, L.; Närhi, V.; Ahonen, T.; Westerholm, J.; Aro, M. Do fatty acids help in overcoming reading difficulties? A double-blind, placebo-controlled study of the effects of eicosapentaenoic acid and carnosine supplementation on children with dyslexia. Child Care Health Dev. 2009, 35, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Harel, Z.; Biro, F.M.; Kottenhahn, R.K.; Rosenthal, S.L. Supplementation with omega-3 polyunsaturated fatty acids in the management of dysmenorrhea in adolescents. Am. J. Obstet. Gynecol. 1996, 174, 1335–1338. [Google Scholar] [CrossRef]
- Reda, D.M.A.; Abd-El-Fatah, N.K.; Omar, T.E.-S.I.; Darwish, O.A.H. Fish oil intake and seizure control in children with medically resistant epilepsy. N. Am. J. Med. Sci. 2015, 7, 317. [Google Scholar] [CrossRef] [PubMed]
- Dalton, A.; Wolmarans, P.; Witthuhn, R.C.; van Stuijvenberg, M.E.; Swanevelder, S.A.; Smuts, C.M. A randomised control trial in schoolchildren showed improvement in cognitive function after consuming a bread spread, containing fish flour from a marine source. Prostaglandin Leukot. Essent. 2009, 80, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Hamazaki, K.; Syafruddin, D.; Tunru, I.S.; Azwir, M.F.; Asih, P.B.; Sawazaki, S.; Hamazaki, T. The effects of docosahexaenoic acid-rich fish oil on behavior, school attendance rate and malaria infection in school children-a double-blind, randomized, placebo-controlled trial in lampung, indonesia. Asia Pac. J. Clin. Nutr. 2008, 17, 258–263. [Google Scholar] [PubMed]
- Itomura, M.; Hamazaki, K.; Sawazaki, S.; Kobayashi, M.; Terasawa, K.; Watanabe, S.; Hamazaki, T. The effect of fish oil on physical aggression in schoolchildren-a randomized, double-blind, placebo-controlled trial. J. Nutr. Biochem. 2005, 16, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.; Fransson, G.; Östlund, S.; Areskoug, B.; Gillberg, C. Omega 3/6 fatty acids for reading in children: A randomized, double-blind, placebo-controlled trial in 9-year-old mainstream schoolchildren in sweden. J. Child Psychol. Psychiatry 2016. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Jackson, P.A.; Elliott, J.M.; Scholey, A.B.; Robertson, B.C.; Greer, J.; Tiplady, B.; Buchanan, T.; Haskell, C.F. Cognitive and mood effects of 8 weeks’ supplementation with 400 mg or 1000 mg of the omega-3 essential fatty acid docosahexaenoic acid (dha) in healthy children aged 10–12 years. Nutr. Neurosci. 2009, 12, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Kirby, A.; Woodward, A.; Jackson, S.; Wang, Y.; Crawford, M.A. A double-blind, placebo-controlled study investigating the effects of omega-3 supplementation in children aged 8-10 years from a mainstream school population. Res. Dev. Disabil. 2010, 31, 718–730. [Google Scholar] [CrossRef] [PubMed]
- Mazurak, V.; Lien, V.; Field, C.; Goruk, S.; Pramuk, K.; Clandinin, M. Long-chain polyunsaturated fat supplementation in children with low docosahexaenoic acid intakes alters immune phenotypes compared with placebo. J. Pediatr. Gastr. Nutr. 2008, 46, 570–579. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K.; Able, J.; Jandacek, R.; Rider, T.; Tso, P.; Eliassen, J.C.; Alfieri, D.; Weber, W.; Jarvis, K.; DelBello, M.P. Docosahexaenoic acid supplementation increases prefrontal cortex activation during sustained attention in healthy boys: A placebo-controlled, dose-ranging, functional magnetic resonance imaging study. Am. J. Clin. Nutr. 2010, 91, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Osendarp, S.J.M.; Baghurst, K.I.; Bryan, J.; Calvaresi, E.; Hughes, D.; Hussaini, M.; Karyadi, E.; Van Klinken, B.J.-W.; Van Der Knaap, H.C.M.; Lukito, W.; et al. Effect of a 12-mo micronutrient intervention on learning and memory in well-nourished and marginally nourished school-aged children: 2 parallel, randomized, placebo-controlled studies in australia and indonesia. Am. J. Clin. Nutr. 2007, 86, 1082–1093. [Google Scholar] [PubMed]
- Parletta, N.; Cooper, P.; Gent, D.N.; Petkov, J.; O’Dea, K. Effects of fish oil supplementation on learning and behaviour of children from australian indigenous remote community schools: A randomised controlled trial. Prostaglandin Leukot. Essent. 2013, 89, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Romeo, J.; Wärnberg, J.; García-Mármol, E.; Rodríguez-Rodríguez, M.; Diaz, L.E.; Gomez-Martínez, S.; Cueto, B.; López-Huertas, E.; Cepero, M.; Boza, J.J. Daily consumption of milk enriched with fish oil, oleic acid, minerals and vitamins reduces cell adhesion molecules in healthy children. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Ryan, A.S.; Nelson, E.B. Assessing the effect of docosahexaenoic acid on cognitive functions in healthy, preschool children: A randomized, placebo-controlled, double-blind study. Clin. Pediatr. 2008, 47, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Seferoğlu, F.; Erman, A.; Şahan, A.; Toktaş, N. The effect of n-3 lc-pufa supplementation on tennis skill acquisition in 10–12 year old girls. Biol. Sport 2012, 29, 241–246. [Google Scholar] [CrossRef]
- Tammam, J.D.; Steinsaltz, D.; Bester, D.; Semb-Andenaes, T.; Stein, J.F. A randomised double-blind placebo-controlled trial investigating the behavioural effects of vitamin, mineral and n-3 fatty acid supplementation in typically developing adolescent schoolchildren. Br. J. Nutr. 2016, 115, 361–373. [Google Scholar] [CrossRef] [PubMed]
- Thienprasert, A.; Samuhaseneetoo, S.; Popplestone, K.; West, A.L.; Miles, E.A.; Calder, P.C. Fish oil n-3 polyunsaturated fatty acids selectively affect plasma cytokines and decrease illness in thai schoolchildren: A randomized, double-blind, placebo-controlled intervention trial. J. Pediatr. 2009, 154, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Verduci, E.; Agostoni, C.; Radaelli, G.; Banderali, G.; Riva, E.; Giovannini, M. Blood lipids profile in hyperlipidemic children undergoing different dietary long chain polyunsaturated supplementations: A preliminary clinical trial. Int. J. Food Sci. Nutr. 2014, 65, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Gidding, S.S.; Prospero, C.; Hossain, J.; Zappalla, F.; Balagopal, P.; Falkner, B.; Kwiterovich, P. A double-blind randomized trial of fish oil to lower triglycerides and improve cardiometabolic risk in adolescents. J. Pediatr. 2014, 165, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, J.; Smuts, C.M.; Malan, L.; Kvalsvig, J.; van Stuijvenberg, M.E.; Hurrell, R.F.; Zimmermann, M.B. Effects of iron and n-3 fatty acid supplementation, alone and in combination, on cognition in school children: A randomized, double-blind, placebo-controlled intervention in south africa. Am. J. Clin. Nutr. 2012, 96, 1327–1338. [Google Scholar] [CrossRef] [PubMed]
- Malan, L.; Baumgartner, J.; Zandberg, L.; Calder, P.C.; Smuts, C.M. Iron and a mixture of dha and epa supplementation, alone and in combination, affect bioactive lipid signalling and morbidity of iron deficient south african school children in a two-by-two randomised controlled trial. Prostaglandin Leukot. Essent. 2016, 105, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Smuts, C.M.; Greeff, J.; Kvalsvig, J.; Zimmermann, M.B.; Baumgartner, J. Long-chain n-3 pufa supplementation decreases physical activity during class time in iron-deficient south african school children. Br. J. Nutr. 2015, 113, 212–224. [Google Scholar] [CrossRef] [PubMed]
- Malan, L.; Baumgartner, J.; Calder, P.C.; Zimmermann, M.B.; Smuts, C.M. N-3 long-chain pufas reduce respiratory morbidity caused by iron supplementation in iron-deficient south african schoolchildren: A randomized, double-blind, placebo-controlled intervention. Am. J. Clin. Nutr. 2015, 101, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.J.; Puri, B.K. A randomized double-blind, placebo-controlled study of the effects of supplementation with highly unsaturated fatty acids on adhd-related symptoms in children with specific learning difficulties. Prog. Neuropsychopharmacol. Biol. Psychiatry 2002, 26, 233–239. [Google Scholar] [CrossRef]
- Portillo-Reyes, V.; Pérez-García, M.; Loya-Méndez, Y.; Puente, A.E. Clinical significance of neuropsychological improvement after supplementation with omega-3 in 8–12 years old malnourished mexican children: A randomized, double-blind, placebo and treatment clinical trial. Res. Dev. Disabil. 2014, 35, 861–870. [Google Scholar] [CrossRef] [PubMed]
- Fayyazi, A.; Khajeh, A.; Ghazavi, A.; Sangestani, M. Omega 3 in childhood migraines: A double blind randomized clinical trial. Iran. J. Child Neurol. 2016, 10, 9–13. [Google Scholar] [PubMed]
- Fristad, M.A.; Young, A.S.; Vesco, A.T.; Nader, E.S.; Healy, K.Z.; Gardner, W.; Wolfson, H.L.; Arnold, L.E. A randomized controlled trial of individual family psychoeducational psychotherapy and omega-3 fatty acids in youth with subsyndromal bipolar disorder. J. Child Adolesc. Psychopharmacol. 2015, 25, 764–774. [Google Scholar] [CrossRef] [PubMed]
- Nemets, H.; Nemets, B.; Apter, A.; Bracha, Z.; Belmaker, R. Omega-3 treatment of childhood depression: A controlled, double-blind pilot study. Am. J. Psychiatry 2006, 163, 1098–1100. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, A.; Gharipour, M.; Arabzadeh, G.; Moin, P.; Hashemipour, M.; Kelishadi, R. The effects of vitamin e and omega-3 pufas on endothelial function among adolescents with metabolic syndrome. Biomed. Res. Int. 2014, 2014, 906019. [Google Scholar] [CrossRef] [PubMed]
- Boyraz, M.; Pirgon, Ö.; Dündar, B.; Çekmez, F.; Hatipoğlu, N. Long-term treatment with n-3 polyunsaturated fatty acids as a monotherapy in children with nonalcoholic fatty liver disease. J. Clin. Res. Pediatr. Endocrinol. 2015, 7, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Janczyk, W.; Lebensztejn, D.; Wierzbicka-Rucińska, A.; Mazur, A.; Neuhoff-Murawska, J.; Matusik, P.; Socha, P. Omega-3 fatty acids therapy in children with nonalcoholic fatty liver disease: A randomized controlled trial. J. Pediatr. 2015, 166, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Janczyk, W.; Socha, P.; Lebensztejn, D.; Wierzbicka, A.; Mazur, A.; Neuhoff-Murawska, J.; Matusik, P. Omega-3 fatty acids for treatment of non-alcoholic fatty liver disease: Design and rationale of randomized controlled trial. BMC Pediatr. 2013, 13, 1. [Google Scholar] [CrossRef] [PubMed]
- Nobili, V.; Bedogni, G.; Donati, B.; Alisi, A.; Valenti, L. The i148m variant of pnpla3 reduces the response to docosahexaenoic acid in children with non-alcoholic fatty liver disease. J. Med. Food 2013, 16, 957–960. [Google Scholar] [CrossRef] [PubMed]
- Nobili, V.; Alisi, A.; Della Corte, C.; Risé, P.; Galli, C.; Agostoni, C.; Bedogni, G. Docosahexaenoic acid for the treatment of fatty liver: Randomised controlled trial in children. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 1066–1070. [Google Scholar] [CrossRef] [PubMed]
- Nobili, V.; Bedogni, G.; Alisi, A.; Pietrobattista, A.; Rise, P.; Galli, C.; Agostoni, C. Docosahexaenoic acid supplementation decreases liver fat content in children with non-alcoholic fatty liver disease: Double-blind randomised controlled clinical trial. Arch. Dis. Child 2011, 96, 350–353. [Google Scholar] [CrossRef] [PubMed]
- Pacifico, L.; Bonci, E.; Di Martino, M.; Versacci, P.; Andreoli, G.; Silvestri, L.; Chiesa, C. A double-blind, placebo-controlled randomized trial to evaluate the efficacy of docosahexaenoic acid supplementation on hepatic fat and associated cardiovascular risk factors in overweight children with nonalcoholic fatty liver disease. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 734–741. [Google Scholar] [CrossRef] [PubMed]
- Spahis, S.; Alvarez, F.; Dubois, J.; Ahmed, N.; Peretti, N.; Levy, E. Plasma fatty acid composition in french-canadian children with non-alcoholic fatty liver disease: Effect of n-3 pufa supplementation. Prostaglandin Leukot. Essent. 2015, 99, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Dangardt, F.; Osika, W.; Chen, Y.; Nilsson, U.; Gan, L.M.; Gronowitz, E.; Strandvik, B.; Friberg, P. Omega-3 fatty acid supplementation improves vascular function and reduces inflammation in obese adolescents. Atherosclerosis 2010, 212, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Dangardt, F.; Chen, Y.; Gronowitz, E.; Dahlgren, J.; Friberg, P.; Strandvik, B. High physiological omega-3 fatty acid supplementation affects muscle fatty acid composition and glucose and insulin homeostasis in obese adolescents. J. Nutr. Metab. 2012, 2012, 395757. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, M.H.; Mølgaard, C.; Hellgren, L.I.; Lauritzen, L. Effects of fish oil supplementation on markers of the metabolic syndrome. J. Pediatr. 2010, 157, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Damsgaard, C.T.; Molgaard, C.; Matthiessen, J.; Gyldenlove, S.N.; Lauritzen, L. The effects of n-3 long-chain polyunsaturated fatty acids on bone formation and growth factors in adolescent boys. Pediatr. Res. 2012, 71, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Alarcon, M.; Martinez-Coronado, A.; Velarde-Castro, O.; Rendon-Macias, E.; Fernandez, J. Supplementation of n3 long-chain polyunsaturated fatty acid synergistically decreases insulin resistance with weight loss of obese prepubertal and pubertal children. Arch. Med. Res. 2011, 42, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Riva, E.; Biasucci, G.; Luotti, D.; Bruzzese, M.G.; Marangoni, F.; Giovannini, M. The effects of n-3 and n-6 polyunsaturated fatty acids on plasma lipids and fatty acids of treated phenylketonuric children. Prostaglandin Leukot. Essent. 1995, 53, 401–404. [Google Scholar] [CrossRef]
- Gabbay, V.; Babb, J.S.; Klein, R.G.; Panzer, A.M.; Katz, Y.; Alonso, C.M.; Petkova, E.; Wang, J.; Coffey, B.J. A double-blind, placebo-controlled trial of omega-3 fatty acids in tourette’s disorder. Pediatrics 2012, 129, e1493–e1500. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Massetto, N.; Biasucci, G.; Rottoli, A.; Bonvissuto, M.; Bruzzese, M.; Giovannini, M.; Riva, E. Effects of long-chain polyunsaturated fatty acid supplementation on fatty acid status and visual function in treated children with hyperphenylalaninemia. J. Pediatr. 2000, 137, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Scaglioni, S.; Bonvissuto, M.; Bruzzese, M.G.; Giovannini, M.; Riva, E. Biochemical effects of supplemented long-chain polyunsaturated fatty acids in hyperphenylalaninemia. Prostaglandin Leukot. Essent. 2001, 64, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Verduci, E.; Massetto, N.; Fiori, L.; Radaelli, G.; Riva, E.; Giovannini, M. Long term effects of long chain polyunsaturated fats in hyperphenylalaninemic children. Arch. Dis. Child 2003, 88, 582–583. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.J.; Burton, J.R.; Sewell, R.P.; Spreckelsen, T.F.; Montgomery, P. Docosahexaenoic acid for reading, cognition and behavior in children aged 7–9 years: A randomized, controlled trial (the dolab study). PLoS ONE 2012, 7, e43909. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, P.; Burton, J.R.; Sewell, R.P.; Spreckelsen, T.F.; Richardson, A.J. Fatty acids and sleep in uk children: Subjective and pilot objective sleep results from the dolab study--A randomized controlled trial. J. Sleep Res. 2014, 23, 364–388. [Google Scholar] [CrossRef] [PubMed]
- Toerien, M.; Brookes, S.T.; Metcalfe, C.; De Salis, I.; Tomlin, Z.; Peters, T.J.; Sterne, J.; Donovan, J.L. A review of reporting of participant recruitment and retention in rcts in six major journals. Trials 2009, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Treweek, S.; Lockhart, P.; Pitkethly, M.; Cook, J.A.; Kjeldstrøm, M.; Johansen, M.; Taskila, T.K.; Sullivan, F.M.; Wilson, S.; Jackson, C.; et al. Methods to improve recruitment to randomised controlled trials: Cochrane systematic review and meta-analysis. BMJ Open 2013, 3, e002360. [Google Scholar] [CrossRef] [PubMed]
- Knox, C.A.; Burkhart, P.V. Issues related to children participating in clinical research. J. Pediatr. Nurs. 2007, 22, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Martinson, B.C.; Lazovich, D.; Lando, H.A.; Perry, C.L.; McGovern, P.G.; Boyle, R.G. Effectiveness of monetary incentives for recruiting adolescents to an intervention trial to reduce smoking. Prev. Med. 2000, 31, 706–713. [Google Scholar] [CrossRef] [PubMed]
- Healy, J.; Hope, R.; Bhabha, J.; Eyal, N. Paying for antiretroviral adherence: Is it unethical when the patient is an adolescent? J. Med. Ethics 2016. [Google Scholar] [CrossRef] [PubMed]
- Wendler, D.; Rackoff, J.E.; Emanuel, E.J.; Grady, C. The ethics of paying for children’s participation in research. J. Pediatr. 2002, 141, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Gul, R.B.; Ali, P.A. Clinical trials: The challenge of recruitment and retention of participants. J. Clin. Nurs. 2010, 19, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.X.; Doku, V.; Tennakoon, L. Challenges in recruitment of research participants. Adv. Psychiatr. Treat. 2003, 9, 229–238. [Google Scholar] [CrossRef]
- Kettler, L.; Sawyer, S.; Winefield, H.; Greville, H. Determinants of adherence in adults with cystic fibrosis. Thorax 2002, 57, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Gau, S.S.; Shen, H.-Y.; Chou, M.-C.; Tang, C.-S.; Chiu, Y.-N.; Gau, C.-S. Determinants of adherence to methylphenidate and the impact of poor adherence on maternal and family measures. J. Child Adolesc. Psychopharmacol. 2006, 16, 286–297. [Google Scholar] [CrossRef] [PubMed]
- Stewart, S.L.; Baiden, P. An exploratory study of the factors associated with medication nonadherence among youth in adult mental health facilities in ontario, canada. Psychiat. Res. 2013, 207, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Bai, G.-N.; Wang, Y.-F.; Yang, L.; Niu, W.-Y. Effectiveness of a focused, brief psychoeducation program for parents of adhd children: Improvement of medication adherence and symptoms. Neuropsych. Dis. Treat. 2015, 11, 2721. [Google Scholar]
- Osterberg, L.; Blaschke, T. Adherence to medication. N. Engl. J. Med. 2005, 353, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Robinson, K.A.; Dennison, C.R.; Wayman, D.M.; Pronovost, P.J.; Needham, D.M. Systematic review identifies number of strategies important for retaining study participants. J. Clin. Epidemiol. 2007, 60, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Coday, M.; Boutin-Foster, C.; Sher, T.G.; Tennant, J.; Greaney, M.L.; Saunders, S.D.; Somes, G.W. Strategies for retaining study participants in behavioral intervention trials: Retention experiences of the nih behavior change consortium. Ann. Behav. Med. 2005, 29, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Hunt, J.R.; White, E. Retaining and tracking cohort study members. Epidemiol. Rev. 1998, 20, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Davis, L.L.; Broome, M.E.; Cox, R.P. Maximizing retention in community-based clinical trials. J. Nurs. Scholarsh. 2002, 34, 47–53. [Google Scholar] [CrossRef] [PubMed]
| Reference | Age Range or Mean (SD) | Gender (%Female) | Population: Healthy, Disorder or Disease | Country |
|---|---|---|---|---|
| [58] | 3–15 | 44 | Acute lymphoblastic leukemia | Egypt |
| [59] | 6–12 | 25 | ADHD | Iran |
| [60] | 7–15 | NR | ADHD | Iran |
| [61] | 11–12 | 31 | ADHD | Canada |
| [62] | 8–14 | 0 | ADHD | The Netherlands |
| [63] | 6–16 | 41 (after intervention) | ADHD | Israel |
| [64] | 7–12 | 20 | ADHD | Sweden |
| [65] | 6–12 | 38 | ADHD | Iran |
| [66] | 6–12 | NR | ADHD | Japan |
| [67,68] | 8–18 | 15 | ADHD | Sweden |
| [69] | 12–16 | 0 | ADHD | UK |
| [70,71] | 6–13 | 23 | ADHD | Australia |
| [72] | 6–12 | 27 | ADHD | Sri Lanka |
| [73] | 7–13 | 41 | ADHD | Israel |
| [74,75] | 7–12 | 23 | ADHD | Australia |
| [76] | 6–13 | 13 | ADHD | USA |
| [77] | 8–13 | 25 | ADHD | Israel |
| [78] | 6–12 | 22 | ADHD | USA |
| [79] | 6–12 | 22 | ADHD | Germany |
| [80,81] | 6–13 | 34 | ADHD | Israel |
| [82] | 7–12 | 43 (after intervention) | ADHD or lower IQ | China |
| [83] | 6–14 | 15 | ADHD | Australia |
| [84] | 6.9–11.9 | NR | ADHD | Canada |
| [85] | 8–16 | 48 | Aggressive behaviour | Mauritius |
| [86] | 6–14 | 42 | Asthma | USA |
| [87] | 8–12 | 56 | Asthma | Australia |
| [88] | 10–12 | 31 | Asthma | Taiwan |
| [89] | 10.2 (2.5) fish oil, 11.9 (3.1) control | 48 | Bronchial asthma | Japan |
| [90] | 3–8 | 11 | Autism | USA |
| [91] | 5–8 | NR | Autism | USA |
| [92] | 2–5 | 26 | Autism | Canada |
| [93] | 3–10 | 17 | Autism | USA |
| [94] | 6–17 | 48% placebo, 46% flax oil | Bipolar disorder | USA |
| [95] | 7.3–9.5 | 54 | CF | Italy |
| [96] | 5–16 | 47 | Crohn’s disease | Italy |
| [97] | 5–12 | 33 | DCD | UK |
| [98] | 10.6 | 43 | Dyslexia | Finland |
| [99] | 15–18 | 100 | Dysmenorrhea | USA |
| [100] | 4–12 | NR | Epilepsy | Egypt |
| [101] | 7–9 | 53 | Healthy | South-Africa |
| [102] | 8–14 | 50 | Healthy | Indonesia |
| [103] | 9–12 | 51 | Healthy | Japan |
| [104] | 9–10 | 50 | Healthy | Sweden |
| [105] | 10–12 | 49 | Healthy | UK |
| [106] | 8–10 | 52 | Healthy | UK |
| [107] | 5–7 | NR | Healthy | Canada |
| [108] | 8–10 | 0 | Healthy | USA |
| [109] | 6–10 | 46 | Healthy | Australia and Indonesia |
| [110] | 3–13 | 46 | Healthy | Australia |
| [111] | 8–14 | 51 | Healthy | Spain |
| [112] | 4 | 47 | Healthy | USA |
| [113] | 10–12 | 100 | Healthy | Turkey |
| [114] | 13–16 | 50 | Healthy | UK |
| [115] | 9–12 | 47 | Healthy | Thailand |
| [116] | 8–13 | 47 | Hyperlipidaemia | Italy |
| [117] | 14 (2) | 31 | Hypertriglyceridemia and low LDL | USA |
| [118,119,120,121] | 6–11 | 49 | Iron deficiency | South-Africa |
| [122] | 8–12 | 15 | Literacy problems | UK |
| [123] | 8–12 | 58 | Malnourished | Mexico |
| [124] | 5–14 | 56 | Migraine | Iran |
| [125] | 7–14 | NR | MDD | USA |
| [126] | 6–12 | NR | MDD | Israel |
| [127] | 10–18 | 59 (after intervention) | Metabolic syndrome | Iran |
| [128] | 9–17 | 47 | NAFL and obesity | Turkey |
| [129,130] | 11–15 | 14 | NAFL and overweight | Poland |
| [131,132,133] | 6–16 | 58 | NAFL | Italy |
| [134] | 10.8 (2.8) | 48 | NAFL and overweight | Italy |
| [135] | 8–18 | 0 | NAFL | Canada |
| [136,137] | 14–17 | 56 | Obesity | Sweden |
| [138,139] | 13–15 | 0 | Overweight | Denmark |
| [140] | 9–18 | NR | Overweight + insulin resistance | Mexico |
| [141] | 5–10 | NR | PKU | Italy |
| [142] | 6–18 | 18 | Tourette’s Disorder | USA |
| [143,144,145] | 10 (7) | 45 | Type-1 type-1 hyperphenylalaninemia, HPA | Italy |
| [146,147] | 6–10 | 47 | Underperforming | UK |
| Reference | Treatment per Day Unless Otherwise Stated | Placebo | Form of Supplementation | Number of Capsules | Duration b (Weeks) |
|---|---|---|---|---|---|
| Healthy | |||||
| [108] | DHASCO a: 400 or 1200 mg DHA | Corn oil | Capsules | 6 | 8 |
| [106] | 800 mg FO: 400 mg DHA, 56 mg EPA | Olive oil | Chewable capsules | 2 | 16 |
| [113] | 670 mg FO | Olive oil | Capsules | 2 | 16 |
| [110] | 2400 mg FO and 600 mg evening primrose oil: 174 mg DHA, 558 mg EPA, 60 mg GLA. | Palm oil | Capsules | 6 | 28.6 |
| [104] | 174 mg DHA, 558 mg EPA, 60 mg GLA | Palm oil | Capsules | 6 | 12 + 12 (open) |
| [102] | 1260 mg DHA rich oil: 652 mg DHA, 101 mg EPA | Placebo oil (656 mg LA, 87 mg ALA) | Capsules | 6 | 12 |
| [101] | Fish flour: 892 mg of DHA per week | Placebo spread contained bread flour | Margarine | NA | 14.9 |
| [107] | 14–21 mg DHA, 20–30 mg AA | Placebo supplement | Sachets to mix into foods | 2–3 sachets | 30 |
| [103] | FO: 3600 mg DHA, 840 mg EPA per week | 50% soybean oil, 50% rapeseed oil (4200 mg LA per week) | Bread and sausages | NA | 12 |
| [114] | 541 mg FO: 116 mg DHA, 165 mg EPA | Sunflower oil | Capsules | 2 | 12 |
| [112] | DHASCO-S a: 400 mg DHA | High oleic sunflower oil | Capsules | 2 | 16 |
| [115] | FO: 1 g DHA, 200 mg EPA | Soybean oil | Chocolate milk | NA | 15.6 |
| [109] | 88 mg DHA, 22 mg EPA | Unclear | Drink | NA | 52 |
| [105] | 500 mg DHASCO-S a: 200 mg DHA, 4 mg EPA | Vegetable oil (15 mg ALA, 250 mg LA) | Capsules | 5 | 8 |
| [111] | FO in dairy drink 120 mg DHA, 60 mg EPA | Whole milk | Milk drink | NA | 20 |
| [117] | 4 g FO: 1.5 g DHA, 1.86 g EPA | Corn oil | Unclear | Unclear | 8 + 8 with 4 weeks wash-out in between |
| [100] | 3 mL dose of 1200 mg FO: 240 mg DHA, 360 mg EPA. | Corn oil | Liquid oil | NA | 12 |
| [88] | FO: 125 mg DHA, 230 mg EPA | Corn oil | Capsules | Dependent on bw | 16 |
| [96] | 3 g O3FA | Olive oil | Capsules | Dependent on bw | 52 |
| [92] | 1.875 mL FO: 0.75 g of DHA + EPA. If well tolerated dose ×2 after 2 weeks. | Olive oil and medium chain triglycerides. | Liquid oil | NA | 24 |
| [65] | 165 mg DHA, 635 mg EPA, 100 mg other O3FA | Olive oil | Capsules | NS | 8 |
| [67,68] | 174 mg DHA, 558 mg EPA, 60 mg GLA | Olive oil | Capsules | 6 | 12 + 12 (open) |
| [97] | FO and EPO: 174 mg DHA, 558 mg EPA, 60 mg GLA | Olive oil | Capsules | 6 | 26 |
| [79] | 120 mg DHA, 600 mg EPA | Olive oil | Capsules | 2 | 16 |
| [122] | 480 mg DHA, 186 mg EPA, 96 mg GLA, 864 mg LA, 42 mg AA, 8 mg thyme oil | Olive oil | Capsules | NR | 12 |
| [66] | DHA-rich fish oil: 3600 mg DHA 700 mg EPA per week. | Olive oil | Milk and bread | NA | 12 |
| [76] | 480 mg DHA, 80 mg EPA, 40 mg AA, 96 mg GLA | Olive oil | Capsules | 8 | 16 |
| [143,144,145] | LCPUFA supplementation: varying dosage | Olive oil | Capsules | 1 per 4 kg of bw | 52 |
| [94] | Flax seed oil: 0.55 to 6.6 g ALA | Olive oil | Capsules | Varying up to 12 | 16 |
| [89] | FO: DHA 7.3 ± 11.5 mg/kg of bw, EPA 17.0 ± 26.8 mg/kg of bw | Olive oil | Capsules | Dependent on bw: 6–12 | 43.6 |
| [83] | PCSO-524 c: 16.5–22 mg DHA, 21.9–29.2 mg EPA | Olive oil, lecithin and coconut oil | Capsules | Dependent on bw: 3–4 | 14 |
| [86] | Drink containing FO (1.6 g DHA, 3 g EPA) and borage oil (3.0 g GLA) | Control drink with high oleic safflower oil | Drink | NA | 12 |
| [70] | EPA-rich FO: 108 mg DHA, 1,109 mg EPA or DHA-rich FO: 1,032 mg DHA, 264 mg EPA | Safflower oil | Capsules | 4 | 16 + 16 + 16 |
| [90] | FO: 1.1 g DHA + EPA | Safflower oil | Pudding packet | 2 pudding packs | 12 |
| [123] | FO: 180 mg DHA, 270 mg EPA | Soybean oil | Capsules | 3 | 12 |
| [135] | 2 g FO: 1200 mg DHA + EPA | Sunflower oil | Capsules | 4 | 24 |
| [87] | FO: 1.2 g O3FA | Sunflower oil | Capsules, salad dressing and margarine | 4 | 24 |
| [72] | FO and EPO oil: 592.74 mg O3FA | Sunflower oil | Capsules | 2 | 26 |
| [58] | 1 g FO:120 mg DHA, 180 mg EPA | Sunflower oil | Capsules | Unclear | 24 |
| [61] | 100–400 mg DHA, 500–100 mg EPA | Sunflower oil | Capsules | Dependent on bw: 2–4 | 16 |
| [129,130] | AO: 450–1300 mg O3FA (DHA: EPA in 3:2 proportion) | Sunflower oil | Capsules | Dependent on bw | 24 |
| [116] | AO: 500 mg DHA or FO:500 mg DHA + EPA | Wheat gern oil | Capsules | 1 | 16 |
| [131,132,133] | AO: 250 or 500 mg DHA | Germ oil | Capsules | 1 | 26.1 |
| [134] | AO: 250 mg DHA | Germ oil | Capsules | NR | 26 |
| [95] | Algae triacylglycerol 100 mg DHA/kg/day 1st month then 1 g DHA/day | Germ oil | Capsules | 4 | 52 |
| [93] | AO: 200 mg DHA | Corn oil + soy bean oil | Capsules | 1 | 26 |
| [146,147] | AO: 600 mg DHA | Corn oil + soy oil | Capsules | 3 | 16 |
| [138,139] | 4.9 g FO: 892 mg DHA, 191 mg EPA | 6:1:1 mix of palm shortening, soy oil, and rapeseed oil | Bread | NA | 16 |
| [141] | 2.5–4 g FO (12% DHA, 18% EPA) | Blackcurrant seed oil (45.7% LA, 18% GLA, 14% ALA) | Capsules | Dependent on bw: 5-8 | 26 |
| [62] | 650 mg DHA, 650 mg EPA | Normal margarine (1 g LA) | Margarine | NA | 16 |
| [99] | FO: 720 mg DHA, 1080 mg EPA | 1800mg lactose | Capsules | 2 | 8+8 |
| [125] | 200 mg DHA, 1400 mg EPA, 400 mg other O3FA | Placebo capsule | Capsules | 2 | 12 |
| [136,137] | FO and EPO: 290 mg DHA, 930 mg EPA, 100 mg GLA | Placebo | Capsules | 10 | 12 + 12 with 6 weeks wash-out in between |
| [91] | FO: 1.1 g DHA + EPA | Identical placebo | Pudding packet | 2 pudding packs | 6 |
| [128] | 1000 mg PUFA | Placebo | Capsule | 1 | 52 |
| [63] | 2 g sage oil: 1 g ALA | Lactose placebo | Capsules | 2 | 8 |
| [60] | 240 mg DHA, 360 mg EPA | Placebo | Capsules | 2 | 8 |
| [64] | FO: 2.7 mg DHA, 500 mg EPA | Placebo | Capsules | 1 | 15 |
| [127] | 2.4 g omega-3 | Vitamin E or placebo | Tablets | NR | 8 |
| [84] | 100 mg DHA, 250 mg EPA, 25 mg phospholipids | Sunflower oil | Capsules | According to bw: 1–2 | 16 |
| [124] | 1 g FO: 120 mg DHA, 180 mg EPA | Placebo capsule | Capsules | 1 | At least 8 weeks |
| [78] | Algae oil: 345 mg DHA | Placebo capsule | Capsules | 1 | 16 |
| [59] | 241 mg DHA, 33 mg EPA, and 180 mg omega-6 | Identical placebo | Capsules | 1 | 10 |
| [118,119,120,121] | FO: 155 mg DHA, 29 mg EPA | Placebo | Capsules | 2 | 15 |
| [142] | Varying 500–6000 mg O3FA | Placebo | Capsules | Varying up to 12 | 20 |
| [140] | Salmon oil: 360 mg DHA, 540 mg EPA | Placebo (corn starch, lactose, magnesium stearate and polyvinyl pyrrolidone) | Capsules | NR | 4 |
| [85] | 300 mg DHA, 200 mg EPA, 400 mg ALA, 100 mg of DPA | Drink without omega-3 | Drink | NA | 24 |
| [77] | FO: 96 mg DHA, 153 mg EPA or n-3 LC-PUFA containing PLs: 95 mg DHA, 156 mg EPA | Rapeseed oil | Chocolate flavoured spread | NA | 13.1 |
| [73] | 240 mg LA, 60 mg ALA, 95 mg mineral oil | Vitamine C capsules | Capsules | 1 | 7 |
| [69] | FO and EPO: 174 mg DHA, 558 mg EPA, 60 mg LA. | Medium chain triglycerides | Capsules | 6 | 12 |
| [126] | 200 mg DHA 400 mg EPA, or 180 mg DHA, 380 mg EPA | Olive oil or safflower oil | Capsules | 1–2 | 16 |
| [74,75] | FO and EPO: 174 mg DHA, 558 mg EPA, 60 mg GLA | Palm oil | Capsules | 6 | 30 |
| [98] | 500 mg ethyl-EPA | Triglycerides and cellulose | Capsules | NR | 12.9 |
| [80,81] | 1–15 weeks: 120 mg EPA + DHA 16–30 weeks: 60 mg EPA + DHA | Cellulose | Capsules | 4 | 15 + 15 |
| [82] | 321 mg DHA, 42.2 g EPA per 100 g egg | Ordinary egg | Egg | 1 | 13.1 |
| Reference | Invited | Responded/Screened | Started | Finished | Started/Invited % | Started/Responded % | Started/Finished % | Recruitment Method | Recruitment Setting | Study Period |
|---|---|---|---|---|---|---|---|---|---|---|
| [141] | NS | NS | 21 | 21 | - | - | 100 | NS | Department of Paediatrics | NS |
| [66] | 46 | 40 | 40 | 40 | 87 | 100 | 100 | Parents of summer camp participants were asked. | Summer camp for children with psychiatric disorders | NS |
| [98] | 107 | 107 | 61 | 61 | 57 | 100 | Teachers nominated children with reading difficulties | School | Autumn 2005–January 2006 | |
| [131,132,133] | NS | NS | 60 | 60 | - | - | 100 | NS | Hospital | NS |
| [115] | NS | NS | 180 | 180 | - | - | 100 | NS | School | NS |
| [116] | NS | NS | 36 | 36 | - | - | 100 | NS | Hospital | 8 month period |
| [146,147] | 1376 | 675 | 362 | 359 | 26 | 54 | 99 | Parents of underperforming children received a letter inviting their children to take part in the formal screening assessments. | School | NS |
| [105] | NS | NS | 90 | 88 | - | - | 98 | Via advertising in newspapers and schools | Community and schools | NS |
| [88] | NS | 298 | 197 | 192 | - | 66 | 98 | Participants with asthma diagnosis were recruited from elementary schools through parent conferences | Schools | NS |
| [82] | 1556 | 1556 | 179 | 171 | 12 | 12 | 96 | Children were screened from students in two township primary schools | Schools | NS |
| [62] | NS | 372 | 79 | 76 | - | 21 | 96 | Via hospital and advertising at schools. | Hospital and schools | NS |
| [72] | NS | 422 | 98 | 94 | - | 23 | 96 | NS | Outpatient treatment program | NS |
| [114] | NS | 408 | 196 | 189 | - | 48 | 96 | NS | School | NS |
| [58] | NS | 100 | 70 | 65 | - | 70 | 93 | NS | Hospital | NS |
| [117] | NS | NS | 42 | 39 | - | - | 93 | NS | Hospital | NS |
| [118,119,120,121] | NS | 926 | 321 | 294 | - | 35 | 92 | Parents were invited to an information meeting. | School | November 2009–November 2010 |
| [127] | NS | NS | 90 | 83 | - | - | 92 | NS | Cardiovascular Research Centre | NS |
| [60] | NS | NS | 75 | 69 | - | - | 92 | NS | Outpatient ADHD clinic | 2007 |
| [103] | NS | 230 | 179 | 166 | - | 78 | 92 | Via advertisements | Community | NS |
| [85] | NS | 938 | 200 | 184 | - | 21 | 92 | Via parents who themselves had participated in a study. | Participants earlier study | November 2009–December 2011 |
| [123] | NS | 59 | 55 | 50 | - | 93 | 91 | Parents were invited to a meeting at which the study procedures were explained and a written informed consent from the tutors and a verbal assent from their children were obtained. | School | NS |
| [95] | NS | NS | 41 | 37 | - | - | 90 | NS | Hospital | NS |
| [101] | NS | NS | 183 | 164 | - | - | 90 | NS | School | NS |
| [138,139] | 3652 | NS | 87 | 78 | 2 | - | 90 | Subjects were recruited via addresses obtained from the Danish Civilian Person Register. | Community | NS |
| [111] | NS | NS | 119 | 107 | - | - | 90 | NS | School | NS |
| [99] | NS | NS | 42 | 37 | - | - | 88 | NS | School | NS |
| [134] | NS | 118 | 58 | 51 | - | 49 | 88 | NS | Hospital | May 2012–September 2014 |
| [113] | NS | 44 | 33 | 29 | - | 75 | 88 | Via public flyers | Community | NS |
| [135] | NS | 30 | 24 | 21 | - | 80 | 88 | NS | Hospital | NS |
| [87] | NS | NS | 45 | 39 | - | - | 87 | NS | NS | Over period of 16 mo. |
| [108] | NS | 48 | 38 | 33 | - | 79 | 87 | NS | NS | NS |
| [112] | NS | 405 | 202 | 175 | - | 50 | 87 | NS | NS | NS |
| [86] | NS | NS | 43 | 37 | - | - | 86 | NS | Outpatient clinic | NS |
| [65] | NS | NS | 120 | 103 | - | - | 86 | NS | Hospital | NS |
| [97] | 189 | 129 | 117 | 100 | 62 | 91 | 86 | Letters of invitation were sent to parents of children who were identified by teachers. | School | NS |
| [78] | NS | 250 | 63 | 54 | - | 25 | 86 | Via advertisements | Community | NS |
| [79] | NS | 334 | 110 | 95 | - | 33 | 86 | Via health professionals, teachers, leaflets handed out to support groups, leaflet distributed at community centres and advertisements in a free of charge regional newspaper. | Community, Health professionals, schools, support groups. | NS |
| [64] | NS | NS | 109 | 92 | - | - | 84 | NS | Hospital and secondary treatment centres | January 2005–June 2007. |
| [129,130] | NS | 86 | 76 | 64 | - | 88 | 84 | NS | Hospital | 2008-2011 |
| [92] | NS | 101 | 38 | 32 | - | 38 | 84 | NS | Hospital | December 2010–December 2013 |
| [107] | NS | NS | 37 | 31 | - | - | 84 | NS | NS | NS |
| [143,144,145] | NS | NS | 24 | 20 | - | - | 83 | NS | NS | Recruited over 6 month |
| [125] | NS | 178 | 23 | 19 | - | 13 | 83 | Via advertisements and clinician referrals. | Community and referral | July 2011–May 2014 |
| [109] | NS | 932 | 780 | 643 | - | 84 | 82 | Via advertisement at schools and media advertisement. | Schools | Auguet 2003–April 2005 |
| [136,137] | 108 | 47 | 31 | 25 | 29 | 66 | 81 | NS. | Outpatient clinic | NS |
| [73] | NS | ~300 | 78 | 63 | - | 26 | 81 | Via advertisement on radio health program, in health newspapers and in ADHD clinics. | Community and ADHD clinic | January 2007–June 2007 |
| [80,81] | NS | 247 | 200 | 162 | - | 81 | 81 | Advertisements in newspapers, on the Internet and medical centres. | Community | NS |
| [91] | 863 | 118 | 57 | 45 | 7 | 48 | 79 | E-mail invitations to in registry and longitudinal study of families of children affected by ASD. | Online registry | 18 September 2012–31 December 2012 |
| [67,68] | NS | NS | 75 | 59 | - | - | 79 | NS | Hospital | October 2004–August 2006 |
| [128] | NS | NS | 138 | 108 | - | - | 78 | NS | Outpatient clinic | March 2010–June 2012 |
| [122] | NS | NS | 41 | 32 | - | - | 78 | NS | School | NS |
| [106] | NS | 511 | 450 | 348 | - | 88 | 77 | Via school | Schools | NS |
| [89] | NS | NS | 30 | 23 | - | - | 77 | NS | Hospital | January 1994–March 1995 |
| [142] | NS | NS | 33 | 25 | - | - | 76 | Via community, hospital and through patient association. | Community and referral | NS |
| [69] | NS | 138 | 76 | 57 | - | 55 | 75 | School and parent group circulated screening information to all potential eligible families | Schools and parent groups | NS |
| [83] | NS | 351 | 144 | 108 | - | 41 | 75 | NS | NS | NS |
| [77] | 250 | 102 | 83 | 60 | 33 | 81 | 72 | Newspaper advertisement | Community | July 2004–January 2005 |
| [126] | NS | NS | 28 | 20 | - | - | 71 | NS | Hospital | NS |
| [93] | NS | 143 | 48 | 34 | - | 34 | 71 | Via recruitment flyers across campus and sent to autism support groups. | Campus, autism support groups | NS |
| [61] | NS | NS | 37 | 26 | - | - | 70 | NS | ADHD clinic | NS |
| [90] | NS | 32 | 27 | 19 | - | 84 | 70 | NS | Outpatient autism clinic | 5 November 2008–25 June 2009 |
| [84] | NR | NR | 37 | 26 | - | - | 70 | NS | NS | NS |
| [104] | NS | 162 | 154 | 105 | - | 95 | 68 | Via teachers who informed families | School | December 2009–July 2011 |
| [76] | NS | 193 | 50 | 33 | - | 26 | 66 | NS | Community | NS |
| [74,75] | NS | 201 | 167 | 109 | - | 83 | 65 | NS | NS | Start March–May 2004 |
| [70] | NS | 199 | 96 | 57 | - | 48 | 59 | Via media releases, television interviews, newspaper advertisements, school newsletters, and flyers. | Community and School | June 2007–June 2009 |
| [110] | 560 | 447 | 408 | 227 | 73 | 91 | 56 | Via information sessions and school newsletters. | Schools | December 2010–May 2011 |
| [94] | NS | NS | 51 | 24 | - | - | 47 | NS | Hospital | November 2001–March 2005 |
| [63] | NS | NS | 40 | 17 | - | - | 43 | NS | ADHD clinic | NS |
| [59] | NS | NS | 40 | NS | - | - | - | NS | Outpatient ADHD clinic | June 2009–March 2010 |
| [124] | NS | NS | 25 | NR | - | - | - | NS | Hospital | NS |
| [140] | 142 | NS | 76 | NS | 54 | - | - | From previous sample children with insulin resistance were identified and invited | Community | NS |
| [102] | NS | NS | 233 | NS | - | - | - | Via school | School | NS |
| [100] | NS | NS | 70 | NS | - | - | - | NS | Hospital | NS |
| [96] | NS | NS | 38 | NS | - | - | - | NS | Hospital | NS |
| Reference | Adherence Assessment | Adherence Mean or nr. of Part. Non-Adherent | Blood FA Determined? | Drop-Out Rate (%) | |
|---|---|---|---|---|---|
| Treatment | Placebo | ||||
| Healthy | |||||
| [114] | Supervision and tick-off form | Active: 88.4%, Placebo: 88.5% | Y | 3.1 | 6.1 |
| [115] | Supervision | NR | Y | 0 | 0 |
| [108] | NR | NR | Y | Low DHA: 20; High DHA: 7.1 | 17 |
| [112] | Capsule count | Nearly 100% | Y | 7.1 | 5.6 |
| [101] | Supervision | Active: 94.8%, Placebo: 94.5% | Y | 11 | 9.8 |
| [106] | Pill diary by teacher or parent | Active: 68.4%, Placebo: 66.7% | Y | 24 | 21 |
| [102] | NR | NR | Y | NR | NR |
| [103] | NR | >90%. | Y | 6.7 | 7.8 |
| [109] | Sachet count and diary (Australia)/Supervision (Indonesia) | Australia: 73-84% | Y | 27 | 34 |
| Indonesia: 85-87% | 3.6 | 5.3 | |||
| [111] | Interview | small increase in DHA in supplemented group | Y | NR | NR |
| [107] | Diary | n = 6 | Y | NR | NR |
| [105] | Parental signing of diary card | >80%. | N | NR | NR |
| [113] | NR | NR | N | 5.9 | 19 |
| [110] | Supervision | Phase 1: 59%, Phase 2: 61% | N | 47 | 42 |
| With disorder or illness | |||||
| [141] | NR | NR | Y | 0 | 0 |
| [140] | Pill count | Active: 93%, Placebo: 96% | Y | NR | NR |
| [82] | Supervision | count of consumed eggs showed good compliance and % of adherence to treatment was 100% | Y | 5.6 | 3.2 |
| [127] | Pill count | pill count revealed no essential irregularities | Y | 13.3 | Vit. E: 0, Placebo: 10 |
| [84] | NR | NR | NR | NR | NR |
| [80,81] | Pill count | n = 14 | N | 20 | 18 |
| [83] | Pill count, compliance diary and telephone call | 96.7% | N | 23 | 23 |
| [95] | NR | n = 2 DHA supplementation induced a median plasma DHA enrichment of 5% suggesting adherence | Y | 14 | 5 |
| [138,139] | Interview | 90% | Y | NR | NR |
| [79] | Capsule count | n = 1 | Y | 13 | 11 |
| [86] | Diary and blood values | 80–85% | Y | 21.1 | 11.1 |
| [77] | Phone calls and product weighting | n = 6 | Y | Phospholips: 38, Fish oil: 25 | 24 |
| [118,119,120,121] | Supervision | 95.4% | Y | 6.9 | 9.9 |
| [61] | Blood | NR | Y | CO | CO |
| [94] | Capsule count and diary | >75% | Y | 42 | 64 |
| [70] | Capsule count | EPA: 83%, DHA: 86% , LA: 85% | Y | CO | CO |
| [87] | Capsule count | 75% | Y | NR | NR |
| [76] | Diary | 88% | Y | 28 | 40 |
| [143,144,145] | NR | NR | Y | 17 | 17 |
| [131,132,133] | Capsule count and interview | excellent in all groups | Y | NR | NR |
| [62] | Product weighting | n = 1 | Y | 0 | 5.1 |
| [135] | Capsule count and interview | NR | Y | 0 | 25 |
| [78] | Capsule count | Active: 96.7%, Placebo: 100% | Y | 15 | 13 |
| [93] | Capsule count | excellent | Y | 21 | 38 |
| [136,137] | Capsule count | n = 1 | Y | CO | CO |
| [90] | Parent interview | Active: 69%, Placebo: 75% | Y | 36 | 23 |
| [69] | Capsule count | FA changed in the expected direction. | Y | 24 | 30 |
| [89] | NR | NR | Y | 27 | 14 |
| [64] | Capsule count | NR | Y | 30 | 19 |
| [116] | Capsule count | DHA: 96.5%, DHA + EPA: 96.9%, Placebo: 96.7% | Y | DHA: 0, DHA + EPA: 0 | 0 |
| [117] | Blood value | NR | Y | CO | CO |
| [129,130] | Capsule count | 95.5% | Y | 21 | 11 |
| [128] | Capsule count | NR | Y | NR | NR |
| [134] | Blood values | n = 5 | Y | 14 | 10 |
| [98] | NR | According to parents children took the capsules carefully | Y | NR | NR |
| [88] | Supervision and capsule count | Pill count: 91% | Y | 0 | 0 |
| [92] | NR | there was no overlap between the distributions of plasma levels between groups at week 24 | Y | 21 | 11 |
| [65] | Capsule count | n = 5 | N | NR | NR |
| [97] | Capsule count and diary | Period 1: 88.7%, Period 2: 85.5% | N | 17 | 12 |
| [91] | Parents e-mail | Active: 69%, Placebo: 83% | N | 28 | 14 |
| [85] | Parent interview and blood values | Average number of drink per week 6.5. | N | 10 | 22 |
| [73] | Capsule count | Active 7.88 capsules left; Placebo: 14 capsules left | N | 18 | 21 |
| [125] | NR | 89–97% | N | 10 | 23 |
| [142] | NR | NR | N | 18 | 31 |
| [67,68] | Parent interview | Period 1: 93.4%, Period 2: 93.3% | N | CO | CO |
| [126] | NR | n = 5 | N | NR | NR |
| [122] | Capsule count | Active: 90.4%, placebo 86.6% | N | 23 | 21 |
| [74,75] | Capsule count and diary | n = 2 | N | CO | CO |
| [63] | Capsule count | NR | N | 60 | 55 |
| [59] | NR | NR | N | NR | NR |
| [124] | NR | NR | N | NR | NR |
| [100] | NR | NR | N | NR | NR |
| [96] | NR | Compliance was optimal | N | NR | NR |
| [66] | NR | NR | N | 0 | 0 |
| [146,147] | Diary | At school: 75% | N | 0.6 | 1.1 |
| [72] | NR | NR | N | 2 | 6.1 |
| [58] | NR | n = 5 | N | 8.6 | 5.7 |
| [60] | NR | NR | N | NR | NR |
| [123] | Diary and capsule count | NR | N | 0 | 20 |
| [104] | Interview | Active: 94%, Placebo: 92%, Period 2: 91% | N | CO | CO |
| [99] | NR | n = 1 | N | CO | CO |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van der Wurff, I.S.M.; Meyer, B.J.; De Groot, R.H.M. A Review of Recruitment, Adherence and Drop-Out Rates in Omega-3 Polyunsaturated Fatty Acid Supplementation Trials in Children and Adolescents. Nutrients 2017, 9, 474. https://doi.org/10.3390/nu9050474
Van der Wurff ISM, Meyer BJ, De Groot RHM. A Review of Recruitment, Adherence and Drop-Out Rates in Omega-3 Polyunsaturated Fatty Acid Supplementation Trials in Children and Adolescents. Nutrients. 2017; 9(5):474. https://doi.org/10.3390/nu9050474
Chicago/Turabian StyleVan der Wurff, Inge S. M., Barbara J. Meyer, and Renate H. M. De Groot. 2017. "A Review of Recruitment, Adherence and Drop-Out Rates in Omega-3 Polyunsaturated Fatty Acid Supplementation Trials in Children and Adolescents" Nutrients 9, no. 5: 474. https://doi.org/10.3390/nu9050474
APA StyleVan der Wurff, I. S. M., Meyer, B. J., & De Groot, R. H. M. (2017). A Review of Recruitment, Adherence and Drop-Out Rates in Omega-3 Polyunsaturated Fatty Acid Supplementation Trials in Children and Adolescents. Nutrients, 9(5), 474. https://doi.org/10.3390/nu9050474







