Low Glycemic Index Prototype Isomaltulose—Update of Clinical Trials
Abstract
:1. Carbohydrates with High and Low Glycemic Index on Postprandial Glucose Homeostasis
2. Isomaltulose—Its Manufacturing and Key Characteristics
3. Postprandial Blood Glucose and Insulin Levels
4. Glucose Turnover in Type 2 Diabetes
5. Sports Nutrition and Cognitive Performance
6. Regulation of Body Weight and Composition
7. Pregnancy Outcome
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ludvik, B.; Nolan, J.J.; Roberts, A.; Baloga, J.; Joyce, M.; Bell, J.M.; Olefsky, J.M. Evidence for decreased splanchnic glucose uptake after oral glucose administration in non-insulin-dependent diabetes mellitus. J. Clin. Invest. 1997, 100, 2354–2361. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Basu, R.; Shah, P.; Vella, A.; Johnson, C.M.; Jensen, M.; Nair, K.S.; Schwenk, W.F.; Rizza, R.A. Type 2 diabetes impairs splanchnic uptake of glucose but does not alter intestinal glucose absorption during enteral glucose feeding: Additional evidence for a defect in hepatic glucokinase activity. Diabetes 2001, 50, 1351–1362. [Google Scholar] [CrossRef] [PubMed]
- Wahren, J.; Ekberg, K. Splanchnic regulation of glucose production. Annu. Rev. Nutr. 2007, 27, 329–345. [Google Scholar] [CrossRef] [PubMed]
- Foster-Powell, K.; Holt, S.H.; Brand-Miller, J.C. International table of glycemic index and glycemic load values: 2002. Am. J. Clin. Nutr. 2002, 76, 5–56. [Google Scholar] [PubMed]
- Thomas, D.; Elliott, E.J. Low glycaemic index, or low glycaemic load, diets for diabetes mellitus. Cochrane Database Syst. Rev. 2009. [Google Scholar] [CrossRef]
- Thomas, D.E.; Elliott, E.J.; Baur, L. Low glycaemic index or low glycaemic load diets for overweight and obesity. Cochrane Database Syst. Rev. 2007. [Google Scholar] [CrossRef]
- Schwarzfuchs, D.; Golan, R.; Shai, I. Four-year follow-up after two-year dietary interventions. N. Engl. J. Med. 2012, 367, 1373–1374. [Google Scholar] [CrossRef] [PubMed]
- Shai, I.; Schwarzfuchs, D.; Henkin, Y.; Shahar, D.R.; Witkow, S.; Greenberg, I.; Golan, R.; Fraser, D.; Bolotin, A.; Vardi, H.; et al. Weight loss with a low-carbohydrate, mediterranean, or low-fat diet. N. Engl. J. Med. 2008, 359, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International tables of glycemic index and glycemic load values: 2008. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes, A. Evidence-based nutrition principles and recommendations for the treatment and prevention of diabetes and related complications. Diabetes Care 2002, 25, 202–212. [Google Scholar] [CrossRef]
- Mann, J.I.; De Leeuw, I.; Hermansen, K.; Karamanos, B.; Karlstrom, B.; Katsilambros, N.; Riccardi, G.; Rivellese, A.A.; Rizkalla, S.; Slama, G.; et al. Evidence-based nutritional approaches to the treatment and prevention of diabetes mellitus. Nutr. Metab. Cardiovasc. Dis. 2004, 14, 373–394. [Google Scholar] [CrossRef]
- Lina, B.A.; Jonker, D.; Kozianowski, G. Isomaltulose (palatinose): A review of biological and toxicological studies. Food Chem. Toxicol. 2002, 40, 1375–1381. [Google Scholar] [CrossRef]
- Weidenhagen, R.L.A.D. Palatinose (6-0-alpha-d-glucopyranosyl-d-fructofuranose), ein neues bakterielles umwandlungsprodukt der saccharose (palatinose (6-0-alpha-d-glucopyranosyl-d-fructofuranose), a new bacterial conversion of sucrose product). Z. Für Die Zuckerind. Fachorg. Für Tecknik Rubenbau Und Wirtsch. 1957, 7, 533–534. [Google Scholar]
- Holub, I.; Gostner, A.; Theis, S.; Nosek, L.; Kudlich, T.; Melcher, R.; Scheppach, W. Novel findings on the metabolic effects of the low glycaemic carbohydrate isomaltulose (palatinose). Br. J. Nutr. 2010, 103, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- Codex, F.C. Monograph on Isomaltulose, 7th ed.; US Pharmacopeial Convention: Rockville, MD, USA, 2010. [Google Scholar]
- Sentko, A.W.-E.I. Isomaltulose. In Sweeteners and Sugar Alternatives in Food Technology, 2nd ed.; Wiley-Blackwell: Oxford, UK, 2012. [Google Scholar]
- Fleddermann, M.; Rauh-Pfeiffer, A.; Demmelmair, H.; Holdt, L.; Teupser, D.; Koletzko, B. Effects of a follow-on formula containing isomaltulose (palatinose) on metabolic response, acceptance, tolerance and safety in infants: A randomized-controlled trial. PLoS ONE 2016, 11, e0151614. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific opinion on the substantiation of health claims related to the sugar replacers xylitol, sorbitol, mannitol, maltitol, lactitol, isomalt, erythritol, d-tagatose, isomaltulose, sucralose and polydextrose and maintenance of tooth mineralisation by decreasing tooth demineralisation (id 463, 464, 563, 618, 647, 1182, 1591, 2907, 2921, 4300), and reduction of post-prandial glycaemic responses (id 617, 619, 669, 1590, 1762, 2903, 2908, 2920) pursuant to article 13 (1) of regulation (ec) No. 1924/2006. EFSA J. 2011, 9, 2076. [Google Scholar]
- Pubmed. Available online: http://www.ncbi.nlm.nih.gov/ (accessed on 1 February 2017).
- Web of Science. Available online: http://webofscience.com/ (accessed on 1 February 2017).
- ScienceDirect. Available online: http://www.sciencedirect.com/ (accessed on 1 February 2017).
- Cochrane Library. Available online: http://www.cochranelibrary.com/ (accessed on 1 February 2017).
- Liao, Z.; Li, Y.; Yao, B.; Fan, H.; Hu, G.L.; Weng, J. The effects of isomaltulose on blood glucose and lipids for diabetic subjects. Diabetes 2001, 50, A1366. [Google Scholar]
- Konig, D.; Theis, S.; Kozianowski, G.; Berg, A. Postprandial substrate use in overweight subjects with the metabolic syndrome after isomaltulose (palatinose) ingestion. Nutrition 2012, 28, 651–656. [Google Scholar] [CrossRef] [PubMed]
- van Can, J.G.; Ijzerman, T.H.; van Loon, L.J.; Brouns, F.; Blaak, E.E. Reduced glycaemic and insulinaemic responses following isomaltulose ingestion: Implications for postprandial substrate use. Br. J. Nutr. 2009, 102, 1408–1413. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, I. The bioavailability of isomaltulose in man and rat. Nutr. Rep. Int. 1983, 28, 1083–1090. [Google Scholar]
- Maeda, A.; Miyagawa, J.; Miuchi, M.; Nagai, E.; Konishi, K.; Matsuo, T.; Tokuda, M.; Kusunoki, Y.; Ochi, H.; Murai, K.; et al. Effects of the naturally-occurring disaccharides, palatinose and sucrose, on incretin secretion in healthy non-obese subjects. J. Diabetes Investig. 2013, 4, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Ang, M.; Linn, T. Comparison of the effects of slowly and rapidly absorbed carbohydrates on postprandial glucose metabolism in type 2 diabetes mellitus patients: A randomized trial. Am. J. Clin. Nutr. 2014, 100, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- Kawai, K.; Okuda, Y.; Yamashita, K. Changes in blood glucose and insulin after an oral palatinose administration in normal subjects. Endocrinol. Jpn. 1985, 32, 933–936. [Google Scholar] [CrossRef] [PubMed]
- Bracken, R.M.; Page, R.; Gray, B.; Kilduff, L.P.; West, D.J.; Stephens, J.W.; Bain, S.C. Isomaltulose improves glycemia and maintains run performance in type 1 diabetes. Med. Sci. Sports Exerc. 2012, 44, 800–808. [Google Scholar] [CrossRef] [PubMed]
- West, D.J.; Stephens, J.W.; Bain, S.C.; Kilduff, L.P.; Luzio, S.; Still, R.; Bracken, R.M. A combined insulin reduction and carbohydrate feeding strategy 30 min before running best preserves blood glucose concentration after exercise through improved fuel oxidation in type 1 diabetes mellitus. J. Sports Sci. 2011, 29, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Achten, J.; Jentjens, R.L.; Brouns, F.; Jeukendrup, A.E. Exogenous oxidation of isomaltulose is lower than that of sucrose during exercise in men. J. Nutr. 2007, 137, 1143–1148. [Google Scholar] [PubMed]
- Konig, D.; Zdzieblik, D.; Holz, A.; Theis, S.; Gollhofer, A. Substrate utilization and cycling performance following palatinose ingestion: A randomized, double-blind, controlled trial. Nutrients 2016, 8, 390. [Google Scholar] [CrossRef] [PubMed]
- König, D.; Luther, W.; Polland, V.; Berg, A. Carbohydrates in sports nutrition impact of the glycemic index. AgroFood 2007, 18, 9–10. [Google Scholar]
- Oosthuyse, T.; Carstens, M.; Millen, A.M. Ingesting isomaltulose versus fructose-maltodextrin during prolonged moderate-heavy exercise increases fat oxidation but impairs gastrointestinal comfort and cycling performance. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 427–438. [Google Scholar] [CrossRef] [PubMed]
- West, D.J.; Morton, R.D.; Stephens, J.W.; Bain, S.C.; Kilduff, L.P.; Luzio, S.; Still, R.; Bracken, R.M. Isomaltulose improves postexercise glycemia by reducing cho oxidation in t1dm. Med. Sci. Sports Exerc. 2011, 43, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Hooper, D.R.; Szivak, T.K.; Kupchak, B.R.; Dunn-Lewis, C.; Comstock, B.A.; Flanagan, S.D.; Looney, D.P.; Sterczala, A.J.; DuPont, W.H.; et al. The addition of beta-hydroxy-beta-methylbutyrate and isomaltulose to whey protein improves recovery from highly demanding resistance exercise. J. Am. Coll. Nutr. 2015, 34, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.D.; Walker, M.; Trenell, M.I.; Stevenson, E.J.; Turner, D.; Bracken, R.M.; Shaw, J.A.; West, D.J. A low-glycemic index meal and bedtime snack prevents postprandial hyperglycemia and associated rises in inflammatory markers, providing protection from early but not late nocturnal hypoglycemia following evening exercise in type 1 diabetes. Diabetes Care 2014, 37, 1845–1853. [Google Scholar] [CrossRef] [PubMed]
- Blaak, E.E.; Antoine, J.M.; Benton, D.; Bjorck, I.; Bozzetto, L.; Brouns, F.; Diamant, M.; Dye, L.; Hulshof, T.; Holst, J.J.; et al. Impact of postprandial glycaemia on health and prevention of disease. Obes. Rev. 2012, 13, 923–984. [Google Scholar] [CrossRef] [PubMed]
- Dye, L.; Gilsenan, M.B.; Quadt, F.; Martens, V.E.; Bot, A.; Lasikiewicz, N.; Camidge, D.; Croden, F.; Lawton, C. Manipulation of glycemic response with isomaltulose in a milk-based drink does not affect cognitive performance in healthy adults. Mol. Nutr. Food Res. 2010, 54, 506–515. [Google Scholar] [CrossRef] [PubMed]
- Sekartini, R.; Wiguna, T.; Bardosono, S.; Novita, D.; Arsianti, T.; Calame, W.; Schaafsma, A. The effect of lactose-isomaltulose-containing growing-up milks on cognitive performance of indonesian children: A cross-over study. Br. J. Nutr. 2013, 110, 1089–1097. [Google Scholar] [CrossRef] [PubMed]
- Taib, M.N.; Shariff, Z.M.; Wesnes, K.A.; Saad, H.A.; Sariman, S. The effect of high lactose-isomaltulose on cognitive performance of young children. A double blind cross-over design study. Appetite 2012, 58, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Young, H.B.; Benton, D. The glycemic load of meals, cognition and mood in middle and older aged adults with differences in glucose tolerance: A randomized trial. e-SPEN J. 2014, 9, e147–e154. [Google Scholar] [CrossRef]
- Young, H.; Benton, D. The effect of using isomaltulose (palatinose) to modulate the glycaemic properties of breakfast on the cognitive performance of children. Eur. J. Nutr. 2015, 54, 1013–1020. [Google Scholar] [CrossRef] [PubMed]
- Kahlhofer, J.; Lagerpusch, M.; Enderle, J.; Eggeling, B.; Braun, W.; Pape, D.; Muller, M.J.; Bosy-Westphal, A. Carbohydrate intake and glycemic index affect substrate oxidation during a controlled weight cycle in healthy men. Eur. J. Clin. Nutr. 2014, 68, 1060–1066. [Google Scholar] [CrossRef] [PubMed]
- Larsen, T.M.; Dalskov, S.M.; van Baak, M.; Jebb, S.A.; Papadaki, A.; Pfeiffer, A.F.; Martinez, J.A.; Handjieva-Darlenska, T.; Kunesova, M.; Pihlsgard, M.; et al. Diets with high or low protein content and glycemic index for weight-loss maintenance. N. Engl. J. Med. 2010, 363, 2102–2113. [Google Scholar] [CrossRef] [PubMed]
- Gower, B.A.; Hunter, G.R.; Chandler-Laney, P.C.; Alvarez, J.A.; Bush, N.C. Glucose metabolism and diet predict changes in adiposity and fat distribution in weight-reduced women. Obesity (Silver Spring) 2010, 18, 1532–1537. [Google Scholar] [CrossRef] [PubMed]
- Feinman, R.D.; Fine, E.J. Nonequilibrium thermodynamics and energy efficiency in weight loss diets. Theor. Biol. Med. Model. 2007, 4, 27. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, D.S. The glycemic index: Physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA 2002, 287, 2414–2423. [Google Scholar] [CrossRef] [PubMed]
- Boule, N.G.; Chaput, J.P.; Doucet, E.; Richard, D.; Despres, J.P.; Bouchard, C.; Tremblay, A. Glucose homeostasis predicts weight gain: Prospective and clinical evidence. Diabetes Metab. Res. Rev. 2008, 24, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Froidevaux, F.; Schutz, Y.; Christin, L.; Jequier, E. Energy expenditure in obese women before and during weight loss, after refeeding, and in the weight-relapse period. Am. J. Clin. Nutr. 1993, 57, 35–42. [Google Scholar] [PubMed]
- Diaz, E.O.; Galgani, J.E.; Aguirre, C.A. Glycaemic index effects on fuel partitioning in humans. Obes. Rev. 2006, 7, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Bosy-Westphal, A.; Hagele, F.; Nas, A. Impact of dietary glycemic challenge on fuel partitioning. Eur. J. Clin. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Casimir, M.; de Andrade, P.B.; Gjinovci, A.; Montani, J.P.; Maechler, P.; Dulloo, A.G. A role for pancreatic beta-cell secretory hyperresponsiveness in catch-up growth hyperinsulinemia: Relevance to thrifty catch-up fat phenotype and risks for type 2 diabetes. Nutr. Metab. (Lond.) 2011, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Reichkendler, M.H.; Auerbach, P.; Rosenkilde, M.; Christensen, A.N.; Holm, S.; Petersen, M.B.; Lagerberg, A.; Larsson, H.B.; Rostrup, E.; Mosbech, T.H.; et al. Exercise training favors increased insulin-stimulated glucose uptake in skeletal muscle in contrast to adipose tissue: A randomized study using fdg pet imaging. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E496–E506. [Google Scholar] [CrossRef] [PubMed]
- Alibegovic, A.C.; Hojbjerre, L.; Sonne, M.P.; van Hall, G.; Stallknecht, B.; Dela, F.; Vaag, A. Impact of 9 days of bed rest on hepatic and peripheral insulin action, insulin secretion, and whole-body lipolysis in healthy young male offspring of patients with type 2 diabetes. Diabetes 2009, 58, 2749–2756. [Google Scholar] [CrossRef] [PubMed]
- Kahlhofer, J.; Karschin, J.; Silberhorn-Buhler, H.; Breusing, N.; Bosy-Westphal, A. Effect of low-glycemic-sugar-sweetened beverages on glucose metabolism and macronutrient oxidation in healthy men. Int. J. Obes. (Lond.) 2016, 40, 990–997. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Gilhooly, C.H.; Golden, J.K.; Pittas, A.G.; Fuss, P.J.; Cheatham, R.A.; Tyler, S.; Tsay, M.; McCrory, M.A.; Lichtenstein, A.H.; et al. Long-term effects of 2 energy-restricted diets differing in glycemic load on dietary adherence, body composition, and metabolism in calerie: A 1-year randomized controlled trial. Am. J. Clin. Nutr. 2007, 85, 1023–1030. [Google Scholar] [PubMed]
- Karl, J.P.; Roberts, S.B.; Schaefer, E.J.; Gleason, J.A.; Fuss, P.; Rasmussen, H.; Saltzman, E.; Das, S.K. Effects of carbohydrate quantity and glycemic index on resting metabolic rate and body composition during weight loss. Obesity (Silver Spring) 2015, 23, 2190–2198. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.A.; Swain, J.; Goldfine, A.B.; Rifai, N.; Ludwig, D.S. Effects of a low-glycemic load diet on resting energy expenditure and heart disease risk factors during weight loss. JAMA 2004, 292, 2482–2490. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Han, S.; Chen, G.C.; Li, Z.N.; Silva-Zolezzi, I.; Pares, G.V.; Wang, Y.; Qin, L.Q. Effects of low-glycemic-index diets in pregnancy on maternal and newborn outcomes in pregnant women: A meta-analysis of randomized controlled trials. Eur. J. Nutr. 2016, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kizirian, N.V.; Kong, Y.; Muirhead, R.; Brodie, S.; Garnett, S.P.; Petocz, P.; Sim, K.A.; Celermajer, D.S.; Louie, J.C.; Markovic, T.P.; et al. Effects of a low-glycemic index diet during pregnancy on offspring growth, body composition, and vascular health: A pilot randomized controlled trial. Am. J. Clin. Nutr. 2016, 103, 1073–1082. [Google Scholar] [CrossRef] [PubMed]
- Kizirian, N.V.; Markovic, T.P.; Muirhead, R.; Brodie, S.; Garnett, S.P.; Louie, J.C.; Petocz, P.; Ross, G.P.; Brand-Miller, J.C. Macronutrient balance and dietary glycemic index in pregnancy predict neonatal body composition. Nutrients 2016, 8, 270. [Google Scholar] [CrossRef] [PubMed]
- Ajala, O.; English, P.; Pinkney, J. Systematic review and meta-analysis of different dietary approaches to the management of type 2 diabetes. Am. J. Clin. Nutr. 2013, 97, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Slame, G.; Lean, M.; Toeller, M.; Chantelau, E.; van Gaal, L.; Heine, R.; Karamanos, B.; Karlstrom, B.; de Leeuw, I.; Librenti, G.; et al. Recommendations for the nutritional management of patients with diabetes mellitus. Diabetes Nutr. Metab. 1995, 8, 186–189. [Google Scholar]
- Brunner, S.; Holub, I.; Theis, S.; Gostner, A.; Melcher, R.; Wolf, P.; Amann-Gassner, U.; Scheppach, W.; Hauner, H. Metabolic effects of replacing sucrose by isomaltulose in subjects with type 2 diabetes: A randomized double-blind trial. Diabetes Care 2012, 35, 1249–1251. [Google Scholar] [CrossRef] [PubMed]
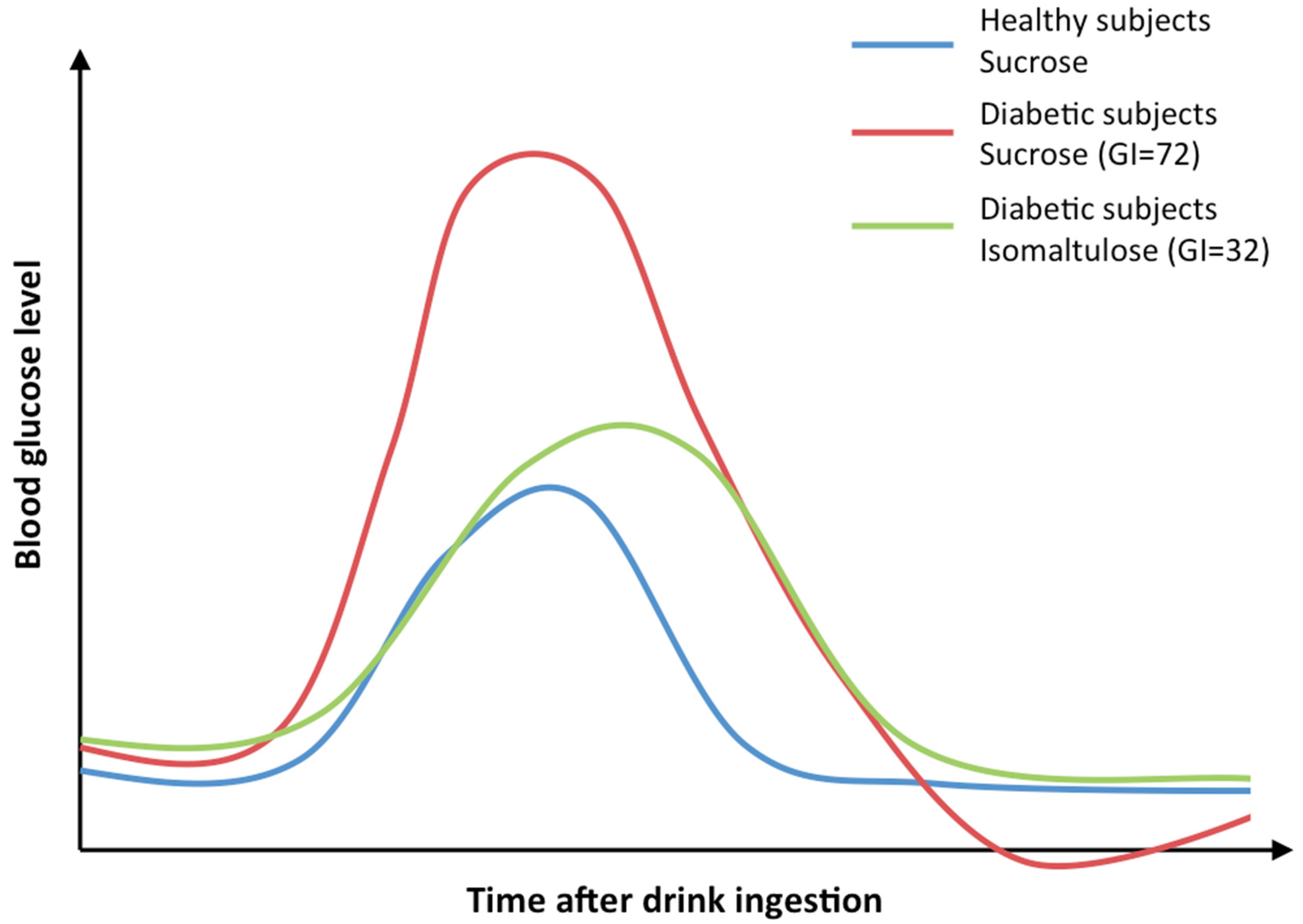
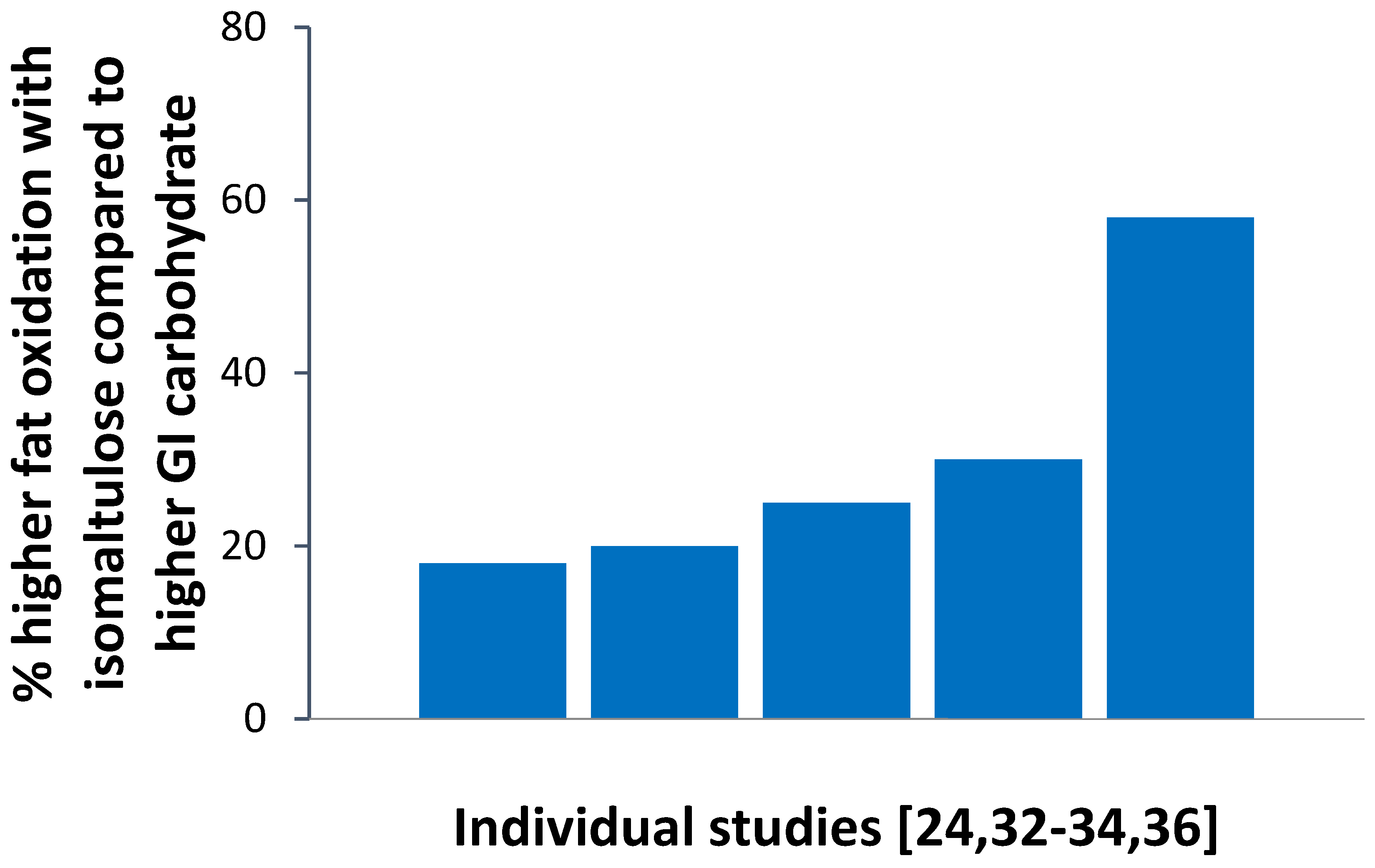
| Application | Observed Effects | Studies Using Isomaltulose Drinks | Studies Using Low-GI Diets |
|---|---|---|---|
| Diabetes mellitus | Isomaltulose drinks: 20%–50% reduced glucose and insulin levels as compared with sucrose or maltodextrin single drink; Delay of peak glucose level; No fermentation up to 75 g per drink; Reduced amount of total glucose in the systemic blood circulation; Increased first-pass splanchnic glucose uptake; Low GI diet: Glycated hemoglobin 1Ac reduced 0.1%–0.5% | [23,24,25,26,27,28,29,30,31] | [5,64,65] |
| Sports | Promotes a higher contribution of fat oxidation in energy metabolism; Improved physical performance; Protection against hypoglycemia during exercise | [24,31,32,33,34,35,36,40] | [24,37,38] |
| Cognitive performance | Positive effects on mood; Improved episodic and working memory; Improved attention speed | [41,42] | [43,44] |
| Body weight and composition | Facilitation of weight maintenance; Increased fat oxidation | [57] | [45,46,47,58,59,60] |
| Pregnancy outcome | Reduced gestational weight gain and birth weight; Reduced proportion of babies born large for gestational age | - | [61,62,63] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maresch, C.C.; Petry, S.F.; Theis, S.; Bosy-Westphal, A.; Linn, T. Low Glycemic Index Prototype Isomaltulose—Update of Clinical Trials. Nutrients 2017, 9, 381. https://doi.org/10.3390/nu9040381
Maresch CC, Petry SF, Theis S, Bosy-Westphal A, Linn T. Low Glycemic Index Prototype Isomaltulose—Update of Clinical Trials. Nutrients. 2017; 9(4):381. https://doi.org/10.3390/nu9040381
Chicago/Turabian StyleMaresch, Constanze Christin, Sebastian Friedrich Petry, Stephan Theis, Anja Bosy-Westphal, and Thomas Linn. 2017. "Low Glycemic Index Prototype Isomaltulose—Update of Clinical Trials" Nutrients 9, no. 4: 381. https://doi.org/10.3390/nu9040381
APA StyleMaresch, C. C., Petry, S. F., Theis, S., Bosy-Westphal, A., & Linn, T. (2017). Low Glycemic Index Prototype Isomaltulose—Update of Clinical Trials. Nutrients, 9(4), 381. https://doi.org/10.3390/nu9040381





