Inverse Association of Plasma Chromium Levels with Newly Diagnosed Type 2 Diabetes: A Case-Control Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Data Collection
2.3. Measurement of Glucose and Lipid Biomarkers
2.4. Measurement of Plasma Chromium Concentrations
2.5. Statistical Analysis
3. Results
Patient Characteristics
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Freund, H.; Atamian, S.; Fischer, J.E. Chromium deficiency during total parenteral nutrition. J. Am. Med. Assoc. 1979, 241, 496–498. [Google Scholar] [CrossRef]
- Jeejeebhoy, K.N.; Chu, R.C.; Marliss, E.B.; Greenberg, G.R.; Bruce-Robertson, A. Chromium deficiency, glucose intolerance, and neuropathy reversed by chromium supplementation, in a patient receiving long-term total parenteral nutrition. Am. J. Clin. Nutr. 1977, 30, 531–538. [Google Scholar] [PubMed]
- Wang, Z.Q.; Zhang, X.H.; Russell, J.C.; Hulver, M.; Cefalu, W.T. Chromium picolinate enhances skeletal muscle cellular insulin signaling in vivo in obese, insulin-resistant JCR:LA-cp rats. J. Nutr. 2006, 136, 415–420. [Google Scholar] [PubMed]
- Sreejayan, N.; Dong, F.; Kandadi, M.R.; Yang, X.; Ren, J. Chromium alleviates glucose intolerance, insulin resistance, and hepatic ER stress in obese mice. Obesity 2008, 16, 1331–1337. [Google Scholar] [CrossRef] [PubMed]
- Cefalu, W.T.; Wang, Z.Q.; Zhang, X.H.; Baldor, L.C.; Russell, J.C. Oral chromium picolinate improves carbohydrate and lipid metabolism and enhances skeletal muscle Glut-4 translocation in obese, hyperinsulinemic (JCR-LA corpulent) rats. J. Nutr. 2002, 132, 1107–1114. [Google Scholar] [PubMed]
- Yang, X.; Li, S.Y.; Dong, F.; Ren, J.; Sreejayan, N. Insulin-sensitizing and cholesterol-lowering effects of chromium (D-Phenylalanine)3. J. Inorg. Biochem. 2006, 100, 1187–1193. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.K.; Rains, J.L.; Croad, J.L. Effect of chromium niacinate and chromium picolinate supplementation on lipid peroxidation, TNF-alpha, IL-6, CRP, glycated hemoglobin, triglycerides, and cholesterol levels in blood of streptozotocin-treated diabetic rats. Free Radic. Biol. Med. 2007, 43, 1124–1131. [Google Scholar] [CrossRef] [PubMed]
- McIver, D.J.; Grizales, A.M.; Brownstein, J.S.; Goldfine, A.B. Risk of type 2 diabetes is lower in US adults taking chromium-containing supplements. J. Nutr. 2015, 145, 2675–2682. [Google Scholar] [CrossRef] [PubMed]
- Balk, E.M.; Tatsioni, A.; Lichtenstein, A.H.; Lau, J.; Pittas, A.G. Effect of chromium supplementation on glucose metabolism and lipids: A systematic review of randomized controlled trials. Diabetes Care 2007, 30, 2154–2163. [Google Scholar] [CrossRef] [PubMed]
- Yin, R.V.; Phung, O.J. Effect of chromium supplementation on glycated hemoglobin and fasting plasma glucose in patients with diabetes mellitus. Nutr. J. 2015, 14, 14. [Google Scholar] [CrossRef] [PubMed]
- Althuis, M.D.; Jordan, N.E.; Ludington, E.A.; Wittes, J.T. Glucose and insulin responses to dietary chromium supplements: A meta-analysis. Am. J. Clin. Nutr. 2002, 76, 148–155. [Google Scholar] [PubMed]
- Evert, A.B.; Boucher, J.L.; Cypress, M.; Dunbar, S.A.; Franz, M.J.; Mayer-Davis, E.J.; Neumiller, J.J.; Nwankwo, R.; Verdi, C.L.; Urbanski, P.; et al. Nutrition therapy recommendations for the management of adults with diabetes. Diabetes Care 2013, 36, 3821–3842. [Google Scholar] [CrossRef] [PubMed]
- Costello, R.B.; Dwyer, J.T.; Bailey, R.L. Chromium supplements for glycemic control in type 2 diabetes: Limited evidence of effectiveness. Nutr. Rev. 2016, 74, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Q.; Cefalu, W.T. Current concepts about chromium supplementation in type 2 diabetes and insulin resistance. Curr. Diabetes Rep. 2010, 10, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Ekmekcioglu, C.; Prohaska, C.; Pomazal, K.; Steffan, I.; Schernthaner, G.; Marktl, W. Concentrations of seven trace elements in different hematological matrices in patients with type 2 diabetes as compared to healthy controls. Biol. Trace Elem. Res. 2001, 79, 205–219. [Google Scholar] [CrossRef]
- Basaki, M.; Saeb, M.; Nazifi, S.; Shamsaei, H.A. Zinc, copper, iron, and chromium concentrations in young patients with type 2 diabetes mellitus. Biol. Trace Elem. Res. 2012, 148, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Nichols, G.A.; Hillier, T.A.; Brown, J.B. Progression from newly acquired impaired fasting glusose to type 2 diabetes. Diabetes Care 2007, 30, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Barr, E.L.; Zimmet, P.Z.; Welborn, T.A.; Jolley, D.; Magliano, D.J.; Dunstan, D.W.; Cameron, A.J.; Dwyer, T.; Taylor, H.R.; Tonkin, A.M.; et al. Risk of cardiovascular and all-cause mortality in individuals with diabetes mellitus, impaired fasting glucose, and impaired glucose tolerance: The Australian Diabetes, Obesity, and Lifestyle Study (AusDiab). Circulation 2007, 116, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.I.; Duncan, B.B.; Bang, H.; Pankow, J.S.; Ballantyne, C.M.; Golden, S.H.; Folsom, A.R.; Chambless, L.E. Identifying individuals at high risk for diabetes: The atherosclerosis risk in communities study. Diabetes Care 2005, 28, 2013–2018. [Google Scholar] [CrossRef] [PubMed]
- Levitzky, Y.S.; Pencina, M.J.; D’Agostino, R.B.; Meigs, J.B.; Murabito, J.M.; Vasan, R.S.; Fox, C.S. Impact of impaired fasting glucose on cardiovascular disease: The Framingham Heart Study. J. Am. Coll. Cardiol. 2008, 51, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Twigg, S.M.; Kamp, M.C.; Davis, T.M.; Neylon, E.K.; Flack, J.R. Prediabetes: A position statement from the Australian Diabetes Society and Australian Diabetes Educators Association. Med. J. Aust. 2007, 186, 461–465. [Google Scholar] [PubMed]
- Alberti, K.G.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet. Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- Song, F.; Jia, W.; Yao, Y.; Hu, Y.; Lei, L.; Lin, J.; Sun, X.; Liu, L. Oxidative stress, antioxidant status and DNA damage in patients with impaired glucose regulation and newly diagnosed type 2 diabetes. Clin. Sci. 2007, 112, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Brune, D.; Aitio, A.; Nordberg, G.; Vesteberg, O.; Gerhardsson, L. Normal concentrations of chromium in serum and urine—A TRACY project. Scand. J. Work Environ. Health 1993, 19 (Suppl. 1), S39–S44. [Google Scholar]
- Lin, C.C.; Tsweng, G.J.; Lee, C.F.; Chen, B.H.; Huang, Y.L. Magnesium, zinc, and chromium levels in children, adolescents, and young adults with type 1 diabetes. Clin. Nutr. 2016, 35, 880–884. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.A.; Bryden, N.A.; Polansky, M.M. Dietary chromium intake. Biol. Trace Elem. Res. 1992, 32, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, G.V.; Kawamura, H.; Parr, R.M.; Miah, F.K.; Wang, J.X.; Dang, H.S.; Djojosubroto, H.; Cho, S.Y.; Akher, P.; Natera, E.S.; et al. Dietary intake of essential minor and trace elements from Asian diets. Food Nutr. Bull. 2002, 23, 124–128. [Google Scholar] [PubMed]
- Kamerud, K.L.; Hobbie, K.A.; Anderson, K.A. Stainless steel leaches nickel and chromium into foods during cooking. J. Agric. Food Chem. 2013, 61, 9495–9501. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Zhou, T.; Li, Q.; Lu, L.; Lin, C. Anthropogenic chromium emissions in China from 1990 to 2009. PLoS ONE 2014, 9, e87753. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Hou, H.; Zhu, F.; Wang, A.; Liu, Y.; Hu, Q. Simultaneous determination of chromium, cadmium, and lead and evaluation of the correlation between chromium and cotinine in Chinese smokers. Biol. Trace Elem. Res. 2014, 158, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Vieira, E.; Soares, M.E.; Kozior, M.; Krejpcio, Z.; Ferreira, I.M.; Bastos, M.L. Quantification of total and hexavalent chromium in lager beers: Variability between styles and estimation of daily intake of chromium from beer. J. Agric. Food Chem. 2014, 62, 9195–9200. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Xun, P.; Morris, S.; Jacobs, D.R., Jr.; Liu, K.; He, K. Chromium exposure and incidence of metabolic syndrome among American young adults over a 23-year follow-up: The CARDIA Trace Element Study. Sci. Rep. 2015, 5, 15606. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.M.; Vincent, J.B. Chromium oligopeptide activates insulin receptor tyrosine kinase activity. Biochemistry 1997, 36, 4382–4385. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Kruszewski, A.; Brautigan, D.L. Cellular chromium enhances activation of insulin receptor kinase. Biochemistry 2005, 44, 8167–8175. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Liu, P.; Pattar, G.R.; Tackett, L.; Bhonagiri, P.; Strawbridge, A.B.; Elmendorf, J.S. Chromium activates glucose transporter 4 trafficking and enhances insulin-stimulated glucose transport in 3T3-L1 adipocytes via a cholesterol-dependent mechanism. Mol. Endocrinol. 2006, 20, 857–870. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Wang, J.; Ma, H.; Xiao, Y.; He, L.; Tong, C.; Wang, Z.; Zheng, Q.; Dolence, E.K.; Nair, S.; et al. A newly synthetic chromium complex-chromium (D-phenylalanine)3 activates AMP-activated protein kinase and stimulates glucose transport. Biochem. Pharmacol. 2009, 77, 1002–1010. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.K.; Kannan, K. Chromium chloride inhibits oxidative stress and TNF-alpha secretion caused by exposure to high glucose in cultured U937 monocytes. Biochem. Biophys. Res. Commun. 2001, 289, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Morris, B.W.; MacNeil, S.; Hardisty, C.A.; Heller, S.; Burgin, C.; Gray, T.A. Chromium homeostasis in patients with type II (NIDDM) diabetes. J. Trace Elem. Med. Biol. 1999, 13, 57–61. [Google Scholar] [CrossRef]
- Minoia, C.; Cavalleri, A. Chromium in urine, serum and red blood cells in the biological monitoring of workers exposed to different chromium valency states. Sci. Total Environ. 1988, 71, 323–327. [Google Scholar] [CrossRef]
- Cefalu, W.T.; Hu, F.B. Role of chromium in human health and in diabetes. Diabetes Care 2004, 27, 2741–2751. [Google Scholar] [CrossRef] [PubMed]
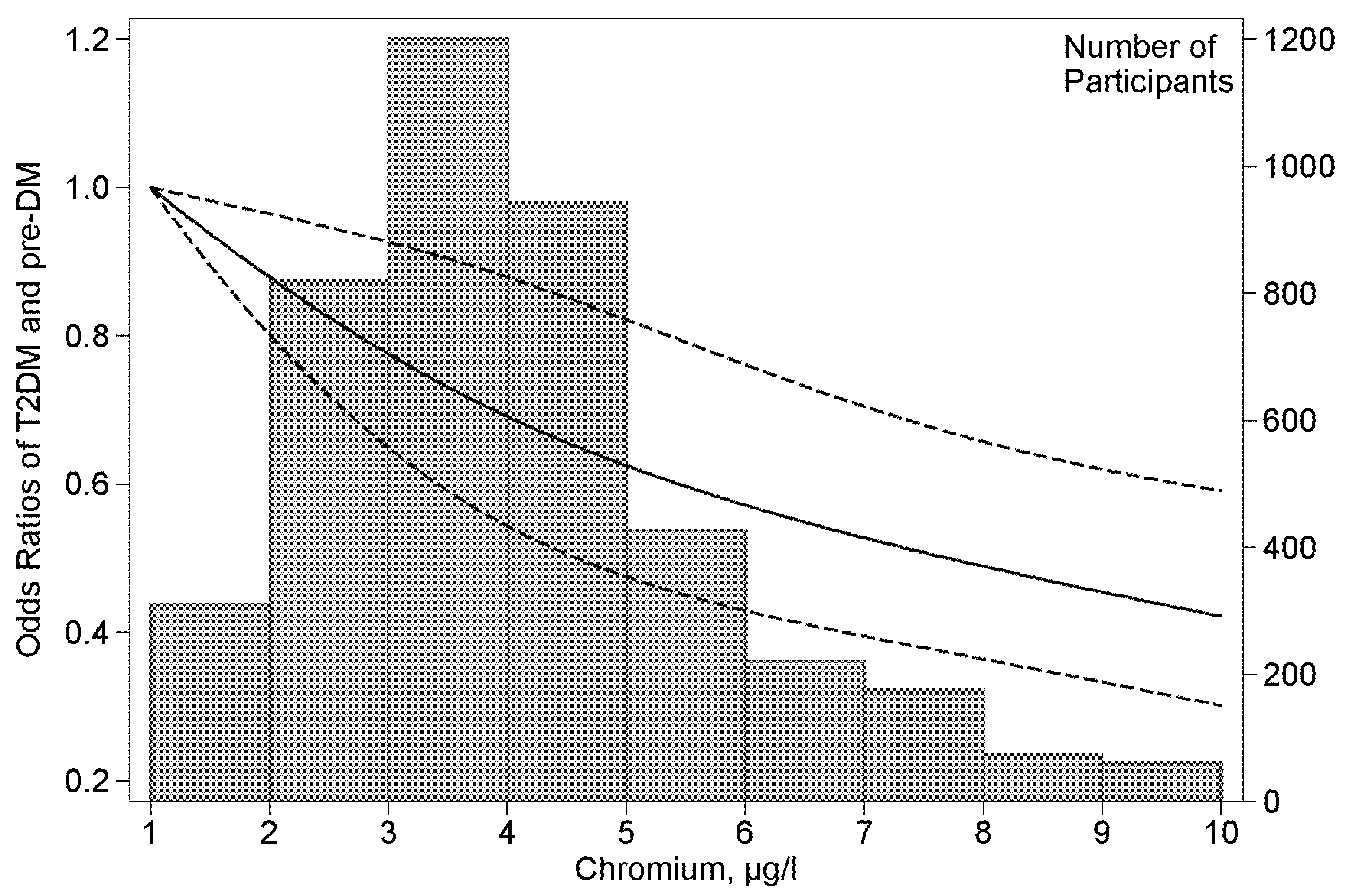
| Parameters | NGT (n = 2290) | Pre-DM (n = 682) | T2DM (n = 1471) | p Value | |
|---|---|---|---|---|---|
| Pre-DM vs. NGT | T2DM vs. NGT | ||||
| Male sex, n (%) | 1481 (64.67%) | 409 (59.97%) | 936 (63.63%) | 0.022 | 0.514 |
| Age (year) | 46.93 (10.28) | 49.47 (9.93) | 48.39 (9.60) | <0.001 | <0.001 |
| BMI (kg/m2) | 23.24 (3.06) | 24.80 (3.28) | 25.20 (3.36) | <0.001 | <0.001 |
| Current smoker, n (%) | 924 (40.34%) | 193 (28.30%) | 562 (38.21%) | <0.001 | 0.322 |
| Current drinker, n (%) | 828 (36.16%) | 210 (30.79%) | 537 (36.51%) | 0.028 | 0.493 |
| Family history of diabetes, n (%) | 239 (10.44%) | 138 (20.23%) | 335 (22.77%) | <0.001 | <0.001 |
| Hypertension, n (%) | 542 (23.67%) | 242 (35.48%) | 524 (35.62%) | <0.001 | <0.001 |
| FPG (mmol/L) | 5.19 (0.75) | 6.11 (0.54) | 9.04 (3.21) | <0.001 | <0.001 |
| OGTT 2 h (mmol/L) | 6.41 (0.95) | 8.81 (1.36) | 16.79 (4.93) | <0.001 | <0.001 |
| FPI (mmol/L) | 6.85 (4.66) | 9.23 (5.86) | 10.65 (8.34) | <0.001 | <0.001 |
| HOMA-IR | 1.67 (1.18) | 2.50 (1.72) | 3.85 (3.01) | <0.001 | <0.001 |
| HOMA-β | 71.34 (57.45) | 73.87 (51.78) | 46.26 (61.67) | 0.612 | 0.879 |
| TC (mmol/L) | 4.40 (1.23) | 4.41 (1.53) | 4.42 (1.58) | 0.069 | 0.909 |
| TG (mmol/L) | 1.30 (0.78) | 1.26 (0.88) | 1.40 (1.02) | 0.831 | <0.001 |
| HDLC (mmol/L) | 1.35 (0.34) | 1.18 (0.53) | 1.12 (0.55) | <0.001 | <0.001 |
| LDLC (mmol/L) | 2.37 (0.97) | 2.43 (1.14) | 2.44 (1.32) | 0.344 | 0.090 |
| Chromium (μg/L) | 3.96 (2.19) | 3.61 (1.77) | 3.68 (1.97) | <0.001 | <0.001 |
| Variables | Quartiles of Plasma Chromium Concentrations | Per 1 SD of Log-Plasma Chromium | p Value for Trend | |||
|---|---|---|---|---|---|---|
| 1 (Lowest) | 2 | 3 | 4 (Highest) | |||
| <3.04 μg/L | 3.04–3.96 μg/L | 3.96–5.22 μg/L | >5.22 μg/L | |||
| T2DM/NGT | ||||||
| Crude | 1 | 0.74 (0.62–0.89) | 0.74 (0.62–0.88) | 0.56 (0.46–0.67) | 0.86 (0.81–0.92) | <0.001 |
| Model 1 | 1 | 0.70 (0.58–0.85) | 0.70 (0.58–0.85) | 0.53 (0.43–0.66) | 0.86 (0.80–0.92) | <0.001 |
| Model 2 | 1 | 0.67 (0.55–0.83) | 0.64 (0.51–0.79) | 0.58 (0.46–0.73) | 0.88 (0.82–0.95) | <0.001 |
| Pre-DM/NGT | ||||||
| Crude | 1 | 0.83 (0.66–1.05) | 0.78 (0.62–0.98) | 0.50 (0.38–0.64) | 0.88 (0.82–0.95) | <0.001 |
| Model 1 | 1 | 0.73 (0.57–0.93) | 0.69 (0.54–0.89) | 0.52 (0.39–0.69) | 0.88 (0.81–0.96) | <0.001 |
| Model 2 | 1 | 0.70 (0.54–0.91) | 0.67 (0.52–0.88) | 0.58 (0.43–0.78) | 0.91 (0.83–0.99) | <0.001 |
| T2DM&Pre-DM/NGT | ||||||
| Crude | 1 | 0.77 (0.65–0.90) | 0.75 (0.64–0.88) | 0.54 (0.45–0.64) | 0.86 (0.81–0.91) | <0.001 |
| Model 1 | 1 | 0.71 (0.60–0.85) | 0.71 (0.59–0.84) | 0.53 (0.44–0.64) | 0.86 (0.80–0.91) | <0.001 |
| Model 2 | 1 | 0.68 (0.57–0.82) | 0.66 (0.54–0.79) | 0.58 (0.47–0.71) | 0.88 (0.82–0.94) | <0.001 |
| Participant | Quartiles of Plasma Chromium Concentrations | Per 1 SD Log-Plasma Cr | p for Interaction | |||
|---|---|---|---|---|---|---|
| 1 (Lowest) | 2 | 3 | 4 (Highest) | |||
| <3.04 μg/L | 3.04–3.96 μg/L | 3.96–5.22 μg/L | >5.22 μg/L | |||
| Subgroup | ||||||
| Age | <0.001 | |||||
| <45 | 1 | 1.00 (0.74–1.36) | 1.08 (0.75–1.23) | 0.45 (0.33–0.62) | 0.88 (0.80–0.97) | |
| ≥45 | 1 | 0.56 (0.44–0.71) | 0.47 (0.37–0.61) | 0.85 (0.63–1.13) | 0.90 (0.80–1.00) | |
| Sex | 0.706 | |||||
| Women | 1 | 0.57 (0.42–0.81) | 0.59 (0.42–0.81) | 0.55 (0.39–0.79) | 0.90 (0.80–1.01) | |
| Men | 1 | 0.76 (0.60–0.95) | 0.69 (0.55–0.88) | 0.59 (0.45–0.76) | 0.87 (0.80–0.95) | |
| BMI | 0.424 | |||||
| <24 | 1 | 0.56 (0.43–0.74) | 0.59 (0.45–0.77) | 0.56 (0.41–0.74) | 0.86 (0.79–0.95) | |
| ≥24 | 1 | 0.82 (0.63–1.06) | 0.75 (0.57–0.97) | 0.62 (0.47–0.83) | 0.91 (0.82–1.00) | |
| Smoking | 0.195 | |||||
| No | 1 | 0.69 (0.55–0.86) | 0.68 (0.54–0.86) | 0.69 (0.53–0.90) | 0.93 (0.85–1.01) | |
| Yes | 1 | 0.68 (0.49–0.95) | 0.61 (0.45–0.83) | 0.44 (0.31–0.61) | 0.81 (0.72–0.71) | |
| Drinking alcohol | 0.026 | |||||
| No | 1 | 0.69 (0.55–0.87) | 0.72 (0.57–0.91) | 0.72 (0.56–0.93) | 0.96 (0.88–1.05) | |
| Yes | 1 | 0.71 (0.51–0.99) | 0.55 (0.40–0.76) | 0.38 (0.26–0.54) | 0.74 (0.65–0.84) | |
| Hypertension | 0.125 | |||||
| No | 1 | 0.66 (0.53–0.82) | 0.71 (0.57–0.89) | 0.53 (0.41–0.67) | 0.87 (0.80–0.94) | |
| Yes | 1 | 0.75 (0.53–1.07) | 0.58 (0.41–0.82) | 0.70 (0.48–1.02) | 0.91 (0.79–1.04) | |
| Overall | 1 | 0.68 (0.57–0.82) | 0.66 (0.54–0.79) | 0.58 (0.47–0.71) | 0.88 (0.82–0.94) | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.; Jin, X.; Shan, Z.; Li, S.; Yin, J.; Sun, T.; Luo, C.; Yang, W.; Yao, P.; Yu, K.; et al. Inverse Association of Plasma Chromium Levels with Newly Diagnosed Type 2 Diabetes: A Case-Control Study. Nutrients 2017, 9, 294. https://doi.org/10.3390/nu9030294
Chen S, Jin X, Shan Z, Li S, Yin J, Sun T, Luo C, Yang W, Yao P, Yu K, et al. Inverse Association of Plasma Chromium Levels with Newly Diagnosed Type 2 Diabetes: A Case-Control Study. Nutrients. 2017; 9(3):294. https://doi.org/10.3390/nu9030294
Chicago/Turabian StyleChen, Sijing, Xiaoling Jin, Zhilei Shan, Shuzhen Li, Jiawei Yin, Taoping Sun, Cheng Luo, Wei Yang, Ping Yao, Kaifeng Yu, and et al. 2017. "Inverse Association of Plasma Chromium Levels with Newly Diagnosed Type 2 Diabetes: A Case-Control Study" Nutrients 9, no. 3: 294. https://doi.org/10.3390/nu9030294
APA StyleChen, S., Jin, X., Shan, Z., Li, S., Yin, J., Sun, T., Luo, C., Yang, W., Yao, P., Yu, K., Zhang, Y., Cheng, Q., Cheng, J., Bao, W., & Liu, L. (2017). Inverse Association of Plasma Chromium Levels with Newly Diagnosed Type 2 Diabetes: A Case-Control Study. Nutrients, 9(3), 294. https://doi.org/10.3390/nu9030294





