We Need Studies of the Mortality Effect of Vitamin A Supplementation, Not Surveys of Vitamin A Deficiency
Abstract
:Acknowledgments
Conflicts of Interest
References
- Sommer, A. Vitamin A deficiency and mortality risk. Lancet 1984, 1, 347–348. [Google Scholar] [CrossRef]
- Beaton, G.H.; Martorell, R.; McCabe, G.; L’abbe, K.A.; Edmonston, B.; Ross, A.C. Effectiveness of Vitamin A Supplementation in the Control of Young Child Morbidity and Mortality in Developing Countries; University of Toronto: Toronto, ON, Canada, 1993. [Google Scholar]
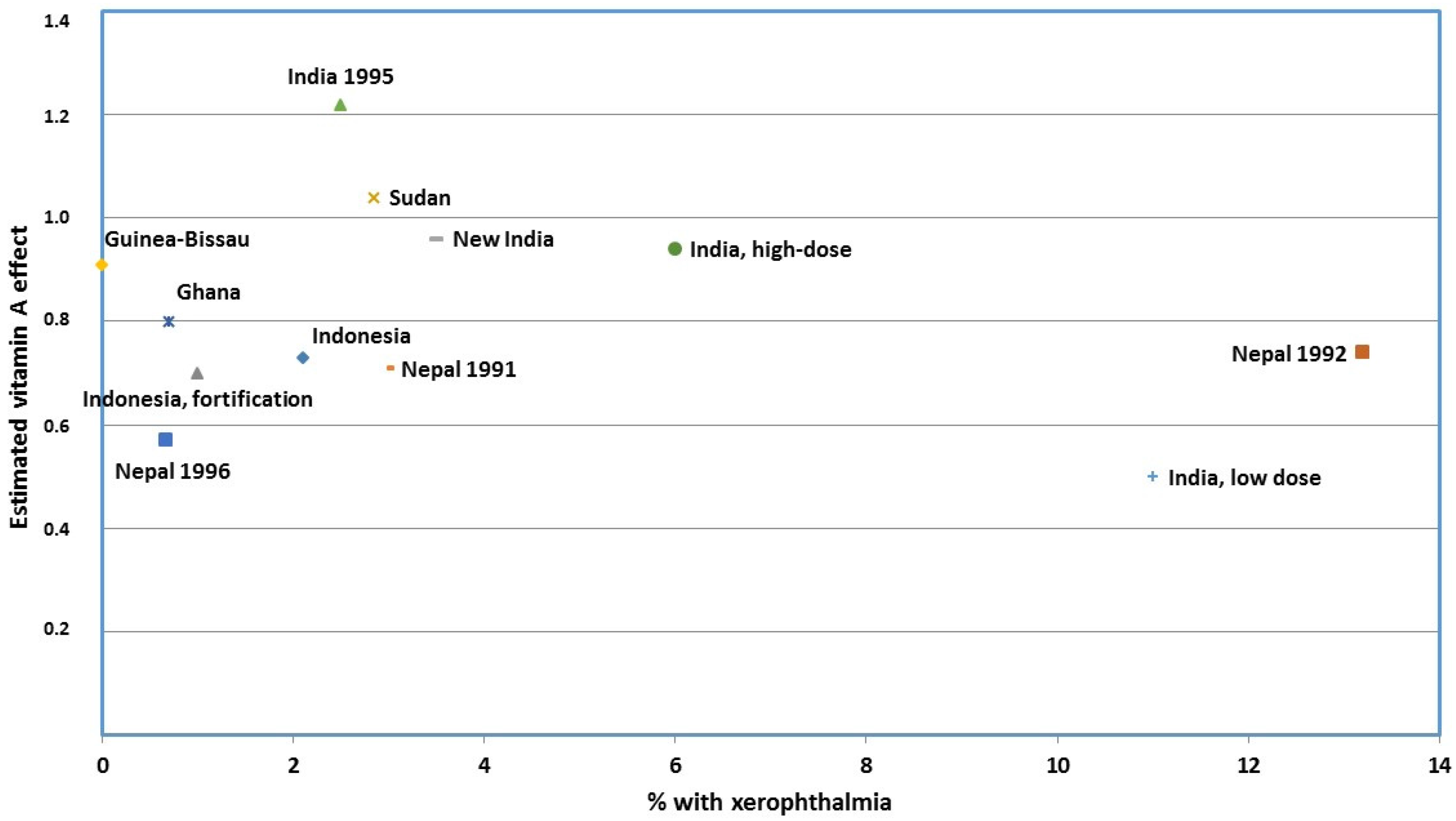
- Mason, J.; Greiner, T.; Shrimpton, R.; Sanders, D.; Yukich, J. Vitamin A policies need rethinking. Int. J. Epidemiol. 2015, 44, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Blomhoff, R.; Green, M.H.; Norum, K.R. Vitamin A: Physiological and biochemical processing. Annu. Rev. Nutr. 1992, 12, 37–57. [Google Scholar] [CrossRef] [PubMed]
- Pedro, M.R.; Madriaga, J.R.; Barba, C.V.; Habito, R.C.; Gana, A.E.; Deitchler, M.; Mason, J.B. The national vitamin A supplementation program and subclinical vitamin a deficiency among preschool children in the Philippines. Food Nutr. Bull. 2004, 25, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.B.; Ramirez, M.A.; Fernandez, C.M.; Pedro, R.; Lloren, T.; Saldanha, L.; Deitchler, M.; Eisele, T. Effects on vitamin A deficiency in children of periodic high-dose supplements and of fortified oil promotion in a deficient area of the Philippines. Int. J. Vitam. Nutr. Res. 2011, 81, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Benn, C.S.; Aaby, P.; Arts, R.J.; Jensen, K.J.; Netea, M.G.; Fisker, A.B. An enigma: Why vitamin A supplementation does not always reduce mortality even though vitamin A deficiency is associated with increased mortality. Int. J. Epidemiol. 2015, 44, 906–918. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, D.K.; Pandey, C.M.; Agarwal, K.N. Vitamin A administration and preschool child mortality. Nutr. Res. 1995, 15, 669–680. [Google Scholar] [CrossRef]
- Pant, C.R.; Pokharel, G.P.; Curtale, F.; Pokhrel, R.P.; Grosse, R.N.; Lepkowski, J.; Muhilal; Bannister, M.; Gorstein, J.; Pak-Gorstein, S.; et al. Impact of nutrition education and mega-dose vitamin A supplementation on the health of children in Nepal. Bull. World Health Organ. 1996, 74, 533–545. [Google Scholar] [PubMed]
- Awasthi, S.; Peto, R.; Read, S.; Clark, S.; Pande, V.; Bundy, D. Vitamin A supplementation every 6 months with retinol in 1 million pre-school children in north India: DEVTA, a cluster-randomised trial. Lancet 2013, 381, 1469–1477. [Google Scholar] [CrossRef]
- Fisker, A.B.; Bale, C.; Rodrigues, A.; Balde, I.; Fernandes, M.; Jorgensen, M.J.; Danneskiold-Samsøe, N.; Hornshøj, L.; Rasmussen, J.; Christensen, E.D.; et al. High-dose vitamin A with vaccination after 6 months of age: A randomized trial. Pediatrics 2014, 134, e739–e748. [Google Scholar] [CrossRef] [PubMed]
- Wirth, J.P.; Petry, N.; Tanumihardjo, S.A.; Rogers, L.M.; McLean, E.; Greig, A.; Garrett, G.S.; Klemm, R.D.; Rohner, F. Vitamin A supplementation programs and country-level evidence of vitamin A deficiency. Nutrients 2017. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benn, C.S. We Need Studies of the Mortality Effect of Vitamin A Supplementation, Not Surveys of Vitamin A Deficiency. Nutrients 2017, 9, 280. https://doi.org/10.3390/nu9030280
Benn CS. We Need Studies of the Mortality Effect of Vitamin A Supplementation, Not Surveys of Vitamin A Deficiency. Nutrients. 2017; 9(3):280. https://doi.org/10.3390/nu9030280
Chicago/Turabian StyleBenn, Christine Stabell. 2017. "We Need Studies of the Mortality Effect of Vitamin A Supplementation, Not Surveys of Vitamin A Deficiency" Nutrients 9, no. 3: 280. https://doi.org/10.3390/nu9030280
APA StyleBenn, C. S. (2017). We Need Studies of the Mortality Effect of Vitamin A Supplementation, Not Surveys of Vitamin A Deficiency. Nutrients, 9(3), 280. https://doi.org/10.3390/nu9030280



