Capsaicin: Friend or Foe in Skin Cancer and Other Related Malignancies?
Abstract
1. Introduction
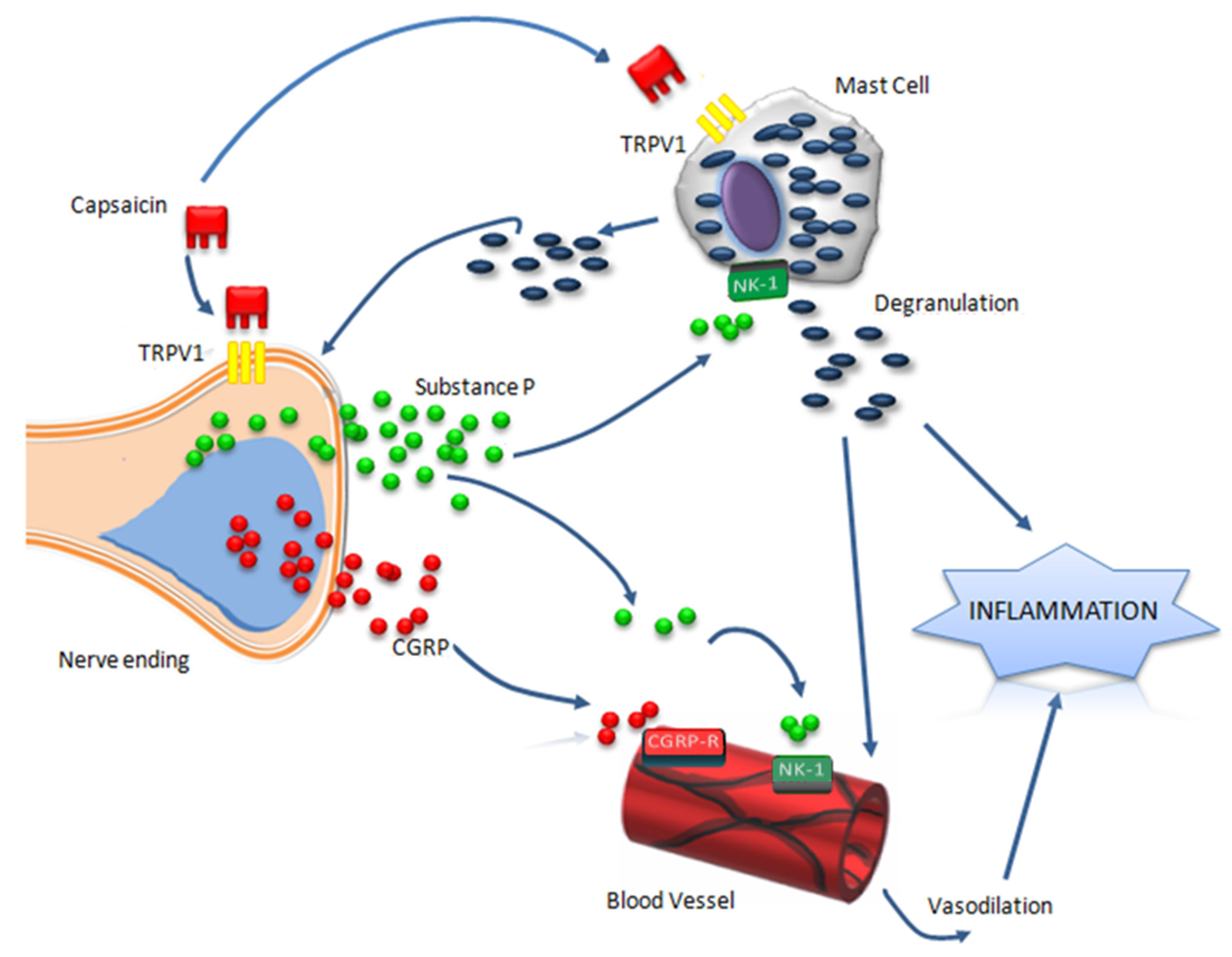
2. Capsaicin and Neurogenic Inflammation
3. Capsaicin and Cancer
3.1. The Impact of Capsaicin on Muco-Cutaneous Squamous Cell Carcinoma
3.2. The Effect of Capsaicin on Melanoma
3.3. Capsaicin’s Involvement in Carcinogenesis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Barceloux, D.G. Pepper and capsaicin (Capsicum and Piper species). Dis. Mon. 2009, 55, 380–390. [Google Scholar] [CrossRef] [PubMed]
- Kiple, K.F. The Cambridge World History of Food.(2-Volume Set); Cambridge University Press: Cambridge, UK, 2001; Volume 2. [Google Scholar]
- Bode, A.M.; Dong, Z. The two faces of capsaicin. Cancer Res. 2011, 71, 2809–2814. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.K. Role of Capsaicin in Oxidative Stress and Cancer; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013; Volume 3. [Google Scholar]
- Dasgupta, P.; Fowler, C.J. Chillies: From antiquity to urology. BJU Int. 1997, 80, 845–852. [Google Scholar] [CrossRef]
- Sharma, S.K.; Vij, A.S.; Sharma, M. Mechanisms and clinical uses of capsaicin. Eur. J. Pharmacol. 2013, 720, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Salam, O.M. Capsaicin as a Therapeutic Molecule; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2014; Volume 68. [Google Scholar]
- Papoiu, A.D.; Yosipovitch, G. Topical capsaicin. The fire of a ‘hot’ medicine is reignited. Expert Opin. Pharmacother. 2010, 11, 1359–1371. [Google Scholar] [CrossRef] [PubMed]
- Thresh, L.T. Isolation of capsaicin. Pharm. J. 1846, 6, 941–942. [Google Scholar]
- Nelson, E.K. The constitution of capsaicin, the pungent principle of capsicum. J. Am. Chem. Soc. 1919, 41, 1115–1121. [Google Scholar] [CrossRef]
- Fan, Y.; Lu, Y.M.; Yu, B.; Tan, C.P.; Cui, B. Extraction and purification of capsaicin from capsicum oleoresin using an aqueous two-phase system combined with chromatography. J. Chromatogr. B. 2017, 1063, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Fayos, O.; De Aguiar, A.C.; Jiménez-Cantizano, A.; Ferreiro-González, M.; Garcés-Claver, A.; Martínez, J.; Mallor, C.; Ruiz-Rodríguez, A.; Palma, M.; Barroso, C.G.; et al. Ontogenetic Variation of Individual and Total Capsaicinoids in Malagueta Peppers (Capsicum frutescens) during Fruit Maturation. Molecules 2017, 22, 736. [Google Scholar] [CrossRef] [PubMed]
- Späth, E.; Darling, S.F. Synthese des capsaicins. Eur. J. Inorg. Chem. 1930, 63, 737–743. [Google Scholar] [CrossRef]
- Zheng, J.; Zhou, Y.; Li, Y.; Xu, D.P.; Li, S.; Li, H.B. Spices for Prevention and Treatment of Cancers. Nutrients 2016, 8, 495. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Ran, L.; Wang, J.; Yu, L.; Lang, H.D.; Wang, X.L.; Mi, M.T.; Zhu, J.D. Capsaicin Supplementation Improved Risk Factors of Coronary Heart Disease in Individuals with Low HDL-C Levels. Nutrients 2017, 9, 1037. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Escogido, M.D.L.; Gonzalez-Mondragon, E.G.; Vazquez-Tzompantzi, E. Chemical and pharmacological aspects of capsaicin. Molecules 2011, 16, 1253–1270. [Google Scholar] [CrossRef] [PubMed]
- Caterina, M.J.; Schumacher, M.A.; Tominaga, M.; Rosen, T.A.; Levine, J.D.; Julius, D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature 1997, 389, 816–824. [Google Scholar] [PubMed]
- Tominaga, M.; Tominaga, T. Structure and function of TRPV1. Pflügers. Arch. 2005, 451, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Premkumar, L.S.; Ahern, G.P. Induction of vanilloid receptor channel activity by protein kinase C. Nature 2000, 408, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, T.; Tominaga, M.; Katsuya, H.; Mizumura, K. Bradykinin lowers the threshold temperature for heat activation of vanilloid receptor 1. J. Neurophysiol. 2002, 88, 544–548. [Google Scholar] [PubMed]
- Schnizler, K.; Shutov, L.P.; Van Kanegan, M.J.; Merrill, M.A.; Nichols, B.; McKnight, G.S.; Usachev, Y.M. Protein kinase A anchoring via AKAP150 is essential for TRPV1 modulation by forskolin and prostaglandin E2 in mouse sensory neurons. J. Neurosci. 2008, 28, 4904–4917. [Google Scholar] [CrossRef] [PubMed]
- Filippi, A.; Caruntu, C.; Gheorghe, R.O.; Deftu, A.; Amuzescu, B.; Ristoiu, V. Catecholamines reduce transient receptor potential vanilloid type 1 desensitization in cultured dorsal root ganglia neurons. J. Physiol. Pharmacol. 2016, 67, 843–850. [Google Scholar] [PubMed]
- Shu, X.; Mendell, L.M. Nerve growth factor acutely sensitizes the response of adult rat sensory neurons to capsaicin. Neurosci. Lett. 1999, 274, 159–162. [Google Scholar] [CrossRef]
- Kajihara, Y.; Murakami, M.; Imagawa, T.; Otsuguro, K.; Ito, S.; Ohta, T. Histamine potentiates acid-induced responses mediating transient receptor potential V1 in mouse primary sensory neurons. Neuroscience 2010, 166, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Hwang, M.K.; Bode, A.M.; Byun, S.; Song, N.R.; Lee, H.J.; Lee, K.W.; Dong, Z. Cocarcinogenic effect of capsaicin involves activation of EGFR signaling but not TRPV1. Cancer Res. 2010, 70, 6859–6869. [Google Scholar] [CrossRef] [PubMed]
- Căruntu, C.; Negrei, C.; Ghiţă, M.A.; Căruntu, A.; Bădărău, A.I.; Buraga, I.; Brănişteanu, D. Capsaicin, a hot topic in skin pharmacology and physiology. Farmacia 2015, 63, 487–491. [Google Scholar]
- Inoue, K.; Koizumi, S.; Fuziwara, S.; Denda, S.; Inoue, K.; Denda, M. Functional vanilloid receptors in cultured normal human epidermal keratinocytes. Biochem. Biophys. Res. Commun. 2002, 291, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Ständer, S.; Moormann, C.; Schumacher, M.; Buddenkotte, J.; Artuc, M.; Shpacovitch, V.; Metze, D. Expression of vanilloid receptor subtype 1 in cutaneous sensory nerve fibers, mast cells, and epithelial cells of appendage structures. Exp. Dermatol. 2004, 13, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Roosterman, D.; Goerge, T.; Schneider, S.W.; Bunnett, N.W.; Steinhoff, M. VI. Role of Capsaicin and transient receptor potential ion channels in the skin. Physiol. Rev. 2006, 86, 1338–1380. [Google Scholar]
- Bı́ró, T.; Maurer, M.; Modarres, S.; Lewin, N.E.; Brodie, C.; Ács, G.; Blumberg, P.M. Characterization of functional vanilloid receptors expressed by mast cells. Blood 1998, 91, 1332–1340. [Google Scholar] [PubMed]
- Southall, M.D.; Li, T.; Gharibova, L.S.; Pei, Y.; Nicol, G.D.; Travers, J.B. Activation of epidermal vanilloid receptor-1 induces release of proinflammatory mediators in human keratinocytes. J. Pharmacol. Exp. Ther. 2003, 304, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Bodó, E.; Bíró, T.; Telek, A.; Czifra, G.; Griger, Z.; Tóth, B.I.; Paus, R. A hot new twist to hair biology: Involvement of vanilloid receptor-1 (VR1/TRPV1) signaling in human hair growth control. Am. J. Pathol. 2005, 166, 985–998. [Google Scholar]
- Geppetti, P.; Nassini, R.; Materazzi, S.; Benemei, S. The concept of neurogenic inflammation. BJU Int. 2008, 101, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Căruntu, C.; Boda, D. Evaluation through in vivo reflectance confocal microscopy of the cutaneous neurogenic inflammatory reaction induced by capsaicin in human subjects. J. Biomed. Opt. 2012, 17, 0850031–0850037. [Google Scholar] [CrossRef] [PubMed]
- Ghita, M.A.; Caruntu, C.; Lixandru, D.; Pitea, A.; Batani, A.; Boda, D. The Quest for Novel Biomarkers in Early Diagnosis of Diabetic Neuropathy. Curr. Proteom. 2017, 14, 86–99. [Google Scholar] [CrossRef]
- Holzer, P. Local effector functions of capsaicin-sensitive sensory nerve endings: Involvement of tachykinins, calcitonin gene-related peptide and other neuropeptides. Neuroscience 1998, 24, 739–768. [Google Scholar] [CrossRef]
- Schmelz, M.; Petersen, L.J. Neurogenic inflammation in human and rodent skin. News Physiol. Sci. 2001, 16, 33–37. [Google Scholar] [PubMed]
- Brain, S.D.; Tippins, J.R.; Morris, H.R.; Maclntyre, I.; Williams, T.J. Potent vasodilator activity of calcitonin gene-related peptide in human skin. J. Investig. Dermatol. 1986, 87, 533–536. [Google Scholar] [CrossRef] [PubMed]
- Ansel, J.C.; Kaynard, A.H.; Armstrong, C.A.; Olerud, J.; Bunnett, N.; Payan, D. Skin-nervous system interactions. J. Investig. Dermatol. 1996, 106, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Ansel, J.C.; Brown, J.R.; Payan, D.G.; Brown, M.A. Substance P selectively activates TNF-alpha gene expression in murine mast cells. J. Immunol. 1993, 150, 4478–4485. [Google Scholar] [PubMed]
- Căruntu, C.; Boda, D.; Musat, S.; Căruntu, A.; Mandache, E. Stress-induced mast cell activation in glabrous and hairy skin. Mediat. Inflamm. 2014, 2014, 105950. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Bley, K. Topical capsaicin for pain management: Therapeutic potential and mechanisms of action of the new high-concentration capsaicin 8% patch. Br. J. Anaesth. 2011, 107, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Frias, B.; Merighi, A. Capsaicin, nociception and pain. Molecules 2016, 21, 797. [Google Scholar] [CrossRef] [PubMed]
- Smutzer, G.; Devassy, R.K. Integrating TRPV1 receptor function with capsaicin psychophysics. Adv. Pharmacol. Sci. 2016, 2016, 1512457. [Google Scholar] [CrossRef] [PubMed]
- Peppin, J.F.; Pappagallo, M. Capsaicinoids in the treatment of neuropathic pain: A review. Ther. Adv. Neurol. Disord. 2014, 7, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Fattori, V.; Hohmann, M.S.; Rossaneis, A.C.; Pinho-Ribeiro, F.A.; Verri, W.A. Capsaicin: Current understanding of its mechanisms and therapy of pain and other pre-clinical and clinical uses. Molecules 2016, 21, 844. [Google Scholar] [CrossRef] [PubMed]
- Holzer, P. Capsaicin: Cellular targets, mechanisms of action, and selectivity for thin sensory neurons. Pharmacol. Rev. 1991, 43, 143–201. [Google Scholar] [PubMed]
- Simone, D.A.; Nolano, M.; Wendelschafer-Crabb, G.; Kennedy, W.R. Intradermal injection of capsaicin in humans: Diminished pain sensation associated with rapid degeneration of intracutaneous nerve fibers. Soc. Neurosci. Abstr. 1996, 22, 1802. [Google Scholar]
- Simone, D.A.; Nolano, M.; Johnson, T.; Wendelschafer-Crabb, G.; Kennedy, W.R. Intradermal injection of capsaicin in humans produces degeneration and subsequent reinnervation of epidermal nerve fibers: Correlation with sensory function. J. Neurosci. 1998, 18, 8947–8959. [Google Scholar] [PubMed]
- Neagu, M.; Caruntu, C.; Constantin, C.; Boda, D.; Zurac, S.; Spandidos, D.A.; Tsatsakis, A.M. Chemically induced skin carcinogenesis: Updates in experimental models. Oncol. Rep. 2016, 35, 2516–2528. [Google Scholar] [CrossRef] [PubMed]
- Neagu, M.; Constantin, C.; Dumitrascu, G.R.; Lupu, A.R.; Caruntu, C.; Boda, D.; Zurac, S. Inflammation markers in cutaneous melanoma-edgy biomarkers for prognosis. Discoveries 2015, 3, e38. [Google Scholar] [CrossRef]
- Mori, A.; Lehmann, S.; O’Kelly, J.; Kumagai, T.; Desmond, J.C.; Pervan, M.; Koeffler, H.P. Capsaicin, a component of red peppers, inhibits the growth of androgen-independent, p53 mutant prostate cancer cells. Cancer Res. 2006, 66, 3222–3229. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.H.; Lai, F.J.; Chen, H.; Luo, J.; Zhang, R.Y.; Bu, H.Q.; Lin, S.Z. Involvement of the phosphoinositide 3-kinase/Akt pathway in apoptosis induced by capsaicin in the human pancreatic cancer cell line PANC-1. Oncol. Lett. 2013, 5, 43–48. [Google Scholar] [PubMed]
- Lee, Y.S.; Kang, Y.S.; Lee, J.S.; Nicolova, S.; Kim, J.A. Involvement of NADPH oxidase-mediated generation of reactive oxygen species in the apototic cell death by capsaicin in HepG2 human hepatoma cells. Free Radic. Res. 2004, 38, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.F.; Chen, Y.L.; Yang, J.S.; Yang, Y.Y.; Liu, J.Y.; Hsu, S.C.; Chung, J.G. Antitumor activity of capsaicin on human colon cancer cells in vitro and colo 205 tumor xenografts in vivo. J. Agric. Food Chem. 2010, 58, 12999–13005. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.C.; Chen, S.T.; Chien, S.Y.; Kuo, S.J.; Tsai, H.T.; Chen, D.R. Capsaicin may induce breast cancer cell death through apoptosis-inducing factor involving mitochondrial dysfunction. Hum. Exp. Toxicol. 2011, 30, 1657–1665. [Google Scholar] [CrossRef] [PubMed]
- Huh, H.C.; Lee, S.Y.; Lee, S.K.; Park, N.H.; Han, I.S. Capsaicin induces apoptosis of cisplatin-resistant stomach cancer cells by causing degradation of cisplatin-inducible Aurora-A protein. Nutr. Cancer 2011, 63, 1095–1103. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.M.; Chuang, S.M.; Su, Y.C.; Li, Y.H.; Chueh, P.J. Down-regulation of tumor-associated NADH oxidase, tNOX (ENOX2), enhances capsaicin-induced inhibition of gastric cancer cell growth. Cell Biochem. Biophys. 2011, 61, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Moon, D.O.; Kang, C.H.; Kang, S.H.; Choi, Y.H.; Hyun, J.W.; Chang, W.Y.; Kim, G.Y. Capsaicin sensitizes TRAIL-induced apoptosis through Sp1-mediated DR5 up-regulation: Involvement of Ca2+influx. Toxicol. Appl. Pharmacol. 2012, 259, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Jun, H.S.; Park, T.; Lee, C.K.; Kang, M.K.; Park, M.S.; Kang, H.I.; Kim, O.H. Capsaicin induced apoptosis of B16-F10 melanoma cells through down-regulation of Bcl-2. Food Chem. Toxicol. 2007, 45, 708–715. [Google Scholar] [CrossRef] [PubMed]
- Le, T.D.; Jin, D.C.; Rho, S.R.; Kim, M.S.; Yu, R.; Yoo, H. Capsaicin-induced apoptosis of FaDu human pharyngeal squamous carcinoma cells. Yonsei Med. J. 2012, 53, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, C.B.; Kirma, N.B.; Jorge, J.; Chen, R.; Henry, M.A.; Luo, S.; Hargreaves, K.M. Vanilloids induce oral cancer apoptosis independent of TRPV1. Oral Oncol. 2014, 50, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Han, S.S.; Keum, Y.S.; Chun, K.S.; Surh, Y.J. Suppression of phorbol ester-induced NF-κB activation by capsaicin in cultured human promyelocytic leukemia cells. Arch. Pharm. Res. 2002, 25, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Surh, Y.J.; Lee, R.C.J.; Park, K.K.; Mayne, S.T.; Liem, A.; Miller, J.A. Chemoprotective effects of capsaicin and diallyl sulfide against mutagenesis or tumorigenesis by vinyl carbamate and N-nitrosodiinethylamine. Carcinogenesis 1995, 16, 2467–2471. [Google Scholar] [CrossRef] [PubMed]
- Morré, D.J.; Sun, E.; Geilen, C.; Wu, L.Y.; De Cabo, R.; Krasagakis, K.; Morre, D.M. Capsaicin inhibits plasma membrane NADH oxidase and growth of human and mouse melanoma lines. Eur. J. Cancer 1996, 32, 1995–2003. [Google Scholar] [CrossRef]
- Brar, S.S.; Kennedy, T.P.; Whorton, A.R.; Sturrock, A.B.; Huecksteadt, T.P.; Ghio, A.J.; Hoidal, J.R. Reactive oxygen species from NAD(P)H: Quinone oxidoreductase constitutively activate NF-κB in malignant melanoma cells. Am. J. Physiol Cell. Physiol. 2001, 280, C659–C676. [Google Scholar] [PubMed]
- Patel, P.S.; Varney, M.L.; Dave, B.J.; Singh, R.K. Regulation of Constitutive and Induced NF-κB Activation in Malignant Melanoma Cells by Capsaicin Modulates Interleukin-8 Production and Cell Proliferation. J. Interferon Cytokine Res. 2002, 22, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.Y. Nitric oxide triggers apoptosis in A375 human melanoma cells treated with capsaicin and resveratrol. Mol. Med. Rep. 2012, 5, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.H.; Kim, O.H.; Jun, H.S.; Kang, M.K. Inhibitory effect of capsaicin on B16-F10 melanoma cell migration via the phosphatidylinositol 3-kinase/Akt/Rac1 signal pathway. Exp. Mol. Med. 2008, 40, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.F.; Wang, M.W.; Ikejima, T. Mechanisms of capsaicin-induced apoptosis of human melanoma A375-S2 cells. Chin. J. Oncol. 2005, 27, 401–403. [Google Scholar]
- Marques, C.M.; Dibden, C.; Danson, S.; Haycock, J.W.; MacNeil, S. Combined effects of capsaicin and HA14-1 in inducing apoptosis in melanoma cells. J. Cosmet. Dermatol. Sci. Appl. 2013, 3, 175–189. [Google Scholar] [CrossRef][Green Version]
- Schwartz, L.; Guais, A.; Israël, M.; Junod, B.; Steyaert, J.M.; Crespi, E.; Abolhassani, M. Tumor regression with a combination of drugs interfering with the tumor metabolism: Efficacy of hydroxycitrate, lipoic acid and capsaicin. Investig. New Drugs 2013, 31, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Bode, A.M.; Cho, Y.Y.; Zheng, D.; Zhu, F.; Ericson, M.E.; Ma, W.Y.; Dong, Z. Transient receptor potential type vanilloid 1 suppresses skin carcinogenesis. Cancer Res. 2009, 69, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Bode, A.M.; Zhu, F.; Liu, K.; Zhang, J.; Kim, M.O.; Langfald, A.K. TRPV1-antagonist AMG9810 promotes mouse skin tumorigenesis through EGFR/Akt signaling. Carcinogenesis 2011, 32, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhu, P.; Tao, Y.; Shen, C.; Wang, S.; Zhao, L.; Zhu, Z. Cancer-promoting effect of capsaicin on DMBA/TPA-induced skin tumorigenesis by modulating inflammation, Erk and p38 in mice. Food Chem. Toxicol. 2015, 81, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.; Ratner, D. Cutaneous squamous-cell carcinoma. N. Engl. J. Med. 2001, 344, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, R.; Zelac, D. Squamous cell carcinoma of the skin. Plast. Reconstr. Surg. 2004, 114, 82e–94e. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, A.S.; Ogle, C.A.; Shim, E.K. Metastatic cutaneous squamous cell carcinoma: An update. Dermatol. Surg. 2007, 33, 885–899. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Shin, H.R.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D.M. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int. J. Cancer 2008, 127, 2893–2917. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.P.; Gil, Z. Current concepts in management of oral cancer–surgery. Oral Oncol. 2009, 45, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Feller, L.; Lemmer, J. Oral squamous cell carcinoma: Epidemiology, clinical presentation and treatment. J. Cancer Ther. 2012, 3, 263–268. [Google Scholar] [CrossRef]
- Matei, C.; Caruntu, C.; Ion, R.M.; Georgescu, S.R.; Dumitrascu, G.R.; Constantin, C.; Neagu, M. Protein microarray for complex apoptosis monitoring of dysplastic oral keratinocytes in experimental photodynamic therapy. Biol. Res. 2014, 47, 33. [Google Scholar] [CrossRef] [PubMed]
- Monta, G.; Caracò, C.; Simeone, E.; Grimaldi, A.M.; Marone, U.; Marzo, M.; Mozzillo, N. Electrochemotherapy efficacy evaluation for treatment of locally advanced stage III cutaneous squamous cell carcinoma: A 22-cases retrospective analysis. J. Transl. Med. 2017, 15, 82. [Google Scholar] [CrossRef] [PubMed]
- Tampa, M.; Matei, C.; Popescu, S.; Georgescu, S.R.; Neagu, M.; Constantin, C.; Ion, R.M. Zinc trisulphonatedphthalocyanine used in photodynamic therapy of dysplastic oral keratinocytes. Rev. Chim. 2013, 64, 639–645. [Google Scholar]
- Matei, C.; Tampa, M.; Ion, R.M.; Neagu, M.; Constantin, C. Photodynamic properties of aluminiumsulphonatedphthalocyanines in human displazic oral keratinocytes experimental model. Dig. J. Nanomater. Biostruct. (DJNB) 2012, 7, 1535–1547. [Google Scholar]
- Yu, C.C.; Hung, S.K.; Lin, H.Y.; Chiou, W.Y.; Lee, M.S.; Liao, H.F.; Su, Y.C. Targeting the PI3K/AKT/mTOR signaling pathway as an effectively radiosensitizing strategy for treating human oral squamous cell carcinoma in vitro and in vivo. Oncotarget 2017, 8, 68641–68653. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, L.A.; Katz, S.I.; Gilchrest, B.A.; Paller, A.S.; Leffell, D.J.; Wolff, K. Fitzpatrick’s Dermatology in General Medicine, 8th ed.; McGrawHill: New York, NY, USA, 2012; ISBN 978-00-7-166904-7. [Google Scholar]
- Bolognia, J.L.; Joseph, L.J.; Schaffer, J.V. Dermatology, 3th ed.; Elsevier: Amsterdam, the Netherlands, 2012; ISBN 978-07-2-343571-6. [Google Scholar]
- Burns, T.; Breathnach, S.; Cox, N.; Griffiths, C. Rook’s Textbook of Dermatology, 8th ed.; Wiley Blackwell: Hoboken, NJ, USA, 2010. [Google Scholar]
- Apalla, Z.; Nashan, D.; Weller, R.B.; Castellsagué, X. Skin cancer: Epidemiology, disease burden, pathophysiology, diagnosis, and therapeutic approaches. Dermatol. Ther. (Heidelb) 2017, 7, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Petti, S. Lifestyle risk factors for oral cancer. Oral Oncol. 2009, 45, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Boda, D.; Neagu, M.; Constantin, C.; Voinescu, R.N.; Caruntu, C.; Zurac, S.; Tsatsakis, A.M. HPV strain distribution in patients with genital warts in a female population sample. Oncol. Lett. 2016, 12, 1779–1782. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Aldabagh, B.; Yu, J.; Arron, S.T. Role of human papillomavirus in cutaneous squamous cell carcinoma: A meta-analysis. J. Am. Acad. Dermatol. 2014, 70, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Lupu, M.; Caruntu, A.; Caruntu, C.; Papagheorghe, L.M.L.; Ilie, M.A.; Voiculescu, V.; Boda, D.; Constantin, C.; Tanase, C.; Sifaki, M.; et al. Neuroendocrine factors: The missing link in non-melanoma skin cancer. Oncol. Rep. 2017, 38, 1327–1340. [Google Scholar] [CrossRef] [PubMed]
- Scholzen, T.; Armstrong, C.A.; Bunnett, N.W.; Luger, T.A.; Olerud, J.E.; Ansel, J.C. Neuropeptides in the skin: Interactions between the neuroendocrine and the skin immune systems. Exp. Dermatol. 1998, 7, 81–96. [Google Scholar] [CrossRef] [PubMed]
- Hara, M.; Toyoda, M.; Yaar, M.; Bhawan, J.; Avila, E.M.; Penner, I.R.; Gilchrest, B.A. Innervation of melanocytes in human skin. J. Exp. Med. 1996, 184, 1385–1395. [Google Scholar] [CrossRef] [PubMed]
- Seiffert, K.; Granstein, R.D. Neuropeptides and neuroendocrine hormones in ultraviolet radiation-induced immunosuppression. Methods 2002, 28, 97–103. [Google Scholar] [CrossRef]
- Weidner, C.; Klede, M.; Rukwied, R.; Lischetzki, G.; Neisius, U.; Schmelz, M.; Petersen, L.J. Acute effects of substance P and calcitonin gene-related peptide in human skin–a microdialysis study. J. Investig. Dermatol. 2000, 115, 1015–1020. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Feng, F.; Xu, G.; Zhang, H.; Hong, L.; Yang, J. Elevated SP/NK-1R in esophageal carcinoma promotes esophageal carcinoma cell proliferation and migration. Gene 2015, 560, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.; Rosso, M.; González-Ortega, A.; Coveñas, R. The NK-1 receptor antagonist L-732,138 induces apoptosis and counteracts substance P-related mitogenesis in human melanoma cell lines. Cancers 2010, 2, 611–623. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.; Rosso, M.; Robles-Frias, M.J.; Salinas-Martín, M.V.; Rosso, R.; González-Ortega, A.; Coveñas, R. The NK-1 receptor is expressed in human melanoma and is involved in the antitumor action of the NK-1 receptor antagonist aprepitant on melanoma cell lines. Lab. Investig. 2010, 90, 1259–1269. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.; Rosso, M.; Robles-Frías, M.J.; Coveñas, R.; Salinas-Martín, M.V. Immunolocalization of the Neurokinin-1 Receptor: A new target in the treatment of the human primary retinoblastoma. In Eye Cancer Res. Progress; Nova Science Publishers: Hauppauge, NY, USA, 2008; pp. 157–178. ISBN 978-1-60456-045-9. [Google Scholar]
- Muñoz, M.; Pérez, A.; Coveñas, R.; Rosso, M.; Castro, E. Antitumoural action of L-733,060 on neuroblastoma and glioma cell lines. Arch. Italiennes Biol. 2004, 142, 105–112. [Google Scholar]
- Brener, S.; González-Moles, M.A.; Tostes, D.; Esteban, F.; Gil-Montoya, J.A.; Ruiz-Avila, I.; Munoz, M. A role for the substance P/NK-1 receptor complex in cell proliferation in oral squamous cell carcinoma. Anticancer Res. 2009, 29, 2323–2329. [Google Scholar] [PubMed]
- Serra, I.; Yamamoto, M.; Calvo, A.; Cavada, G.; Baez, S.; Endoh, K.; Tajima, K. Association of chili pepper consumption, low socioeconomic status and longstanding gallstones with gallbladder cancer in a Chilean population. Int. J. Cancer 2002, 102, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Toth, B.; Gannett, P. Carcinogenicity of lifelong administration of capsaicin of hot pepper in mice. In Vivo 1992, 6, 59–63. [Google Scholar] [PubMed]
- Chanda, S.; Erexson, G.; Frost, D.; Babbar, S.; Burlew, J.A.; Bley, K. 26-Week dermal oncogenicity study evaluating pure trans-capsaicin in Tg. AC hemizygous mice (FBV/N). Int. J. Toxicol. 2007, 26, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Surh, Y.J.; Lee, S.S. Capsaicin, a double-edged sword: Toxicity, metabolism, and chemopreventive potential. Life Sci. 1995, 56, 1845–1855. [Google Scholar] [CrossRef]
- Park, K.K.; Surh, Y.J. Effects of capsaicin on chemically-induced two-stage mouse skin carcinogenesis. Cancer Lett. 1997, 114, 183–184. [Google Scholar] [CrossRef]
- Park, K.K.; Chun, K.S.; Yook, J.I.; Surh, Y.J. Lack of tumor promoting activity of capsaicin, a principal pungent ingredient of red pepper, in mouse skin carcinogenesis. Anticancer Res. 1998, 18, 4201–4205. [Google Scholar] [PubMed]
- Surh, Y.J.; Han, S.S.; Keum, Y.S.; Seo, H.J.; Lee, S.S. Inhibitory effects of curcumin and capsaicin on phorbol ester-induced activation of eukaryotic transcription factors, NF-kappaBand AP-1. Biofactors 2000, 12, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Braun-Falco, O.; Plewig, G.; Wolff, H.H.; Landthaler, M. Braun-Falco’s Dermatology, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 978-3-540-29312-5. [Google Scholar]
- Sterry, W.; Paus, R.; Burgdorf, W.; Holtermann, H. Thieme Clinical Companions Dermatology; Thieme: Stuttgart, Germany, 2006. [Google Scholar]
- Tampa, M.; Matei, C.; Caruntu, C.; Poteca, T.; Mihaila, D.; Paunescu, C.; Neagu, M. Cellular impedance measurement–novel method for in vitro investigation of drug efficacy. Farmacia 2016, 5, 430–434. [Google Scholar]
- Niezgoda, A.; Niezgoda, P.; Czajkowski, R. Novel approaches to treatment of advanced melanoma: A review on targeted therapy and immunotherapy. BioMed Res. Int. 2015, 2015, 851387. [Google Scholar] [CrossRef] [PubMed]
- Caruntu, C.; Mirica, A.; Roca, A.E.; Mirica, R.; Caruntu, A.; Matei, C.; Moraru, L. The Role of Estrogens and Estrogen Receptors in Melanoma Development and Progression. ActaEndocrinol. 2016, 12, 234–241. [Google Scholar] [CrossRef]
- Caruntu, C.; Boda, D.; Constantin, C.; Caruntu, A.; Neagu, M. Catecholamines Increase in vitro Proliferation of Murine B16F10 Melanoma Cells. ActaEndocrinol. 2014, 10, 545–558. [Google Scholar] [CrossRef]
- Borrego, J.F.; Huelsmeyer, M.K.; Pinkerton, M.E.; Muszynski, J.L.; Miller, S.A.K.; Kurzman, I.D.; Vail, D.M. Neurokinin-1 receptor expression and antagonism by the NK-1R antagonist maropitant in canine melanoma cell lines and primary tumour tissues. Vet. Comp. Oncol. 2016, 14, 210–224. [Google Scholar] [CrossRef] [PubMed]
- Khare, V.K.; Albino, A.P.; Reed, J.A. The neuropeptide/mast cell secretagogue substance P is expressed in cutaneous melanocytic lesions. J. Cutan. Pathol. 1998, 25, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Geng, K.K.; Ping, F.F.; Gao, Y.Y.; Liu, L.; Feng, B.N. Cross-talk between 5-hydroxytryptamine and substance P in the melanogensis and apoptosis of B16F10 melanoma cells. Eur. J. Pharmacol. 2016, 775, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Ping, F.; Shang, J.; Zhou, J.; Song, J.; Zhang, L. Activation of neurokinin-1 receptor by substance P inhibits melanogenesis in B16-F10 melanoma cells. Int. J. Biochem. Cell Biol. 2012, 44, 2342–2348. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.; Bernabeu-Wittel, J.; Coveñas, R. NK-1 as a melanoma target. Expert Opin. Ther. Targets 2011, 15, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.; Pérez, A.; Rosso, M.; Zamarriego, C.; Rosso, R. Antitumoral action of the neurokinin-1 receptor antagonist L-733 060 on human melanoma cell lines. Melanoma Res. 2004, 14, 183–188. [Google Scholar] [PubMed]
- Ueda, Y.; Richmond, A. NF-κB activation in melanoma. Pigment. Cell. Melanoma Res. 2006, 19, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Neagu, M.; Constantin, C.; Longo, C. Chemokines in the melanoma metastasis biomarkers portrait. J. Immunoass. Immunochem. 2015, 36, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Oyagbemi, A.A.; Saba, A.B.; Azeez, O.I. Capsaicin: A novel chemopreventive molecule and its underlying molecular mechanisms of action. Indian J. Cancer 2010, 47, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.A. The role of the PI3K-AKT pathway in melanoma. Cancer J. 2012, 18, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Kwong, L.N.; Davies, M.A. Navigating the therapeutic complexity of PI3K pathway inhibition in melanoma. Clin. Cancer Res. 2013, 19, 5310–5319. [Google Scholar] [CrossRef] [PubMed]
- Price, L.S.; Collard, J.G. Regulation of the cytoskeleton by Rho-family GTPases: Implications for tumour cell invasion. Semin. Cancer Biol. 2001, 11, 167–173. [Google Scholar] [CrossRef] [PubMed]
| Effect of Capsaicin | Primary Pathway through Which the Effect Is Occurring | Model Used to Demonstrate the Effect | References |
|---|---|---|---|
| Anticarcinogenic | Mitochondrial pathway-dependent apoptosis: ↓Bcl-2 ↑Bax, ↑Bad | human pharyngeal SCC cells (FaDu) | Le et al. [61] |
| Induction of reactive oxygen species; apoptosis independent oftransient receptor potential vanilloid 1 receptor (TRPV1) | oral squamous cell carcinoma (OSCC) cell lines | Gonzales et al. [62] | |
| Nuclear factor kappaB (NF-kB), activator protein 1 (AP-1) | ICR mouse model; humanpromyelocytic leukemiaHL-60 cells | Han et al. [63] | |
| Inhibition of the cytochrome P-450 IIE1 isoform | ICR mouse model | Surh et al. [64] | |
| ↓nicotinamide adenine dinucleotide (NADH) oxidase activity; ↑apoptosis | A375, SK-MEL-28 human melanoma cell lines; B16 murine melanoma cell line | Morré et al. [65] | |
| ↓nicotinamide adenine dinucleotide phosphate-reduced(NAD(P)H): quinone oxidoreductase ; ↓NF-κB | CRL 1585 and CRL 1619 human melanoma cell lines | Brar et al. [66] | |
| ↓activation of constitutive and IL-1beta-induced NF-κB | Human melanoma cells | Patel et al. [67] | |
| ↑p53, induces apoptosis via Bcl-2, Bax, caspases 3,8,9 | A375 human melanoma cell line | Kim [68] | |
| Down-regulation of PI3-K/Akt pathway | B16-F10 mouse melanoma cells | Shin et al. [69] | |
| Downregulation of Bcl-2; induction of apoptosis | B16-F10 mouse melanoma cells | Jun et al. [60] | |
| ↓caspase-activated DNase inhibitor(ICAD)expression; induction of apoptosis | human melanoma A375-S2 cell line | Gong et al. [70] | |
| Induction of apoptosis | melanocytes and HBL, A375SM, C8161 melanoma cell lines | Marques et al. [71] | |
| Delays tumor growth | melanoma B16-F10; mouse model | Schwartz et al. [72] | |
| Cocarcinogenic | Epidermal growth factor receptor(EGFR) pathway | DMBA/TPA mouse model | Bode et al. [73] |
| EGFR pathway; ↑cyclo-oxygenase-2 (COX-2) | DMBA/TPA mouse model | Hwang et al. [25] | |
| EGFR/Akt/mTOR signaling pathway | DMBA/TPA mouse model | Li et al. [74] | |
| Erk/p38 signaling pathway | DMBA/TPA mouse model | Liu et al. [75] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Georgescu, S.-R.; Sârbu, M.-I.; Matei, C.; Ilie, M.A.; Caruntu, C.; Constantin, C.; Neagu, M.; Tampa, M. Capsaicin: Friend or Foe in Skin Cancer and Other Related Malignancies? Nutrients 2017, 9, 1365. https://doi.org/10.3390/nu9121365
Georgescu S-R, Sârbu M-I, Matei C, Ilie MA, Caruntu C, Constantin C, Neagu M, Tampa M. Capsaicin: Friend or Foe in Skin Cancer and Other Related Malignancies? Nutrients. 2017; 9(12):1365. https://doi.org/10.3390/nu9121365
Chicago/Turabian StyleGeorgescu, Simona-Roxana, Maria-Isabela Sârbu, Clara Matei, Mihaela Adriana Ilie, Constantin Caruntu, Carolina Constantin, Monica Neagu, and Mircea Tampa. 2017. "Capsaicin: Friend or Foe in Skin Cancer and Other Related Malignancies?" Nutrients 9, no. 12: 1365. https://doi.org/10.3390/nu9121365
APA StyleGeorgescu, S.-R., Sârbu, M.-I., Matei, C., Ilie, M. A., Caruntu, C., Constantin, C., Neagu, M., & Tampa, M. (2017). Capsaicin: Friend or Foe in Skin Cancer and Other Related Malignancies? Nutrients, 9(12), 1365. https://doi.org/10.3390/nu9121365










