Anserine/Carnosine Supplementation Suppresses the Expression of the Inflammatory Chemokine CCL24 in Peripheral Blood Mononuclear Cells from Elderly People
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Test Formulae
2.3. Inventory of Food Intake during the 3-Month Test Period
2.4. Cognitive Testing
2.5. Microarray Analysis
2.6. qRT-PCR
3. Results
3.1. Participants
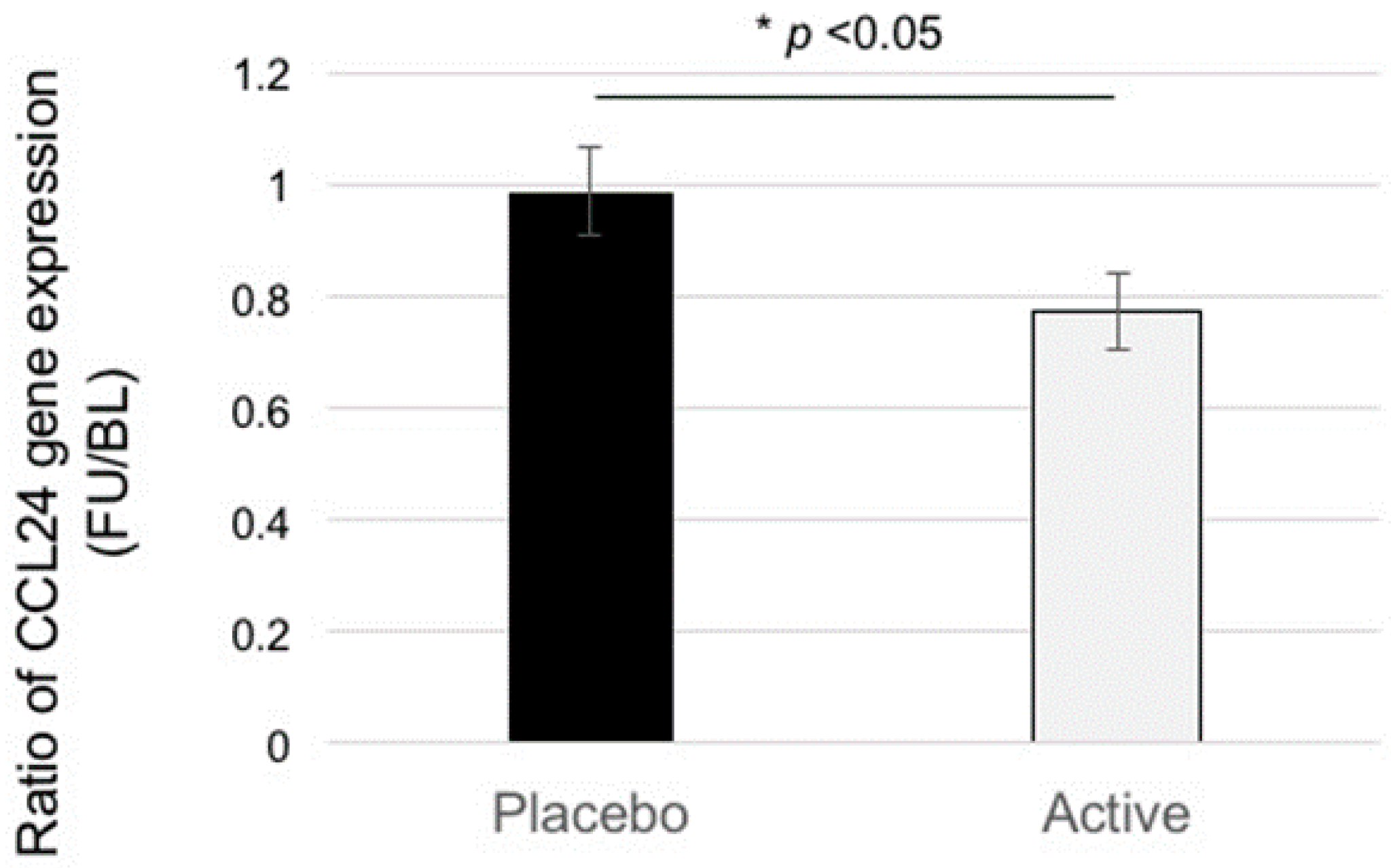
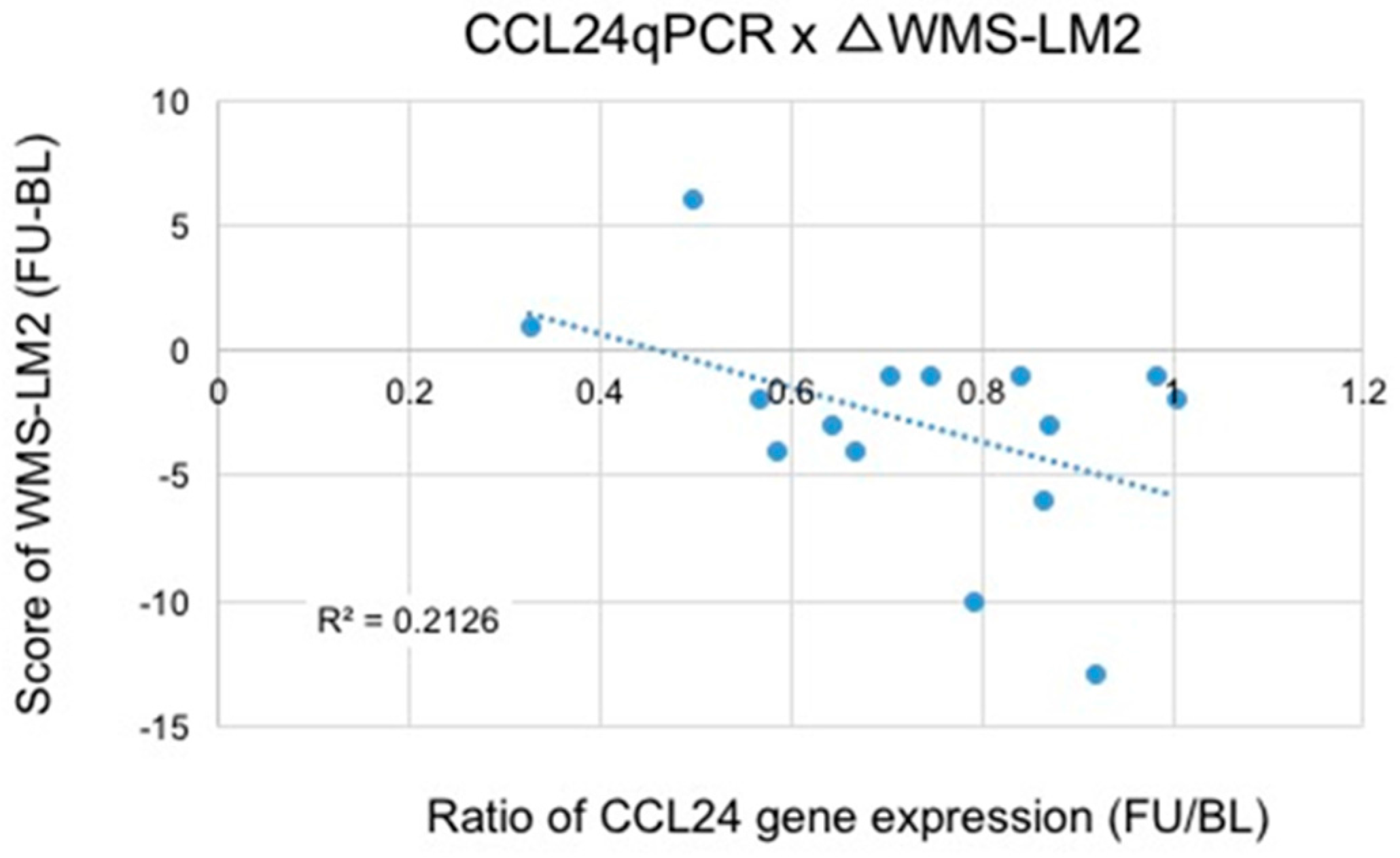
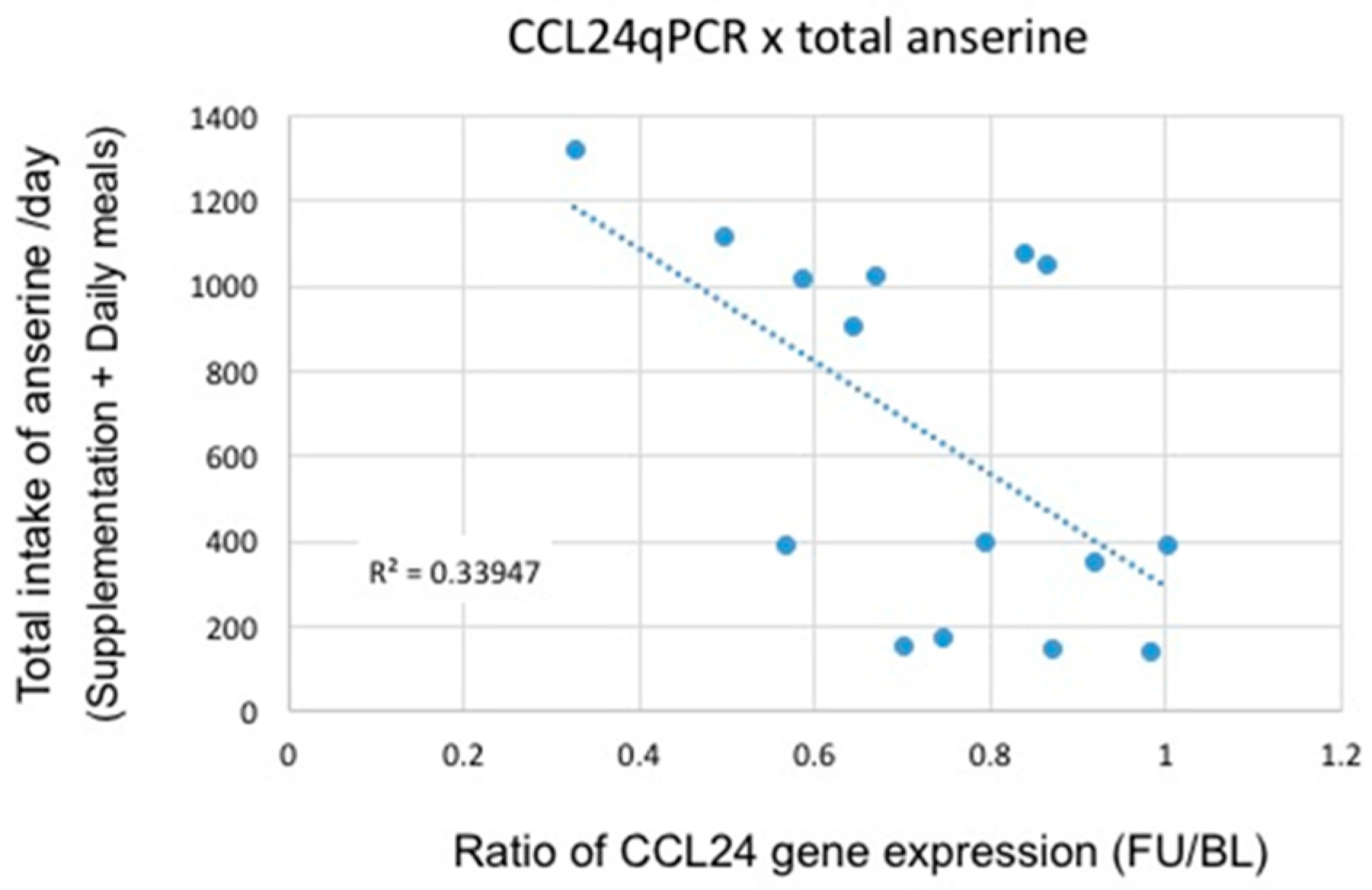
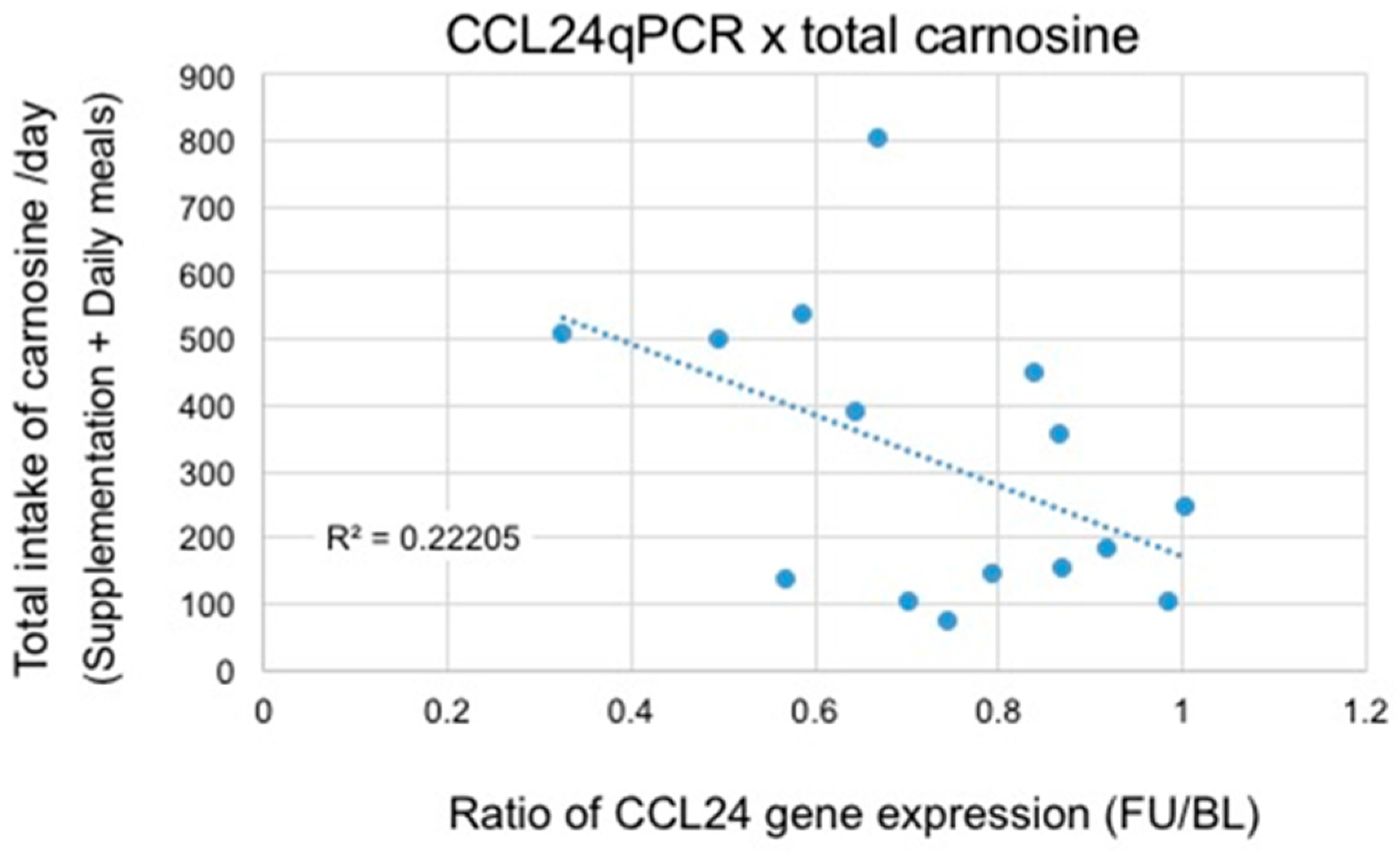
3.2. Microarray and qRT-PCR Analysis
3.3. Cognitive Tests
3.4. Age-Restricted Sub-Analysis (Participants in Their 60s and 70s)
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Baumgart, M.; Snyder, H.M.; Carrillo, M.C.; Fazio, S.; Kim, H.; Johns, H. Summary of the evidence on modifiable risk factors for cognitive decline and dementia: A population-based perspective. Alzheimer’s Dement. 2015, 11, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Lindenberger, U. Human cognitive aging: corriger la fortune? Science 2014, 346, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Gutchess, A. Plasticity of the aging brain: new directions in cognitive neuroscience. Science 2014, 346, 579–582. [Google Scholar] [CrossRef] [PubMed]
- Farías, G.A.; Guzmán-Martínez, L.; Delgado, C.; Maccioni, R.B. Nutraceuticals: A novel concept in prevention and treatment of Alzheimer’s disease and related disorders. J. Alzheimer’s Dis. 2014, 42, 357–367. [Google Scholar]
- Scheltens, P.; Kamphuis, P.J.; Verhey, F.R.; Rikkert, M.G.O.; Wurtman, R.J.; Wilkinson, D.; Twisk, J.W.; Kurz, A. Efficacy of a medical food in mild Alzheimer’s disease: A randomized, controlled trial. Alzheimer’s Dement. 2010, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Boldyrev, A.A.; Aldini, G.; Derave, W. Physiology and pathophysiology of carnosine. Physiol. Rev. 2013, 93, 1803–1845. [Google Scholar] [CrossRef] [PubMed]
- Szcześniak, D.; Budzeń, S.; Kopeć, W.; Rymaszewska, J. Anserine and carnosine supplementation in the elderly: Effects on cognitive functioning and physical capacity. Arch. Gerontol. Geriatr. 2014, 59, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Rokicki, J.; Li, L.; Imabayashi, E.; Kaneko, J.; Hisatsune, T.; Matsuda, H. Daily carnosine and anserine supplementation alters verbal episodic memory and resting state network connectivity in healthy elderly adults. Front. Aging Neurosci. 2015, 7, 219. [Google Scholar] [CrossRef] [PubMed]
- Hisatsune, T.; Kaneko, J.; Kurashige, H.; Cao, Y.; Satsu, H.; Totsuka, M.; Katakura, Y.; Imabayashi, E.; Matsuda, H. Effect of anserine/carnosine supplementation on verbal episodic memory in elderly people. J. Alzheimer’s Dis. 2016, 50, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Kawano, N.; Awata, S.; Ijuin, M.; Iwamoto, K.; Ozaki, N. Necessity of normative data on the Japanese version of the Wechsler Memory Scale-Revised Logical Memory subtest for old-old people. Geriatr. Gerontol. Int. 2013, 13, 726–730. [Google Scholar] [CrossRef] [PubMed]
- Villeda, S.A.; Luo, J.; Mosher, K.I.; Zou, B.; Britschgi, M.; Bieri, G.; Stan, T.M.; Fainberg, N.; Ding, Z.; Eggel, A.; et al. The ageing systemic milieu negatively regulates neurogenesis and cognitive function. Nature 2011, 477, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Huber, A.K.; Giles, D.A.; Segal, B.M.; Irani, D.N. An emerging role for eotaxins in neurodegenerative disease. Clin. Immunol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Aoyagi, S.; Sugino, T.; Kajimoto, Y.; Nishitani, M. Safety of long-term administration of CBEX-Dr-containing drink of healthy people. Jpn. Pharmacol. Ther. 2008, 36, 213–224. [Google Scholar]
- Aoyagi, S.; Sugino, T.; Kajimoto, Y.; Nishitani, M. Safety of excess administration of CBEX-Dr-containing drink of healthy people. Jpn. Pharmacol. Ther. 2008, 36, 225–235. [Google Scholar]
- Kiyohara, Y.; Shinohara, A.; Kato, I.; Shirota, T.; Kubo, M.; Tanizaki, Y.; Fujishima, M.; Iida, M. Dietary factors and development of impaired glucose tolerance and diabetes in a general Japanese population: The Hisayama Study. J. Epidemiol. 2003, 13, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Homma, A.; Fukuzawa, K.; Tsukada, Y.; Ishii, T.; Hasegawa, K.; Mohs, R.C. Development of a Japanese version of Alzheimer’s disease Assessment Scale (ADAS). Jpn. J. Geriatr. Psychiatry 1992, 3, 647–655. [Google Scholar]
- Mohs, R.C.; Rosen, W.G.; Davis, K.L. The Alzheimer’s disease assessment scale: An instrument for assessing treatment efficacy. Psychopharmacol. Bull. 1983, 19, 448–450. [Google Scholar] [PubMed]
- Beck, A.T.; Steer, R.A.; Brown, G.K. Manual for the Beck Depression Inventory, 2nd ed.; Pearson: San Antonio, TX, USA, 1996. [Google Scholar]
- Kojima, M.; Furukawa, T.A. Japanese Manual of the Beck Depression Inventory, 2nd ed.; Nihon Bunka Kagakusha: Tokyo, Japan, 2003. [Google Scholar]
- Ware, J.E.; Sherbourne, C.D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 1992, 30, 474–483. [Google Scholar] [CrossRef]
- Lu, Y.; Nyunt, M.S.; Gwee, X.; Feng, L.; Feng, L.; Kua, E.H.; Kumar, R.; Ng, T.P. Life event stress and chronic obstructive pulmonary disease (COPD): Associations with mental well-being and quality of life in a population-based study. BMJ Open 2012, 2, e001674. [Google Scholar] [CrossRef] [PubMed]
- Uchida, K.; Shan, L.; Suzuki, H.; Tabuse, Y.; Nishimura, Y.; Hirokawa, Y.; Mizukami, K.; Akatsu, H.; Meno, K.; Asada, T. Amyloid-β sequester proteins as blood-based biomarkers of cognitive decline. Alzheimer’s Dement. 2015, 1, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Da Conceicao, V.N.; Dyer, W.B.; Gandhi, K.; Gupta, P.; Saksena, N.K. Genome-wide analysis of primary peripheral blood mononuclear cells from HIV+ patients-pre-and post- HAART show immune activation and inflammation the main drivers of host gene expression. Mol. Cell. Ther. 2014, 2, 11. [Google Scholar] [CrossRef] [PubMed]
- Bolstad, B.M.; Irizarry, R.A.; Astrand, M.; Speed, T.P. A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics 2003, 19, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Gentleman, R.C.; Carey, V.J.; Bates, D.M.; Bolstad, B.; Dettling, M.; Dudoit, S.; Ellis, B.; Gautier, L.; Ge, Y.; Gentry, J.; et al. Bioconductor: open software development for computational biology and bioinformatics. Genome Biol. 2004, 5, R80. [Google Scholar] [CrossRef] [PubMed]
- Saeed, A.I.; Sharov, V.; White, J.; Li, J.; Liang, W.; Bhagabati, N.; Braisted, J.; Klapa, M.; Currier, T.; Thiagarajan, M.; et al. TM4: A free, open-source system for microarray data management and analysis. Biotechniques 2003, 34, 374–378. [Google Scholar] [PubMed]
- Goto, M.; Abe, O.; Miyati, T.; Yoshikawa, T.; Hayashi, N.; Takao, H.; Inano, S.; Kabasawa, H.; Mori, H.; Kunimatsu, A.; et al. Entorhinal cortex volume measured with 3T MRI is positively correlated with the Wechsler Memory Scale-Revised logical/verbal memory score for healthy subjects. Neuroradiology 2011, 53, 617–622. [Google Scholar] [CrossRef] [PubMed]
- Lonie, J.A.; Tierney, K.M.; Ebmeier, K.P. Screening for mild cognitive impairment: A systematic review. Int. J. Geriatr. Psychiatry 2009, 24, 902–915. [Google Scholar] [CrossRef] [PubMed]
- Grober, E.; Sanders, A.E.; Hall, C.; Lipton, R.B. Free and cued selective reminding identifies very mild dementia in primary care. Alzheimer Dis. Assoc. Disord. 2010, 24, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Derby, C.A.; Burns, L.C.; Wang, C.; Katz, M.J.; Zimmerman, M.E.; L’italien, G.; Guo, Z.; Berman, R.M.; Lipton, R.B. Screening for predementia AD: Time-dependent operating characteristics of episodic memory tests. Neurology 2013, 80, 1307–1314. [Google Scholar] [CrossRef] [PubMed]
- Son, D.O.; Satsu, H.; Kiso, Y.; Totsuka, M.; Shimizu, M. Inhibitory effect of carnosine on interleukin-8 production in intestinal epithelial cells through translational regulation. Cytokine 2008, 42, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Crush, K.G. Carnosine and related substances in animal tissues. Comp. Biochem. Physiol. 1970, 34, 3–30. [Google Scholar] [CrossRef]
- Suzuki, T.; Hirano, T.; Suyama, M. Free imidazole compounds in white and dark muscles of migratory marine fish. Comp. Biochem. Physiol. B 1987, 87, 615–619. [Google Scholar] [CrossRef]
- Herculano, B.; Tamura, M.; Ohba, A.; Shimatani, M.; Kutsuna, N.; Hisatsune, T. β-alanyl-l-histidine rescues cognitive deficits caused by feeding a high fat diet in a transgenic mouse model of Alzheimer’s disease. J. Alzheimer’s Dis. 2013, 33, 983–997. [Google Scholar]
- Kaneko, J.; Enya, A.; Enomoto, K.; Ding, Q.; Hisatsune, T. Anserine (beta-alanyl-3-methyl-l-histidine) improves neurovascular-unit dysfunction and spatial memory in aged AβPPswe/PSEN1dE9 Alzheimer’s-model mice. Sci. Rep. 2017. [Google Scholar] [CrossRef] [PubMed]
- Bell, R.D.; Winkler, E.A.; Singh, I.; Sagare, A.P.; Deane, R.; Wu, Z.; Holtzman, D.M.; Betsholtz, C.; Armulik, A.; Sallstrom, J.; et al. Apolipoprotein E controls cerebrovascular integrity via cyclophilin A. Nature 2012, 485, 512–516. [Google Scholar] [CrossRef] [PubMed]
- Montagne, A.; Barnes, S.R.; Sweeney, M.D.; Halliday, M.R.; Sagare, A.P.; Zhao, Z.; Toga, A.W.; Jacobs, R.E.; Liu, C.Y.; Amezcua, L.; et al. Blood-brain barrier breakdown in the aging human hippocampus. Neuron 2015, 85, 296–302. [Google Scholar] [CrossRef] [PubMed]
| Demographics | Active Group | Placebo Group | p Value |
|---|---|---|---|
| Age | 60.4 (2.1) | 65.3 (1.6) | 0.07 |
| Sex (M/F) | 10/20 | 10/20 | - |
| BMI | 21.0 (0.51) | 21.1(0.86) | 0.97 |
| Years of education | 14.9 (0.4) | 14.9 (0.4) | 0.995 |
| MMSE | 28.7 (0.3) | 29.1 (0.2) | 0.29 |
| APOE4+/APOE4− | 4/26 | 4/26 | - |
| Imidazoledipeptide | Active Group | Placebo Group | p Value |
|---|---|---|---|
| Anserine | 384 ± 45 1 | 358 ± 48 1 | 0.699 |
| Carnosine | 259 ± 25 1 | 199 ± 19 1 | 0.058 |
| Test | Baseline | Follow-Up | Change | ||||
|---|---|---|---|---|---|---|---|
| Active | Placebo | Active | Placebo | Active | Placebo | p Value 2 | |
| WMS-1 3 | 14.8 (0.8) | 14.4 (0.7) | 12.2 (0.7) | 12.1 (0.8) | −2.6 (0.7) | −2.4 (0.5) | 0.39 |
| WMS-2 3 | 13.6 (0.8) | 13.9 (0.6) | 12.0 (0.7) | 11.0 (0.8) | 1.4 (3.0) | 0.7 (3.0) | 0.078 ## |
| ADAS | 8.3 (0.7) | 7.2 (0.8) | 7.3 (0.8) | 7.2 (0.9) | −0.9 (0.6) | −0.04 (0.8) | 0.18 # |
| SF-36 (PCS) | 47.3 (1.5) | 47.6 (1.2) | 48.5 (1.4) | 48.3 (1.5) | 1.2 (1.3) | 0.7 (1.4) | 0.40 |
| SF-36 (MCS) | 50.4 (1.8) | 49.5 (1.4) | 52.2 (1.4) | 51.5 (1.2) | 1.7 (1.2) | 2.0 (1.1) | 0.44 |
| BDI | 9.8 (1.2) | 9.5 (1.2) | 6.9 (0.9) | 7.7 (0.9) | −2.9 (1.0) | −1.8 (0.9) | 0.19 # |
| 60s Group | Baseline | Follow-Up | Change | ||||
|---|---|---|---|---|---|---|---|
| Active | Placebo | Active | Placebo | Active | Placebo | p Value 2 | |
| WMS-1 3 | 13.5 (0.8) | 13.4 (0.8) | 11.3 (0.6) | 10.2 (0.7) | −2.3 (0.9) | −3.2 (0.6) | 0.31 |
| WMS-2 3 | 12.1 (0.8) | 12.7 (0.6) | 11.2 (0.6) | 9.2 (0.6) | −0.9 (0.7) | −3.4 (0.5) | 0.048 * |
| ADAS | 9.5 (0.8) | 7.6 (0.7) | 8.1 (0.7) | 6.4 (0.7) | −1.4 (0.6) | −1.2 (0.9) | 0.47 |
| SF-36 (PCS) | 48.6 (1.3) | 49.6 (1.0) | 52.1 (0.4) | 48.0 (2.1) | 3.4 (1.4) | −1.6 (1.5) | 0.08 ## |
| SF-36 (MCS) | 53.3 (1.6) | 51.0 (1.6) | 52.1 (1.4) | 51.6 (1.0) | −1.1 (1.2) | 0.6 (1.6) | 0.30 |
| BDI | 9.4 (1.2) | 8.9 (1.1) | 6.6 (0.9) | 8.6 (0.7) | −2.8 (0.8) | −0.3 (1.1) | 0.14 # |
| CCL24 post/pre | - | - | - | - | 0.86 (0.07) | 1.09 (0.09) | 0.14 # |
| 70s Group | Baseline | Follow-Up | Change | ||||
|---|---|---|---|---|---|---|---|
| Active | Placebo | Active | Placebo | Active | Placebo | p Value 2 | |
| WMS-1 3 | 13.2 (0.7) | 13.0 (0.7) | 11.6 (0.8) | 10.2 (0.7) | −1.6 (0.4) | −2.8 (0.5) | 0.15 # |
| WMS-2 3 | 11.6 (0.7) | 13.5 (0.8) | 11.7 (0.9) | 8.9 (0.9) | 0.14 (3.0) | −4.55 (3.0) | 0.017 * |
| ADAS | 9.9 (0.6) | 10.3 (0.7) | 10.0 (1.1) | 11.5 (0.7) | 0.1 (0.6) | 1.3 (0.9) | 0.30 |
| SF-36 (PCS) | 45.4 (1.7) | 46.7 (1.6) | 42.3 (0.9) | 46.7 (1.3) | −3.0 (1.4) | 0.1 (1.5) | 0.21 |
| SF-36 (MCS) | 55.3 (1.6) | 50.3 (1.0) | 57.2 (1.3) | 53.7 (1.2) | 1.9 (1.1) | 3.3 (0.8) | 0.28 |
| BDI | 11.6 (1.1) | 8.7 (1.4) | 7.1 (0.8) | 6.5 (0.8) | −4.4 (0.8) | −2.3 (0.9) | 0.18 # |
| CCL24 post/pre | - | - | - | - | 0.59 (0.03) | 0.83 (0.03) | 0.006 ** |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katakura, Y.; Totsuka, M.; Imabayashi, E.; Matsuda, H.; Hisatsune, T. Anserine/Carnosine Supplementation Suppresses the Expression of the Inflammatory Chemokine CCL24 in Peripheral Blood Mononuclear Cells from Elderly People. Nutrients 2017, 9, 1199. https://doi.org/10.3390/nu9111199
Katakura Y, Totsuka M, Imabayashi E, Matsuda H, Hisatsune T. Anserine/Carnosine Supplementation Suppresses the Expression of the Inflammatory Chemokine CCL24 in Peripheral Blood Mononuclear Cells from Elderly People. Nutrients. 2017; 9(11):1199. https://doi.org/10.3390/nu9111199
Chicago/Turabian StyleKatakura, Yoshinori, Mamoru Totsuka, Etsuko Imabayashi, Hiroshi Matsuda, and Tatsuhiro Hisatsune. 2017. "Anserine/Carnosine Supplementation Suppresses the Expression of the Inflammatory Chemokine CCL24 in Peripheral Blood Mononuclear Cells from Elderly People" Nutrients 9, no. 11: 1199. https://doi.org/10.3390/nu9111199
APA StyleKatakura, Y., Totsuka, M., Imabayashi, E., Matsuda, H., & Hisatsune, T. (2017). Anserine/Carnosine Supplementation Suppresses the Expression of the Inflammatory Chemokine CCL24 in Peripheral Blood Mononuclear Cells from Elderly People. Nutrients, 9(11), 1199. https://doi.org/10.3390/nu9111199




