Diosgenin-Rich Yam Extract Enhances Cognitive Function: A Placebo-Controlled, Randomized, Double-Blind, Crossover Study of Healthy Adults
Abstract
1. Introduction
2. Methods
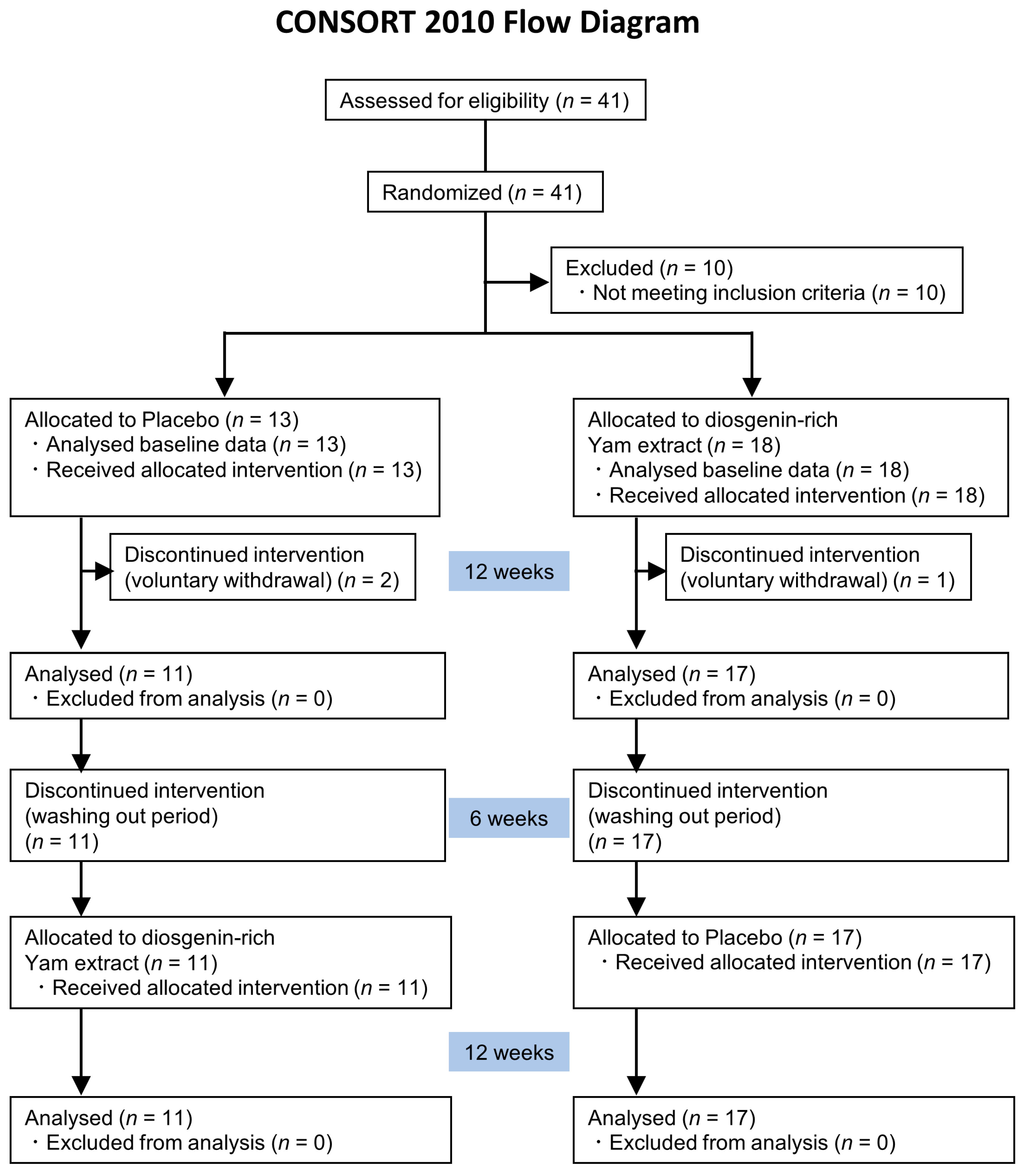
2.1. Trial Design
2.2. Participants
2.3. Intervention
2.4. Outcomes and Assessments
2.5. Neurocognitive Assessments
2.6. Safety Assessment
2.7. Randomization
2.8. Animal Experiments: Animals and Materials
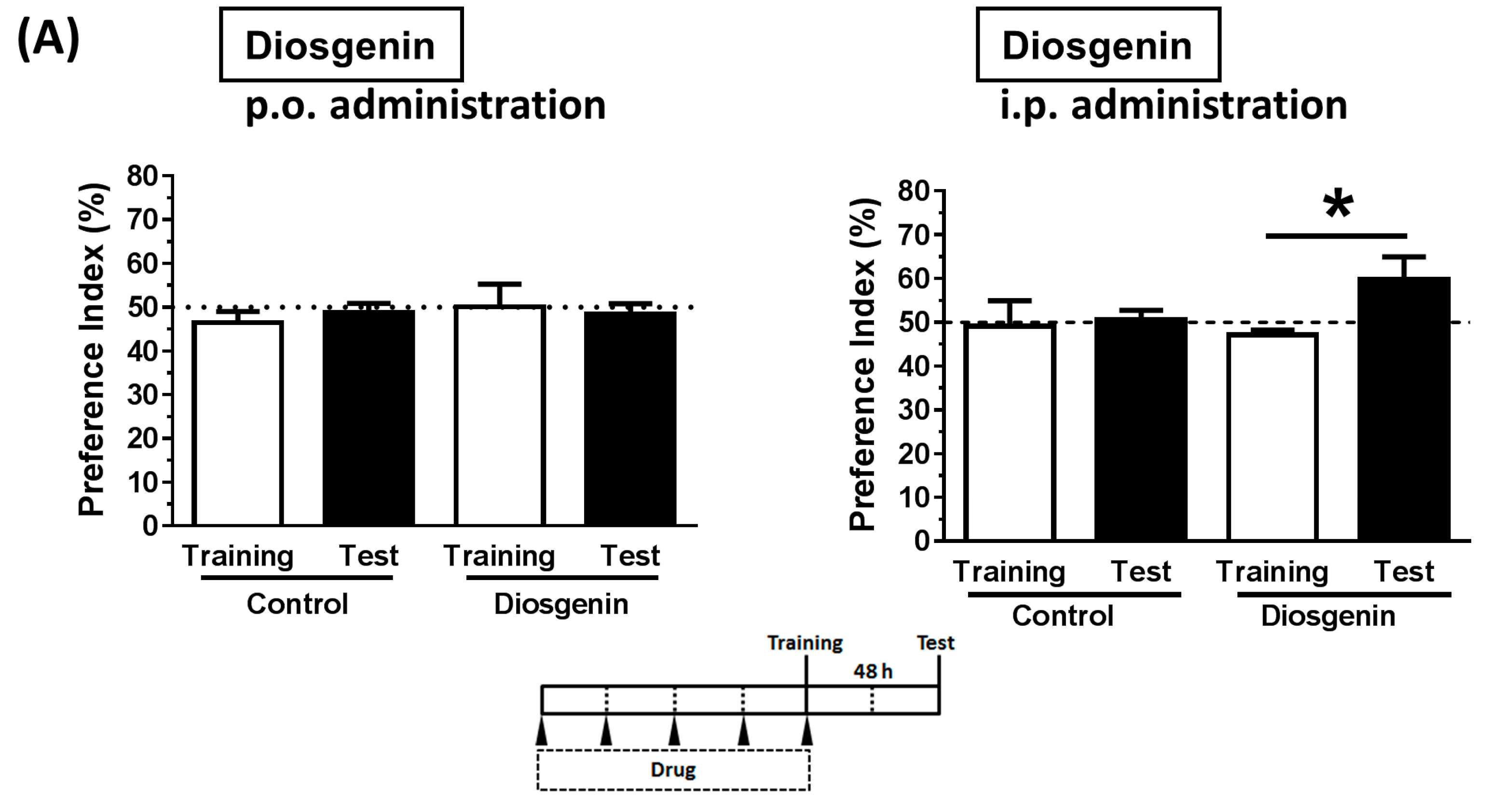
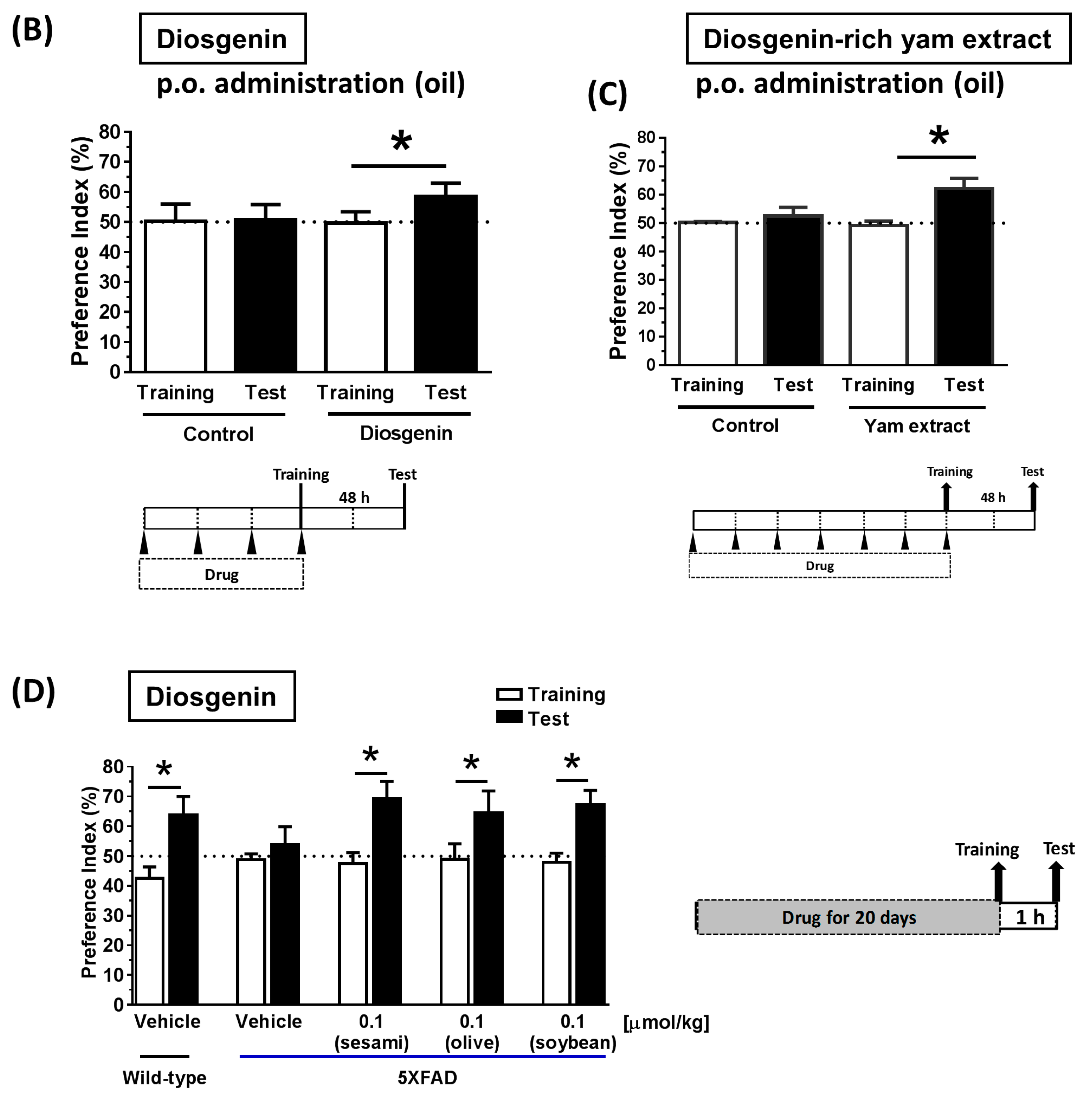
2.9. Animal Experiments: Object Recognition Test
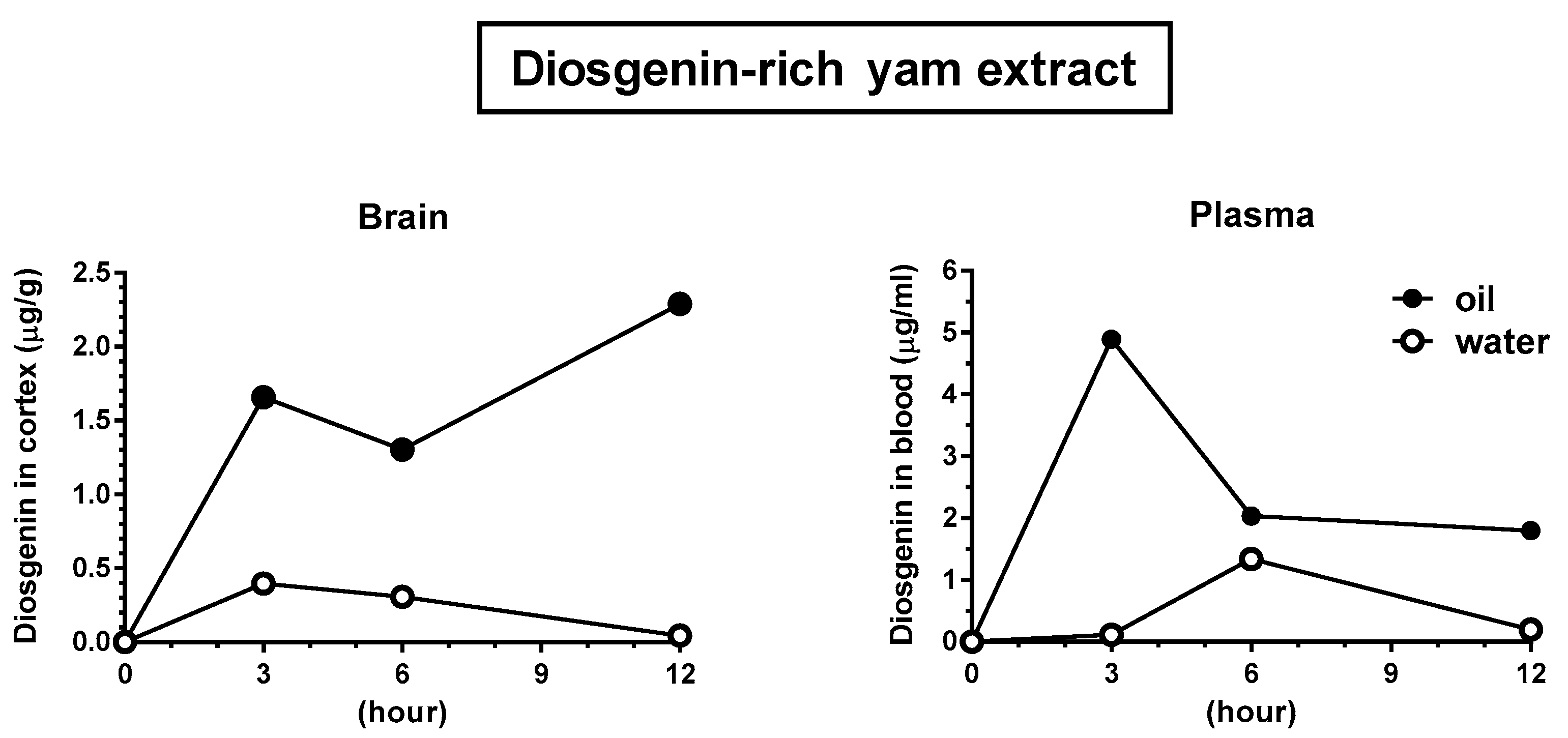
2.10. Animal Experiments: Brain Penetration of Diosgenin after the Oral Administration of a Diosgenin-Rich Extract
2.11. Statistical Analysis
3. Results
3.1. Baseline Characteristics of the Study Groups
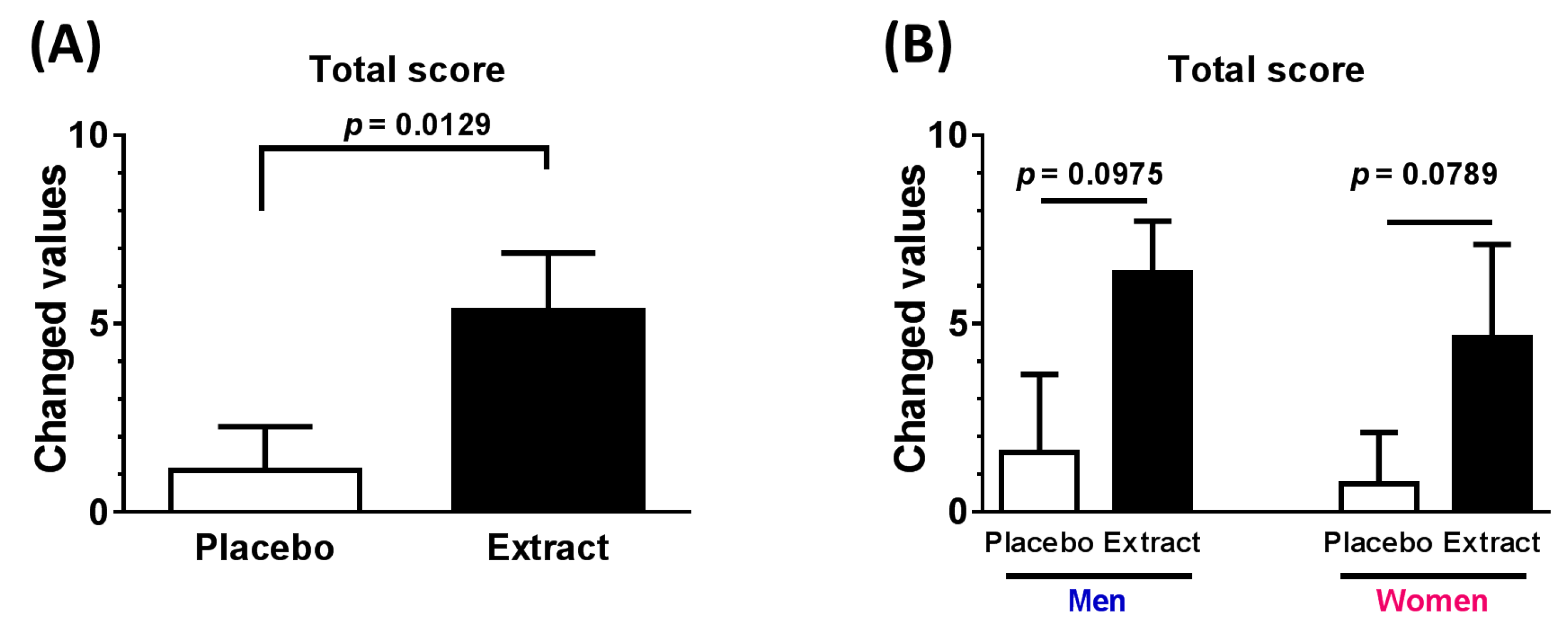
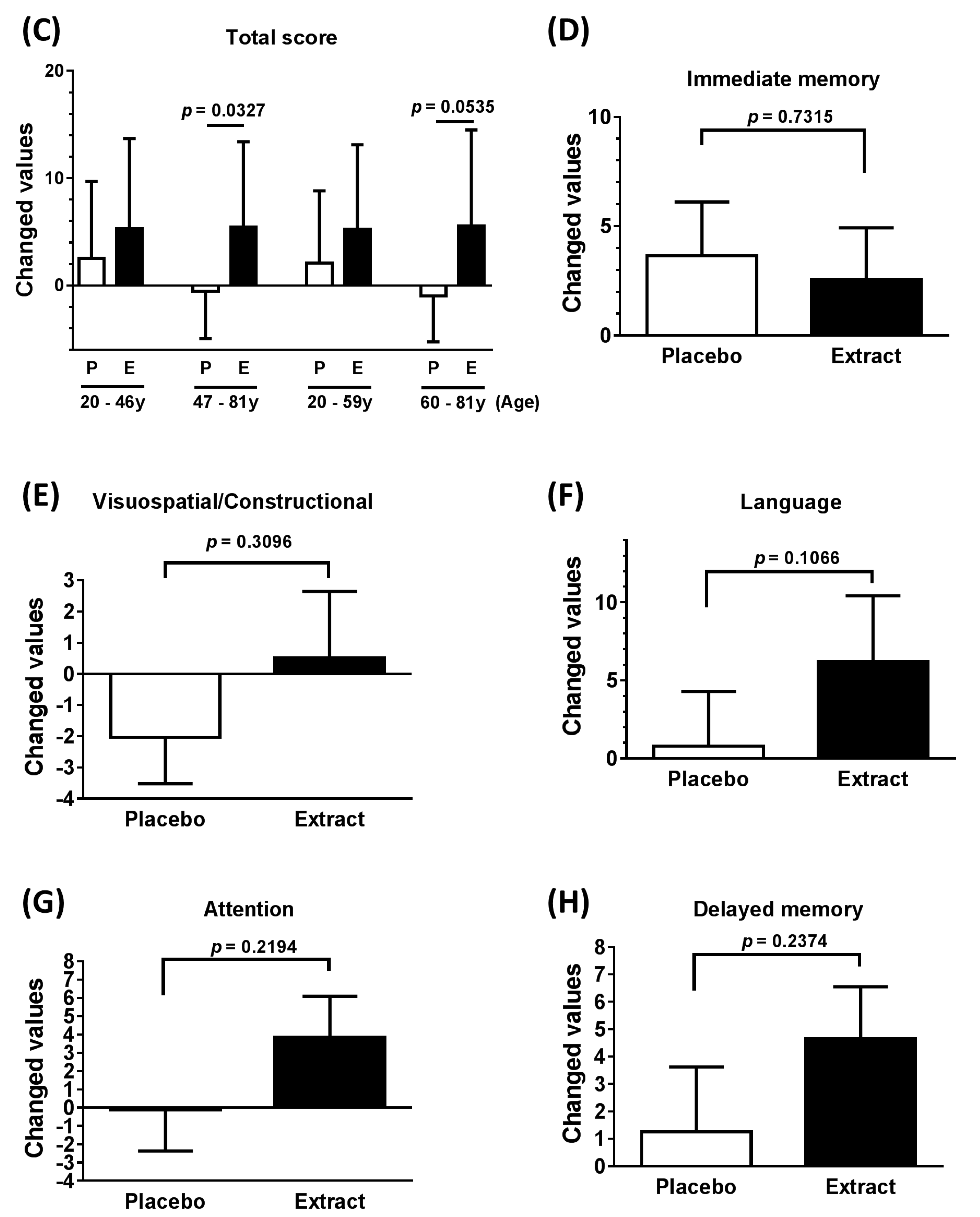
3.2. Neuropsychological Functioning
3.3. Safety Measures
3.4. Solubilization in Oil Facilitated Diosgenin Distribution in the Brain
4. Discussion
5. Limitations
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tan, C.C.; Yu, J.T.; Wang, H.F.; Tan, M.S.; Meng, X.F.; Wang, C.; Jiang, T.; Zhu, X.C.; Tan, L. Efficacy and safety of donepezil, galantamine, rivastigmine, and memantine for the treatment of Alzheimer’s disease: A systematic review and meta-analysis. J. Alzheimer’s Dis. 2014, 41, 615–631. [Google Scholar]
- Mehta, D.; Jackson, R.; Paul, G.; Shi, J.; Sabbagh, M. Why do trials for Alzheimer’s disease drugs keep failing? A discontinued drug perspective for 2010–2015. Expert Opin. Investig. Drugs 2017, 26, 735–739. [Google Scholar] [CrossRef] [PubMed]
- Insel, P.S.; Ossenkoppele, R.; Gessert, D.; Jagust, W.; Landau, S.; Hansson, O.; Weiner, M.W.; Mattsson, N. Alzheimer’s Disease Neuroimaging Initiative. Time to amyloid positivity and preclinical changes in brain metabolism, atrophy, and cognition: Evidence for emerging amyloid pathology in Alzheimer’s disease. Front. Neurosci. 2017, 11, 281. [Google Scholar] [CrossRef] [PubMed]
- Tohda, C.; Urano, T.; Umezaki, M.; Nemere, I.; Kuboyama, T. Diosgenin is an exogenous activator of 1,25D3-MARRS/Pdia3/ERp57 and improves Alzheimer’s disease pathologies in 5XFAD mice. Sci. Rep. 2012, 2, 535. [Google Scholar] [CrossRef] [PubMed]
- Tohda, C.; Lee, Y.A.; Goto, Y.; Nemere, I. Diosgenin-induced cognitive enhancement in normal mice is mediated by 1,25D3-MARRS. Sci. Rep. 2013, 3, 3395. [Google Scholar] [CrossRef] [PubMed]
- Uhlén, M.; Björling, E.; Agaton, C.; Szigyarto, C.A.; Amini, B.; Andersen, E.; Andersson, A.C.; Angelidou, P.; Asplund, A.; Asplund, C.; et al. A human protein atlas for normal and cancer tissues based on antibody proteomics. Mol. Cell Proteom. 2005, 4, 1920–1932. [Google Scholar] [CrossRef] [PubMed]
- Laws, K.R.; Sweetnam, H.; Kondel, T.K. Is Ginkgo biloba a cognitive enhancer in healthy individuals? A meta-analysis. Hum. Psychopharmacol. 2012, 27, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Kaschel, R. Specific memory effects of Ginkgo biloba extract EGb 761 in middle-aged healthy volunteers. Phytomedicine 2011, 18, 1202–1207. [Google Scholar] [CrossRef] [PubMed]
- Stonehouse, W.; Conlon, C.A.; Podd, J.; Hill, S.R.; Minihane, A.M.; Haskell, C.; Kennedy, D. DHA supplementation improved both memory and reaction time in healthy young adults: A randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 1134–1143. [Google Scholar] [CrossRef] [PubMed]
- Jackson, P.A.; Forster, J.S.; Bell, J.G.; Dick, J.R.; Younger, I.; Kennedy, D.O. DHA supplementation alone or in combination with other nutrients does not modulate cerebral hemodynamics or cognitive function in healthy older adults. Nutrients 2016, 8, 86. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.N.; Pang, Y.X.; He, X.C.; Zhang, Y.Z.; Zhang, J.Y.; Zhao, Z.Z.; Yi, T.; Chen, H.B. UPLC-QTOF-MS identification of metabolites in rat biosamples after oral administration of Dioscorea saponins: A comparative study. J. Ethnopharmacol. 2015, 165, 127–140. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.S.; Chiu, Y.J.; Wu, L.Y.; Lu, T.C.; Huang, T.H.; Hsieh, M.T.; Lu, C.Y.; Peng, W.H. Diosgenin ameliorates cognition deficit and attenuates oxidative damage in senescent mice induced by d-galactose. Am. J. Chin. Med. 2011, 39, 551–563. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.S.; Deng, J.S.; Hsieh, M.T.; Fan, M.J.; Lee, M.M.; Chueh, F.S.; Han, C.K.; Lin, Y.C.; Peng, W.H. Yam (Dioscorea pseudojaponica Yamamoto) ameliorates cognition deficit and attenuates oxidative damage in senescent mice induced by d-galactose. Am. J. Chin. Med. 2009, 37, 889–902. [Google Scholar] [CrossRef] [PubMed]
- Randolph, C.; Tierney, M.C.; Mohr, E.; Chase, T.N. The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS): Preliminary clinical validity. J. Clin. Exp. Neuropsychol. 1998, 20, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Matsui, M.; Kasai, Y.; Nagasaki, M. Reliability and validity for the Japanese version of the repeatable battery for the assessment of neuropsychological status (RBANS). Toyama Med. J. 2010, 21, 31–36. (In Japanese) [Google Scholar]
- Maki, N.; Ikeda, M.; Hokoishi, K.; Nebu, A.; Hirono, N.; Tanabe, H. Japanese version of the Short-Memory Questionnaire and Mini-Mental State Examination in relation to demographic variables: Community survey of elderly healthy residents in Nakayama, Japan. No To Shinkei 1999, 51, 209–213. (In Japanese) [Google Scholar] [PubMed]
- Chen, Y.; Tang, Y.M.; Yu, S.L.; Han, Y.W.; Kou, J.P.; Liu, B.L.; Yu, B.Y. Advances in the pharmacological activities and mechanisms of diosgenin. Chin. J. Nat. Med. 2015, 13, 578–587. [Google Scholar] [CrossRef]
- Okawara, M.; Hashimoto, F.; Todo, H.; Sugibayashi, K.; Tokudome, Y. Effect of liquid crystals with cyclodextrin on the bioavailability of a poorly water-soluble compound, diosgenin, after its oral administration to rats. Int. J. Pharm. 2014, 472, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Louveau, A.; Smirnov, I.; Keyes, T.J.; Eccles, J.D.; Rouhani, S.J.; Peske, J.D.; Derecki, N.C.; Castle, D.; Mandell, J.W.; Lee, K.S.; et al. Structural and functional features of central nervous system lymphatic vessels. Nature 2015, 523, 337–341. [Google Scholar] [CrossRef] [PubMed]
| Values | |||
|---|---|---|---|
| n | Mean | SD | |
| Subjects (Men/Women) | 28 (12/16) | ||
| Age (years) | 46.50 | 18.67 | |
| Race | Asian | ||
| Educated period (years) | 16.29 | 2.37 | |
| Estimated intelligence quotients (IQ) | 110.79 | 6.49 | |
| MMSE-J | 29.39 | 1.03 | |
| Total cholesterol (mg/dL) | 203.36 | 35.94 |
| Cognitive Domain | Mean of Difference | SD of Difference | 95% CI | p Value |
|---|---|---|---|---|
| Total score (All) | 4.250 | 8.949 | 0.974 to 7.526 | 0.0129 * |
| Total score (Men) | 4.750 | 9.087 | −1.023 to 10.520 | 0.0975 |
| Total score (Women) | 3.875 | 8.221 | −5.056 to 8.256 | 0.0789 |
| Total score (47–81 years) | 6.000 | 8.963 | 0.5838 to 11.420 | 0.0327 * |
| Total score (60–81 years) | 6.556 | 8.691 | −0.125 to 13.240 | 0.0535 * |
| Immediate memory | −1.107 | 16.900 | −7.659 to 5.445 | 0.7315 |
| Visuospatial/Constructional | 2.536 | 12.960 | −2.489 to 7.560 | 0.3096 |
| Language | 5.429 | 17.210 | −1.243 to 12.100 | 0.1066 |
| Attention | 3.964 | 16.690 | −2.506 to 10.430 | 0.2194 |
| Delayed memory | 3.393 | 14.860 | −2.369 to 9.154 | 0.2374 |
| MMSE-J | −0.107 | 1.100 | −0.5337 to 0.3194 | 0.6105 |
| Cognitive Domain | Subtest | Mean of Difference | SD of Difference | 95% CI | p Value |
|---|---|---|---|---|---|
| Immediate memory | List learning | 0.3214 | 3.1860 | −9.1410 to 1.5570 | 0.5978 |
| Story memory | −0.6071 | 3.3260 | −1.8970 to 0.6825 | 0.3426 | |
| Visuospatial/Constructional | Figure copy | 0.1071 | 2.1140 | −0.7126 to 0.9269 | 0.7906 |
| Line orientation | 0.5000 | 2.4420 | −0.4469 to 1.4470 | 0.2882 | |
| Language | Picture naming | 0.1429 | 2.0850 | −0.6658 to 0.9515 | 0.7198 |
| Semantic fluency | 1.3210 | 3.1280 | 0.1087 to 2.5340 | 0.0338 * | |
| Attention | Digit span | 1.1070 | 4.2720 | −0.5492 to 2.7640 | 0.1815 |
| Digit symbol coding | 0.5357 | 3.3160 | −0.7503 to 1.8220 | 0.4002 | |
| Delayed memory | List recall | 0.0714 | 3.4950 | −1.2840 to 1.4270 | 0.9147 |
| List recognition | 0.0714 | 2.7070 | −0.9783 to 1.1210 | 0.8900 | |
| Story recall | 0.3571 | 1.7890 | −0.3366 to 1.0510 | 0.3002 | |
| Figure recall | 0.9643 | 3.2940 | −0.3130 to 2.2420 | 0.1330 |
| Changed Values | |||||
|---|---|---|---|---|---|
| Placebo | Diosgenin-Rich Yam Extract | ||||
| Mean | SD | Mean | SD | p Value | |
| Total protein | −0.0393 | 0.3190 | 0.0464 | 0.3533 | 0.6102 |
| Total cholesterol | −10.0000 | 22.9331 | 0.0357 | 25.1123 | 0.2765 |
| HDL-cholesterol | −1.0357 | 13.3569 | 0.6786 | 8.9238 | 0.9651 |
| Triglyceride | −13.8214 | 88.0980 | −36.4286 | 72.4980 | 0.0674 |
| Glucose (non-fasting) | −3.2143 | 29.0496 | 12.2500 | 28.1644 | 0.1105 |
| AST (GOT) | 1.4643 | 4.9026 | −0.7500 | 4.2828 | 0.3281 |
| ALT (GPT) | 0.4643 | 5.4670 | −0.1429 | 4.2922 | 0.9946 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tohda, C.; Yang, X.; Matsui, M.; Inada, Y.; Kadomoto, E.; Nakada, S.; Watari, H.; Shibahara, N. Diosgenin-Rich Yam Extract Enhances Cognitive Function: A Placebo-Controlled, Randomized, Double-Blind, Crossover Study of Healthy Adults. Nutrients 2017, 9, 1160. https://doi.org/10.3390/nu9101160
Tohda C, Yang X, Matsui M, Inada Y, Kadomoto E, Nakada S, Watari H, Shibahara N. Diosgenin-Rich Yam Extract Enhances Cognitive Function: A Placebo-Controlled, Randomized, Double-Blind, Crossover Study of Healthy Adults. Nutrients. 2017; 9(10):1160. https://doi.org/10.3390/nu9101160
Chicago/Turabian StyleTohda, Chihiro, Ximeng Yang, Mie Matsui, Yuna Inada, Emika Kadomoto, Shotaro Nakada, Hidetoshi Watari, and Naotoshi Shibahara. 2017. "Diosgenin-Rich Yam Extract Enhances Cognitive Function: A Placebo-Controlled, Randomized, Double-Blind, Crossover Study of Healthy Adults" Nutrients 9, no. 10: 1160. https://doi.org/10.3390/nu9101160
APA StyleTohda, C., Yang, X., Matsui, M., Inada, Y., Kadomoto, E., Nakada, S., Watari, H., & Shibahara, N. (2017). Diosgenin-Rich Yam Extract Enhances Cognitive Function: A Placebo-Controlled, Randomized, Double-Blind, Crossover Study of Healthy Adults. Nutrients, 9(10), 1160. https://doi.org/10.3390/nu9101160





