A New Approach to Assess Lifetime Dietary Patterns Finds Lower Consumption of Animal Foods with Aging in a Longitudinal Analysis of a Health-Oriented Adventist Population
Abstract
:1. Introduction
2. Materials and Methods
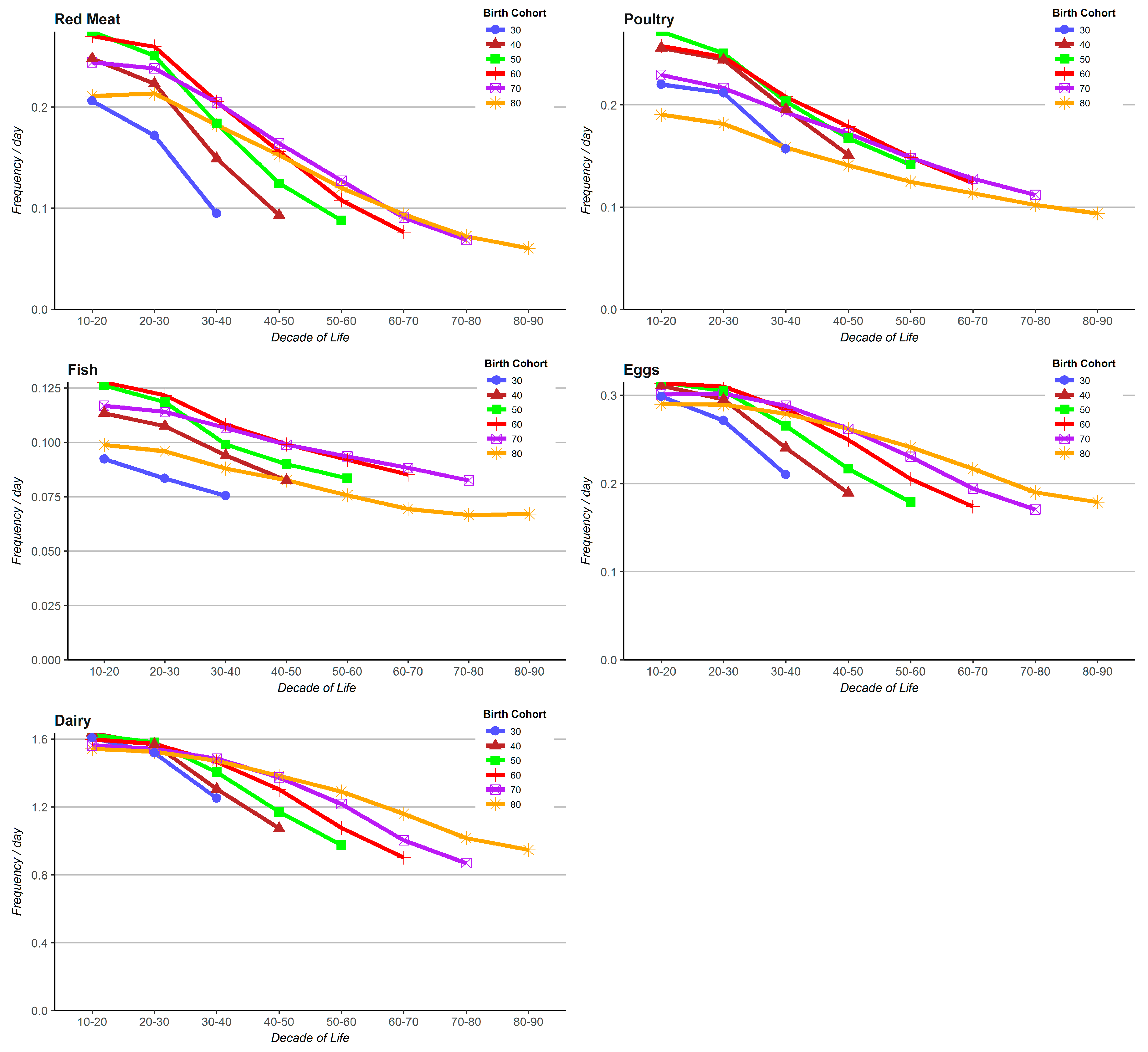
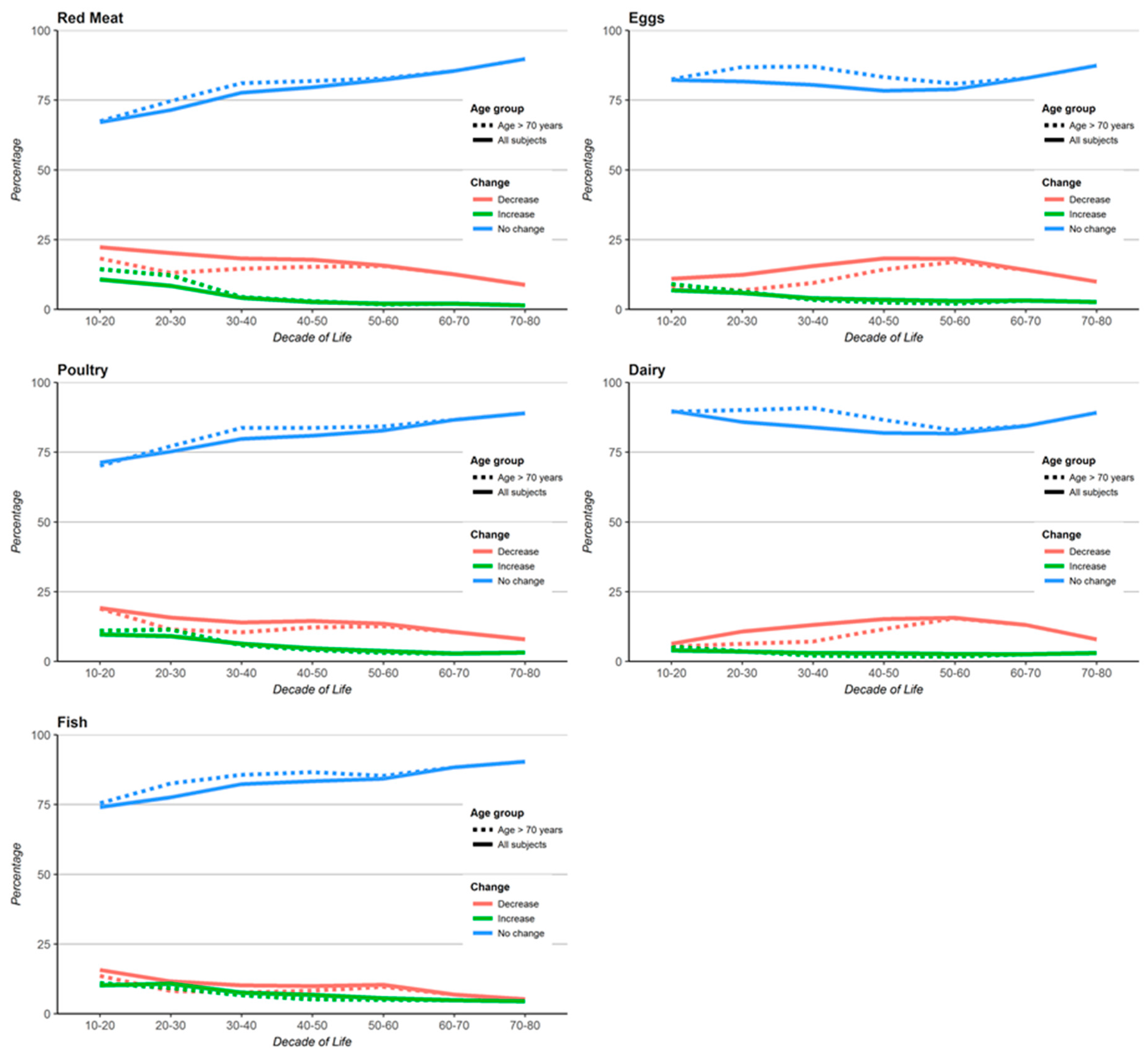
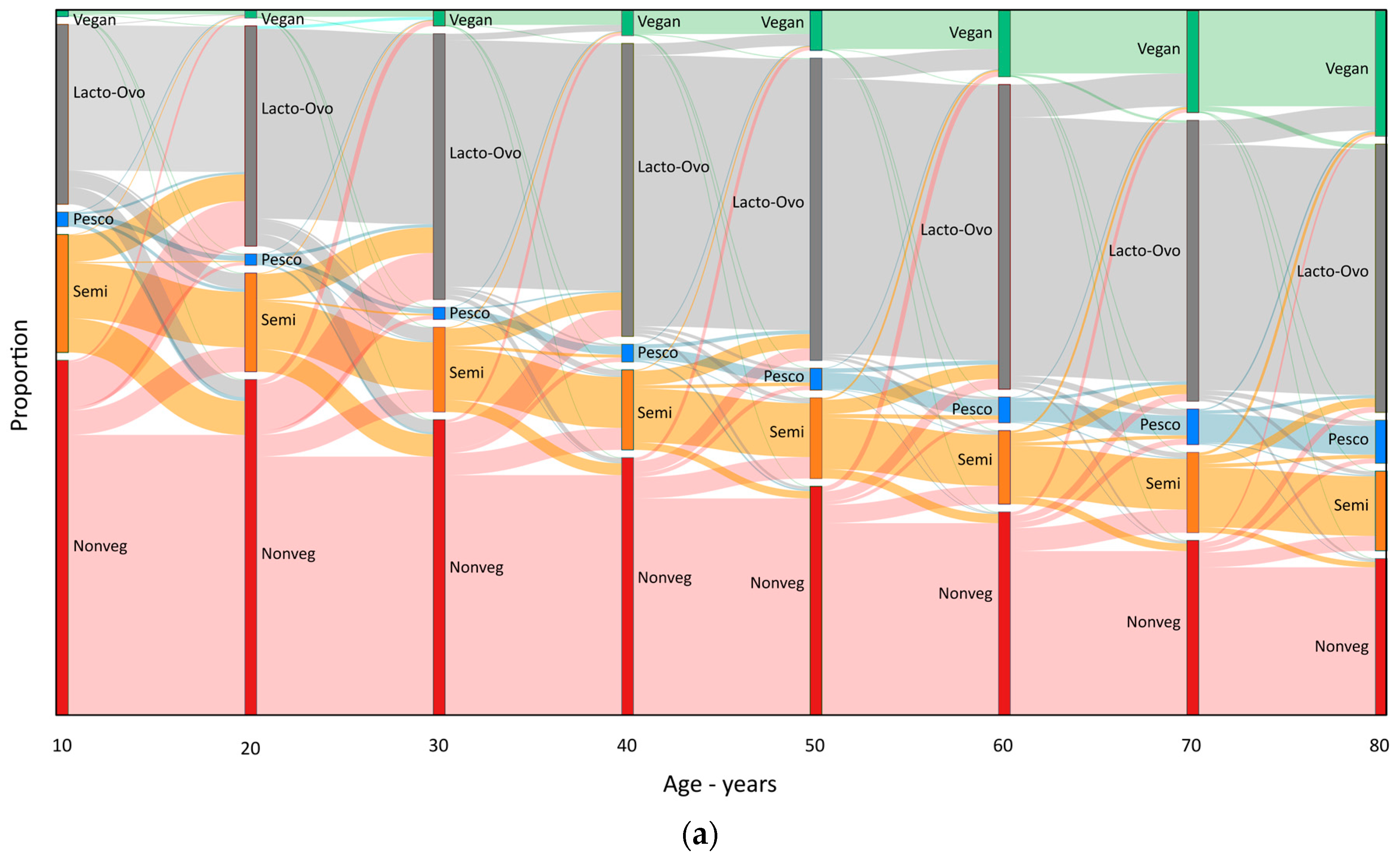
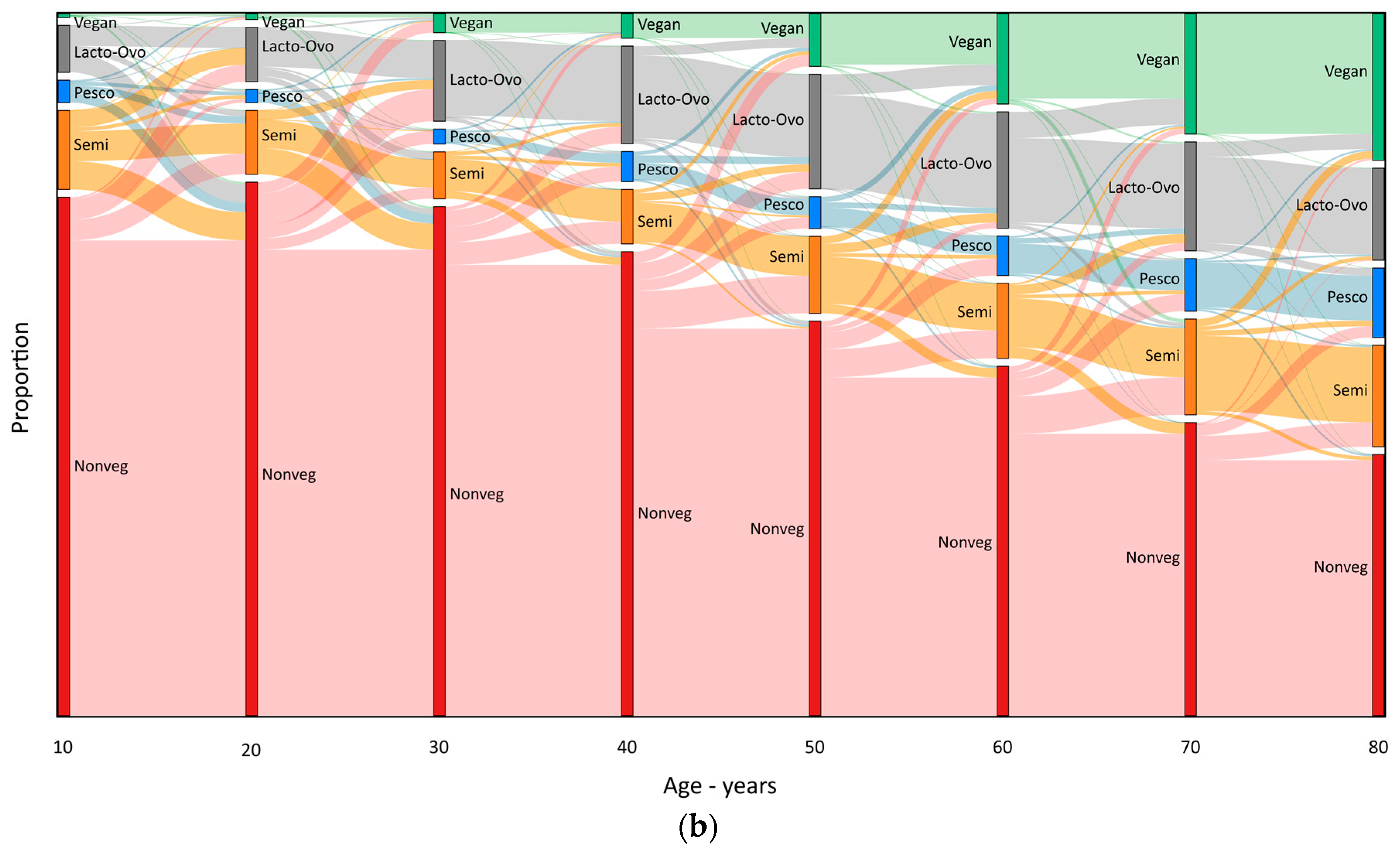
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mudaliar, U.; Zabetian, A.; Goodman, M.; Echouffo-Tcheugui, J.B.; Albright, A.L.; Gregg, E.W.; Ali, M.K. Cardiometabolic risk factor changes observed in diabetes prevention programs in us settings: A systematic review and meta-analysis. PLoS Med. 2016, 13, e1002095. [Google Scholar] [CrossRef] [PubMed]
- Patnode, C.D.; Evans, C.V.; Senger, C.A.; Redmond, N.; Lin, J.S. Behavioral counseling to promote a healthful diet and physical activity for cardiovascular disease prevention in adults without known cardiovascular disease risk factors: Updated evidence report and systematic review for the us preventive services task force. JAMA 2017, 318, 175–193. [Google Scholar] [PubMed]
- Ford, E.S.; Bergmann, M.M.; Boeing, H.; Li, C.; Capewell, S. Healthy lifestyle behaviors and all-cause mortality among adults in the united states. Prev. Med. 2012, 55, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Kvaavik, E.; Batty, G.D.; Ursin, G.; Huxley, R.; Gale, C.R. Influence of individual and combined health behaviors on total and cause-specific mortality in men and women: The united kingdom health and lifestyle survey. Arch. Intern. Med. 2010, 170, 711–718. [Google Scholar]
- Nechuta, S.J.; Shu, X.O.; Li, H.L.; Yang, G.; Xiang, Y.B.; Cai, H.; Chow, W.H.; Ji, B.; Zhang, X.; Wen, W.; et al. Combined impact of lifestyle-related factors on total and cause-specific mortality among chinese women: Prospective cohort study. PLoS Med. 2010, 7, e1000339. [Google Scholar] [CrossRef] [PubMed]
- Tonstad, S.; Stewart, K.; Oda, K.; Batech, M.; Herring, R.P.; Fraser, G.E. Vegetarian diets and incidence of diabetes in the adventist health study-2. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 292–299. [Google Scholar] [CrossRef] [PubMed]
- King, V.; Dakin, R.S.; Liu, L.; Hadoke, P.W.; Walker, B.R.; Seckl, J.R.; Norman, J.E.; Drake, A.J. Maternal obesity has little effect on the immediate offspring but impacts on the next generation. Endocrinology 2013, 154, 2514–2524. [Google Scholar] [CrossRef] [PubMed]
- Roseboom, T.J.; Watson, E.D. The next generation of disease risk: Are the effects of prenatal nutrition transmitted across generations? Evidence from animal and human studies. Placenta 2012, 33 (Suppl. S2), e40–e44. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Afshin, A.; Benowitz, N.L.; Bittner, V.; Daniels, S.R.; Franch, H.A.; Jacobs, D.R., Jr.; Kraus, W.E.; Kris-Etherton, P.M.; Krummel, D.A.; et al. Population approaches to improve diet, physical activity, and smoking habits: A scientific statement from the american heart association. Circulation 2012, 126, 1514–1563. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Ocke, M.C. Evaluation of methodologies for assessing the overall diet: Dietary quality scores and dietary pattern analysis. Proc. Nutr. Soc. 2013, 72, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Orlich, M.J.; Fraser, G.E. Vegetarian diets in the adventist health study 2: A review of initial published findings. Am. J. Clin. Nutr. 2014, 100 (Suppl. S1), 353S–358S. [Google Scholar] [CrossRef] [PubMed]
- Dinu, M.; Abbate, R.; Gensini, G.F.; Casini, A.; Sofi, F. Vegetarian, vegan diets and multiple health outcomes: A systematic review with meta-analysis of observational studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 3640–3649. [Google Scholar] [CrossRef] [PubMed]
- Tonstad, S.; Butler, T.; Yan, R.; Fraser, G.E. Type of vegetarian diet, body weight, and prevalence of type 2 diabetes. Diabetes Care 2009, 32, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Rosell, M.; Appleby, P.; Spencer, E.; Key, T. Weight gain over 5 years in 21,966 meat-eating, fish-eating, vegetarian, and vegan men and women in EPIC-Oxford. Int. J. Obes. 2006, 30, 1389–1396. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, N.S.; Sabate, J.; Jaceldo-Siegl, K.; Fraser, G.E. Vegetarian dietary patterns are associated with a lower risk of metabolic syndrome: The adventist health study-2. Diabetes Care 2011, 34, 1225–1227. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, B.J.; Anousheh, R.; Fan, J.; Jaceldo-Siegl, K.; Fraser, G.E. Vegetarian diets and blood pressure among white subjects: Results from the adventist health study-2 (AHS-2). Public Health Nutr. 2012, 15, 1909–1916. [Google Scholar] [CrossRef] [PubMed]
- Fraser, G.; Katuli, S.; Anousheh, R.; Knutsen, S.; Herring, P.; Fan, J. Vegetarian diets and cardiovascular risk factors in black members of the adventist health study-2. Public Health Nutr. 2015, 18, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Orlich, M.J.; Singh, P.N.; Sabate, J.; Fan, J.; Sveen, L.; Bennett, H.; Knutsen, S.F.; Beeson, W.L.; Jaceldo-Siegl, K.; Butler, T.L.; et al. Vegetarian dietary patterns and the risk of colorectal cancers. JAMA Intern. Med. 2015, 175, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Tantamango-Bartley, Y.; Knutsen, S.F.; Knutsen, R.; Jacobsen, B.K.; Fan, J.; Beeson, W.L.; Sabate, J.; Hadley, D.; Jaceldo-Siegl, K.; Penniecook, J.; et al. Are strict vegetarians protected against prostate cancer? Am. J. Clin. Nutr. 2016, 103, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Tantamango-Bartley, Y.; Jaceldo-Siegl, K.; Fan, J.; Fraser, G. Vegetarian diets and the incidence of cancer in a low-risk population. Cancer Epidemiol. Biomarkers Prev. 2013, 22, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Orlich, M.J.; Jaceldo-Siegl, K.; Sabate, J.; Fan, J.; Singh, P.N.; Fraser, G.E. Patterns of food consumption among vegetarians and non-vegetarians. Br. J. Nutr. 2014, 112, 1644–1653. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, N.S.; Jaceldo-Siegl, K.; Sabate, J.; Fraser, G.E. Nutrient profiles of vegetarian and nonvegetarian dietary patterns. J. Acad. Nutr. Diet. 2013, 113, 1610–1619. [Google Scholar] [CrossRef] [PubMed]
- Darnton-Hill, I.; Nishida, C.; James, W.P. A life course approach to diet, nutrition and the prevention of chronic diseases. Public Health Nutr. 2004, 7, 101–121. [Google Scholar] [CrossRef] [PubMed]
- Parekh, N.; Zizza, C. Life course epidemiology in nutrition and chronic disease research: A timely discussion. Adv. Nutr. 2013, 4, 551–553. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.N.; Batech, M.; Faed, P.; Jaceldo-Siegl, K.; Martins, M.; Fraser, G.E. Reliability of meat, fish, dairy, and egg intake over a 33-year interval in adventist health study 2. Nutr. Cancer 2014, 66, 1315–1321. [Google Scholar] [CrossRef] [PubMed]
- Teixeira Martins, M.C.; Jaceldo-Siegl, K.; Fan, J.; Singh, P.; Fraser, G.E. Short- and long-term reliability of adult recall of vegetarian dietary patterns in the adventist health study-2 (AHS-2). J. Nutr. Sci. 2015, 4, e11. [Google Scholar] [CrossRef] [PubMed]
- Butler, T.L.; Fraser, G.E.; Beeson, W.L.; Knutsen, S.F.; Herring, R.P.; Chan, J.; Sabate, J.; Montgomery, S.; Haddad, E.; Preston-Martin, S.; et al. Cohort profile: The adventist health study-2 (AHS-2). Int. J. Epidemiol. 2008, 37, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Schafer, J.L. Inference by data augmentation. In Analysis of Incomplete Multivariate Data; Chapman and Hall/CRC: Boca Raton, FL, USA, 1997. [Google Scholar]
- Jaceldo-Siegl, K.; Knutsen, S.F.; Sabate, J.; Beeson, W.L.; Chan, J.; Herring, R.P.; Butler, T.L.; Haddad, E.; Bennett, H.; Montgomery, S.; et al. Validation of nutrient intake using an ffq and repeated 24 h recalls in black and white subjects of the adventist health study-2 (AHS-2). Public Health Nutr. 2010, 13, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Jaceldo-Siegl, K.; Fan, J.; Sabate, J.; Knutsen, S.F.; Haddad, E.; Beeson, W.L.; Herring, R.P.; Butler, T.L.; Bennett, H.; Fraser, G.E. Race-specific validation of food intake obtained from a comprehensive FFQ: The adventist health study-2. Public Health Nutr. 2011, 14, 1988–1997. [Google Scholar] [CrossRef] [PubMed]
- Fraser, G.E. Diet, Life Expectancy, and Chronic Disease: Studies of Seventh-Day Adventists and Other Vegetarians; Oxford University Press: Oxford, NY, USA, 2003; p. 1. [Google Scholar]
- Lee, J.; Stacey, G.; Fraser, G. Social support, religiosity, other psychological factors, and health. In Diet, Life Expectancy, and Chronic Disease, 1st ed.; Oxford University Press: Oxford, NY, USA, 2003; pp. 149–175. [Google Scholar]
- Derbyshire, E.J. Flexitarian diets and health: A review of the evidence-based literature. Front. Nutr. 2016, 3, 55. [Google Scholar] [CrossRef] [PubMed]
- Caini, S.; Masala, G.; Gnagnarella, P.; Ermini, I.; Russell-Edu, W.; Palli, D.; Gandini, S. Food of animal origin and risk of non-hodgkin lymphoma and multiple myeloma: A review of the literature and meta-analysis. Crit. Rev. Oncol. Hematol. 2016, 100, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Kouvari, M.; Tyrovolas, S.; Panagiotakos, D.B. Red meat consumption and healthy ageing: A review. Maturitas 2016, 84, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Battaglia Richi, E.; Baumer, B.; Conrad, B.; Darioli, R.; Schmid, A.; Keller, U. Health risks associated with meat consumption: A review of epidemiological studies. Int. J. Vitam. Nutr. Res. 2015, 85, 70–78. [Google Scholar] [CrossRef] [PubMed]
- McAfee, A.J.; McSorley, E.M.; Cuskelly, G.J.; Moss, B.W.; Wallace, J.M.; Bonham, M.P.; Fearon, A.M. Red meat consumption: An overview of the risks and benefits. Meat Sci. 2010, 84, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Antunes, P.; Mourao, J.; Campos, J.; Peixe, L. Salmonellosis: The role of poultry meat. Clin. Microbiol. Infect. 2016, 22, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Whiley, H.; Ross, K. Salmonella and eggs: From production to plate. Int. J. Environ. Res. Public Health 2015, 12, 2543–2556. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Liu, X.; Zhang, D. Fish consumption and risk of depression: A meta-analysis. J. Epidemiol. Community Health 2016, 70, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Bosch, A.C.; O’Neill, B.; Sigge, G.O.; Kerwath, S.E.; Hoffman, L.C. Heavy metals in marine fish meat and consumer health: A review. J. Sci. Food Agric. 2016, 96, 32–48. [Google Scholar] [CrossRef] [PubMed]
- Fuller, N.R.; Sainsbury, A.; Caterson, I.D.; Markovic, T.P. Egg consumption and human cardio-metabolic health in people with and without diabetes. Nutrients 2015, 7, 7399–7420. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.C. Milk nutritional composition and its role in human health. Nutrition 2014, 30, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Astrup, A. Yogurt and dairy product consumption to prevent cardiometabolic diseases: Epidemiologic and experimental studies. Am. J. Clin. Nutr. 2014, 99, 1235S–1242S. [Google Scholar] [CrossRef] [PubMed]
- Forouhi, N.G. Association between consumption of dairy products and incident type 2 diabetes—Insights from the european prospective investigation into cancer study. Nutr. Rev. 2015, 73 (Suppl. S1), 15S–22S. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Dairy products and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Am. J. Clin. Nutr. 2013, 98, 1066–1083. [Google Scholar] [CrossRef] [PubMed]
- Soret, S.; Mejia, A.; Batech, M.; Jaceldo-Siegl, K.; Harwatt, H.; Sabate, J. Climate change mitigation and health effects of varied dietary patterns in real-life settings throughout North America. Am. J. Clin. Nutr. 2014, 100 (Suppl. S1), 490S–495S. [Google Scholar] [CrossRef] [PubMed]
- Koenig, H.G. Religion, spirituality, and health: A review and update. Adv. Mind Body Med. 2015, 29, 19–26. [Google Scholar] [PubMed]
- Dudley, R.L.; Mutch, P.B.; Cruise, R.J. Religious factors and drug usage among seventh-day adventist youth in North America. J. Sci. Study Relig. 1987, 26, 218–233. [Google Scholar] [CrossRef]
- Lee, J.W.; Rice, G.T.; Gillespie, V.B. Family worship patterns and their correlation with adolescent behavior and beliefs. J. Sci. Study Relig. 1997, 36, 372–381. [Google Scholar] [CrossRef]
- Morton, K.R.; Lee, J.W.; Martin, L.R. Pathways from religion to health: Mediation by psychosocial and lifestyle mechanisms. Psychol. Relig. Spiritual. 2017, 9, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Hopp-Marshak, H.; Freier, K.; Fraser, G. Changing a population’s diet: A behavioral view of the adventist experience. In Diet, Life Expectancy, and Chronic Disease, 1st ed.; Oxford University Press: Oxford, NY, USA, 2003; pp. 241–247. [Google Scholar]
- Orlich, M.J.; Singh, P.N.; Sabate, J.; Jaceldo-Siegl, K.; Fan, J.; Knutsen, S.; Beeson, W.L.; Fraser, G.E. Vegetarian dietary patterns and mortality in adventist health study 2. JAMA Intern. Med. 2013, 173, 1230–1238. [Google Scholar] [CrossRef] [PubMed]
- Chang-Claude, J.; Frentzel-Beyme, R. Dietary and lifestyle determinants of mortality among german vegetarians. Int. J. Epidemiol. 1993, 22, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Chang-Claude, J.; Frentzel-Beyme, R.; Eilber, U. Mortality pattern of german vegetarians after 11 years of follow-up. Epidemiology 1992, 3, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Fraser, G.E. Associations between diet and cancer, ischemic heart disease, and all-cause mortality in non-hispanic white california seventh-day adventists. Am. J. Clin. Nutr. 1999, 70, 532S–538S. [Google Scholar] [PubMed]
- Fraser, G.E.; Shavlik, D.J. Ten years of life: Is it a matter of choice? Arch. Intern. Med. 2001, 161, 1645–1652. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.N.; Sabate, J.; Fraser, G.E. Does low meat consumption increase life expectancy in humans? Am. J. Clin. Nutr. 2003, 78, 526S–532S. [Google Scholar] [PubMed]
- Key, T.J.; Fraser, G.E.; Thorogood, M.; Appleby, P.N.; Beral, V.; Reeves, G.; Burr, M.L.; Chang-Claude, J.; Frentzel-Beyme, R.; Kuzma, J.W.; et al. Mortality in vegetarians and nonvegetarians: Detailed findings from a collaborative analysis of 5 prospective studies. Am. J. Clin. Nutr. 1999, 70, 516S–524S. [Google Scholar] [PubMed]
- Montero, P.; Bernis, C.; Varea, C.; Arias, S. Lifetime dietary change and its relation to increase in weight in Spanish women. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Hosking, D.; Danthiir, V.; Nettelbeck, T.; Wilson, C. Assessing lifetime diet: Reproducibility of a self-administered, non-quantitative ffq. Public Health Nutr. 2011, 14, 801–808. [Google Scholar] [CrossRef] [PubMed]
- Halioua, L.; Anderson, J.J. Lifetime calcium intake and physical activity habits: Independent and combined effects on the radial bone of healthy premenopausal Caucasian women. Am. J. Clin. Nutr. 1989, 49, 534–541. [Google Scholar] [PubMed]
- Soroko, S.; Holbrook, T.L.; Edelstein, S.; Barrett-Connor, E. Lifetime milk consumption and bone mineral density in older women. Am. J. Public Health 1994, 84, 1319–1322. [Google Scholar] [CrossRef] [PubMed]
- Steck, S.E.; Gaudet, M.M.; Eng, S.M.; Britton, J.A.; Teitelbaum, S.L.; Neugut, A.I.; Santella, R.M.; Gammon, M.D. Cooked meat and risk of breast cancer—Lifetime versus recent dietary intake. Epidemiology 2007, 18, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, C.M.; Georgiou, C.C.; Snow-Harter, C.M.; Gillis, D.E. Bone mineral density in mother-daughter pairs: Relations to lifetime exercise, lifetime milk consumption, and calcium supplements. Am. J. Clin. Nutr. 1996, 63, 72–79. [Google Scholar] [PubMed]
- Wallace, L.S.; Ballard, J.E. Lifetime physical activity and calcium intake related to bone density in young women. J. Women's Health Gend. Based Med. 2002, 11, 389–398. [Google Scholar] [CrossRef] [PubMed]

| LTS-Vegan | LTS-Lacto | LTS-Semi | LTS-Pesco | LTS-Non-veg | Switchers | p | |
|---|---|---|---|---|---|---|---|
| n | 52 | 1403 | 165 | 22 | 2856 | 10,834 | |
| Age in years, mean (SD) | 78.85 (5.22) | 78.45 (5.40) | 78.87 (5.30) | 78.23 (5.07) | 76.78 (4.93) | 77.52 (5.20) | <0.001 |
| Gender, % | <0.001 | ||||||
| Male | 36.5 | 36.3 | 43.6 | 27.3 | 42.5 | 38.3 | |
| Female | 63.5 | 63.7 | 56.4 | 72.7 | 57.5 | 61.7 | |
| Race, % | <0.001 | ||||||
| Non-black | 86.5 | 98.9 | 94.5 | 95.5 | 82.6 | 91.0 | |
| Black | 13.5 | 1.1 | 5.5 | 4.5 | 17.4 | 9.0 | |
| BMI in kg/m2, % | <0.001 | ||||||
| Underweight (<18.5) | 2.0 | 2.1 | 1.3 | 9.5 | 1.0 | 2.2 | |
| Normal (18.5–24.9) | 55.1 | 46.6 | 29.9 | 28.6 | 26.1 | 43.6 | |
| Overweight (25–29.9) | 34.7 | 37 | 49 | 33.3 | 43.6 | 36.6 | |
| Obese (≥30) | 8.2 | 14.3 | 19.7 | 28.6 | 29.3 | 17.7 | |
| Education, % | <0.001 | ||||||
| High school or less | 26.9 | 12.4 | 28.5 | 31.8 | 34.5 | 23.6 | |
| Some college | 48.1 | 35.3 | 38.2 | 31.8 | 40.8 | 39.1 | |
| College or higher | 25 | 52.3 | 33.3 | 36.4 | 24.7 | 37.4 | |
| Exercise, % | 0.027 | ||||||
| None | 63.2 | 69.8 | 70.8 | 60.0 | 68.7 | 69.7 | |
| 1–20 min/week | 10.5 | 15.3 | 11.8 | 6.7 | 13.3 | 13.9 | |
| 21–60 min/week | 7.9 | 9.2 | 11.8 | 26.7 | 10.4 | 9.8 | |
| 61–150 min/week | 7.9 | 3.6 | 2.8 | 0 | 5.3 | 4.6 | |
| ≥151 min/week | 10.5 | 2.1 | 2.8 | 6.7 | 2.2 | 2 | |
| TV watching, % | <0.001 | ||||||
| None to <1 h/day | 38.5 | 26.3 | 12.1 | 22.7 | 11.1 | 22.5 | |
| 1–2 h/day | 48.1 | 52.8 | 54.5 | 54.5 | 44.3 | 49.4 | |
| ≥3 h/day | 13.5 | 20.9 | 33.3 | 22.7 | 44.6 | 28 | |
| Sleep, % | <0.001 | ||||||
| <6 h/day | 21.2 | 21.9 | 23 | 45.5 | 32.2 | 27.9 | |
| 7 h/day | 42.3 | 36.4 | 31.5 | 22.7 | 31.6 | 34.8 | |
| ≥8 h/day | 36.5 | 41.8 | 45.5 | 31.8 | 36.2 | 37.3 | |
| Alcohol, past year, % | <0.001 | ||||||
| No | 100 | 99.2 | 95.2 | 100 | 91.2 | 97.5 | |
| Yes | 0 | 0.8 | 4.8 | 0 | 8.8 | 2.5 | |
| Cigarette smoking, % | <0.001 | ||||||
| Never | 96.2 | 98.5 | 89.7 | 100 | 67.1 | 83.3 | |
| Ever | 3.8 | 1.5 | 10.3 | 0 | 32.9 | 16.7 | |
| Years since baptism into Adventist church, mean (SD) | 60.18 (13.73) | 65.54 (6.37) | 59.44 (14.41) | 60.25 (14.89) | 44.80 (20.52) | 56.56 (14.75) | <0.001 |
| Lifetime Adventists, % | <0.001 | ||||||
| No | 28.8 | 5.1 | 26.7 | 22.7 | 63.7 | 41.8 | |
| Yes | 71.2 | 94.9 | 73.3 | 77.3 | 36.3 | 58.2 |
| Level | Converter | Multiverter | Reverter | p | |
|---|---|---|---|---|---|
| n | 6467 | 3384 | 983 | ||
| Age in years, mean (SD) | 77.46 (5.19) | 77.63 (5.26) | 77.56 (5.10) | 0.288 | |
| Gender, % | Male | 40.1 | 35.5 | 36.1 | <0.001 |
| Female | 59.9 | 64.5 | 63.9 | ||
| Race, % | Non-black | 90.6 | 91.1 | 93.5 | 0.011 |
| Black | 9.4 | 8.9 | 6.5 | ||
| BMI in kg/m2, % | Underweight (<18.5) | 2.6 | 1.9 | 0.8 | <0.001 |
| Normal (18.5–24.9) | 46.9 | 40.9 | 30.5 | ||
| Overweight (25–29.9) | 34.4 | 38.8 | 43.1 | ||
| Obese (≥30) | 16.1 | 18.3 | 25.5 | ||
| Education, % | High school or less | 23.5 | 23.1 | 25.4 | 0.001 |
| Some college | 37.8 | 40.6 | 41.8 | ||
| College or higher | 38.7 | 36.2 | 32.8 | ||
| Exercise, % | None | 68.6 | 70.1 | 75.7 | 0.002 |
| 1–20 min/week | 14.3 | 13.5 | 13 | ||
| 21–60 min/week | 10.1 | 9.8 | 7.6 | ||
| 61–150 min/week | 4.7 | 4.8 | 2.6 | ||
| ≥151 min/week | 2.3 | 1.9 | 1.1 | ||
| TV watching, % | None to <1 h/day | 24.7 | 20.9 | 14.4 | <0.001 |
| 1–2 h/day | 48.7 | 51 | 48.9 | ||
| ≥3 h/day | 26.6 | 28.2 | 36.6 | ||
| Sleep, % | <6 h/day | 26.9 | 29.2 | 29.6 | 0.001 |
| 7 h/day | 36.4 | 32.7 | 31.8 | ||
| ≥8 h/day | 36.7 | 38.1 | 38.6 | ||
| Alcohol, past year, % | No | 98.8 | 96.7 | 92 | <0.001 |
| Yes | 1.2 | 3.3 | 8 | ||
| Cigarette smoking, % | Never | 83.5 | 83.2 | 82.4 | 0.678 |
| Ever | 16.5 | 16.8 | 17.6 | ||
| Years since baptism into Adventist church, mean (SD) | 55.71 (14.94) | 57.73 (14.03) | 58.12 (15.56) | <0.001 | |
| Lifetime Adventists, % | No | 46.5 | 37.5 | 26.4 | <0.001 |
| Yes | 53.5 | 62.5 | 73.6 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, M.C.T.; Jaceldo-Siegl, K.; Orlich, M.; Fan, J.; Mashchak, A.; Fraser, G.E. A New Approach to Assess Lifetime Dietary Patterns Finds Lower Consumption of Animal Foods with Aging in a Longitudinal Analysis of a Health-Oriented Adventist Population. Nutrients 2017, 9, 1118. https://doi.org/10.3390/nu9101118
Martins MCT, Jaceldo-Siegl K, Orlich M, Fan J, Mashchak A, Fraser GE. A New Approach to Assess Lifetime Dietary Patterns Finds Lower Consumption of Animal Foods with Aging in a Longitudinal Analysis of a Health-Oriented Adventist Population. Nutrients. 2017; 9(10):1118. https://doi.org/10.3390/nu9101118
Chicago/Turabian StyleMartins, Marcia C. T., Karen Jaceldo-Siegl, Michael Orlich, Jing Fan, Andrew Mashchak, and Gary E. Fraser. 2017. "A New Approach to Assess Lifetime Dietary Patterns Finds Lower Consumption of Animal Foods with Aging in a Longitudinal Analysis of a Health-Oriented Adventist Population" Nutrients 9, no. 10: 1118. https://doi.org/10.3390/nu9101118
APA StyleMartins, M. C. T., Jaceldo-Siegl, K., Orlich, M., Fan, J., Mashchak, A., & Fraser, G. E. (2017). A New Approach to Assess Lifetime Dietary Patterns Finds Lower Consumption of Animal Foods with Aging in a Longitudinal Analysis of a Health-Oriented Adventist Population. Nutrients, 9(10), 1118. https://doi.org/10.3390/nu9101118





