1. Introduction
Maternal malnutrition in pregnancy can cause maternal endocrine and metabolic disorders, further damaging the growth of the placenta, thereby affecting the mother-to-fetus nutrient transport and weakening the growth and development of the fetus [
1]. Pregnancy malnutrition will not only delay the growth of the fetus, but also damage many of the fetus’ organs, as well as their organizational structure and function [
2]. In animal and human nutrition, the gastrointestinal tract is responsible for the introduction of nutrients into the body cells. However, the intestine is not only a major part of the digestion and absorption of nutrients, but is also a key part of the process of immune challenge, because it is constantly interacting with foreign substances [
3]. A large amount of experimental data has shown that nutritional factors, including energy, protein, fat and fatty acids, affect intestinal function [
4], and some studies have also shown that maternal malnutrition during pregnancy may result in poor development of the intestinal tract and intestinal function damage [
5,
6]. Due to development and growth constraints, compared with normal newborn piglets, intrauterine growth restriction (IUGR) piglets often manifest immune dysfunctions [
7,
8]. Neonates with IUGR show substantially higher rates of perinatal mortality and morbidity, partly due to impairment of cell proliferation, and nutrient digestion, absorption, and metabolism in the small intestine (SI) [
9,
10,
11]. At birth, the intestinal and immune systems of the newborn piglets are complete, but the functions of the small intestine and the immune system are immature, and are susceptible to infection and damage by harmful antigens. Interactions between the microbiota and the host are being considered as potential players in the early programming of gut functions. Increasing evidence indicates that the gut microbiota programs the functions and metabolism of host organs, including the GIT [
12,
13]. Thus, early nutrition plays a very important role in protecting the development of the small intestine and adjusting the immune response after the challenge of the antigen [
14]. However, the effect of maternal low-energy intake on intestinal immune function of offspring is not known. Many studies on early programming have been conducted in rodents and sheep, and a recent literature survey suggests that swine are a good translational model for humans [
15]. In this study, therefore, we investigated whether providing maternal low-energy intake would affect intestinal development and immune function in offspring.
2. Materials and Methods
The study protocol was reviewed and approved by the Care and Use Committee of Sichuan Agricultural University, and followed the current laws of animal protection (Ethic Approval Code: SCAUAC201408-3).
2.1. Animals and Diets
A total of thirty-six nine-parity sows (Landrace × Yorkshire), with initial body weights (BW) of 233.56 ± 2.77 kg and backfat thickness (BF) <14 mm at day 0 of gestation were used in this study. Sows were artificially inseminated with pooled semen obtained from a littermate boar (housed on the research farm) on the day of estrus, and then 12 and 24 h later. After artificial insemination, sows were randomly assigned to one of two diets: the control (CON) diet was designed as per the recommendations of the National Research Council (NRC; 2012) for gestating sows, and contained 3.40 MCal digestible energy (DE) per kilogram, and 7.3% crude protein, together with 5% crude fiber; while the low-energy diet (LED) contained 3.00 MCal DE/kg, with the same crude protein and crude fiber as CON. Sows were fed 2.2 kg/day during early pregnancy (days 1–30 of gestation), 2.4 kg/day mid-pregnancy (days 31–90), and 2.8 kg/day late pregnancy (days 91 to parturition). After farrowing, sows were fed a lactation diet in accordance with NRC 2012, and were offered the diet three times per day (i.e., at 0800, 1200, and 1500 h), starting at 2.0 kg/day, then gradually increasing by 1 kg/day until the sixth day, after which the sows could feed ad libitum. Throughout the experiment, all animals had free access to water. The ambient temperature for sows was maintained at 20–25 °C. Heating light and pads were provided for suckling piglets, and the temperature was maintained at 26–32 °C, which was gradually decreased with increase in age. All sows were housed in individual feed stalls during gestation and, during lactation, were housed in farrowing pens.
At birth, six piglets (one piglet from each of six litters), each having approximately the same body weight (±10%) as the mean body weight of the litter, were selected from each group and slaughtered before suckling. At the day of weaning (day 28 post-birth), twelve healthy male piglets were selected from each of the two treatments. Six piglets per treatment were intraperitoneally injected with LPS solution to administer LPS at 50 µg/kg body weight, in order to make a model of immunological challenge, and another six piglets in each treatment were intraperitoneally injected with an equivalent amount of sterile saline. Feed was removed 4 h before the injection. The rectal temperature of each piglet was measured every 2 h. LPS (
Escherichia coli L2880, Sigma-Aldrich, Los Angeles, CA, USA) was dissolved in sterile saline (9 g/L) to make the LPS solution (500 mg LPS per liter of saline). The dosage of the LPS injection and the time to sacrifice piglets were described previously [
16].
2.2. Sample Collection
The pigs were anesthetized with an intravenous injection of pentobarbital sodium (50 mg/kg BW) before blood sample collection, and then slaughtered. From each group, six newborn piglets without eating colostrum (from six sows and close to the average body weight in each treatment group, three males and three females) were slaughtered, as well as 12 weaning piglets deprived of breast nursing from sows for 4 h before the LPS injection (12 males from 12 different sows, and close to the average body weight in each treatment group). On the day of birth and at 0 and 4 h after injection of LPS or normal saline, blood samples were collected from the anterior vena cava and placed into heparinized tubes, centrifuged (2550×
g, 4 °C, 10 min) and stored at −20 °C until analysis. Immediately after collection of blood samples at 4 h, piglets were slaughtered by jugular puncture, and the entire intestine was removed as quickly as possible. The small intestine (SI) was quickly measured for weight and length. The middle portion (approximately 4 cm) of each segment of the small intestine (duodenum, jejunum, and ileum) was rapidly obtained and placed in liquid nitrogen, and then stored at −80 °C for subsequent analysis, as previously described [
17]. In addition, about 1 cm each of integral duodenum, jejunum, and ileum tissue were cut out, fixed with 4% paraformaldehyde for intestinal slice production and morphology observation. The intestinal mucosa of the remaining duodenum, jejunum, and ileum segment was obtained carefully with a glass slide scraping, frozen in liquid nitrogen as soon as possible, and then stored at −80 °C for future analysis of intestinal enzyme activity. Colonic and caecal content samples were collected quickly, as previously described [
18], and then stored at −80 °C for future analysis of short-chain fatty acids and intestinal microflora.
2.3. Intestinal Morphology Analysis
Intestinal tissue samples were fixed with 4% formaldehyde, dehydrated stepwise with ethanol (from low concentration to high concentration, up to 100%), and then embedded in transparent paraffin and sliced [
17]. All tissue sections (5 μm) were stained with hematoxylin and eosin (HE) and mounted with gum. The slice images were captured at 100× magnification using an Olympus BX51 microscope equipped with a DP70 digital camera (Olympus, Tokyo, Japan). Crypt depth (μm) and villus length (μm) were measured with Image-Pro Plus 6.0 software (Media Cybernetics, Bethesda, MD, USA). At least eight pairs of villus and crypts were observed in each slice, and the average was the final value.
2.4. Analysis of Intestinal Enzyme Activity
Intestinal enzyme activity was measured as previously described [
17]. After thawing, 0.2–0.5 g of mucosa samples were homogenized according to the weight of intestine mucosa (g): volume of physiological saline pre-cooled on ice (mL) = 1:9, centrifuged at 2500 g/min for 10 min, the supernatant was used to measure enzymatic activity. The enzyme activity was measured strictly in accordance with the respective instructions of the kit (Jiancheng Bioengineering Ltd., Nanjing, China).
2.5. RT-PCR Analysis
Gene expression levels were detected by using a reverse transcription polymerase chain reaction (RT-PCR) similar to the procedure previously described [
19]. Briefly, total RNA was extracted from frozen ileum samples with TRIzol reagent (Invitrogen, Carlsbad, CA, USA), synthesis of cDNA from isolated total RNA was carried out with Prime-Script™ RT reagent kit (Takara, Dalian, China) according to the instructions of the manufacturer. Real-time PCR assays were performed on complementary DNA samples in 384-well optical plates on a 7900HT ABI Prism Sequence Detection System (Applied Biosystems, Foster City, CA, USA) using the SYBR green system (Takara). Primers for individual genes were designed using Primer Express 3.0 (Applied Biosystems, Foster City, CA, USA). The reaction mixture (10 μL) contained 5 μL of fresh SYBR
®Premix Ex Taq II (Tli RNaseH Plus) and 0.2 μL ROX Reference Dye II (50×), 0.8 μL of the primers, 1 μL of RT products and 3 μL diethylpyrocarbonate-treated water. The PCR protocol was used as follows: 1 cycle (95 °C for 30 s), 40 cycles (95 °C for 5 s, 60 °C for 31 s) and 1 cycle (95 °C for 15 s, 60 °C for 1 min and 95 °C for 15 s). The relative mRNA abundance of the analyzed genes was calculated using the 2
−ΔΔCt method, as described previously by Mou et al. [
19]. The most stable housekeeping genes (β-actin and GADPH) were chosen for normalization. Finally, the mRNA level of each target gene for the CON group was set to 1. The primer sequence is shown in
Table 1.
2.6. Plasma TNF-α, IL-1β, IL-10, IgA and GLP-2 Analysis
Plasma TNF-α, IL-1β, IL-10, IgA and GLP-2 concentrations were measured by using ELISA kits suitable for pigs (Nanjing JianCheng Bioengineering Institute, Inc., Nanjing, China) according to the manufacturer’s protocol.
2.7. Plasma IgG and IgM Analysis
Routine analysis of blood samples in heparinized-plasma venipuncture vials were performed by automatic blood analyzer (Alfa Basic 16p, Boule Medical AB, Stockholm, Sweden).
2.8. Short Chain Fatty Acid (SCFA) Analysis
SCFAs, including acetate, propionate and butyrate in digesta and mucosa samples, were analyzed with a modification of the previous method [
18]. Briefly, 2 g of digesta or mucosa samples were weighed into a 10-mL centrifuge tube and added to 5 mL of deionized water. After the tube was capped, the contents were vortex mixed for 30 s, left to stand for 30 min at 4 °C and then centrifuged (1000×
g, 4 °C) for 10 min. The supernatant (1.2 mL) was removed by aspiration into another 5-mL centrifuge tube, added to 0.24 mL of 25% metaphosphate, vortex mixed for 30 s, and then left to stand for 30 min at 4 °C. Next, the contents were centrifuged (1000×
g, 4 °C) for 10 min, and then 1.2 mL of the supernatant was removed by aspiration, added to 23.3 μL of 210 mmol/L cortonic acid, and vortex mixed for 30 s. Then, 0.3 mL of the mixed solution was removed and placed into another 2-mL tube, added to 0.9 mL Carbinol, and vortex mixed for 30 s for subsequent gas chromatography analysis. The samples were analyzed by CP-3800 gas chromatography (Varian, Inc., Artaud, CA, USA) equipped with a micro-injector (10 μL), a flame ionization detector and a capillary chromatographic column (CP-FFAP, 25 m × 0.32 mm × 0.3 μm). The injector temperature was 220 °C, the detector temperature was 250 °C, hydrogen flux was 40 mL/min, and air flux was 450 mL/min. The temperature program was as follows: 100 °C, hold 1 min, increase to 190 °C at 20 °C/min, increase to 190 °C, hold 3 min. Peaks were identified by comparing their retention times with individual reference standard fatty acids.
2.9. PCR Analysis of Bacteria
Quantitative RT-PCR of total bacteria was performed with SYBR
® Green PCR reagents (TaKaRa, Kyoto, Japan), whereas quantitative RT-PCR for Bifidobacterium, Lactobacillus, and Escherichia coli were performed with Taq primers and fluorescent oligonucleotide probes that had been commercially synthesized (Life Technologies Ltd., Beijing, China), as listed in
Table 2 [
20]. The PCR reaction of total bacteria was set up in a total volume of 15 μL and contained 7.5 μL of SYBR Premix Ex TaqII, 0.6 μL of each primer, 0.6 μL of genomic DNA, and 5.7 μL of sterile deionized water; whereas the PCR reactions for
Bifidobacterium,
Lactobacillus, and
Escherichia coli were set up in a total volume of 10 μL, and contained 0.15 μL of probe, 0.5 μL of each primer, 0.5 μL of probe enhancer solution, 4 μL of Real Master Mix, 0.5 μL of genomic DNA, and 3.85 μL of sterile deionized water. Amplification was performed using a CFX 96 System (Bio-Rad Laboratories, Inc., Hercules, CA, USA). As for total bacteria, the amplification program consisted of one cycle of 95 °C for 30 s and then 40 cycles of 95 °C for 5 s, 60 °C for 34 s, and one cycle of 95 °C for 15 s, 60 °C for 1 min, 95 °C for 15 s. For
Bifidobacterium,
Lactobacillus, and
Escherichia coli, the amplification program consisted of one cycle of 50 °C for 2 min, 95 °C for 10 min and then 40 cycles of 95 °C for 15 s and 55.6–59.5 °C for 1 min. Melting curve analysis and size determination of PCR fragment on agarose gels verified amplification of the target fragments. Standard curves were generated as previously described [
20].
2.10. Statistical Analyses
SPSS 21.0 (IBM SPSS Company, Chicago, IL, USA) was used for all statistical analyses. Newborn piglets’ results were tested for variance using Levene’s test; a two-tailed t-test was used to compare the differences between the dietary groups. CON and the LED results after LPS challenge were performed by two-way ANOVA; data was tested for normality and homogeneity of variances (Shapiroe–Wilk and Levene tests, respectively) and, when necessary, data was normalized (arcsine, square root, or logarithm normalized) to achieve ANOVA assumptions. Additionally, Duncan’s multiple range tests were conducted to determine the difference among the treatments. All values are means with their standard errors (SE), and differences between treatments were considered significant when p < 0.05.
4. Discussion
In the embryonic period, the gastrointestinal system is one of the earliest systems of polarization, and the last few weeks before the birth of the fetus are a very important period for intestinal potential and digestive function maturity [
21]. Some studies have shown that maternal nutrition affects the development and function of the gut of the offspring [
5,
6,
16]. In this study, maternal LED intake during gestation significantly decreased piglet BW and SI indices, such as SI length and the SI weight-to-length ratio in newborn piglets. Piglets with lower birth weight may suffer from IUGR, which impairs growth, muscle accretion, duodenal mucosa morphology and carcass traits [
22]. Previous studies have shown that IUGR piglets’ SI weight-to-length ratio is only 61–76% that of normal pigs [
23,
24], and that they have a lower SI mass [
7] and SI length [
25] at birth. Maternal pregnancy intake of nutrients, in addition to meeting their own needs, must also provide the fetus with nutrients to meet its growth and development [
26]. Studies have shown that, compared with normal-weight piglets, IUGR reduced the growth rate of piglets one month after birth by about 15–30% [
21]. The results of our study showed that maternal LED diets significantly decreased weaning weight, SI weight and length, SI unit length and SI weight to length ratio in weaned piglets. Studies in rats also showed a significant reduction in body weight, body length and intestinal length in low protein-to-energy rats compared to the control group, and decreased bowel weight and DNA in malnourished rats [
27].
Integrity of the intestinal structure is required for the maintenance of intestinal nutrition [
28]. Fetuses receive constant nutrients from mothers via the placenta, whereas pups must take up nutrients from food via the small intestine post-birth; therefore, small intestine development during the gestation period plays an important role. Previous studies have shown that low birth weight piglets have significantly decreased duodenal VCR, jejunum villus height and VCR compared with normal-weight piglets [
29,
30]. Some studies also showed that the small intestine surface area of IUGR piglets is smaller than that of normal pigs, mainly due to the lower average number of villi per unit area and low villus height. In our study, LED in sows during pregnancy significantly decreased the VCR of duodenum and ileum in newborn piglets, which was consistent with previous reports. Similarly, the results of this test found that sows fed a low-energy diet during pregnancy significantly decreased ileum VCR of weaning piglets. To our knowledge, the VCR represents the capacity of the small intestine for nutrient digestion and absorption [
31]. The proliferation and differentiation of intestinal epithelial cells is accompanied by their migration along the crypt-villus axis [
32]. Thus, a lower VCR may indicate less proliferation and differentiation of intestinal epithelial cells. It is logical to consider that maternal LED may suppress digestion and absorption function by inhibiting the proliferation and differentiation of intestinal epithelial cells in offspring. Consistently with the intestinal morphology results, we found jejunum mucosal lactase activity was significantly decreased in newborn piglets, which indicated digestion ability for lactose. Meanwhile, we also found jejunum mucosal sucrase and maltase activity were significantly decreased in weaning piglets. In terms of digestive capacity, intestinal enzymes are partially responsible for food processing and hydrolyzing macromolecule nutrients to small molecules for intestinal absorption [
15]. During intrauterine life, glucose and amino acids are the main nutrients that provide energy for intrauterine growth; however, piglets utilize the abundant lactose in milk to provide most of their energy during neonatal life [
33,
34]. Based on this information, it may be concluded that intestinal digestion and absorption ability could be significantly decreased by a maternal low-energy diet during gestation.
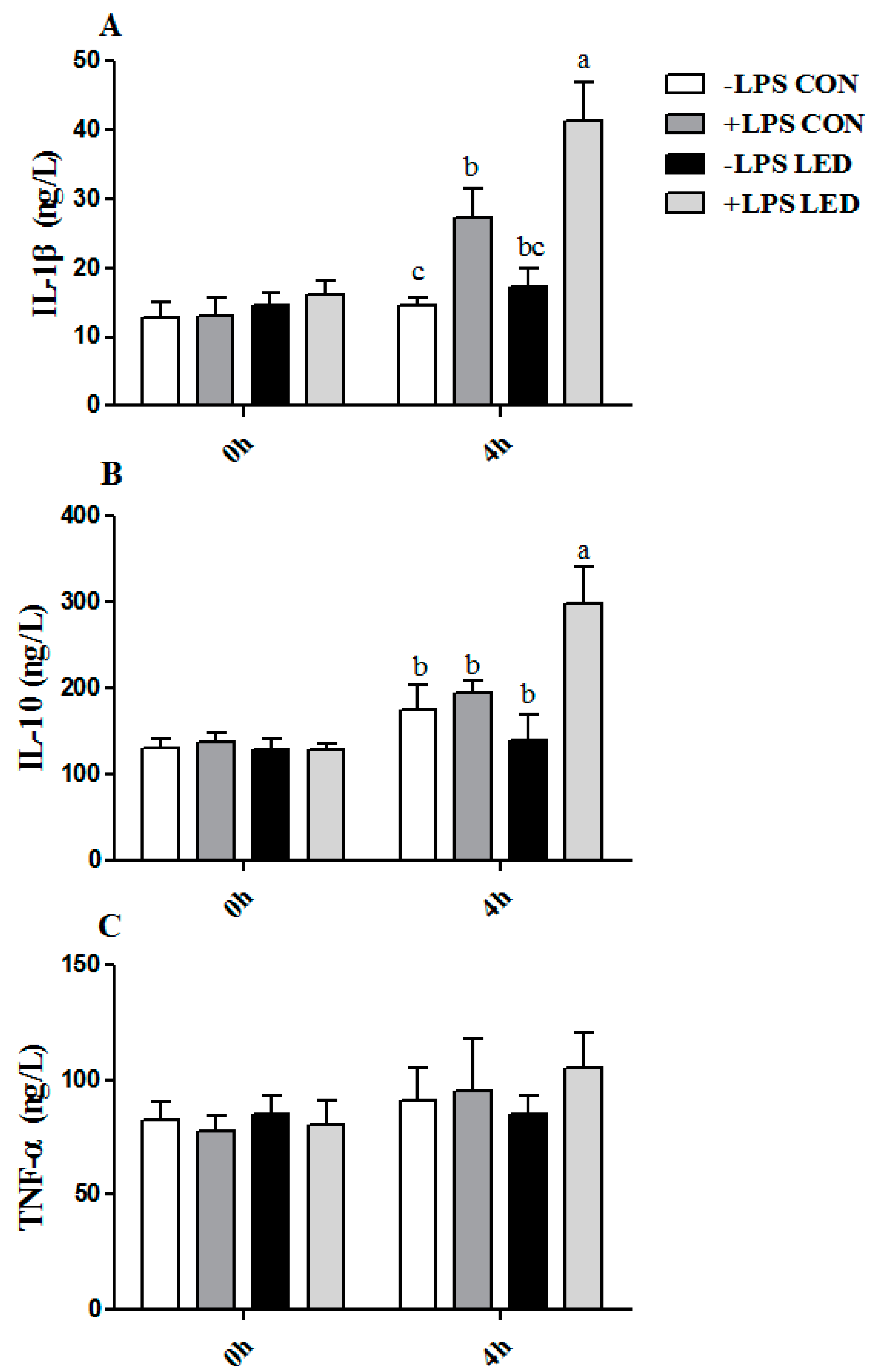
Nutritional and other molecular events in fetal and neonatal life lay the foundation for future health, which includes effects on immune system function. The second aim of this study was to determine the potential effect of maternal LED on the immune responses of LPS-challenged piglets. The increased body temperature, plasma TNF-α, and IL-1β concentrations indicated the successful establishment of the immune model following LPS challenge. It has been demonstrated that the TLR-4–Myd88–NF-kB signal pathway is involved in inflammation [
35]. Moreover, the gastrointestinal tract is the largest immune organ in the body and, as such, is the location for the majority of lymphocytes and immune effector cells with pattern recognition receptors [
36], which sense luminal antigens and mediate the inflammatory response [
37]. In the present study, ileal mRNA abundance of pro-inflammatory cytokines, including
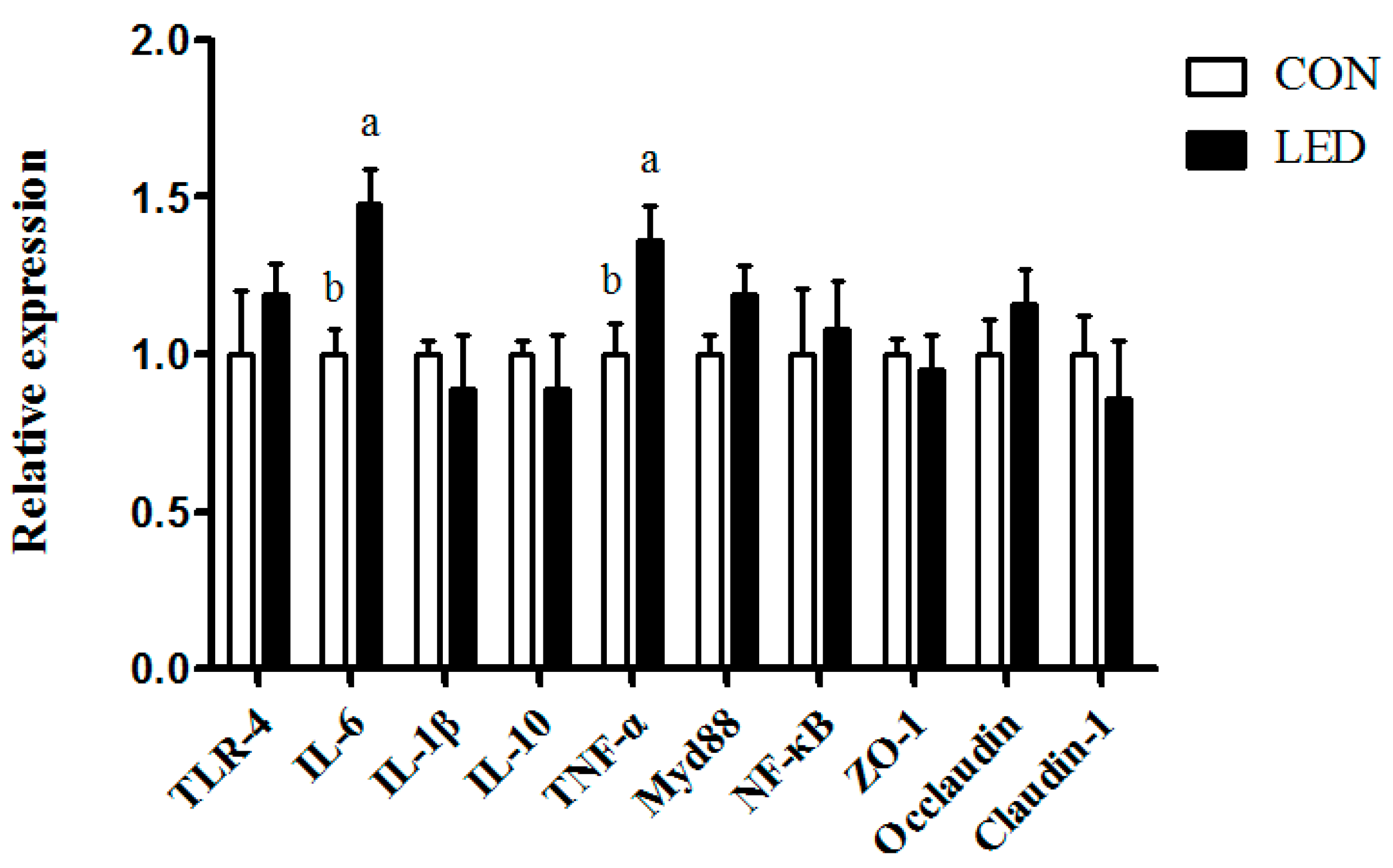
TNF-α and
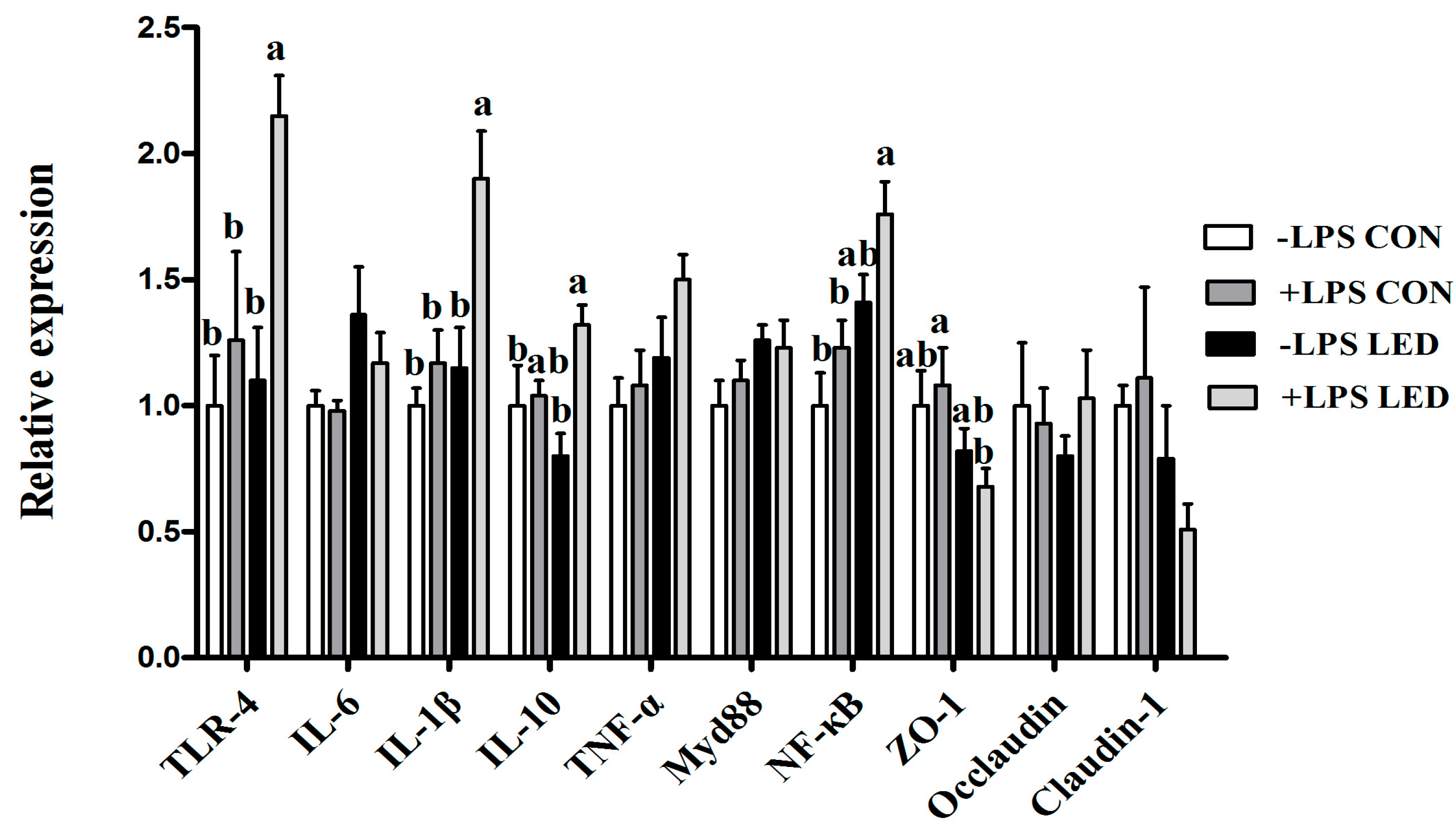
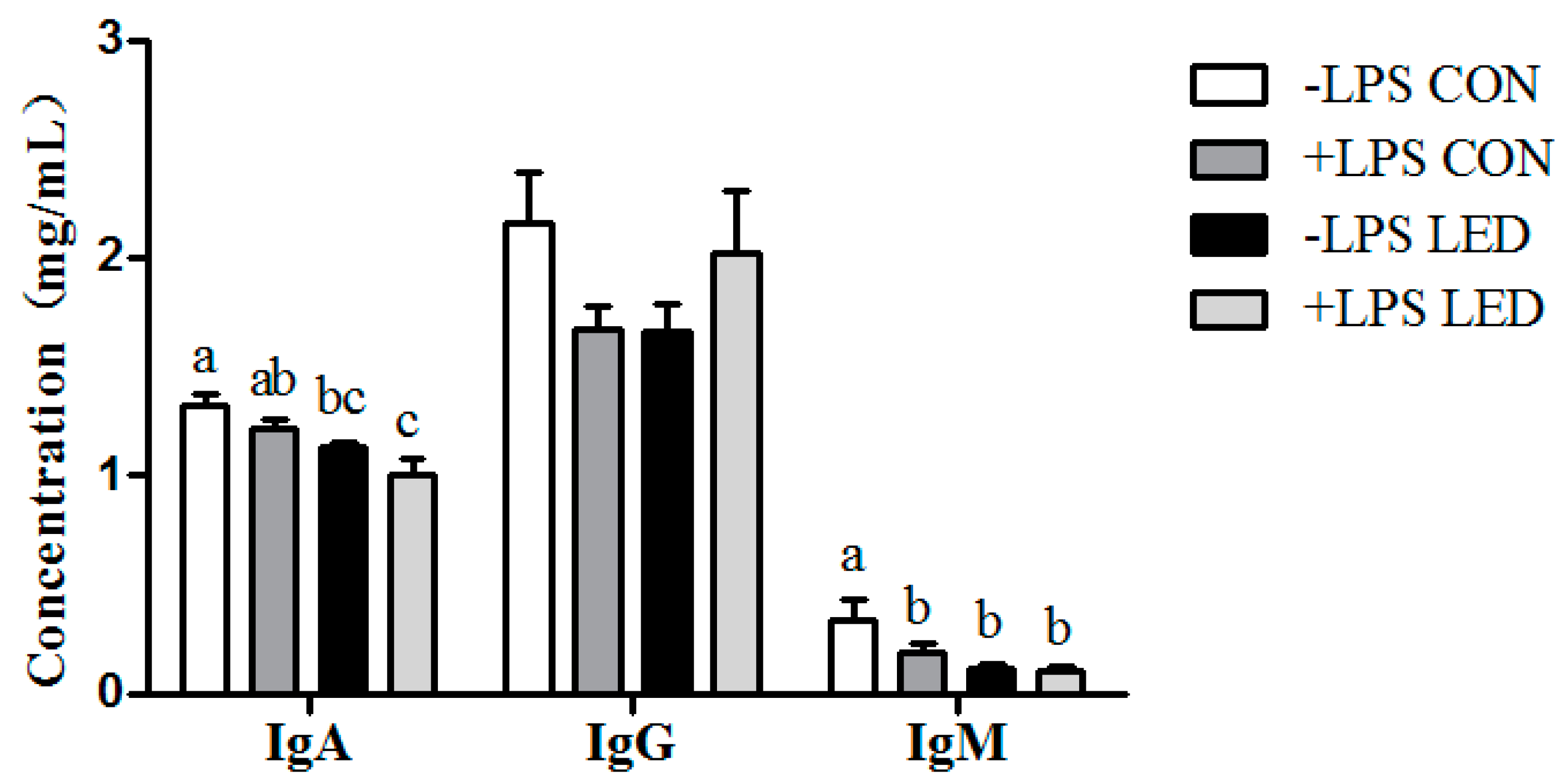
IL-6, were significantly up-regulated by maternal LED in newborn piglets, and plasma IgA and IgM were significantly reduced by the maternal dietary treatment in weaning piglets. Meanwhile, it was observed that LPS challenge resulted in increased plasma IL-1β concentration, which was consistent with gene expression results. Meanwhile, LPS challenge and low-energy diets increased
TLR-4 and
NF-kB gene expression in weaning pigs’ ileum, which corresponded positively with inflammatory response. Previous studies also found maternal restraint stress during gestation in pigs enhanced the magnitude of the TNF-α and IL-6 responses to LPS in the offspring [
38]. Therefore, maternal LED increased systemic inflammation, reduced the amount of immunoglobulin in offspring and up-regulated the expression of inflammation-related genes in offspring, resulting in increased sensitivity to LPS.
The intestinal epithelial barrier includes the presence of a single epithelial cell (EC) layer, tight junctions (TJs) between ECs, and the presence of associated immune cells [
39]. ZO-1, Occludin and Claudin-1 are the most important tight junction proteins, and up-regulation of expression of these proteins means a reduction in intestinal inflammatory disease risk [
40]. On the contrary, down-regulated expression of these proteins will lead to increased intestinal permeability and harmful bacteria in the intestine, whereupon LPS and other harmful substances will pass easily through the intestinal epithelium and into the submucosa, causing intestinal inflammation, damaging the health of the body [
41]. In addition, earlier studies have reported that the production of excessive proinflammatory cytokines can activate the apoptotic pathway, induce apoptosis, and reduce the tight junction barrier of the intestinal epithelium [
42]. In the present study, maternal LED significantly down-regulated the expression of the
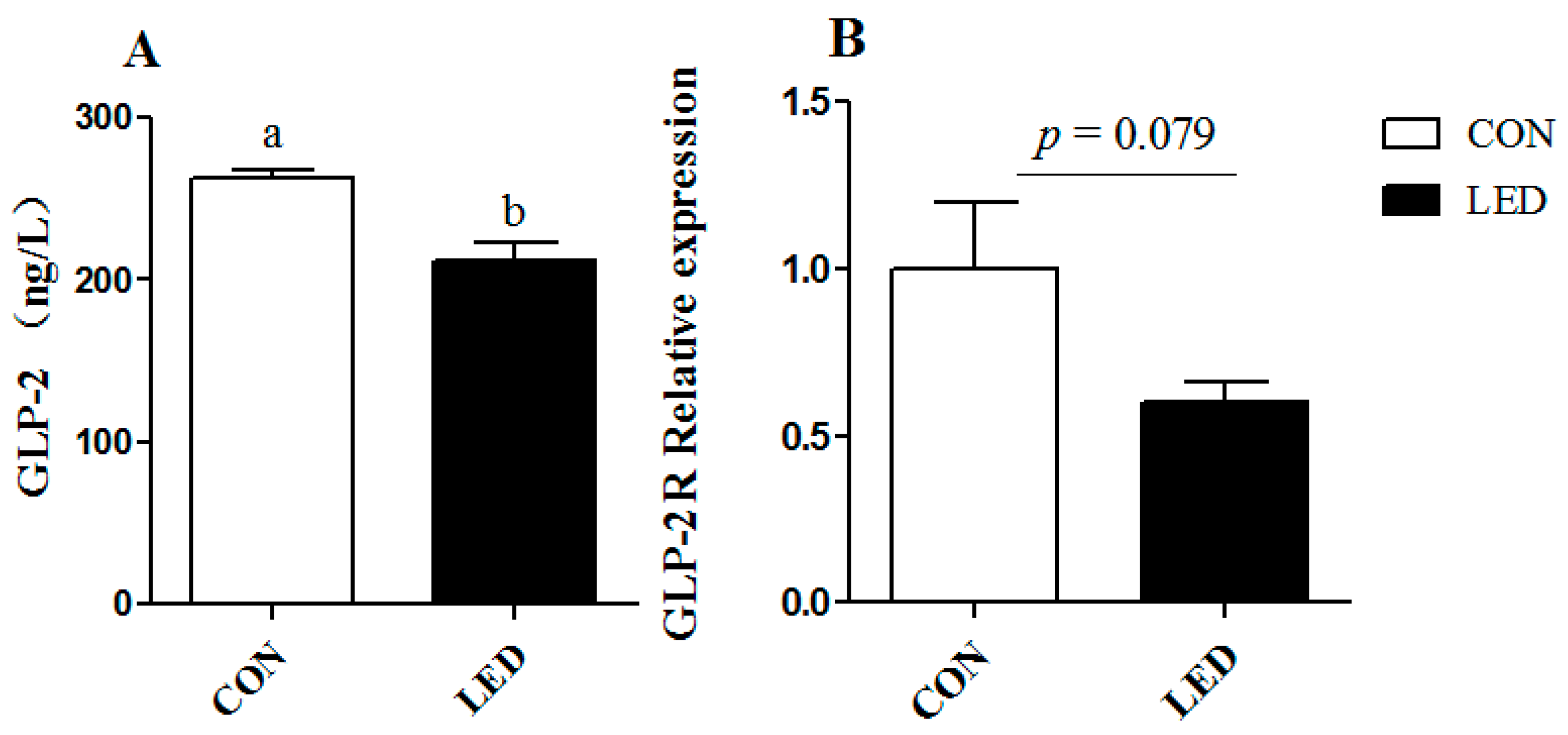
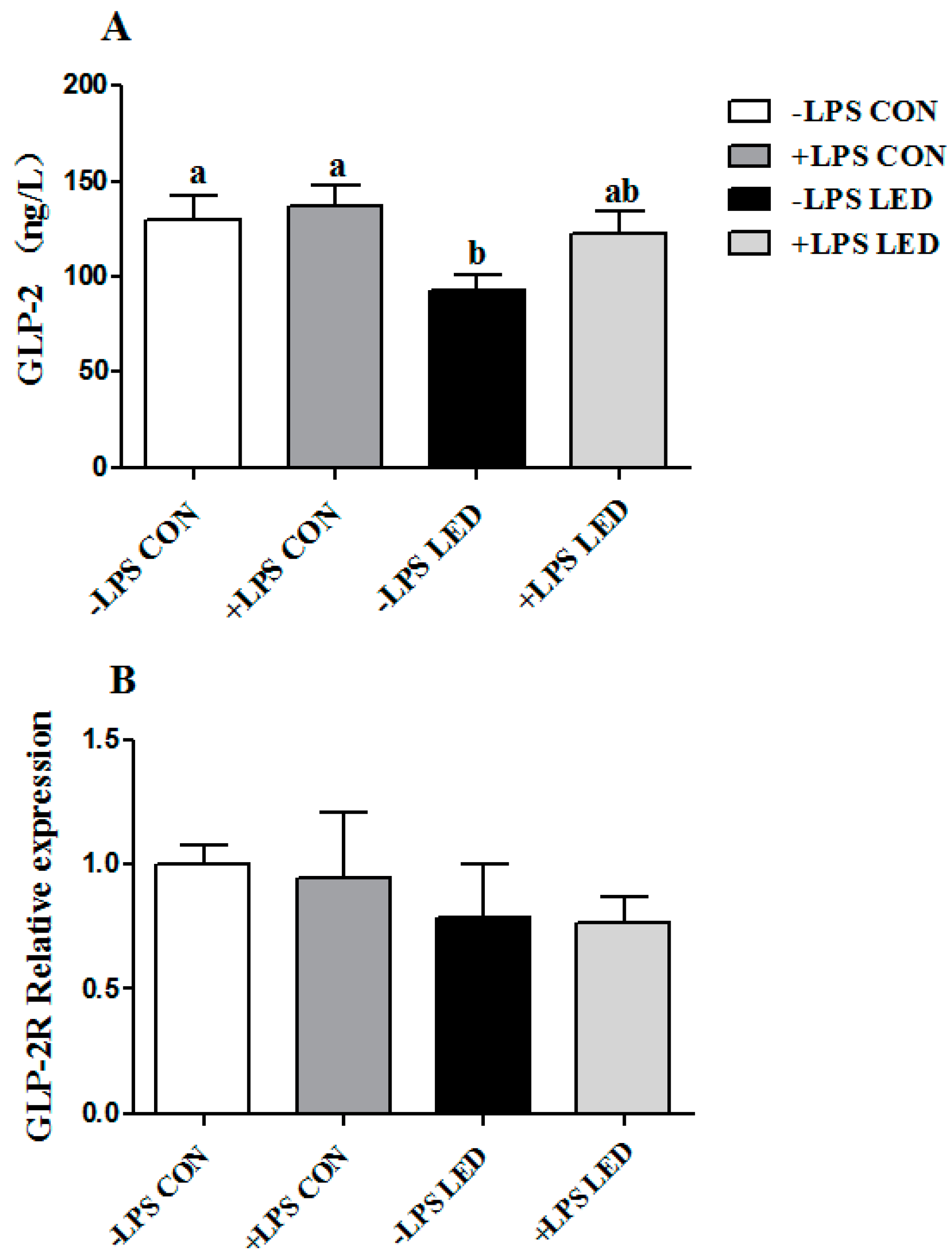
ZO-1 gene in weaning piglets. At the same time, maternal LED significantly reduced the GLP-2 levels in newborn and weaning piglet plasma. GLP-2 increases the rate of crypt cell proliferation and villus elongation, and reduces apoptosis, leading to improved barrier function [
43]. Meanwhile, endogenous GLP-2 production increased to improve the intestinal barrier function. SCFA (acetate, propionic acid and butyric acid) are the major end products of bacterial metabolism in the large intestine [
44]. In the present study, LPS challenge resulted in the suppression of microbial fermentation, which was evidenced by the acetate and propionic acid concentrations in the caecal content, which were decreased by LPS challenge. A novel study in a mouse model demonstrated that increased production of acetate inhibits translocation of the E. coli toxin from the gut lumen to the blood, which improves intestinal defense mediated by epithelial cells, and thereby protects the host against lethal infection [
45]. The decreased acetate content might, in part, account for the serious congestion in the intestine and destruction of the intestinal epithelial barrier. A previous study has shown that IUGR alters the GIT barrier and affects the gut immune response, as well as whole-body metabolism [
25]. These results suggested sows fed low-energy diets during pregnancy may cause adverse intestinal epithelial barrier function, leading to increased intestinal permeability and greater susceptibility to inflammation.