Anti-Inflammatory Thioredoxin Family Proteins for Medicare, Healthcare and Aging Care
Abstract
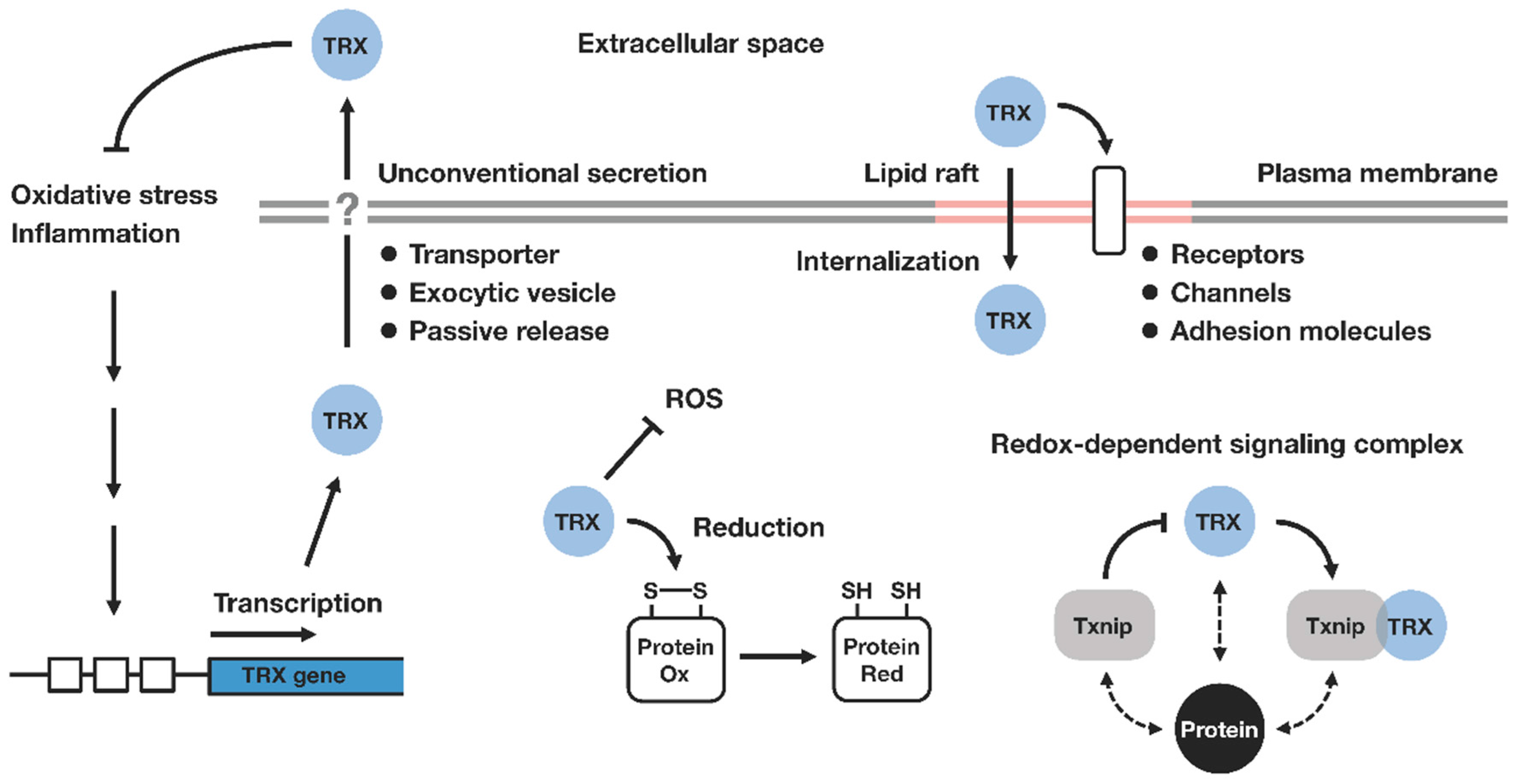
1. Introduction
2. Background of TRX Research Associated with Human Diseases
3. Mucosa and Skin Inflammation
4. Oral Delivery of TRX
5. TRX-Inducing Principles
6. Thioredoxin Interacting Protein (Txnip/TBP-2/VDUP1)
7. TRX/Txnip; Redoxisome, a Redox-Related Signal Complex
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Holmgren, A.; Lu, J. Thioredoxin and thioredoxin reductase: Current research with special reference to human disease. Biochem. Biophys. Res. Commun. 2010, 396, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Lillig, C.H.; Holmgren, A. Thioredoxin and related molecules—From biology to health and disease. Antioxid. Redox Signal. 2007, 9, 25–47. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, D.F.; Abderrazak, A.; El Hadri, K.; Simmet, T.; Rouis, M. The thioredoxin system as a therapeutic target in human health and disease. Antioxid. Redox Signal. 2013, 19, 1266–1303. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.G.; Chae, H.Z.; Kim, K. Peroxiredoxins: A historical overview and speculative preview of novel mechanisms and emerging concepts in cell signaling. Free Radic. Biol. Med. 2005, 38, 1543–1552. [Google Scholar] [CrossRef] [PubMed]
- Laurent, T.C.; Moore, E.C.; Reichard, P. Enzymatic Synthesis of Deoxyribonucleotides. IV. Isolation and Characterization of Thioredoxin, the Hydrogen Donor from Escherichia Coli B. J. Biol. Chem. 1964, 239, 3436–3444. [Google Scholar] [PubMed]
- Ishizaka, K.; Ishizaka, T.; Hornbrook, M.M. Physicochemical properties of reaginic antibody. V. Correlation of reaginic activity wth gamma-E-globulin antibody. J. Immunol. 1966, 97, 840–853. [Google Scholar] [PubMed]
- Johansson, S.G.; Bennich, H. Immunological studies of an atypical (myeloma) immunoglobulin. Immunology 1967, 13, 381–394. [Google Scholar] [PubMed]
- Tagaya, Y.; Maeda, Y.; Mitsui, A.; Kondo, N.; Matsui, H.; Hamuro, J.; Brown, N.; Arai, K.; Yokota, T.; Wakasugi, H.; et al. ATL-derived factor (ADF), an IL-2 receptor/Tac inducer homologous to thioredoxin; possible involvement of dithiol-reduction in the IL-2 receptor induction. EMBO J. 1989, 8, 757–764. [Google Scholar] [PubMed]
- Yodoi, J.; Takatsuki, K.; Masuda, T. Letter: Two cases of T-cell chronic lymphocytic leukemia in Japan. N. Engl. J. Med. 1974, 290, 572–573. [Google Scholar] [PubMed]
- Uchiyama, T.; Yodoi, J.; Sagawa, K.; Takatsuki, K.; Uchino, H. Adult T-cell leukemia: Clinical and hematologic features of 16 cases. Blood 1977, 50, 481–492. [Google Scholar] [CrossRef]
- Masutani, H.; Hirota, K.; Sasada, T.; Ueda-Taniguchi, Y.; Taniguchi, Y.; Sono, H.; Yodoi, J. Transactivation of an inducible anti-oxidative stress protein, human thioredoxin by HTLV-I Tax. Immunol. Lett. 1996, 54, 67–71. [Google Scholar] [CrossRef]
- Kondo, N.; Nakamura, H.; Masutani, H.; Yodoi, J. Redox regulation of human thioredoxin network. Antioxid. Redox Signal. 2006, 8, 1881–1890. [Google Scholar] [CrossRef] [PubMed]
- Matthias, L.J.; Yam, P.T.; Jiang, X.M.; Vandegraaff, N.; Li, P.; Poumbourios, P.; Donoghue, N.; Hogg, P.J. Disulfide exchange in domain 2 of CD4 is required for entry of HIV-1. Nat. Immunol. 2002, 3, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Schwertassek, U.; Balmer, Y.; Gutscher, M.; Weingarten, L.; Preuss, M.; Engelhard, J.; Winkler, M.; Dick, T.P. Selective redox regulation of cytokine receptor signaling by extracellular thioredoxin-1. EMBO J. 2007, 26, 3086–3097. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.Z.; Sukumar, P.; Zeng, F.; Li, J.; Jairaman, A.; English, A.; Naylor, J.; Ciurtin, C.; Majeed, Y.; Milligan, C.J.; et al. TRPC channel activation by extracellular thioredoxin. Nature 2008, 451, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Kondo, N.; Ishii, Y.; Kwon, Y.W.; Tanito, M.; Sakakura-Nishiyama, J.; Mochizuki, M.; Maeda, M.; Suzuki, S.; Kojima, M.; Kim, Y.C.; et al. Lipid raft-mediated uptake of cysteine-modified thioredoxin-1: Apoptosis enhancement by inhibiting the endogenous thioredoxin-1. Antioxid. Redox Signal. 2007, 9, 1439–1448. [Google Scholar] [CrossRef] [PubMed]
- Manabe, Y.; Takagi, M.; Nakamura-Yamada, M.; Goto-Inoue, N.; Taoka, M.; Isobe, T.; Fujii, N.L. Redox proteins are constitutively secreted by skeletal muscle. J. Physiol. Sci. 2014, 64, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Rubartelli, A.; Bajetto, A.; Allavena, G.; Wollman, E.; Sitia, R. Secretion of thioredoxin by normal and neoplastic cells through a leaderless secretory pathway. J. Biol. Chem. 1992, 267, 24161–24164. [Google Scholar] [PubMed]
- Keller, M.; Ruegg, A.; Werner, S.; Beer, H.D. Active caspase-1 is a regulator of unconventional protein secretion. Cell 2008, 132, 818–831. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; De Rosa, S.; Roederer, M.; Anderson, M.T.; Dubs, J.G.; Yodoi, J.; Holmgren, A.; Herzenberg, L.A.; Herzenberg, L.A. Elevation of plasma thioredoxin levels in HIV-infected individuals. Int. Immunol. 1996, 8, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; De Rosa, S.C.; Yodoi, J.; Holmgren, A.; Ghezzi, P.; Herzenberg, L.A.; Herzenberg, L.A. Chronic elevation of plasma thioredoxin: Inhibition of chemotaxis and curtailment of life expectancy in AIDS. Proc. Natl. Acad. Sci. USA 2001, 98, 2688–2693. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, C.; Shioji, K.; Nakamura, H.; Nakayama, Y.; Yodoi, J.; Sasayama, S. Serum thioredoxin (TRX) levels in patients with heart failure. Jpn. Circ. J. 2001, 65, 491–494. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, S.; Sakamoto, T.; Soejima, H.; Shimomura, H.; Kajiwara, I.; Kojima, S.; Hokamaki, J.; Sugiyama, S.; Yoshimura, M.; Ozaki, Y.; et al. Plasma thioredoxin levels and platelet aggregability in patients with acute myocardial infarction. Am. Heart J. 2003, 146, 465–471. [Google Scholar] [CrossRef]
- Soejima, H.; Suefuji, H.; Miyamoto, S.; Kajiwaram, I.; Kojima, S.; Hokamaki, J.; Sakamoto, T.; Yoshimura, M.; Nakamura, H.; Yodoi, J.; et al. Increased plasma thioredoxin in patients with acute myocardial infarction. Clin. Cardiol. 2003, 26, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, S.; Nishio, A.; Nakamura, H.; Kido, M.; Kiriya, K.; Asada, M.; Tamaki, H.; Fukui, T.; Kawasaki, K.; Watanabe, N.; et al. Clinical significance of serum thioredoxin 1 levels in patients with acute pancreatitis. Pancreas 2006, 32, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Callister, M.E.; Burke-Gaffney, A.; Quinlan, G.J.; Nicholson, A.G.; Florio, R.; Nakamura, H.; Yodoi, J.; Evans, T.W. Extracellular thioredoxin levels are increased in patients with acute lung injury. Thorax 2006, 61, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Nakamura, H.; Adachi, T.; Sannohe, S.; Oyamada, H.; Kayaba, H.; Yodoi, J.; Chihara, J. Elevated serum levels of thioredoxin in patients with acute exacerbation of asthma. Immunol. Lett. 2003, 86, 199–205. [Google Scholar] [CrossRef]
- Madrigal-Matute, J.; Fernandez-Garcia, C.E.; Blanco-Colio, L.M.; Burillo, E.; Fortuno, A.; Martinez-Pinna, R.; Llamas-Granda, P.; Beloqui, O.; Egido, J.; Zalba, G.; et al. Thioredoxin-1/peroxiredoxin-1 as sensors of oxidative stress mediated by NADPH oxidase activity in atherosclerosis. Free Radic. Biol. Med. 2015, 86, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Abdiu, A.; Nakamura, H.; Sahaf, B.; Yodoi, J.; Holmgren, A.; Rosen, A. Thioredoxin blood level increases after severe burn injury. Antioxid. Redox Signal. 2000, 2, 707–716. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Vaage, J.; Valen, G.; Padilla, C.A.; Bjornstedt, M.; Holmgren, A. Measurements of plasma glutaredoxin and thioredoxin in healthy volunteers and during open-heart surgery. Free Radic. Biol. Med. 1998, 24, 1176–1186. [Google Scholar] [CrossRef]
- Jekell, A.; Hossain, A.; Alehagen, U.; Dahlstrom, U.; Rosen, A. Elevated circulating levels of thioredoxin and stress in chronic heart failure. Eur. J. Heart Fail. 2004, 6, 883–890. [Google Scholar] [CrossRef] [PubMed]
- Kakisaka, Y.; Nakashima, T.; Sumida, Y.; Yoh, T.; Nakamura, H.; Yodoi, J.; Senmaru, H. Elevation of serum thioredoxin levels in patients with type 2 diabetes. Horm. Metab. Res. 2002, 34, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, S.; Kawano, H.; Hokamaki, J.; Soejima, H.; Kojima, S.; Kudoh, T.; Nagayoshi, Y.; Sugiyama, S.; Sakamoto, T.; Yoshimura, M.; et al. Increased plasma levels of thioredoxin in patients with glucose intolerance. Intern. Med. 2005, 44, 1127–1132. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Cheng, Z.J.; Liu, Y.; Yan, Z.L.; Wang, K.; Wu, D.; Wan, X.Y.; Xia, Y.; Lau, W.Y.; Wu, M.C.; et al. Serum thioredoxin is a diagnostic marker for hepatocellular carcinoma. Oncotarget 2015, 6, 9551–9563. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, K.; Noda, N.; Okada, S.; Hagiwara, Y.; Miyata, M.; Sakurabayashi, I.; Yamaguchi, N.; Sugimura, T.; Terada, M.; Wakasugi, H. Elevated serum level of thioredoxin in patients with hepatocellular carcinoma. Biotherapy 1998, 11, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Sumida, Y.; Nakashima, T.; Yoh, T.; Nakajima, Y.; Ishikawa, H.; Mitsuyoshi, H.; Sakamoto, Y.; Okanoue, T.; Kashima, K.; Nakamura, H.; et al. Serum thioredoxin levels as an indicator of oxidative stress in patients with hepatitis C virus infection. J. Hepatol. 2000, 33, 616–622. [Google Scholar] [CrossRef]
- Tamaki, H.; Nakamura, H.; Nishio, A.; Nakase, H.; Ueno, S.; Uza, N.; Kido, M.; Inoue, S.; Mikami, S.; Asada, M.; et al. Human thioredoxin-1 ameliorates experimental murine colitis in association with suppressed macrophage inhibitory factor production. Gastroenterology 2006, 131, 1110–1121. [Google Scholar] [CrossRef] [PubMed]
- Sakuma, K.; Nakamura, H.; Nakamura, T.; Hoshino, Y.; Ueda, S.; Ichikawa, M.; Tabata, C.; Fujita, S.; Masago, K.; Yodoi, J.; et al. Elevation of serum thioredoxin in patients with gefitinib-induced interstitial lung disease. Intern. Med. 2007, 46, 1905–1909. [Google Scholar] [CrossRef] [PubMed]
- Sumida, Y.; Nakashima, T.; Yoh, T.; Furutani, M.; Hirohama, A.; Kakisaka, Y.; Nakajima, Y.; Ishikawa, H.; Mitsuyoshi, H.; Okanoue, T.; et al. Serum thioredoxin levels as a predictor of steatohepatitis in patients with nonalcoholic fatty liver disease. J. Hepatol. 2003, 38, 32–38. [Google Scholar] [CrossRef]
- Okamoto, M.; Azuma, K.; Hoshino, T.; Imaoka, H.; Ikeda, J.; Kinoshita, T.; Takamori, S.; Ohshima, K.; Edakuni, N.; Kato, S.; et al. Correlation of decreased survival and IL-18 in bone metastasis. Intern. Med. 2009, 48, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Chin, K.; Nakamura, H.; Morita, S.; Sumi, K.; Oga, T.; Matsumoto, H.; Niimi, A.; Fukuhara, S.; Yodoi, J.; et al. Plasma thioredoxin, a novel oxidative stress marker, in patients with obstructive sleep apnea before and after nasal continuous positive airway pressure. Antioxid. Redox Signal. 2008, 10, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Csosz, E.; Labiscsak, P.; Kallo, G.; Markus, B.; Emri, M.; Szabo, A.; Tar, I.; Tozser, J.; Kiss, C.; Marton, I. Proteomics investigation of OSCC-specific salivary biomarkers in a Hungarian population highlights the importance of identification of population-tailored biomarkers. PLoS ONE 2017, 12, e0177282. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Bai, J.; Nishinaka, Y.; Ueda, S.; Sasada, T.; Ohshio, G.; Imamura, M.; Takabayashi, A.; Yamaoka, Y.; Yodoi, J. Expression of thioredoxin and glutaredoxin, redox-regulating proteins, in pancreatic cancer. Cancer Detect. Prev. 2000, 24, 53–60. [Google Scholar] [PubMed]
- Koura, T.; Gon, Y.; Hashimoto, S.; Azuma, A.; Kudoh, S.; Fukuda, Y.; Sugawara, I.; Yodoi, J.; Horie, T. Expression of thioredoxin in granulomas of sarcoidosis: Possible role in the development of T lymphocyte activation. Thorax 2000, 55, 755–761. [Google Scholar] [CrossRef] [PubMed]
- Jikimoto, T.; Nishikubo, Y.; Koshiba, M.; Kanagawa, S.; Morinobu, S.; Morinobu, A.; Saura, R.; Mizuno, K.; Kondo, S.; Toyokuni, S.; et al. Thioredoxin as a biomarker for oxidative stress in patients with rheumatoid arthritis. Mol. Immunol. 2002, 38, 765–772. [Google Scholar] [CrossRef]
- Maurice, M.M.; Nakamura, H.; Gringhuis, S.; Okamoto, T.; Yoshida, S.; Kullmann, F.; Lechner, S.; van der Voort, E.A.; Leow, A.; Versendaal, J.; et al. Expression of the thioredoxin-thioredoxin reductase system in the inflamed joints of patients with rheumatoid arthritis. Arthritis Rheumatol. 1999, 42, 2430–2439. [Google Scholar] [CrossRef]
- Yoshida, S.; Katoh, T.; Tetsuka, T.; Uno, K.; Matsui, N.; Okamoto, T. Involvement of thioredoxin in rheumatoid arthritis: Its costimulatory roles in the TNF-alpha-induced production of IL-6 and IL-8 from cultured synovial fibroblasts. J. Immunol. 1999, 163, 351–358. [Google Scholar] [PubMed]
- Kurimoto, C.; Kawano, S.; Tsuji, G.; Hatachi, S.; Jikimoto, T.; Sugiyama, D.; Kasagi, S.; Komori, T.; Nakamura, H.; Yodoi, J.; et al. Thioredoxin may exert a protective effect against tissue damage caused by oxidative stress in salivary glands of patients with Sjogren’s syndrome. J. Rheumatol. 2007, 34, 2035–2043. [Google Scholar] [PubMed]
- Dai, J.X.; Cai, J.Y.; Lin, Q.; Chen, X.D.; Lu, C.; Sun, J.; Ba, H.J. Thioredoxin as a marker for severity and prognosis of aneurysmal subarachnoid hemorrhage. J. Neurol. Sci. 2016, 363, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, J.; Gidlof, A.; Eriksson, M.; Larsson, E.; Brattstrom, O.; Oldner, A. Thioredoxin a novel biomarker of post-injury sepsis. Free Radic. Biol. Med. 2017, 104, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Hokamaki, J.; Kawano, H.; Soejima, H.; Miyamoto, S.; Kajiwara, I.; Kojima, S.; Sakamoto, T.; Sugiyama, S.; Yoshimura, M.; Nakamura, H.; et al. Plasma thioredoxin levels in patients with unstable angina. Int. J. Cardiol. 2005, 99, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Burke-Gaffney, A.; Callister, M.E.; Nakamura, H. Thioredoxin: Friend or foe in human disease? Trends Pharmacol. Sci. 2005, 26, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Benhar, M.; Shytaj, I.L.; Stamler, J.S.; Savarino, A. Dual targeting of the thioredoxin and glutathione systems in cancer and HIV. J. Clin. Investig. 2016, 126, 1630–1639. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Holmgren, A. A novel antioxidant mechanism of ebselen involving ebselen diselenide, a substrate of mammalian thioredoxin and thioredoxin reductase. J. Biol. Chem. 2002, 277, 39456–39462. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Masayasu, H.; Holmgren, A. Ebselen: A substrate for human thioredoxin reductase strongly stimulating its hydroperoxide reductase activity and a superfast thioredoxin oxidant. Proc. Natl. Acad. Sci. USA 2002, 99, 8579–8584. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, Y.; Yodoi, J. Extracellular thioredoxin: A therapeutic tool to combat inflammation. Cytokine Growth Factor Rev. 2013, 24, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Hoshino, Y.; Okuyama, H.; Matsuo, Y.; Yodoi, J. Thioredoxin 1 delivery as new therapeutics. Adv. Drug Deliv. Rev. 2009, 61, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Masutani, H.; Yodoi, J. Extracellular thioredoxin and thioredoxin-binding protein 2 in control of cancer. Semin. Cancer Biol. 2006, 16, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Yodoi, J.; Tian, H.; Masutani, H.; Nakamura, H. Thiol redox barrier; local and systemic surveillance against stress and inflammatory diseases. Arch. Biochem. Biophys. 2016, 595, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Bachnoff, N.; Trus, M.; Atlas, D. Alleviation of oxidative stress by potent and selective thioredoxin-mimetic peptides. Free Radic. Biol. Med. 2011, 50, 1355–1367. [Google Scholar] [CrossRef] [PubMed]
- Fukunaga, A.; Horikawa, T.; Ogura, K.; Taguchi, K.; Yu, X.; Funasaka, Y.; Takeda, M.; Nakamura, H.; Yodoi, J.; Nishigori, C. Thioredoxin suppresses the contact hypersensitivity response by inhibiting leukocyte recruitment during the elicitation phase. Antioxid. Redox Signal. 2009, 11, 1227–1235. [Google Scholar] [CrossRef] [PubMed]
- Ono, R.; Masaki, T.; Dien, S.; Yu, X.; Fukunaga, A.; Yodoi, J.; Nishigori, C. Suppressive effect of recombinant human thioredoxin on ultraviolet light-induced inflammation and apoptosis in murine skin. J. Dermatol. 2012, 39, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Son, A.; Nakamura, H.; Kondo, N.; Matsuo, Y.; Liu, W.; Oka, S.; Ishii, Y.; Yodoi, J. Redox regulation of mast cell histamine release in thioredoxin-1 (TRX) transgenic mice. Cell Res. 2006, 16, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Gabor, M. Models of acute inflammation in the ear. Methods Mol. Biol. 2003, 225, 129–137. [Google Scholar] [PubMed]
- Tian, H.; Matsuo, Y.; Fukunaga, A.; Ono, R.; Nishigori, C.; Yodoi, J. Thioredoxin ameliorates cutaneous inflammation by regulating the epithelial production and release of pro-inflammatory cytokines. Front. Immunol. 2013, 4, 269. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Matsui, M.; Iwata, S.; Nishiyama, A.; Mori, K.; Yodoi, J. AP-1 transcriptional activity is regulated by a direct association between thioredoxin and Ref-1. Proc. Natl. Acad. Sci. USA 1997, 94, 3633–3638. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.R.; Wakasugi, N.; Virelizier, J.L.; Yodoi, J.; Hay, R.T. Thioredoxin regulates the DNA-binding activity of NF-kappa B by reduction of a disulphide bond involving cysteine 62. Nucleic Acids Res. 1992, 20, 3821–3830. [Google Scholar] [CrossRef] [PubMed]
- Saitoh, M.; Nishitoh, H.; Fujii, M.; Takeda, K.; Tobiume, K.; Sawada, Y.; Kawabata, M.; Miyazono, K.; Ichijo, H. Mammalian thioredoxin is a direct inhibitor of apoptosis signal-regulating kinase (ASK) 1. EMBO J. 1998, 17, 2596–2606. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, K.; Nishio, A.; Nakamura, H.; Uchida, K.; Fukui, T.; Ohana, M.; Yoshizawa, H.; Ohashi, S.; Tamaki, H.; Matsuura, M.; et al. Helicobacter felis-induced gastritis was suppressed in mice overexpressing thioredoxin-1. Lab. Investig. 2005, 85, 1104–1117. [Google Scholar] [CrossRef] [PubMed]
- Tan, A.; Nakamura, H.; Kondo, N.; Tanito, M.; Kwon, Y.W.; Ahsan, M.K.; Matsui, H.; Narita, M.; Yodoi, J. Thioredoxin-1 attenuates indomethacin-induced gastric mucosal injury in mice. Free Radic. Res. 2007, 41, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, A.; Fukui, T.; Takahashi, Y.; Kishimoto, M.; Yamashina, M.; Nakayama, S.; Sakaguchi, Y.; Yoshida, K.; Uchida, K.; Nishio, A.; et al. Attenuation of indomethacin-induced gastric mucosal injury by prophylactic administration of sake yeast-derived thioredoxin. J. Gastroenterol. 2012, 47, 978–987. [Google Scholar] [CrossRef] [PubMed]
- Taketani, Y.; Kinugasa, K.; Kitajima, R.; Nishiumi, S.; Ashida, H.; Nakamura, H.; Fujita, T.; Kanzaki, K.; Masutani, H.; Yodoi, J. Protective effects of oral administration of yeast thioredoxin against gastric mucosal injury. Biosci. Biotechnol. Biochem. 2014, 78, 1221–1230. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.; Ashida, H.; Watanabe, R.; Inai, K.; Kim, Y.S.; Mukougawa, K.; Fukuda, H.; Tomizawa, K.; Ushiyama, K.; Asao, H.; et al. Production of biologically active human thioredoxin 1 protein in lettuce chloroplasts. Plant Mol. Biol. 2011, 76, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Nakamura, K.; Yodoi, J. Redox regulation of cellular activation. Annu. Rev. Immunol. 1997, 15, 351–369. [Google Scholar] [CrossRef] [PubMed]
- Masutani, H.; Ueda, S.; Yodoi, J. The thioredoxin system in retroviral infection and apoptosis. Cell Death Differ. 2005, 12 (Suppl. 1), 991–998. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Nakamura, H.; Kwon, Y.W.; Hattori, I.; Yamaguchi, Y.; Kim, Y.C.; Kondo, N.; Oka, S.; Ueda, S.; Masutani, H.; et al. Critical roles of thioredoxin in nerve growth factor-mediated signal transduction and neurite outgrowth in PC12 cells. J. Neurosci. 2003, 23, 503–509. [Google Scholar] [PubMed]
- Kim, Y.C.; Masutani, H.; Yamaguchi, Y.; Itoh, K.; Yamamoto, M.; Yodoi, J. Hemin-induced activation of the thioredoxin gene by Nrf2. A differential regulation of the antioxidant responsive element by a switch of its binding factors. J. Biol. Chem. 2001, 276, 18399–18406. [Google Scholar] [CrossRef] [PubMed]
- Tanito, M.; Masutani, H.; Kim, Y.C.; Nishikawa, M.; Ohira, A.; Yodoi, J. Sulforaphane induces thioredoxin through the antioxidant-responsive element and attenuates retinal light damage in mice. Investig. Ophthalmol. Vis. Sci. 2005, 46, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Dekigai, H.; Nakamura, H.; Bai, J.; Tanito, M.; Masutani, H.; Hirota, K.; Matsui, H.; Murakami, M.; Yodoi, J. Geranylgeranylacetone promotes induction and secretion of thioredoxin in gastric mucosal cells and peripheral blood lymphocytes. Free Radic. Res. 2001, 35, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Nakamura, H.; Arai, T.; Ishii, H.; Bai, J.; Itoh, T.; Fukuda, K.; Yodoi, J. Geranylgeranylacetone enhances expression of thioredoxin and suppresses ethanol-induced cytotoxicity in cultured hepatocytes. Biochem. Biophys. Res. Commun. 2000, 275, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.C.; Yamaguchi, Y.; Kondo, N.; Masutani, H.; Yodoi, J. Thioredoxin-dependent redox regulation of the antioxidant responsive element (ARE) in electrophile response. Oncogene 2003, 22, 1860–1865. [Google Scholar] [CrossRef] [PubMed]
- Tonissen, K.F.; Wells, J.R. Isolation and characterization of human thioredoxin-encoding genes. Gene 1991, 102, 221–228. [Google Scholar] [CrossRef]
- Yodoi, J.; Nakamura, H.; Masutani, H. Redox regulation of stress signals: Possible roles of dendritic stellate TRX producer cells (DST cell types). Biol. Chem. 2002, 383, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Masutani, H.; Otsuki, R.; Yamaguchi, Y.; Takenaka, M.; Kanoh, N.; Takatera, K.; Kunimoto, Y.; Yodoi, J. Fragrant unsaturated aldehydes elicit activation of the Keap1/Nrf2 system leading to the upregulation of thioredoxin expression and protection against oxidative stress. Antioxid. Redox Signal. 2009, 11, 949–962. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, A.; Matsui, M.; Iwata, S.; Hirota, K.; Masutani, H.; Nakamura, H.; Takagi, Y.; Sono, H.; Gon, Y.; Yodoi, J. Identification of thioredoxin-binding protein-2/vitamin D(3) up-regulated protein 1 as a negative regulator of thioredoxin function and expression. J. Biol. Chem. 1999, 274, 21645–21650. [Google Scholar] [CrossRef] [PubMed]
- Patwari, P.; Higgins, L.J.; Chutkow, W.A.; Yoshioka, J.; Lee, R.T. The interaction of thioredoxin with Txnip. Evidence for formation of a mixed disulfide by disulfide exchange. J. Biol. Chem. 2006, 281, 21884–21891. [Google Scholar] [CrossRef] [PubMed]
- Oka, S.; Masutani, H.; Liu, W.; Horita, H.; Wang, D.; Kizaka-Kondoh, S.; Yodoi, J. Thioredoxin-binding protein-2-like inducible membrane protein is a novel vitamin D3 and peroxisome proliferator-activated receptor (PPAR)gamma ligand target protein that regulates PPARgamma signaling. Endocrinology 2006, 147, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Patwari, P.; Chutkow, W.A.; Cummings, K.; Verstraeten, V.L.; Lammerding, J.; Schreiter, E.R.; Lee, R.T. Thioredoxin-independent regulation of metabolism by the alpha-arrestin proteins. J. Biol. Chem. 2009, 284, 24996–25003. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, C.E. On the origins of arrestin and rhodopsin. BMC Evol. Biol. 2008, 8, 222. [Google Scholar] [CrossRef] [PubMed]
- Masutani, H.; Yoshihara, E.; Masaki, S.; Chen, Z.; Yodoi, J. Thioredoxin binding protein (TBP)-2/Txnip and alpha-arrestin proteins in cancer and diabetes mellitus. J. Clin. Biochem. Nutr. 2012, 50, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.H.; Lee, K.N.; Hwang, C.Y.; Kwon, K.S.; You, K.H.; Choi, I. Tumor suppressor VDUP1 increases p27(kip1) stability by inhibiting JAB1. Cancer Res. 2005, 65, 4485–4489. [Google Scholar] [CrossRef] [PubMed]
- Nishinaka, Y.; Masutani, H.; Oka, S.; Matsuo, Y.; Yamaguchi, Y.; Nishio, K.; Ishii, Y.; Yodoi, J. Importin alpha1 (Rch1) mediates nuclear translocation of thioredoxin-binding protein-2/vitamin D(3)-up-regulated protein 1. J. Biol. Chem. 2004, 279, 37559–37565. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, F.; Takata, M.; Kamitori, K.; Nonaka, M.; Dong, Y.; Sui, L.; Tokuda, M. Rare sugar D-allose induces specific up-regulation of TXNIP and subsequent G1 cell cycle arrest in hepatocellular carcinoma cells by stabilization of p27kip1. Int. J. Oncol. 2008, 32, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, E.; Fujimoto, S.; Inagaki, N.; Okawa, K.; Masaki, S.; Yodoi, J.; Masutani, H. Disruption of TBP-2 ameliorates insulin sensitivity and secretion without affecting obesity. Nat. Commun. 2010, 1, 127. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Tardivel, A.; Thorens, B.; Choi, I.; Tschopp, J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat. Immunol. 2010, 11, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Bodnar, J.S.; Chatterjee, A.; Castellani, L.W.; Ross, D.A.; Ohmen, J.; Cavalcoli, J.; Wu, C.; Dains, K.M.; Catanese, J.; Chu, M.; et al. Positional cloning of the combined hyperlipidemia gene Hyplip1. Nat. Genet. 2002, 30, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, K.L.; Margosian, M.R.; Sheth, S.S.; Lusis, A.J.; Parks, E.J. Increased lipogenesis and fatty acid reesterification contribute to hepatic triacylglycerol stores in hyperlipidemic Txnip-/- mice. J. Nutr. 2004, 134, 1475–1480. [Google Scholar] [PubMed]
- Oka, S.; Liu, W.; Masutani, H.; Hirata, H.; Shinkai, Y.; Yamada, S.; Yoshida, T.; Nakamura, H.; Yodoi, J. Impaired fatty acid utilization in thioredoxin binding protein-2 (TBP-2)-deficient mice: A unique animal model of Reye syndrome. FASEB J. 2006, 20, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Oka, S.; Yoshihara, E.; Bizen-Abe, A.; Liu, W.; Watanabe, M.; Yodoi, J.; Masutani, H. Thioredoxin binding protein-2/thioredoxin-interacting protein is a critical regulator of insulin secretion and peroxisome proliferator-activated receptor function. Endocrinology 2009, 150, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Lopez-Ramos, D.A.; Yoshihara, E.; Maeda, Y.; Masutani, H.; Sugie, K.; Maeda, M.; Yodoi, J. Thioredoxin-binding protein-2 (TBP-2/VDUP1/TXNIP) regulates T-cell sensitivity to glucocorticoid during HTLV-I-induced transformation. Leukemia 2011, 25, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Yoshihara, E.; Son, A.; Matsuo, Y.; Masutani, H.; Sugie, K.; Maeda, M.; Yodoi, J. Differential roles of Annexin A1 (ANXA1/lipocortin-1/lipomodulin) and thioredoxin binding protein-2 (TBP-2/VDUP1/TXNIP) in glucocorticoid signaling of HTLV-I-transformed T cells. Immunol. Lett. 2010, 131, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Ling, X.; Xiong, Y.; Xu, D. Molecular characterization of differentially expressed TXNIP gene and its association with porcine carcass traits. Mol. Biol. Rep. 2012, 39, 10439–10446. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Rong, Y.P.; Malone, M.H.; Davis, M.C.; Zhong, F.; Distelhorst, C.W. Thioredoxin-interacting protein (txnip) is a glucocorticoid-regulated primary response gene involved in mediating glucocorticoid-induced apoptosis. Oncogene 2006, 25, 1903–1913. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Zheng, B.; Shaywitz, A.; Dagon, Y.; Tower, C.; Bellinger, G.; Shen, C.H.; Wen, J.; Asara, J.; McGraw, T.E.; et al. AMPK-dependent degradation of TXNIP upon energy stress leads to enhanced glucose uptake via GLUT1. Mol. Cell 2013, 49, 1167–1175. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.S.; DeLuca, H.F. Isolation and characterization of a novel cDNA from HL-60 cells treated with 1,25-dihydroxyvitamin D-3. Biochim. Biophys. Acta 1994, 1219, 26–32. [Google Scholar] [CrossRef]

| Disease | Sample | References |
|---|---|---|
| Acquired immunodeficiency syndrome (AIDS) | Plasma | [20,21] |
| Acute coronary syndrome | Serum | [22] |
| Acute myocardial infarction | Plasma | [23,24] |
| Acute pancreatitis | Serum | [25] |
| Acute respiratory distress syndrome (ARDS) | BALF/Plasma | [26] |
| Asthma | Serum | [27] |
| Atherosclerosis | Plasma | [28] |
| Burns | Serum | [29] |
| Cardiac surgery with cardiopulmonary bypass | Plasma | [30] |
| Chronic heart failure | Plasma | [31] |
| Diabetes mellitus | Plasma/Serum | [32,33] |
| Dilated cardiomyopathy | Serum | [22] |
| Hepatocellular carcinoma | Serum | [34,35] |
| Hepatitis C | Serum | [36] |
| Inflammatory bowel disease | Serum | [37] |
| Interstitial lung disease | Serum | [38] |
| Nonalcoholic steatohepatitis (NASH) | Serum | [39] |
| Non-small cell lung cancer | Serum | [40] |
| Obstructive sleep apnea (OSA) | Plasma | [41] |
| Oral squamous cell carcinoma | Saliva | [42] |
| Pancreatic ductal carcinoma | Plasma | [43] |
| Pulmonary sarcoidosis | BALF | [44] |
| Rheumatoid arthritis | Plasma/Serum/SF | [45,46,47] |
| Sjögren’s syndrome | Saliva | [48] |
| Subarachnoid hemorrhage | Serum | [49] |
| Trauma | Plasma | [50] |
| Unstable angina | Plasma | [51] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yodoi, J.; Matsuo, Y.; Tian, H.; Masutani, H.; Inamoto, T. Anti-Inflammatory Thioredoxin Family Proteins for Medicare, Healthcare and Aging Care. Nutrients 2017, 9, 1081. https://doi.org/10.3390/nu9101081
Yodoi J, Matsuo Y, Tian H, Masutani H, Inamoto T. Anti-Inflammatory Thioredoxin Family Proteins for Medicare, Healthcare and Aging Care. Nutrients. 2017; 9(10):1081. https://doi.org/10.3390/nu9101081
Chicago/Turabian StyleYodoi, Junji, Yoshiyuki Matsuo, Hai Tian, Hiroshi Masutani, and Takashi Inamoto. 2017. "Anti-Inflammatory Thioredoxin Family Proteins for Medicare, Healthcare and Aging Care" Nutrients 9, no. 10: 1081. https://doi.org/10.3390/nu9101081
APA StyleYodoi, J., Matsuo, Y., Tian, H., Masutani, H., & Inamoto, T. (2017). Anti-Inflammatory Thioredoxin Family Proteins for Medicare, Healthcare and Aging Care. Nutrients, 9(10), 1081. https://doi.org/10.3390/nu9101081





