Soluble Fibre Meal Challenge Reduces Airway Inflammation and Expression of GPR43 and GPR41 in Asthma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Design
2.3. Hypertonic Saline Challenge
2.4. Sputum Processing
2.5. Sputum Supernatant Measurements
2.6. RNA Extraction and cDNA Synthesis
2.7. mRNA Quantification by Real-Time qRT-PCR
2.8. Statistical Analysis
3. Results
3.1. Baseline Characteristics
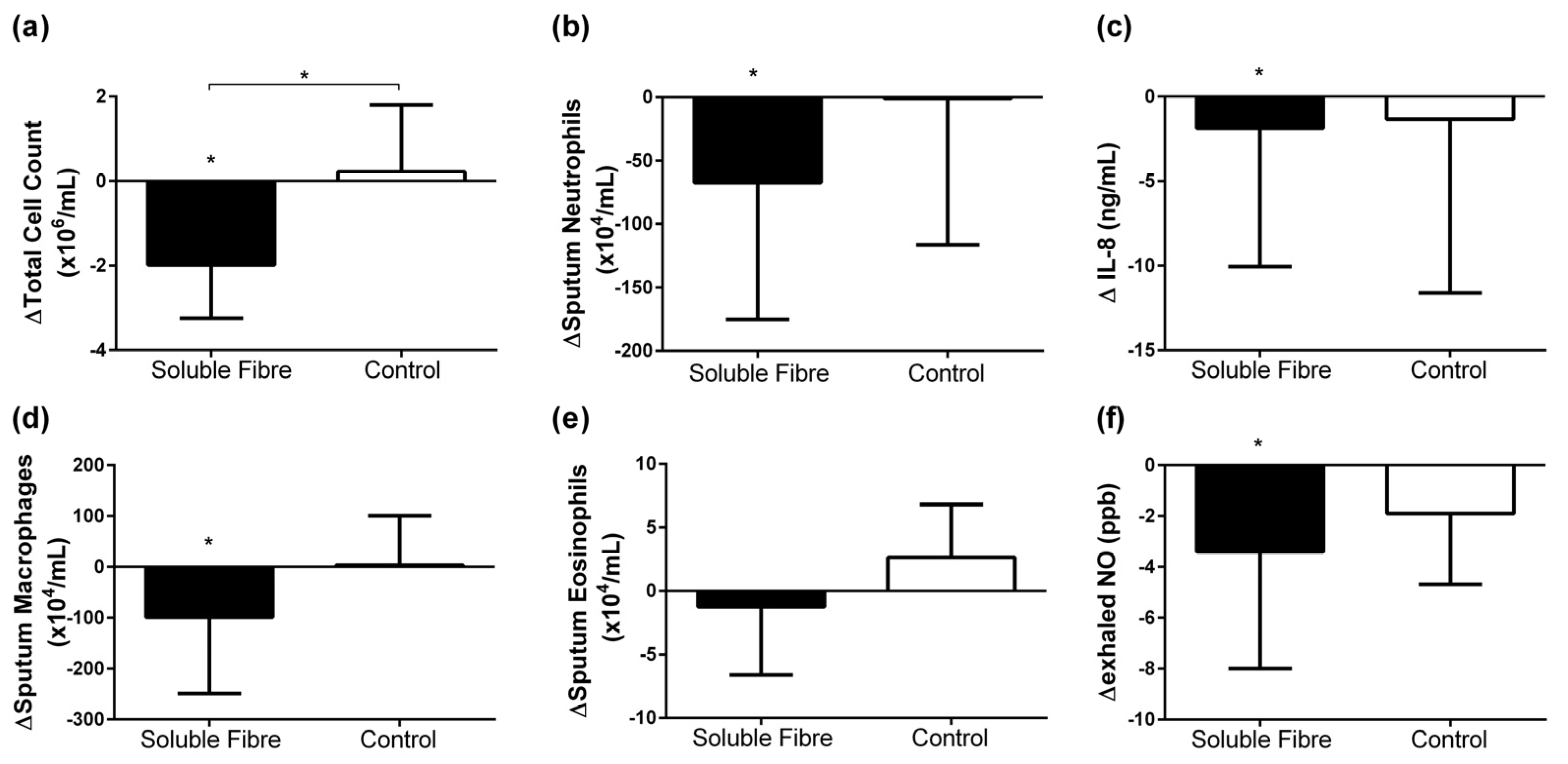
3.2. Airway Inflammatory Markers
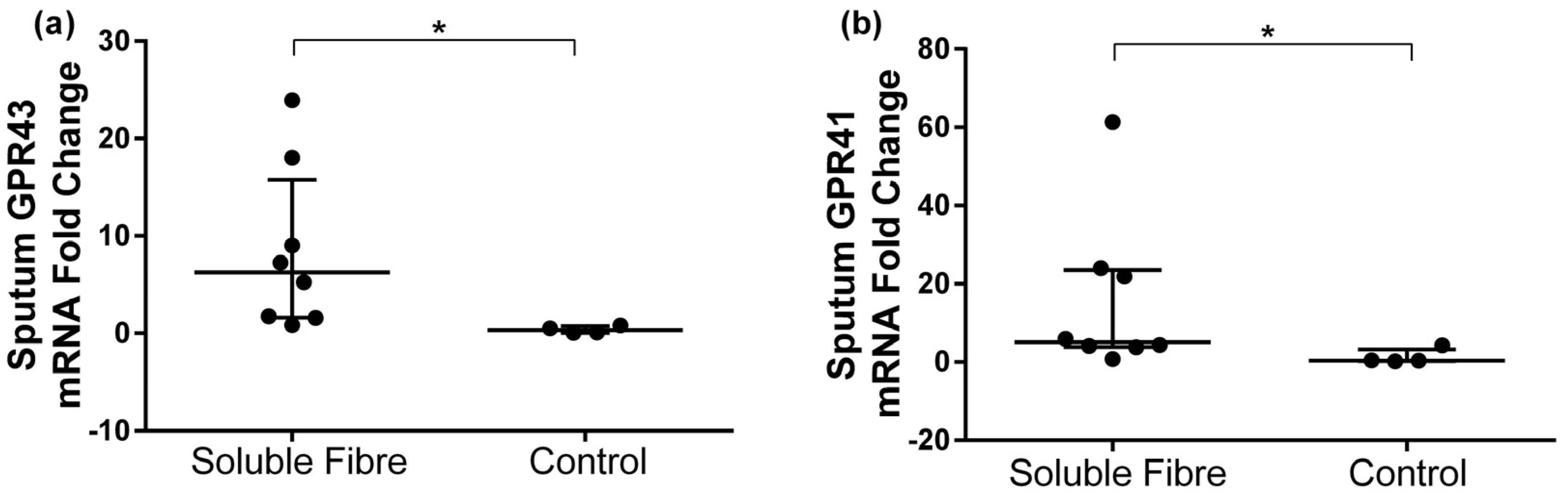
3.3. GPR41 and GPR43 Sputum Gene Expression
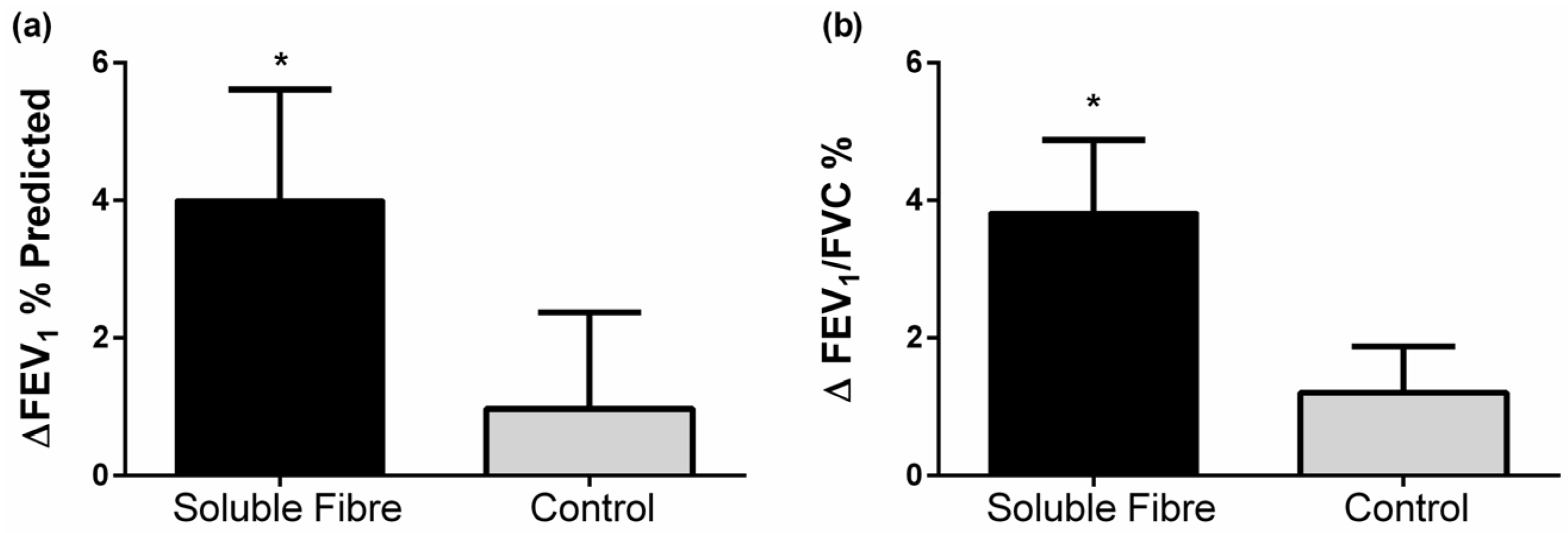
3.4. Lung Function
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Masoli, M.; Fabian, D.; Holt, S.; Beasley, R. Global initiative for asthma (GINA) program. The global burden of asthma: Executive summary of the GINA dissemination committee report. Allergy 2004, 59, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Australian Centre for Asthma Monitoring. Asthma in Australia 2011. In AIHW Asthma Series No. 4. Cat. No. ACM 22; Australian Institute of Health and Welfare (AIHW): Canberra, Australia, 2011. [Google Scholar]
- Barnes, P.J.; Adcock, I.M. Glucocorticoid resistance in inflammatory diseases. Lancet 2009, 373, 1905–1917. [Google Scholar] [CrossRef]
- Wickens, K.; Barry, D.; Friezema, A.; Rhodius, R.; Bone, N.; Purdie, G.; Crane, J. Fast foods—Are they a risk factor for asthma? Allergy 2005, 60, 1537–1541. [Google Scholar] [CrossRef] [PubMed]
- Hijazi, N.; Abalkhail, B.; Seaton, A. Diet and childhood asthma in a society in transition: A study in urban and rural Saudi Arabia. Thorax 2000, 55, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Carey, O.J.; Cookson, J.B.; Britton, J.; Tattersfield, A.E. The effect of lifestyle on wheeze, atopy, and bronchial hyperreactivity in Asian and white children. Am. J. Respir. Crit. Care Med. 1996, 154, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.-L.; Pan, W.-H. Dietary fats and asthma in teenagers: Analyses of the first Nutrition and Health Survey in Taiwan (NAHSIT). Clin. Exp. Allergy 2001, 31, 1875–1880. [Google Scholar] [CrossRef] [PubMed]
- Rolfes, S.R.; Pinna, K.; Whitney, E. Understanding Normal and Clinical Nutrition, 8th ed.; Wadsworth: Belmont, CA, USA, 2009. [Google Scholar]
- Ganapathy, V.; Thangaraju, M.; Prasad, P.D.; Martin, P.M.; Singh, N. Transporters and receptors for short-chain fatty acids as the molecular link between colonic bacteria and the host. Curr. Opin. Pharmacol. 2013, 13, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Vinolo, M.A.R.; Rodrigues, H.G.; Nachbar, R.T.; Curi, R. Regulation of Inflammation by Short Chain Fatty Acids. Nutrients 2011, 3, 858–876. [Google Scholar] [CrossRef] [PubMed]
- Root, M.M.; Houser, S.M.; Anderson, J.J.; Dawson, H.R. Healthy Eating Index 2005 and selected macronutrients are correlated with improved lung function in humans. Nutr. Res. 2014, 34, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Berthon, B.S.; Macdonald-Wicks, L.K.; Gibson, P.G.; Wood, L.G. Investigation of the association between dietary intake, disease severity and airway inflammation in asthma. Respirology 2013, 18, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.; Goldsworthy, S.M.; Barnes, A.A.; Eilert, M.M.; Tcheang, L.; Daniels, D.; Muir, A.I.; Wigglesworth, M.J.; Kinghorn, I.; Fraser, N.J.; et al. The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J. Biol. Chem. 2003, 278, 11312–11319. [Google Scholar] [CrossRef] [PubMed]
- Baines, K.J.; Simpson, J.L.; Wood, L.G.; Scott, R.J.; Gibson, P.G. Transcriptional phenotypes of asthma defined by gene expression profiling of induced sputum samples. J. Allergy Clin. Immunol. 2011, 127, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Fahy, J.V.; Liu, J.; Wong, H.; Boushey, H.A. Analysis of cellular and biochemical constituents of induced sputum after allergen challenge: A method for studying allergic airway inflammation. J. Allergy Clin. Immunol. 1994, 93, 1031–1039. [Google Scholar] [CrossRef]
- Wood, L.G.; Garg, M.L.; Gibson, P.G. A high fat challenge increases airway inflammation and impairs bronchodilator recovery in asthma. J. Allergy Clin. Immunol. 2011, 127, 1133–1140. [Google Scholar] [CrossRef] [PubMed]
- Juniper, E.F.; Bousquet, J.; Abetz, L.; Bateman, E.D. Identifying ‘well-controlled’ and ‘not well-controlled’ asthma using the Asthma Control Questionnaire. Respir. Med. 2006, 100, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention 2012 (Update). Available online: http://wwwginasthmaorg (accessed on 30 July 2012).
- Hankinson, J.; Odencrantz, J.; Fedan, K. Spirometric reference values from a sample of the general U.S. population. Am. J. Respir. Crit. Care Med. 1999, 159, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Stanojevic, S.; Wade, A.; Stocks, J. Reference values for lung function: Past, present and future. Eur. Respir. J. 2010, 36, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.G.; Wlodarczyk, J.W.; Hensley, M.J.; Gleeson, M.; Henry, R.L.; Cripps, A.W.; Clancy, R.L. Epidemiological Association of Airway Inflammation with Asthma Symptoms and Airway Hyperresponsiveness in Childhood. Am. J. Respir. Crit. Care Med. 1998, 158, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.G.; Henry, R.L.; Thomas, P. Noninvasive assessment of airway inflammation in children: Induced sputum, exhaled nitric oxide, and breath condensate. Eur. Respir. J. 2000, 16, 1008–1015. [Google Scholar] [PubMed]
- Simpson, J.L.; Scott, R.J.; Boyle, M.J.; Gibson, P.G. Differential proteolytic enzyme activity in eosinophilic and neutrophilic asthma. Am. J. Respir. Crit. Care Med. 2005, 172, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Wood, L.G.; Garg, M.L.; Simpson, J.L.; Mori, T.A.; Croft, K.D.; Wark, P.A.B.; Gibson, P.G. Induced Sputum 8-Isoprostane Concentrations in Inflammatory Airway Diseases. Am. J. Respir. Crit. Care Med. 2005, 171, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Altalag, A.; Road, J.; Wilcox, P. Pulmonary Function Tests in Clinical Practice; Springer: London, UK, 2009. [Google Scholar]
- Ma, Y.; Griffith, J.A.; Chasan-Taber, L.; Olendzki, B.C.; Jackson, E.; Stanek, E.J.; Li, W.; Pagoto, S.L.; Hafner, A.R.; Ockene, I.S. Association between dietary fiber and serum C-reactive protein. Am. J. Clin. Nutr. 2006, 83, 760–766. [Google Scholar] [PubMed]
- Ajani, U.A.; Ford, E.S.; Mokdad, A.H. Dietary fiber and C-reactive protein: Findings from national health and nutrition examination survey data. J. Nutr. 2004, 134, 1181–1185. [Google Scholar] [PubMed]
- Taylor, D.R.; Pijnenburg, M.W.; Smith, A.D.; Jongste, J.C.D. Exhaled nitric oxide measurements: Clinical application and interpretation. Thorax 2006, 61, 817–827. [Google Scholar] [CrossRef] [PubMed]
- Maslowski, K.M.; Vieira, A.T.; Ng, A.; Kranich, J.; Sierro, F.; Yu, D.; Schilter, H.C.; Rolph, M.S.; Mackay, F.; Artis, D.; et al. Regulation of inflammatory responses by gut microbiota and chemoattractant receptor GPR43. Nature 2009, 461, 1282–1286. [Google Scholar] [CrossRef] [PubMed]
- Trompette, A.; Gollwitzer, E.S.; Yadava, K.; Sichelstiel, A.K.; Sprenger, N.; Ngom-Bru, C.; Blanchard, C.; Junt, T.; Nicod, L.P.; Harris, N.L.; et al. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat. Med. 2014, 20, 159–166. [Google Scholar] [CrossRef] [PubMed]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, S.; Cleary, S.; Bahrami, B.; Reynolds, N.; Macfarlane, G.T. Synbiotic consumption changes the metabolism and composition of the gut microbiota in older people and modifies inflammatory processes: A randomised, double-blind, placebo-controlled crossover study. Aliment. Pharmacol. Ther. 2013, 38, 804–816. [Google Scholar] [CrossRef] [PubMed]
- Nappo, F.; Esposito, K.; Cioffi, M.; Giugliano, G.; Molinari, A.M.; Paolisso, G.; Marfella, R.; Giugliano, D. Postprandial endothelial activation in healthy subjects and in Type 2 diabetic patients: Role of fat and carbohydrate meals. J. Am. Coll. Cardiol. 2002, 39, 1145–1150. [Google Scholar] [CrossRef]
- Tarini, J.; Wolever, T.M. The fermentable fibre inulin increases postprandial serum short-chain fatty acids and reduces free-fatty acids and ghrelin in healthy subjects. Appl. Physiol. Nutr. Metab. 2010, 35, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Jeukendrup, A.E.; Ball, D. Sodium acetate induces a metabolic alkalosis but not the increase in fatty acid oxidation observed following bicarbonate ingestion in humans. J. Nutr. 2007, 137, 1750–1756. [Google Scholar] [PubMed]
- Roberfroid, M.B. Introducing inulin-type fructans. Br. J. Nutr. 2005, 93, S13–S25. [Google Scholar] [CrossRef] [PubMed]
| Clinical Characteristic | Soluble Fibre Group (n = 17) | Control Group (n = 12) | p * |
|---|---|---|---|
| Gender Male n (%) | 7 (41) | 6 (50) | 0.716 |
| Female n (%) | 10 (59) | 6 (50) | |
| Age (years) | 42.1 ± 3.4 | 40.4 ±4.6 | 0.770 |
| BMI (kg/m2) | 24.9 ± 0.7 | 26.6 ± 0.7 | 0.099 |
| Ex-smokers n (%) | 6 (35) | 3 (25) | 0.694 |
| ACQ-7 (units) | 0.7 ± 0.1 | 0.7 ± 0.2 | 0.941 |
| Atopy n (%) | 14 (82) | 8 (67) | 0.403 |
| AHR n (%) | 10 (59] | 5 (41.7) | 0.462 |
| DRS (%∆FEV1/mL), med [IQR] | 2.2 [0.4, 6.4] | 0.6 [0.2, 2.4] | 0.109 |
| ICS dose † (μg/day), med [IQR] | 1000 [400, 1000] | 1000 [563, 1000] | 0.679 |
| Airway Inflammation | Soluble Fibre Group | p † | Control Group | p † | p * | ||
|---|---|---|---|---|---|---|---|
| 0 h | ∆4 h | 0 h | ∆4 h | ||||
| Sputum (n) | 16 | 14 | 10 | 9 | |||
| TCC (×106/mL) | 3.7 [2.2, 6.3] | −2.0 [−2.8, −1.1] | 0.013 | 3.0 [1.4, 6.0] | 0.2 [−0.8, 1.4] | 0.833 | 0.037 |
| Neutrophils (×104/mL) | 76.2 [54.6, 249.4] | −67.5 [−158.0, −6.4] | 0.033 | 92.4 [7.2, 342.7] | −1.1 [−94.9, 105.1] | 1.000 | 0.148 |
| Eosinophils (×104/mL) | 3.3 [1.3, 16.0] | −1.2 [−5.8, 1.8] | 0.490 | 2.4 [0.0, 4.5] | 2.6 [−1.7, 6.7] | 0.440 | 0.393 |
| Macrophages (×104/mL) | 144.0 [93.0, 386.3] | −99.1 [−244.6, −12.2] | 0.030 | 124.5 [104.5, 211.5] | 3.4 [−47.6, 85.1] | 0.779 | 0.117 |
| Lymphocytes (×104/mL) | 1.6 [0.2, 7.1] | −1.4 [−3.9, −0.3] | 0.002 | 4.1 [0.4, 6.7] | 2.1 [−1.5, 3.9] | 0.401 | 0.034 |
| Sputum IL-8 (ng/mL) | 5.7 [3.9, 9.7] | −1.9 [−7.6, −1.1] | 0.005 | 3.7 [3.0, 22.6] | −1.3 [−10.1, 1.2] | 0.208 | 0.539 |
| Exhaled NO (ppb) | 17.5 [14.5, 70.5] | −3.4 [−7.9, −0.6] | 0.028 | 14.9 [11.0, 51.0] | −1.9 [−4.6, 1.0] | 0.170 | 0.301 |
| Gene | Soluble Fibre Group (n = 8) | p † | Control Group (n = 4) | p † | p * | ||||
|---|---|---|---|---|---|---|---|---|---|
| 0 h | 4 h | FC | 0 h | 4 h | FC | ||||
| GPR43 | 0.07 [0.05, 0.29] | 0.79 [0.26, 2.01] | 6.26 [1.66, 13.5] | 0.124 | 1.53 [0.29, 3.64] | 0.24 [0.17, 0.27] | 0.31 [0.07, 0.67] | 0.068 | 0.007 |
| GPR41 | 0.07 [0.04, 0.35] | 1.70 [0.14, 5.18] | 5.15 [3.96, 22.9] | 0.161 | 1.27 [0.80, 1.42] | 0.58 [0.41, 1.22] | 0.41 [0.31, 2.37] | 0.715 | 0.027 |
| Lung Function | Soluble Fibre Group | p † | Control Group | p † | p * | ||
|---|---|---|---|---|---|---|---|
| 0 h | ∆4 h | 0 h | ∆4 h | ||||
| (n) | 17 | 17 | 12 | 12 | |||
| FEV1 (L) ^ | 2.7 [2.2, 3.6] | 0.1 [0.0, 0.2] | 0.022 | 2.9 [2.0, 3.5] | 0.1 [−0.1, 0.2] | 0.347 | 0.341 |
| FEV1 (% predicted) # | 82.4 ± 4.4 | 4.0 ± 1.6 | 0.024 | 77.7 ± 6.8 | 1.0 ± 1.4 | 0.506 | 0.190 |
| FVC (L) ^ | 4.1 [3.4, 4.7] | 0.0 [−0.1, 0.1] | 0.331 | 4.0 [3.5, 4.9] | 0.01 [−0.04, 0.1] | 0.503 | 0.595 |
| FVC (% predicted) # | 98.9 ± 3.4 § | 1.6 ± 1.2 | 0.193 | 86.9 ± 4.5 | −0.2 ± 1.0 | 0.859 | 0.285 |
| FEV1/FVC (%) # | 69.2 ± 2.5 | 3.8 ± 1.1 | 0.002 | 71.9 ± 4.6 | 1.2 ± 0.7 | 0.101 | 0.070 |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Halnes, I.; Baines, K.J.; Berthon, B.S.; MacDonald-Wicks, L.K.; Gibson, P.G.; Wood, L.G. Soluble Fibre Meal Challenge Reduces Airway Inflammation and Expression of GPR43 and GPR41 in Asthma. Nutrients 2017, 9, 57. https://doi.org/10.3390/nu9010057
Halnes I, Baines KJ, Berthon BS, MacDonald-Wicks LK, Gibson PG, Wood LG. Soluble Fibre Meal Challenge Reduces Airway Inflammation and Expression of GPR43 and GPR41 in Asthma. Nutrients. 2017; 9(1):57. https://doi.org/10.3390/nu9010057
Chicago/Turabian StyleHalnes, Isabel, Katherine J. Baines, Bronwyn S. Berthon, Lesley K. MacDonald-Wicks, Peter G. Gibson, and Lisa G. Wood. 2017. "Soluble Fibre Meal Challenge Reduces Airway Inflammation and Expression of GPR43 and GPR41 in Asthma" Nutrients 9, no. 1: 57. https://doi.org/10.3390/nu9010057
APA StyleHalnes, I., Baines, K. J., Berthon, B. S., MacDonald-Wicks, L. K., Gibson, P. G., & Wood, L. G. (2017). Soluble Fibre Meal Challenge Reduces Airway Inflammation and Expression of GPR43 and GPR41 in Asthma. Nutrients, 9(1), 57. https://doi.org/10.3390/nu9010057







