Calorie Restricted High Protein Diets Downregulate Lipogenesis and Lower Intrahepatic Triglyceride Concentrations in Male Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Dietary Intervention
2.3. Total Body and Fat Mass
2.4. Metabolic Profile
2.5. Hepatic Triglyceride Concentrations
2.6. mRNA Expression
2.7. Protein Expression
2.8. Statistical Analysis
3. Results
3.1. Total Body and Fat Mass
3.2. Metabolic Profile
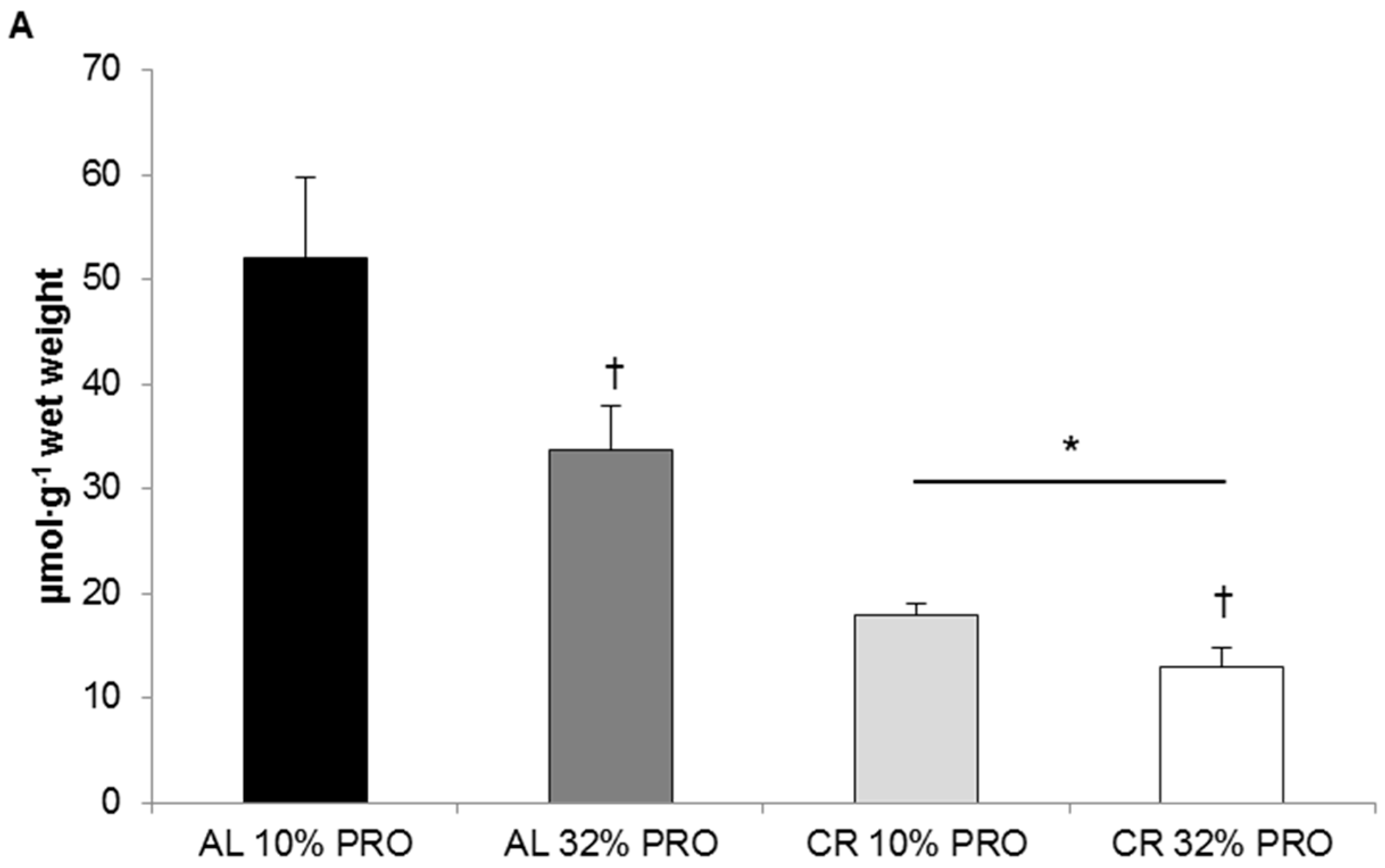
3.3. Hepatic Triglyceride Concentration
3.4. mRNA Expression
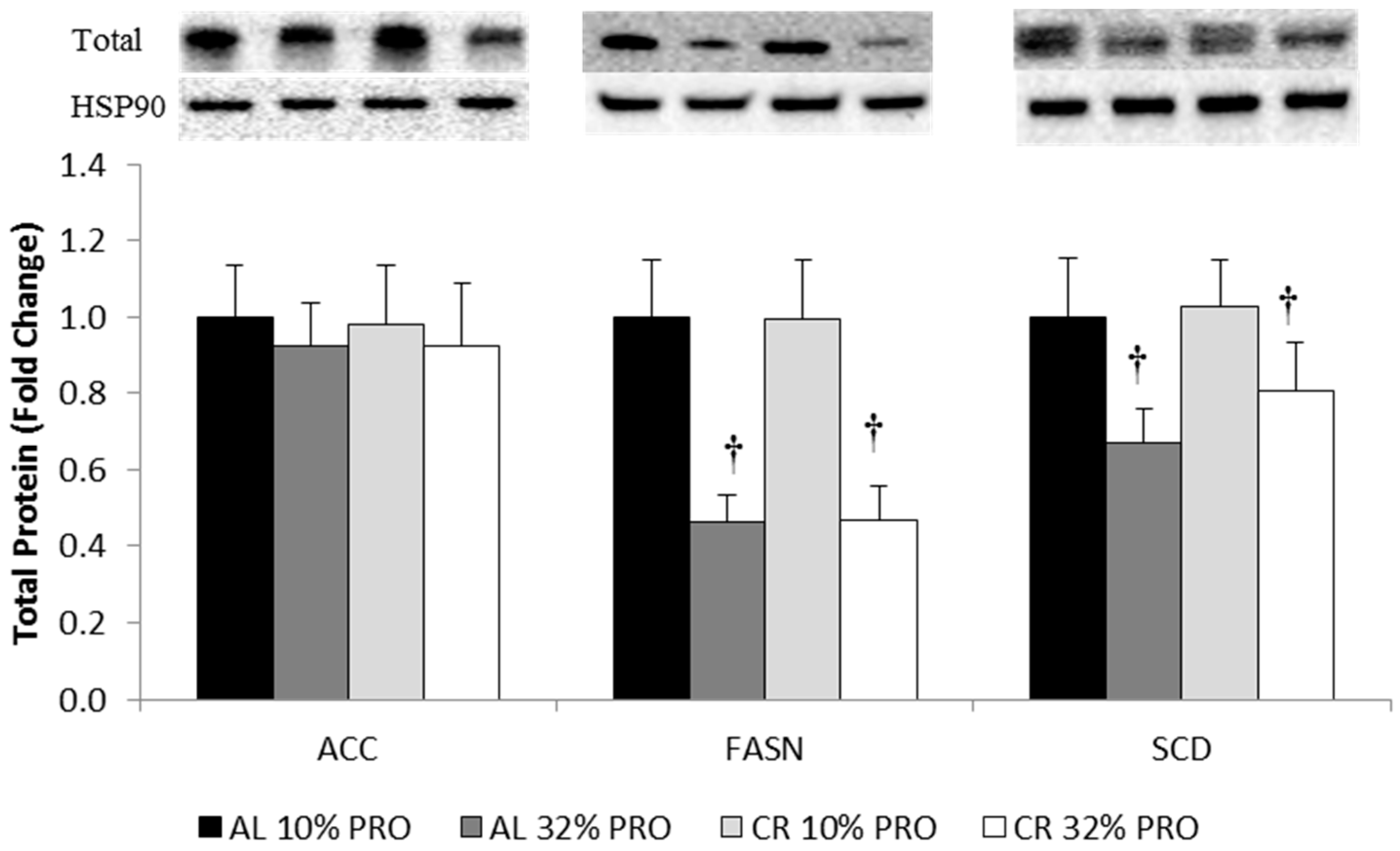
3.5. Protein Expression
3.6. Relationship of Whole-Body Fat, Intrahepatic Triglycerides, and Gene Expression
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ACC | Acetyl CoA Carboxylase |
| AL | Ad libitum |
| BCAA | Branched chain amino acids |
| CR | Calorie restriction |
| DNL | de novo lipogenesis |
| DXA | Dual Energy X-ray Absorptiometry |
| FASN | Fatty acid synthase |
| FM | Fat mass |
| HOMA-IR | Homeostatic model assessment of insulin resistance |
| HOMA-β | Homeostatic model assessment of β-cell function |
| HSP90 | Heat shock protein 90 |
| IHTG | Intrahepatic triglycerides |
| mTORC1 | mechanistic target of rapamycin complex 1 |
| PDK4 | Pyruvate dehydrogenase kinase, isozyme 4 |
| PRO | Protein |
| SCD1 | Stearoyl-CoA destaurase |
References
- Menezes, A.L.; Pereira, M.P.; Buzelle, S.L.; Dos Santos, M.P.; de Franca, S.A.; Baviera, A.M.; Andrade, C.M.; Garofalo, M.A.; Kettelhut Ido, C.; Chaves, V.E.; et al. A low-protein, high-carbohydrate diet increases de novo fatty acid synthesis from glycerol and glycerokinase content in the liver of growing rats. Nutr. Res. 2013, 33, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Marchesini, G.; Brizi, M.; Morselli-Labate, A.M.; Bianchi, G.; Bugianesi, E.; McCullough, A.J.; Forlani, G.; Melchionda, N. Association of nonalcoholic fatty liver disease with insulin resistance. Am. J. Med. 1999, 107, 450–455. [Google Scholar] [CrossRef]
- Sanyal, A.J.; Campbell-Sargent, C.; Mirshahi, F.; Rizzo, W.B.; Contos, M.J.; Sterling, R.K.; Luketic, V.A.; Shiffman, M.L.; Clore, J.N. Nonalcoholic steatohepatitis: Association of insulin resistance and mitochondrial abnormalities. Gastroenterology 2001, 120, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Boden, G. Fatty acid-induced inflammation and insulin resistance in skeletal muscle and liver. Curr. Diab. Rep. 2006, 6, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Boden, G.; Chen, X.; Ruiz, J.; White, J.V.; Rossetti, L. Mechanisms of fatty acid-induced inhibition of glucose uptake. J. Clin. Investig. 1994, 93, 2438–2446. [Google Scholar] [CrossRef] [PubMed]
- Browning, J.D.; Baker, J.A.; Rogers, T.; Davis, J.; Satapati, S.; Burgess, S.C. Short-term weight loss and hepatic triglyceride reduction: Evidence of a metabolic advantage with dietary carbohydrate restriction. Am. J. Clin. Nutr. 2011, 93, 1048–1052. [Google Scholar] [CrossRef] [PubMed]
- Layman, D.K.; Clifton, P.; Gannon, M.C.; Krauss, R.M.; Nuttall, F.Q. Protein in optimal health: Heart disease and type 2 diabetes. Am. J. Clin. Nutr. 2008, 87, 1571S–1575S. [Google Scholar] [PubMed]
- Layman, D.K.; Boileau, R.A.; Erickson, D.J.; Painter, J.E.; Shiue, H.; Sather, C.; Christou, D.D. A reduced ratio of dietary carbohydrate to protein improves body composition and blood lipid profiles during weight loss in adult women. J. Nutr. 2003, 133, 411–417. [Google Scholar] [PubMed]
- Farnsworth, E.; Luscombe, N.D.; Noakes, M.; Wittert, G.; Argyiou, E.; Clifton, P.M. Effect of a high-protein, energy-restricted diet on body composition, glycemic control, and lipid concentrations in overweight and obese hyperinsulinemic men and women. Am. J. Clin. Nutr. 2003, 78, 31–39. [Google Scholar] [PubMed]
- Gannon, M.C.; Nuttall, F.Q.; Saeed, A.; Jordan, K.; Hoover, H. An increase in dietary protein improves the blood glucose response in persons with type 2 diabetes. Am. J. Clin. Nutr. 2003, 78, 734–741. [Google Scholar] [PubMed]
- Larsen, T.M.; Dalskov, S.M.; van Baak, M.; Jebb, S.A.; Papadaki, A.; Pfeiffer, A.F.; Martinez, J.A.; Handjieva-Darlenska, T.; Kunesova, M.; Pihlsgard, M.; et al. Diets with high or low protein content and glycemic index for weight-loss maintenance. N. Engl. J. Med. 2010, 363, 2102–2113. [Google Scholar] [CrossRef] [PubMed]
- Gaffney-Stomberg, E.; Cao, J.J.; Lin, G.G.; Wulff, C.R.; Murphy, N.E.; Young, A.J.; McClung, J.P.; Pasiakos, S.M. Dietary protein level and source differentially affect bone metabolism, strength, and intestinal calcium transporter expression during ad libitum and food-restricted conditions in male rats. J. Nutr. 2014, 144, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Van der Poorten, D.; Milner, K.L.; Hui, J.; Hodge, A.; Trenell, M.I.; Kench, J.G.; London, R.; Peduto, T.; Chisholm, D.J.; George, J. Visceral fat: A key mediator of steatohepatitis in metabolic liver disease. Hepatology 2008, 48, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Perry, R.J.; Samuel, V.T.; Petersen, K.F.; Shulman, G.I. The role of hepatic lipids in hepatic insulin resistance and type 2 diabetes. Nature 2014, 510, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Shulman, G.I. Cellular mechanisms of insulin resistance. J. Clin. Investig. 2000, 106, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Birkenfeld, A.L.; Shulman, G.I. Nonalcoholic fatty liver disease, hepatic insulin resistance, and type 2 diabetes. Hepatology 2014, 59, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Rietman, A.; Schwarz, J.; Blokker, B.A.; Siebelink, E.; Kok, F.J.; Afman, L.A.; Tome, D.; Mensink, M. Increasing protein intake modulates lipid metabolism in healthy young men and women consuming a high-fat hypercaloric diet. J. Nutr. 2014, 144, 1174–1180. [Google Scholar] [CrossRef] [PubMed]
- Westerbacka, J.; Lammi, K.; Hakkinen, A.M.; Rissanen, A.; Salminen, I.; Aro, A.; Yki-Jarvinen, H. Dietary fat content modifies liver fat in overweight nondiabetic subjects. J. Clin. Endocrinol. Metabol. 2005, 90, 2804–2809. [Google Scholar] [CrossRef] [PubMed]
- Krebs, M.; Krssak, M.; Bernroider, E.; Anderwald, C.; Brehm, A.; Meyerspeer, M.; Nowotny, P.; Roth, E.; Waldhausl, W.; Roden, M. Mechanism of amino acid-induced skeletal muscle insulin resistance in humans. Diabetes 2002, 51, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Solon-Biet, S.M.; Mitchell, S.J.; Coogan, S.C.; Cogger, V.C.; Gokarn, R.; McMahon, A.C.; Raubenheimer, D.; de Cabo, R.; Simpson, S.J.; Le Couteur, D.G. Dietary protein to carbohydrate ratio and caloric restriction: Comparing metabolic outcomes in mice. Cell Rep. 2015, 11, 1529–1534. [Google Scholar] [CrossRef] [PubMed]
- Huffman, K.M.; Shah, S.H.; Stevens, R.D.; Bain, J.R.; Muehlbauer, M.; Slentz, C.A.; Tanner, C.J.; Kuchibhatla, M.; Houmard, J.A.; Newgard, C.B.; et al. Relationships between circulating metabolic intermediates and insulin action in overweight to obese, inactive men and women. Diabetes Care 2009, 32, 1678–1683. [Google Scholar] [CrossRef] [PubMed]
- Shaham, O.; Wei, R.; Wang, T.J.; Ricciardi, C.; Lewis, G.D.; Vasan, R.S.; Carr, S.A.; Thadhani, R.; Gerszten, R.E.; Mootha, V.K. Metabolic profiling of the human response to a glucose challenge reveals distinct axes of insulin sensitivity. Mol. Syst. Biol. 2008, 4, 214. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B.; An, J.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Lien, L.F.; Haqq, A.M.; Shah, S.H.; Arlotto, M.; Slentz, C.A.; et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metabol. 2009, 9, 311–326. [Google Scholar] [CrossRef] [PubMed]
- Krezowski, P.A.; Nuttall, F.Q.; Gannon, M.C.; Bartosh, N.H. The effect of protein ingestion on the metabolic response to oral glucose in normal individuals. Am. J. Clin. Nutr. 1986, 44, 847–856. [Google Scholar] [PubMed]
- Baum, J.I.; Layman, D.K.; Freund, G.G.; Rahn, K.A.; Nakamura, M.T.; Yudell, B.E. A reduced carbohydrate, increased protein diet stabilizes glycemic control and minimizes adipose tissue glucose disposal in rats. J. Nutr. 2006, 136, 1855–1861. [Google Scholar] [PubMed]
- Guo, F.; Cavener, D.R. The GCN2 eIF2α kinase regulates fatty-acid homeostasis in the liver during deprivation of an essential amino acid. Cell Metabol. 2007, 5, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F.; Huang, Z.; Li, H.; Yu, J.; Wang, C.; Chen, S.; Meng, Q.; Cheng, Y.; Gao, X.; Li, J.; et al. Leucine deprivation increases hepatic insulin sensitivity via GCN2/mTOR/S6K1 and AMPK pathways. Diabetes 2011, 60, 746–756. [Google Scholar] [CrossRef] [PubMed]
- Bowker-Kinley, M.M.; Davis, W.I.; Wu, P.; Harris, R.A.; Popov, K.M. Evidence for existence of tissue-specific regulation of the mammalian pyruvate dehydrogenase complex. Biochem. J. 1998, 329 Pt 1, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.I.; Lee, F.N.; Choi, W.S.; Lee, S.; Youn, J.H. Insulin regulation of skeletal muscle PDK4 mRNA expression is impaired in acute insulin-resistant states. Diabetes 2006, 55, 2311–2317. [Google Scholar] [CrossRef] [PubMed]
- Rosa, G.; Mingrone, G.; Manco, M.; Euthine, V.; Gniuli, D.; Calvani, R.; Calvani, M.; Favuzzi, A.M.; Castagneto, M.; Vidal, H. Molecular mechanisms of diabetes reversibility after bariatric surgery. Int. J. Obes. 2007, 31, 1429–1436. [Google Scholar] [CrossRef] [PubMed]
- Rosa, G.; Di Rocco, P.; Manco, M.; Greco, A.V.; Castagneto, M.; Vidal, H.; Mingrone, G. Reduced PDK4 expression associates with increased insulin sensitivity in postobese patients. Obes. Res. 2003, 11, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, J.M.; Neese, R.A.; Turner, S.; Dare, D.; Hellerstein, M.K. Short-term alterations in carbohydrate energy intake in humans. Striking effects on hepatic glucose production, de novo lipogenesis, lipolysis, and whole-body fuel selection. J. Clin. Investig. 1995, 96, 2735–2743. [Google Scholar] [CrossRef] [PubMed]
- Semenkovich, C.F. Regulation of fatty acid synthase (FAS). Prog. Lipid Res. 1997, 36, 43–53. [Google Scholar] [CrossRef]
- Mangravite, L.M.; Dawson, K.; Davis, R.R.; Gregg, J.P.; Krauss, R.M. Fatty acid desaturase regulation in adipose tissue by dietary composition is independent of weight loss and is correlated with the plasma triacylglycerol response. Am. J. Clin. Nutr. 2007, 86, 759–767. [Google Scholar] [PubMed]
- McDevitt, R.M.; Bott, S.J.; Harding, M.; Coward, W.A.; Bluck, L.J.; Prentice, A.M. De novo lipogenesis during controlled overfeeding with sucrose or glucose in lean and obese women. Am. J. Clin. Nutr. 2001, 74, 737–746. [Google Scholar] [PubMed]
- Parks, E.J.; Hellerstein, M.K. Carbohydrate-induced hypertriacylglycerolemia: Historical perspective and review of biological mechanisms. Am. J. Clin. Nutr. 2000, 71, 412–433. [Google Scholar] [PubMed]
- Blouet, C.; Mariotti, F.; Azzout-Marniche, D.; Bos, C.; Mathe, V.; Tome, D.; Huneau, J.F. The reduced energy intake of rats fed a high-protein low-carbohydrate diet explains the lower fat deposition, but macronutrient substitution accounts for the improved glycemic control. J. Nutr. 2006, 136, 1849–1854. [Google Scholar] [PubMed]
- Uebanso, T.; Taketani, Y.; Fukaya, M.; Sato, K.; Takei, Y.; Sato, T.; Sawada, N.; Amo, K.; Harada, N.; Arai, H.; et al. Hypocaloric high-protein diet improves fatty liver and hypertriglyceridemia in sucrose-fed obese rats via two pathways. Am. J. Physiol. Endocrinol. Metab. 2009, 297, 76–84. [Google Scholar] [CrossRef] [PubMed]


| Analytes | Ad Libitum | Calorie Restriction | p Value | ||||
|---|---|---|---|---|---|---|---|
| 10% PRO | 32% PRO | 10% PRO | 32% PRO | Calorie | Protein | C × P 2 | |
| Glucagon (ng·L−1) | 50.7 ± 4.2 | 55.3 ± 5.4 | 49.8 ± 3.2 | 50.3 ± 3.3 | 0.48 | 0.55 | 0.62 |
| Leptin (mmol·L−1) | 3912.4 ± 336.7 | 3019.4 ± 327.8 | 2035.0 ± 184.7 | 1756.3 ± 184.7 | <0.01 * | 0.03 † | 0.26 |
| Triglycerides (mmol·L−1) | 0.95 ± 0.12 | 0.83 ± 0.11 | 0.69 ± 0.03 | 0.57 ± 0.07 | <0.01 * | 0.22 | 0.98 |
| Glucose (mmol·L−1) | 8.0 ± 0.9 | 7.0 ± 0.8 | 8.3 ± 1.0 | 7.0 ± 0.8 | 0.83 | 0.19 | 0.90 |
| Insulin (pmol·L−1) | 234.1 ± 37.7 | 161.7 ± 26.9 | 144.3 ± 24.5 | 137.6 ± 24.5 | 0.04 * | 0.15 | 0.23 |
| HOMA-IR 3 | 12.7 ± 3.3 | 7.2 ± 1.2 | 8.4 ± 2.0 | 6.3 ± 1.0 | 0.22 | 0.08 | 0.40 |
| HOMA-B 4 | 58.5 ± 9.0 | 49.3 ± 8.1 | 35.4 ± 5.5 | 41.4 ± 5.5 | 0.04 * | 0.83 | 0.30 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Margolis, L.M.; Rivas, D.A.; Ezzyat, Y.; Gaffney-Stomberg, E.; Young, A.J.; McClung, J.P.; Fielding, R.A.; Pasiakos, S.M. Calorie Restricted High Protein Diets Downregulate Lipogenesis and Lower Intrahepatic Triglyceride Concentrations in Male Rats. Nutrients 2016, 8, 571. https://doi.org/10.3390/nu8090571
Margolis LM, Rivas DA, Ezzyat Y, Gaffney-Stomberg E, Young AJ, McClung JP, Fielding RA, Pasiakos SM. Calorie Restricted High Protein Diets Downregulate Lipogenesis and Lower Intrahepatic Triglyceride Concentrations in Male Rats. Nutrients. 2016; 8(9):571. https://doi.org/10.3390/nu8090571
Chicago/Turabian StyleMargolis, Lee M., Donato A. Rivas, Yassine Ezzyat, Erin Gaffney-Stomberg, Andrew J. Young, James P. McClung, Roger A. Fielding, and Stefan M. Pasiakos. 2016. "Calorie Restricted High Protein Diets Downregulate Lipogenesis and Lower Intrahepatic Triglyceride Concentrations in Male Rats" Nutrients 8, no. 9: 571. https://doi.org/10.3390/nu8090571
APA StyleMargolis, L. M., Rivas, D. A., Ezzyat, Y., Gaffney-Stomberg, E., Young, A. J., McClung, J. P., Fielding, R. A., & Pasiakos, S. M. (2016). Calorie Restricted High Protein Diets Downregulate Lipogenesis and Lower Intrahepatic Triglyceride Concentrations in Male Rats. Nutrients, 8(9), 571. https://doi.org/10.3390/nu8090571




