The Effect of Casein Protein Prior to Sleep on Fat Metabolism in Obese Men
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.2.1. Morning Visits
Anthropometrics and Body Composition
Appetite
Metabolism
Blood Sampling and Analysis
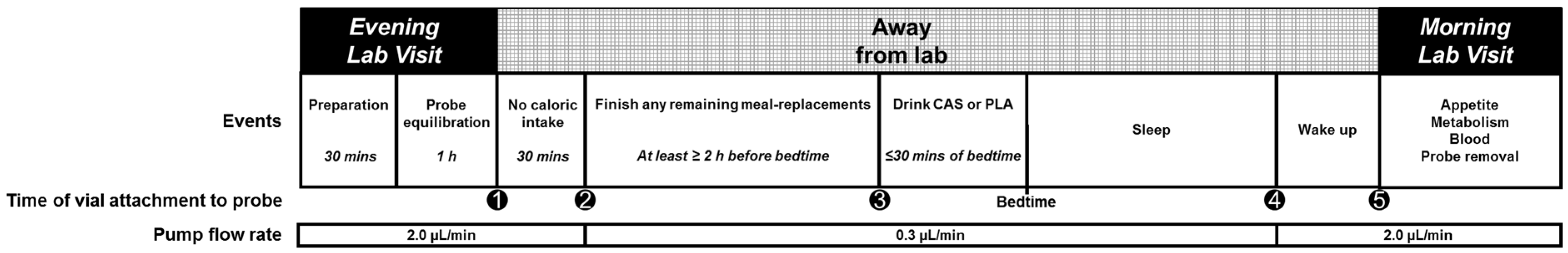
2.2.2. Evening Visits
Microdialysis
Calculation for Interstitial Glycerol and Glucose
Blood Flow
2.2.3. Dietary Logs and Standardization
Nighttime Supplementation
2.2.4. Sleep Quantity and Quality Assessment
2.2.5. Statistical Analyses
3. Results
3.1. Descriptive Characteristics
3.2. Appetite
3.3. Metabolism
3.4. Blood Markers
3.5. Sleep Quantity and Quality
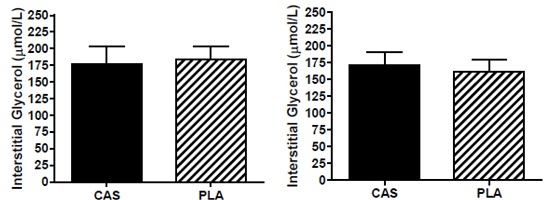
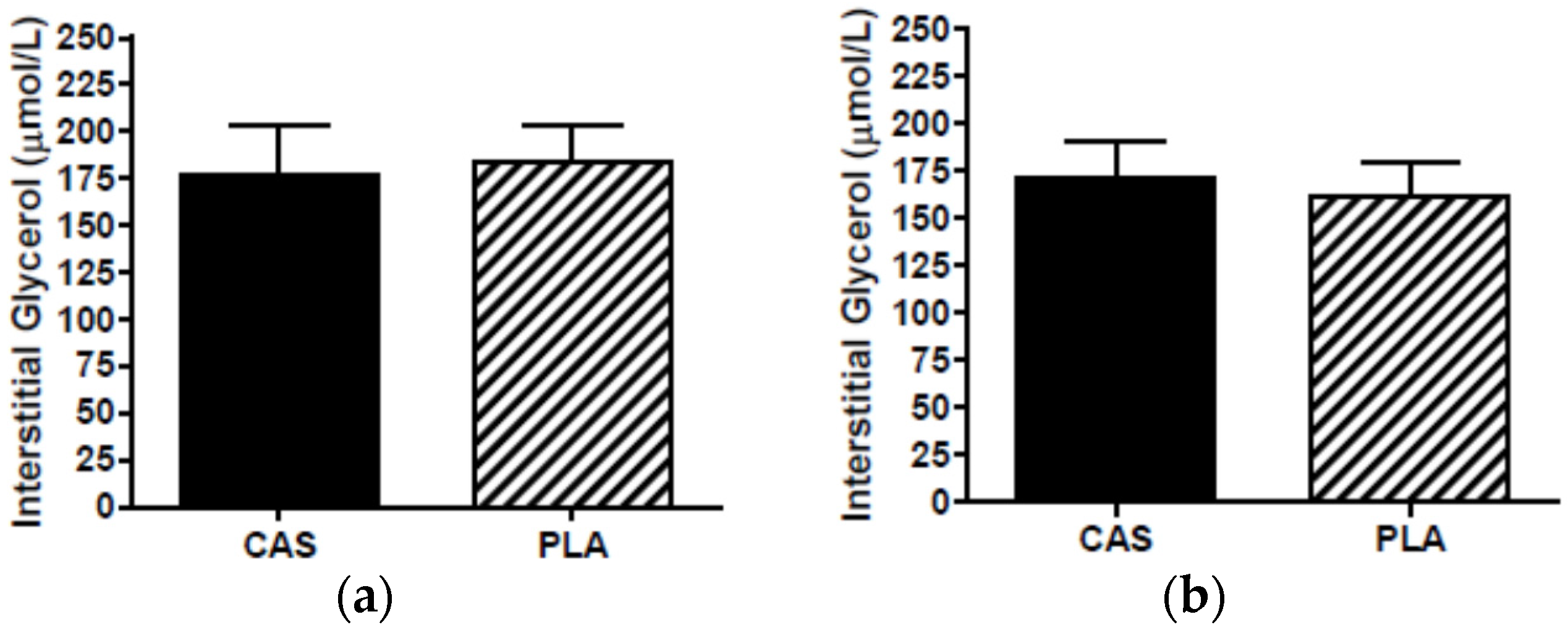
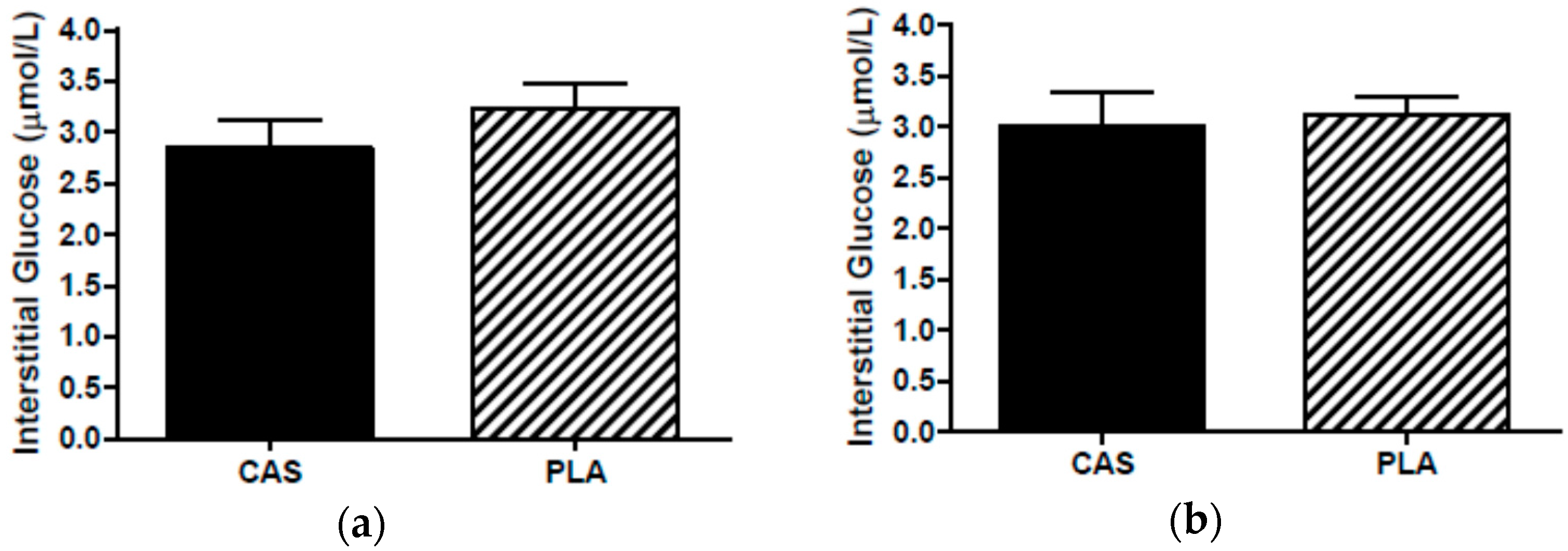
3.6. SCAAT Interstitial Glycerol and Glucose
3.7. Blood Flow
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CAS | Casein protein |
| REE | Resting energy expenditure |
| PLA | Placebo |
| SCAAT | Subcutaneous abdominal adipose tissue |
| DXA | Dual energy X-ray absorptiometry |
| VAS | Visual analog scale |
| RER | Respiratory exchange ratio |
| HOMA-IR | Homeostatic model assessment of insulin resistance |
References
- Andersen, G.S.; Stunkard, A.J.; Sørensen, T.I.; Petersen, L.; Heitmann, B.L. Night eating and weight change in middle-aged men and women. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 1338–1343. [Google Scholar] [CrossRef] [PubMed]
- Gluck, M.E.; Venti, C.A.; Salbe, A.D.; Krakoff, J. Nighttime eating: Commonly observed and related to weight gain in an inpatient food intake study. Am. J. Clin. Nutr. 2008, 88, 900–905. [Google Scholar] [PubMed]
- Arble, D.M.; Bass, J.; Laposky, A.D.; Vitaterna, M.H.; Turek, F.W. Circadian timing of food intake contributes to weight gain. Obesity 2009, 17, 2100–2102. [Google Scholar] [CrossRef] [PubMed]
- Costa, G. The problem: Shiftwork. Chronobiol. Int. 1997, 14, 89–98. [Google Scholar] [CrossRef] [PubMed]
- De Zwaan, M.; Burgard, M.A.; Schenck, C.H.; Mitchell, J.E. Night time eating: A review of the literature. Eur. Eat. Disord. Rev. 2003, 24, 7–24. [Google Scholar] [CrossRef]
- Kinsey, A.W.; Ormsbee, M.J. The health impact of nighttime eating: Old and new perspectives. Nutrients 2015, 7, 2648–2662. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, A.; Wong, A.; Kinsey, A.; Kalfon, R.; Eddy, W.; Ormsbee, M.J. Effects of milk proteins and combined exercise training on aortic hemodynamics and arterial stiffness in young obese women with high blood pressure. Am. J. Hypertens. 2014, 27, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Kinsey, A.W.; Eddy, W.R.; Madzima, T.A.; Panton, L.B.; Arciero, P.J.; Kim, J.-S.; Ormsbee, M.J. Influence of night-time protein and carbohydrate intake on appetite and cardiometabolic risk in sedentary overweight and obese women. Br. J. Nutr. 2014, 112, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Madzima, T.A.; Panton, L.B.; Fretti, S.K.; Kinsey, A.W.; Ormsbee, M.J. Night-time consumption of protein or carbohydrate results in increased morning resting energy expenditure in active college-aged men. Br. J. Nutr. 2014, 111, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Ormsbee, M.J.; Kinsey, A.W.; Eddy, W.R.; Madzima, T.A.; Arciero, P.J.; Figueroa, A.; Panton, L.B. The influence of nighttime feeding of carbohydrate or protein combined with exercise training on appetite and cardiometabolic risk in young obese women. Appl. Physiol. Nutr. Metab. 2015, 40, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Res, P.T.; Groen, B.; Pennings, B.; Beelen, M.; Wallis, G.A.; Gijsen, A.P.; Senden, J.M.; van Loon, L.J. Protein 481 ingestion before sleep improves postexercise overnight recovery. Med. Sci. Sports Exerc. 2012, 44, 1560–1569. [Google Scholar] [CrossRef] [PubMed]
- Groen, B.B.; Res, P.T.; Pennings, B.; Hertle, E.; Senden, J.M.; Saris, W.H.; van Loon, L.J. Intragastric protein administration stimulates overnight muscle protein synthesis in elderly men. Am. J. Physiol. Endocrinol. Metab. 2012, 302, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Snijders, T.; Res, P.T.; Smeets, J.S.; van Vliet, S.; van Kranenburg, J.; Maase, K.; Kies, A.K.; Verdijk, L.B.; van Loon, L.J. Protein ingestion before sleep increases muscle mass and strength gains during prolonged resistance-type exercise training in healthy young men. J. Nutr. 2015, 145, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Astrup, A. The satiating power of protein—A key to obesity prevention? Am. J. Clin. Nutr. 2005, 82, 1–2. [Google Scholar] [PubMed]
- Lorenzen, J.; Frederiksen, R.; Hoppe, C.; Hvid, R.; Astrup, A. The effect of milk proteins on appetite regulation and diet-induced thermogenesis. Eur. J. Clin. Nutr. 2012, 66, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Pennings, B.; Boirie, Y.; Senden, J.M.G.; Gijsen, A.P.; Kuipers, H.; van Loon, L.J. Whey protein stimulates postprandial muscle protein accretion more effectively than do casein and casein hydrolysate in older men. Am. J. Clin. Nutr. 2011, 93, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- Arciero, P.J.; Ormsbee, M.J.; Gentile, C.L.; Nindl, B.C.; Brestoff, J.R.; Ruby, M. Increased protein intake and meal frequency reduces abdominal fat during energy balance and energy deficit. Obesity 2013, 21, 1357–1366. [Google Scholar] [CrossRef] [PubMed]
- Marsset-Baglieri, A.; Fromentin, G.; Airinei, G.; Pedersen, C.; Léonil, J.; Piedcoq, J.; Rémond, D.; Benamouzig, R.; Tomé, D.; Gaudichon, C. Milk protein fractions moderately extend the duration of satiety compared with carbohydrates independently of their digestive kinetics in overweight subjects. Br. J. Nutr. 2014, 112, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Boirie, Y.; Dangin, M.; Gachon, P.; Vasson, M.P.; Maubois, J.L.; Beaufrère, B. Slow and fast dietary proteins differently modulate postprandial protein accretion. Proc. Natl. Acad. Sci. USA 1997, 94, 14930–14935. [Google Scholar] [CrossRef] [PubMed]
- Dangin, M.; Boirie, Y.; Garcia-Rodenas, C.; Gachon, P.; Fauquant, J.; Callier, P.; Ballèvre, O.; Beaufrère, B. The digestion rate of protein is an independent regulating factor of postprandial protein retention. Am. J. Physiol. Endocrinol. Metab. 2001, 280, 340–348. [Google Scholar]
- Hibi, M.; Masumoto, A.; Naito, Y.; Kiuchi, K.; Yoshimoto, Y.; Matsumoto, M.; Katashima, M.; Oka, J.; Ikemoto, S. Nighttime snacking reduces whole body fat oxidation and increases LDL cholesterol in healthy young women. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.D. Adipose tissue and fatty acid metabolism in humans. J. R. Soc. Med. 2002, 95, 3–7. [Google Scholar] [PubMed]
- Random.org. Availible online: https://www.random.org/ (accessed on 9 November 2014).
- Flint, A.; Raben, A.; Blundell, J.E.; Astrup, A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.A. Measurement of energy expenditure. Public Health Nutr. 2005, 8, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Frayn, K.N. Calculation of substrate oxidation rates in vivo from gaseous exchange. J. Appl. Physiol. 1983, 55, 628–634. [Google Scholar] [PubMed]
- Achten, J.; Jeukendrup, A.E. Maximal fat oxidation during exercise in trained men. Int. J. Sports Med. 2003, 24, 603–608. [Google Scholar] [PubMed]
- Lima-Silva, A.E.; Bertuzzi, R.C.; Pires, F.O.; Gagliardi, J.F.; Barros, R.V.; Hammond, J.; Kiss, M.A. Relationship between training status and maximal fat oxidation rate. J. Sports Sci. Med. 2010, 9, 31–35. [Google Scholar] [PubMed]
- Liu, J.; Prudom, C.E.; Nass, R.; Pezzoli, S.S.; Oliveri, M.C.; Johnson, M.L.; Veldhuis, P.; Gordon, D.A.; Howard, A.D.; Witcher, D.R.; et al. Novel ghrelin assays provide evidence for independent regulation of ghrelin acylation and secretion in healthy young men. J. Clin. Endocrinol. Metab. 2008, 93, 1980–1987. [Google Scholar] [CrossRef] [PubMed]
- Prudom, C.; Liu, J.; Patrie, J.; Gaylinn, B.D.; Foster-Schubert, K.E.; Cummings, D.E.; Thorner, M.O.; Geysen, H.M. Comparison of competitive radioimmunoassays and two-site sandwich assays for the measurement and interpretation of plasma ghrelin levels. J. Clin. Endocrinol. Metab. 2010, 95, 2351–2358. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Hickner, R.C.; Rosdahl, H.; Borg, I.; Ungerstedt, U.; Jorfeldt, L.; Henriksson, J. The ethanol technique of monitoring local blood flow changes in rat skeletal muscle: Implications for microdialysis. Acta Physiol. Scand. 1992, 146, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Howe, H.R.; Heidal, K.; Choi, M.D.; Kraus, R.M.; Boyle, K.; Hickner, R.C. Increased adipose tissue lipolysis after a 2-week high-fat diet in sedentary overweight/obese men. Metabolism 2011, 60, 976–981. [Google Scholar] [CrossRef] [PubMed]
- Shippenberg, T.S.; Thompson, A.C. Overview of microdialysis. Curr. Protoc. Neurosci. 2001. [Google Scholar] [CrossRef]
- De Lange, E.C.M. Recovery and calibration techniques: Toward quantitative microdialysis. In Microdialysis in Drug Development; Springer: New York, NY, USA, 2013; Volume 4, pp. 13–33. [Google Scholar]
- Ormsbee, M.J.; Choi, M.D.; Medlin, J.K.; Geyer, G.H.; Trantham, L.H.; Dubis, G.S.; Hickner, R.C. Regulation of fat metabolism during resistance exercise in sedentary lean and obese men. J. Appl. Physiol. 2009, 106, 1529–1537. [Google Scholar] [CrossRef] [PubMed]
- Hickner, R.C.; Rosdahl, H.; Borg, I.; Ungerstedt, U.; Jorfeldt, L.; Henriksson, J. Ethanol may be used with the microdialysis technique to monitor blood flow changes in skeletal muscle: Dialysate glucose concentration is blood-flow-dependent. Acta Physiol. Scand. 1991, 143, 355–356. [Google Scholar] [CrossRef] [PubMed]
- United States Department of Agriculture. Super Tracker. Availible online: https://www.supertracker.usda.gov/default.aspx (accessed on 14 May 2015).
- Gropper, S.S.; Smith, J.L. Advanced Nutrition and Human Metabolism, 6th ed.; Wadsworth, Cengage Learning: Belmont, CA, USA, 2012. [Google Scholar]
- Noor, Z.M.; Smith, A.J.; Smith, S.S.; Nissen, L.M. Feasibility and acceptability of wrist actigraph in assessing sleep quality and sleep quantity: A home-based pilot study in healthy volunteers. Health 2013, 5, 63–72. [Google Scholar] [CrossRef]
- Russell, C.A.; Caldwell, J.A.; Myers, L.J.; Wubbels, P.; Downs, H. Validation of the Fatigue Science Readiband Actigraph and Associated Sleep/Wake Classification Algorithms. Available online: http://static1.squarespace.com/static/550af02ae4b0cf85628d981a/t/5526c99ee4b019412c323758/1428605342303/Readiband_Validation.pdf (accessed on 25 July 2016).
- Peters, B. What is Sleep Efficiency? Availible online: https://www.verywell.com/sleep-efficiency-3014912 (accessed on 25 April 2016).
- Lavinsky, L.; Oliveira, M.W.; Bassanesi, H.J.C.; D’Avila, C.; Lavinsky, M. Hyperinsulinemia and tinnitus: A historical cohort. Int. Tinnitus J. 2004, 10, 24–30. [Google Scholar] [PubMed]
- Ravussin, E.; Lillioja, S.; Knowler, W.C.; Christin, L.; Freymond, D.; Abbott, W.G.; Boyce, V.; Howard, B.V.; Bogardus, C. Reduced rate of energy expenditure as a risk factor for body-weight gain. N. Engl. J. Med. 1988, 318, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Acheson, K.J.; Blondel-Lubrano, A.; Oguey-Araymon, S.; Beaumont, M.; Emady-Azar, S.; Ammon-Zufferey, C.; Monnard, I.; Pinaud, S.; Nielsen-Moennoz, C.; Bovetto, L. Protein choices targeting thermogenesis and metabolism. Am. J. Clin. Nutr. 2011, 93, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Langin, D. Control of fatty acid and glycerol release in adipose tissue lipolysis. C. R. Biol. 2006, 329, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Boyle, P.J.; Avogaro, A.; Smith, L.; Bier, D.M.; Pappu, A.S.; Illingworth, D.R.; Cryer, P.E. Role of GH in regulating nocturnal rates of lipolysis and plasma mevalonate levels in normal and diabetic humans. Am. J. Physiol. 1992, 263, 168–172. [Google Scholar]
- Jensen, M.D.; Bajnárek, J.; Lee, S.Y.; Nielsen, S.; Koutsari, C. Relationship between postabsorptive respiratory exchange ratio and plasma free fatty acid concentrations. J. Lipid Res. 2009, 50, 1863–1869. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.N.; Neylan, T.C.; Grunfeld, C.; Mulligan, K.; Schambelan, M.; Schwarz, J.-M. Subchronic sleep restriction causes tissue-specific insulin resistance. J. Clin. Endocrinol. Metab. 2015, 100, 1664–1671. [Google Scholar] [CrossRef] [PubMed]
- Tschop, M.; Weyer, C.; Tataranni, P.A.; Devanarayan, V.; Ravussin, E.; Heiman, M.L. Circulating ghrelin levels are decreased in human obesity. Diabetes 2001, 50, 707–709. [Google Scholar] [CrossRef] [PubMed]
- Druce, M.R.; Wren, A.M.; Park, A.J.; Milton, J.E.; Patterson, M.; Frost, G.; Ghatei, M.A.; Small, C.; Bloom, S.R. Ghrelin increases food intake in obese as well as lean subjects. Int. J. Obes. 2005, 29, 1130–1136. [Google Scholar] [CrossRef] [PubMed]
- Pencharz, P.B.; Elango, R.; Wolfe, R.R. Recent developments in understanding protein needs—How much and what kind should we eat? Appl. Physiol. Nutr. Metab. 2016, 41, 577–580. [Google Scholar] [CrossRef] [PubMed]
| n = 12 | |
|---|---|
| Age (years) | 27.0 ± 2.2 |
| Height (cm) | 176.0 ± 2.0 |
| Weight (kg) | 112.0 ± 6.6 |
| BMI (kg/m2) | 36.1 ± 1.9 |
| Body Fat (%) | 36.7 ± 1.8 |
| Android Fat (%) | 43.8 ± 1.9 |
| A/G ratio | 1.19 ± 0.03 |
| Fat Mass (kg) | 41.4 ± 4.3 |
| Lean Mass (kg) | 66.0 ± 2.7 |
| Habitual | Controlled | |
|---|---|---|
| Food Intake (kcals) | 2489 ± 191 ^ | 2795 ± 154 |
| Carbohydrate | ||
| % | 43.8 ± 2.0 ^ | 57.0 |
| grams | 274.2 ± 25.9 | 398.2 ± 22.0 * |
| Protein | ||
| % | 16.8 ± 1.1 ^ | 15.0 |
| grams | 103.2 ± 9.3 | 104.8 ± 5.8 |
| grams/kg/day | 0.97 ± 0.1 | 0.95 ± 0.1 |
| Fat | ||
| % | 39.2 ± 2.2 ^ | 28.0 |
| grams | 107.7 ± 10.2 | 86.9 ± 16.6 |
| Baseline | CAS | PLA | |
|---|---|---|---|
| Insulin (pmol/L) | 202.2 ± 38.4 | 197.4 ± 37.2 | 189.6 ± 29.4 |
| Glucose (mmol/L) ^ | 5.0 ± 0.2 | 4.9 ± 0.2 | 5.0 ± 0.2 |
| HOMA-IR ^ | 7.9 ± 1.9 | 7.3 ± 1.6 | 7.1 ± 1.3 |
| Growth Hormone (μg/L) # | 0.16 ± 0.08 | 0.15 ± 0.07 | 0.11 ± 0.03 |
| Acyl Ghrelin (pmol/L) | 184.7 ± 53.3 | 183.0 ± 33.7 | 206.6 ± 51.2 |
| Des Ghrelin (pmol/L) | 148.0 ± 30.9 | 132.0 ± 24.5 | 143.5 ± 31.6 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kinsey, A.W.; Cappadona, S.R.; Panton, L.B.; Allman, B.R.; Contreras, R.J.; Hickner, R.C.; Ormsbee, M.J. The Effect of Casein Protein Prior to Sleep on Fat Metabolism in Obese Men. Nutrients 2016, 8, 452. https://doi.org/10.3390/nu8080452
Kinsey AW, Cappadona SR, Panton LB, Allman BR, Contreras RJ, Hickner RC, Ormsbee MJ. The Effect of Casein Protein Prior to Sleep on Fat Metabolism in Obese Men. Nutrients. 2016; 8(8):452. https://doi.org/10.3390/nu8080452
Chicago/Turabian StyleKinsey, Amber W., Stacy R. Cappadona, Lynn B. Panton, Brittany R. Allman, Robert J. Contreras, Robert C. Hickner, and Michael J. Ormsbee. 2016. "The Effect of Casein Protein Prior to Sleep on Fat Metabolism in Obese Men" Nutrients 8, no. 8: 452. https://doi.org/10.3390/nu8080452
APA StyleKinsey, A. W., Cappadona, S. R., Panton, L. B., Allman, B. R., Contreras, R. J., Hickner, R. C., & Ormsbee, M. J. (2016). The Effect of Casein Protein Prior to Sleep on Fat Metabolism in Obese Men. Nutrients, 8(8), 452. https://doi.org/10.3390/nu8080452