Chemopreventive Effect of Dietary Glutamineon Colitis-Associated Colorectal Cancer Is Associated with Modulation of the DEPTOR/mTOR Signaling Pathway
Abstract
:1. Introduction
2. Experimental Section
2.1. Animal Experiments
2.2. Pathological Examination
2.3. Western Blot Analysis
2.4. Statistical Analyses
3. Results
3.1. GLN Protects against CAC
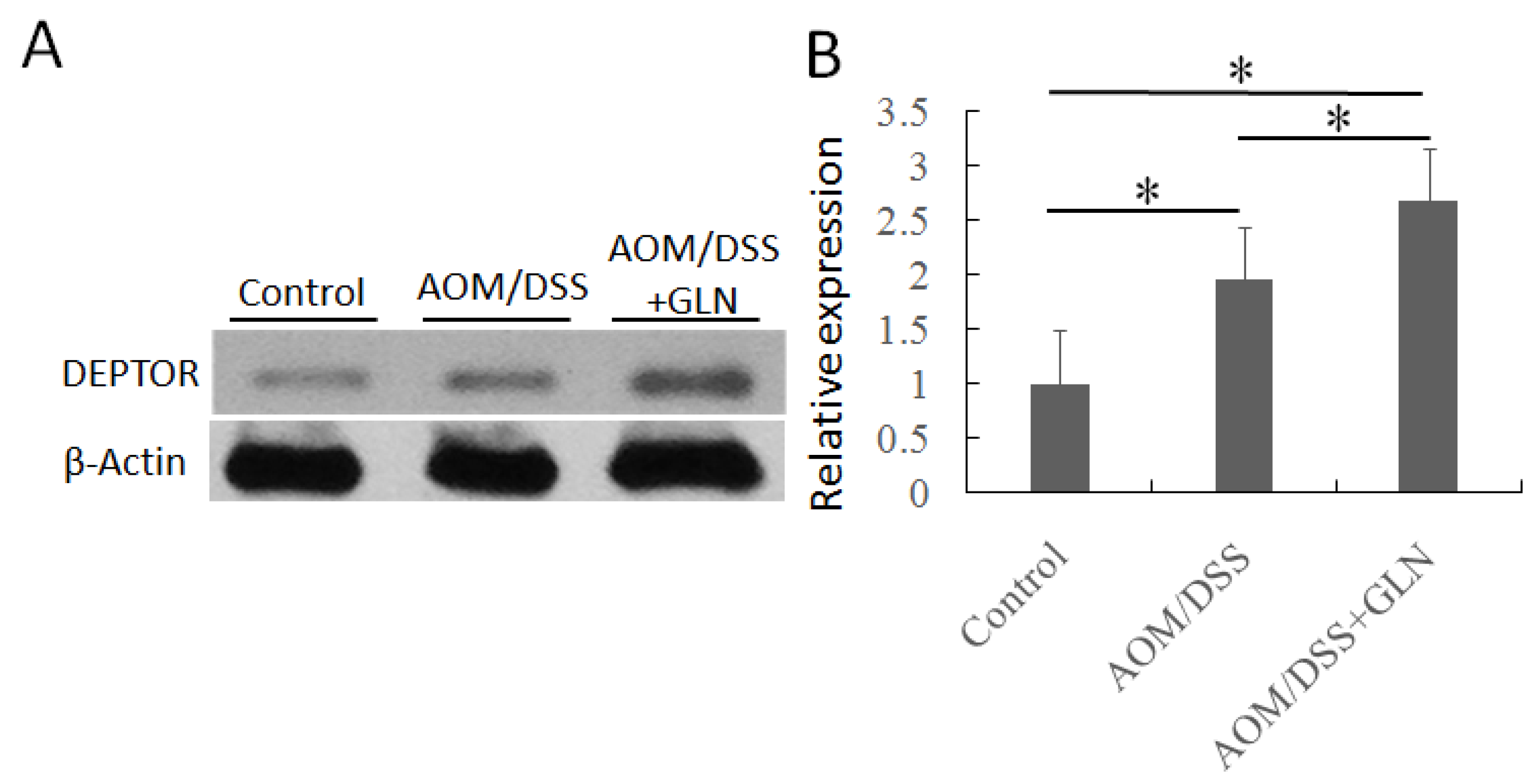
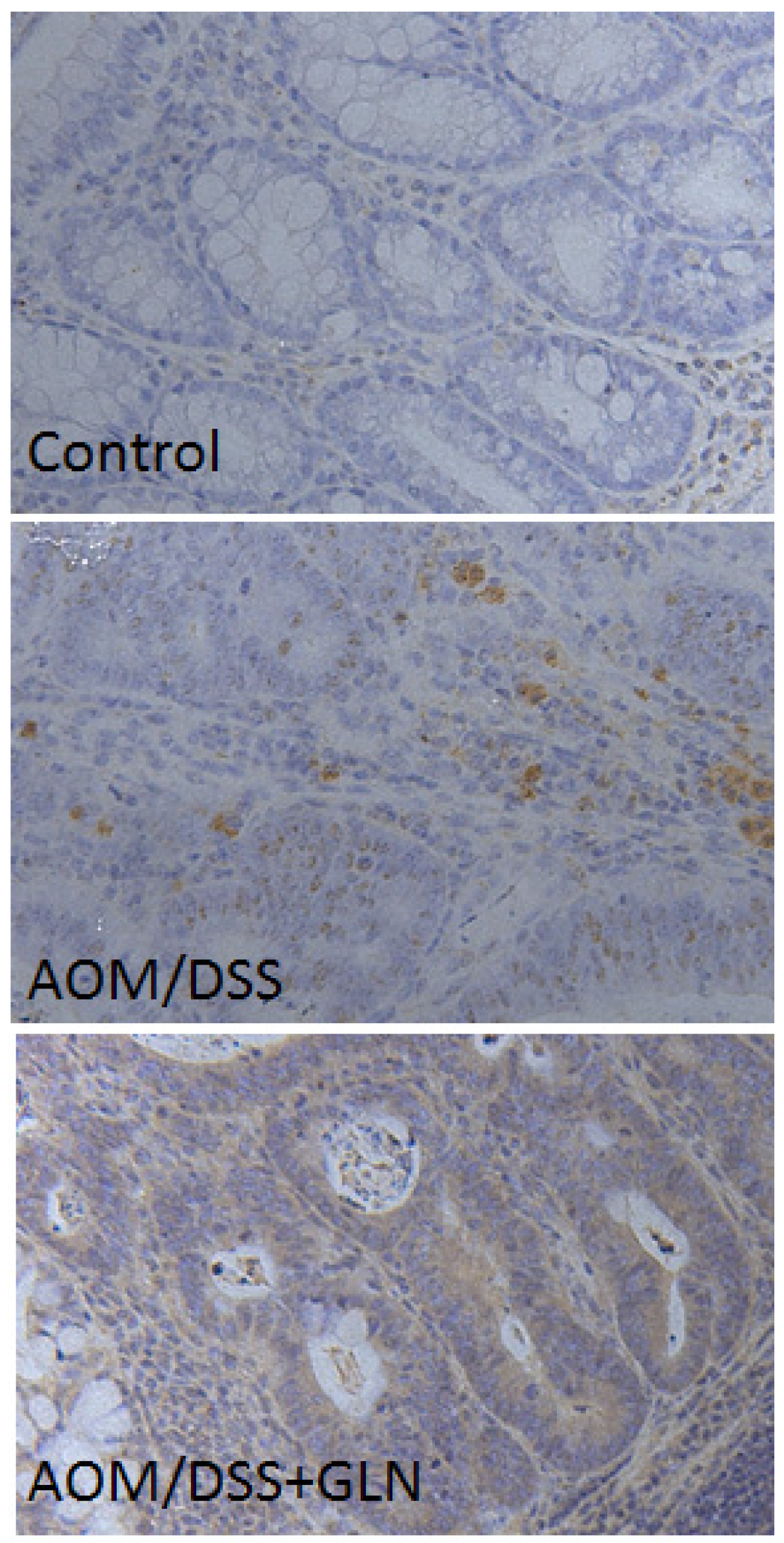
3.2. GLN Promotes DEPTOR Expression
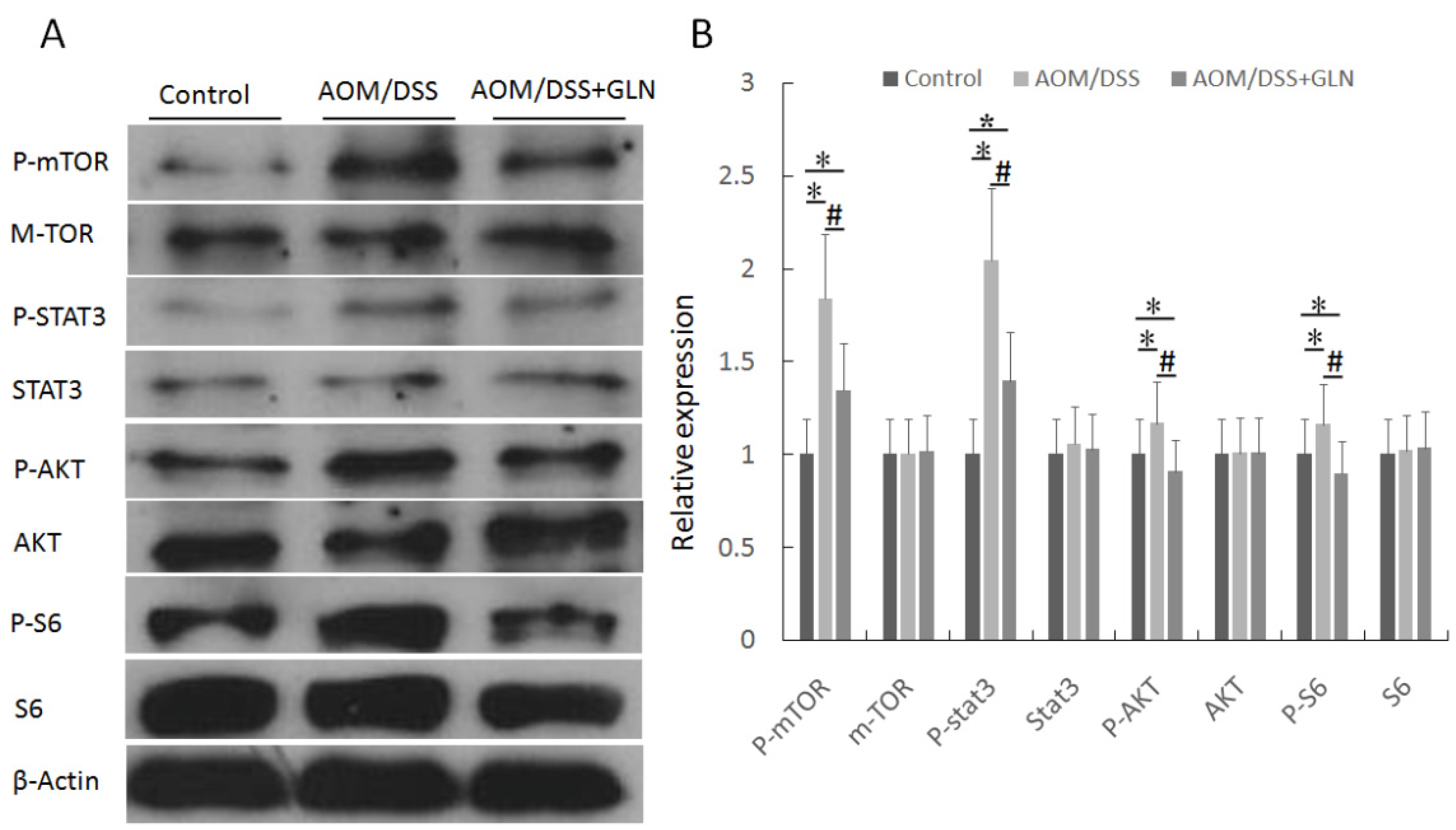
3.3. GLN Regulates mTOR Signaling
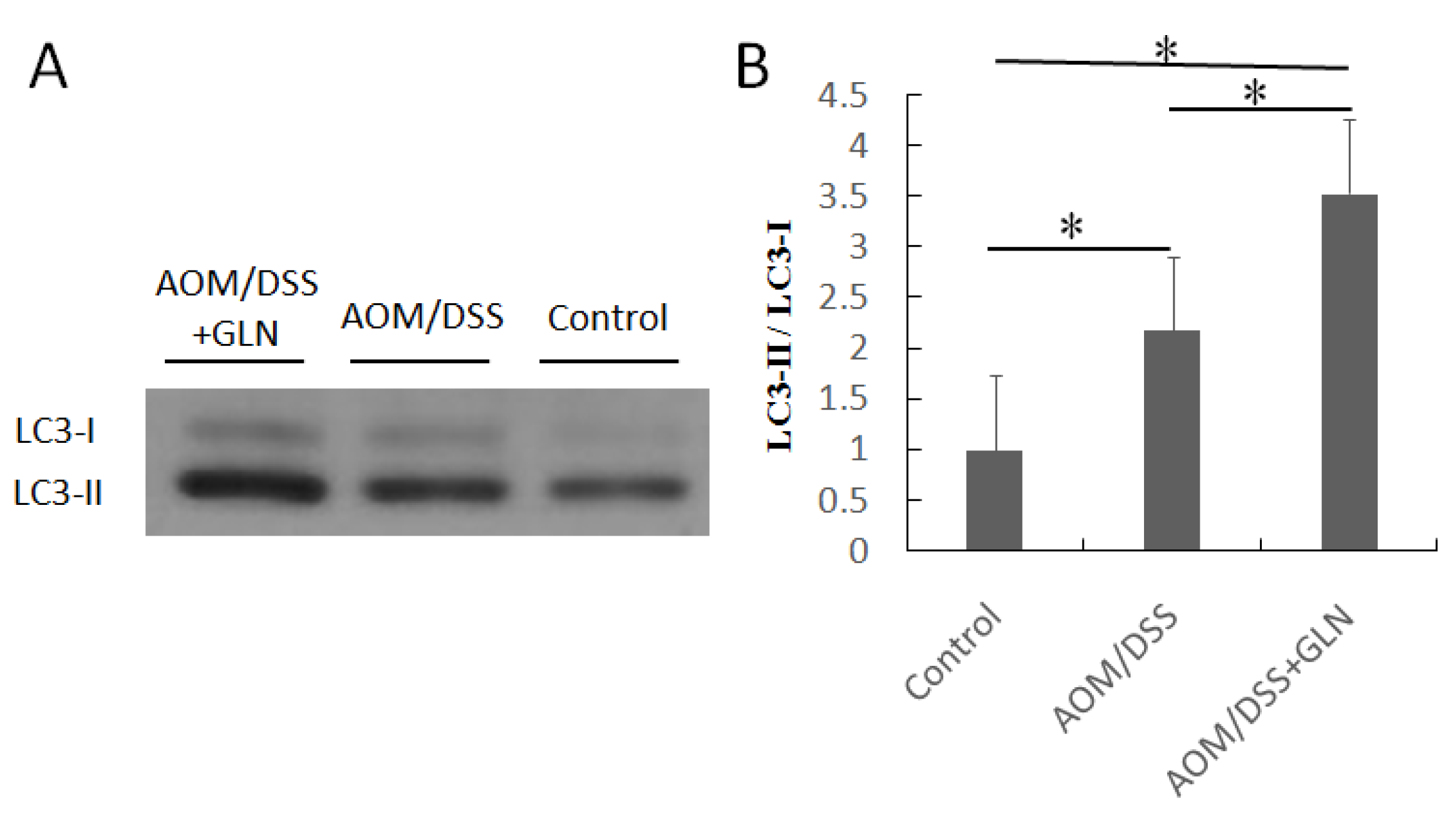
3.4. GLN Induces Autophagy
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
References
- Brenner, H.; Kloor, M.; Pox, C.P. Colorectal cancer. Lancet 2014, 383, 1490–1502. [Google Scholar] [CrossRef]
- Itzkowitz, S.H.; Yio, X. Inflammation and cancer IV. Colorectal cancer in inflammatory bowel disease: The role of inflammation. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 287, G7–G17. [Google Scholar] [CrossRef] [PubMed]
- Guertin, D.A.; Sabatini, D.M. Defining the role of mTOR in cancer. Cancer Cell 2007, 12, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Dancey, J. mTOR signaling and drug development in cancer. Nat. Rev. Clin. Oncol. 2010, 7, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, S.; Peterson, T.R.; Laplante, M.; Oh, S.; Sabatini, D.M. mTORC1 controls fasting-induced ketogenesis and its modulation by ageing. Nature 2010, 468, 1100–1104. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, S.; Peterson, T.R.; Sabatini, D.M. Regulation of the mTOR complex 1 pathway by nutrients, growth factors, and stress. Mol. Cell 2010, 40, 310–322. [Google Scholar] [CrossRef] [PubMed]
- Sabatini, D.M. mTOR and cancer: Insights into a complex relationship. Nat. Rev. Cancer 2006, 6, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zheng, X.F. mTOR-independent 4E-BP1 phosphorylation is associated with cancer resistance to mTOR kinase inhibitors. Cell Cycle 2012, 11, 594–603. [Google Scholar] [CrossRef] [PubMed]
- Peterson, T.R.; Laplante, M.; Thoreen, C.C.; Sancak, Y.; Kang, S.A.; Kuehl, W.M.; Gray, N.S.; Sabatini, D.M. DEPTOR is an mTOR inhibitor frequently overexpressed in multiple myeloma cells and required for their survival. Cell 2009, 137, 873–886. [Google Scholar] [CrossRef] [PubMed]
- Swaid, F.; Sukhotnik, I.; Matter, I.; Berkowitz, D.; Hadjittofi, C.; Pollak, Y.; Lavy, A. Dietary glutamine supplementation prevents mucosal injury and modulates intestinal epithelial restitution following acetic acid induced intestinal injury in rats. Nutr. Metab. 2013, 10, 53. [Google Scholar] [CrossRef] [PubMed]
- Sukhotnik, I.; Khateeb, K.; Mogilner, J.G.; Helou, H.; Lurie, M.; Coran, A.G.; Shiloni, N. Dietary glutamine supplementation prevents mucosal injury and modulates intestinal epithelial restitution following ischemia-reperfusion injury in the rat. Dig. Dis. Sci. 2007, 52, 1497–1504. [Google Scholar] [CrossRef] [PubMed]
- Kouznetsova, L.; Bijlsma, P.B.; van Leeuwen, P.A.; Groot, J.A.; Houdijk, A.P. Glutamine reduces phorbol-12,13-dibutyrate-induced macromolecular hyperpermeability in HT-29Cl.19A intestinal cells. JPEN J. Parenter. Enter. Nutr. 1999, 23, 136–139. [Google Scholar] [CrossRef]
- Boukhettala, N.; Claeyssens, S.; Bensifi, M.; Maurer, B.; Abed, J.; Lavoinne, A.; Déchelotte, P.; Coëffier, M. Effects of essential amino acids or glutamine deprivation on intestinal permeability and protein synthesis in HCT-8 cells: Involvement of GCN2 and mTOR pathways. Amino Acids 2010, 42, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Van der Vos, K.E.; Coffer, P.J. Glutamine metabolism links growth factor signaling to the regulation of autophagy. Autophagy 2012, 8, 1862–1864. [Google Scholar] [CrossRef] [PubMed]
- Xi, P.; Jiang, Z.; Dai, Z.; Li, X.; Yao, K.; Zheng, C.; Lin, Y.; Wang, J.; Wu, G. Regulation of protein turnover by L-glutamine in porcine intestinal epithelial cells. J. Nutr. Biochem. 2012, 23, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Sakiyama, T.; Musch, M.W.; Ropeleski, M.J.; Tsubouchi, H.; Chang, E.B. Glutamine increases autophagy under Basal and stressed conditions in intestinal epithelial cells. Gastroenterology 2009, 136, 924–932. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Wang, K.; Wang, Z.; Li, N.; Ji, G. Chemopreventive effect of dietary glutamine on colitis-associated colon tumorigenesis in mice. Carcinogenesis 2013, 34, 1593–1600. [Google Scholar] [CrossRef] [PubMed]
- Cooper, H.S.; Murthy, S.N.; Shah, R.S.; Sedergran, D.J. Clinicopathologic study of dextran sulfate sodium experimental murine colitis. Lab. Investgi. 1993, 69, 238–249. [Google Scholar]
- Wouters, B.G.; Koritzinsky, M. Hypoxia signalling through mTOR and the unfolded protein response in cancer. Nat. Rev. Cancer 2008, 8, 851–864. [Google Scholar] [CrossRef] [PubMed]
- Zoncu, R.; Efeyan, A.; Sabatini, D.M. mTOR: From growth signal integration to cancer, diabetes and ageing. Nat. Rev. Mol. Cell Biol. 2011, 12, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Gulhati, P.; Bowen, K.A.; Liu, J.; Stevens, P.D.; Rychahou, P.G.; Chen, M.; Lee, E.Y.; Weiss, H.L.; O’Connor, K.L.; Gao, T.; et al. mTORC1 and mTORC2 regulate EMT, motility, and metastasis of colorectal cancer via RhoA and Rac1 signaling pathways. Cancer Res. 2011, 71, 3246–3256. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Zhou, J.F.; Sellers, R.S.; Li, J.F.; Nguyen, A.V.; Wang, Y.; Orlofsky, A.; Liu, Q.; Hume, D.A.; Pollard, J.W.; et al. A novel mouse model of inflammatory bowel disease links mammalian target of rapamycin-dependent hyperproliferation of colonic epithelium to inflammation-associated tumorigenesis. Am. J. Pathol. 2010, 176, 952–967. [Google Scholar] [CrossRef] [PubMed]
- Bhonde, M.R.; Gupte, R.D.; Dadarkar, S.D.; Jadhav, M.G.; Tannu, A.A.; Bhatt, P.; Bhatia, D.R.; Desai, N.K.; Deore, V.; Yewalkar, N.; et al. A novel mTOR inhibitor is efficacious in a murine model of colitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 295, G1237–G1245. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhong, J.; Inuzuka, H.; Gao, D.; Shaik, S.; Sarkar, F.H.; Wei, W. An evolving role for DEPTOR in tumor development and progression. Neoplasia 2012, 14, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Inuzuka, H.; Tan, M.K.; Fukushima, H.; Locasale, J.W.; Liu, P.; Wan, L.; Zhai, B.; Chin, Y.R.; Shaik, S.; et al. mTOR drives its own activation via SCF(betaTrCP)-dependent degradation of the mTOR inhibitor DEPTOR. Mol. Cell 2011, 44, 290–303. [Google Scholar] [CrossRef] [PubMed]
- Manhart, N.; Vierlinger, K.; Spittler, A.; Bergmeister, H.; Sautner, T.; Roth, E. Oral feeding with glutamine prevents lymphocyte and glutathione depletion of Peyer’s patches in endotoxemic mice. Ann. Surg. 2001, 234, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Xue, H.; Sufit, A.J.; Wischmeyer, P.E. Glutamine therapy improves outcome of in vitro and in vivo experimental colitis models. JPEN J. Parenter. Enter. Nutr. 2011, 35, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Marc Rhoads, J.; Wu, G. Glutamine, arginine, and leucine signaling in the intestine. Amino Acids 2009, 37, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Nakajo, T.; Yamatsuji, T.; Ban, H.; Shigemitsu, K.; Haisa, M.; Motoki, T.; Noma, K.; Nobuhisa, T.; Matsuoka, J.; Gunduz, M.; et al. Glutamine is a key regulator for amino acid-controlled cell growth through the mTOR signaling pathway in rat intestinal epithelial cells. Biochem. Biophys. Res. Commun. 2005, 326, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Ge, W.; Ren, J. mTOR-STAT3-notch signalling contributes to ALDH2-induced protection against cardiac contractile dysfunction and autophagy under alcoholism. J. Cell. Mol. Med. 2012, 16, 616–626. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, M.A.; de Jong, J.; Vaessen, M.J.; van Leeuwen, P.A.; Houdijk, A.P. Glutamate reduces experimental intestinal hyperpermeability and facilitates glutamine support of gut integrity. World J. Gastroenterol. 2011, 17, 1569–1573. [Google Scholar] [CrossRef] [PubMed]
| AOM/DSS | AOM/DSS + GLN | p Value | |
|---|---|---|---|
| Colonic inflammation score | 1.9 ± 0.7 | 1.0 ± 0.7 | 0.010 |
| Adenoma, n/N, mean ± SD | 8/10, 4.7 ± 3.3 | 6/10, 1.9 ± 1.7 | 0.028 a |
| Adenocarcinoma, n/N, mean ± SD | 10/10, 3.6 ± 1.7 | 5/10, 1.1 ± 1.3 | 0.002 a |
| Total tumors, n/N, mean ± SD | 10/10, 8.3 ± 4.1 | 7/10, 3.0 ± 2.6 | 0.003 a |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, Y.; Wang, K.; Fan, Y.; Wang, Y.; Sun, L.; Wang, L.; Wang, J.; Wang, Z.; Li, J.; Ye, Y.; et al. Chemopreventive Effect of Dietary Glutamineon Colitis-Associated Colorectal Cancer Is Associated with Modulation of the DEPTOR/mTOR Signaling Pathway. Nutrients 2016, 8, 261. https://doi.org/10.3390/nu8050261
Tian Y, Wang K, Fan Y, Wang Y, Sun L, Wang L, Wang J, Wang Z, Li J, Ye Y, et al. Chemopreventive Effect of Dietary Glutamineon Colitis-Associated Colorectal Cancer Is Associated with Modulation of the DEPTOR/mTOR Signaling Pathway. Nutrients. 2016; 8(5):261. https://doi.org/10.3390/nu8050261
Chicago/Turabian StyleTian, Yun, Keming Wang, Yingrui Fan, Yan Wang, Liqun Sun, Li Wang, Jirong Wang, Zhaoxia Wang, Juan Li, Ying Ye, and et al. 2016. "Chemopreventive Effect of Dietary Glutamineon Colitis-Associated Colorectal Cancer Is Associated with Modulation of the DEPTOR/mTOR Signaling Pathway" Nutrients 8, no. 5: 261. https://doi.org/10.3390/nu8050261
APA StyleTian, Y., Wang, K., Fan, Y., Wang, Y., Sun, L., Wang, L., Wang, J., Wang, Z., Li, J., Ye, Y., & Ji, G. (2016). Chemopreventive Effect of Dietary Glutamineon Colitis-Associated Colorectal Cancer Is Associated with Modulation of the DEPTOR/mTOR Signaling Pathway. Nutrients, 8(5), 261. https://doi.org/10.3390/nu8050261




