Long Chain Omega-3 Polyunsaturated Fatty Acid Supplementation Alleviates Doxorubicin-Induced Depressive-Like Behaviors and Neurotoxicity in Rats: Involvement of Oxidative Stress and Neuroinflammation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Experimental Design
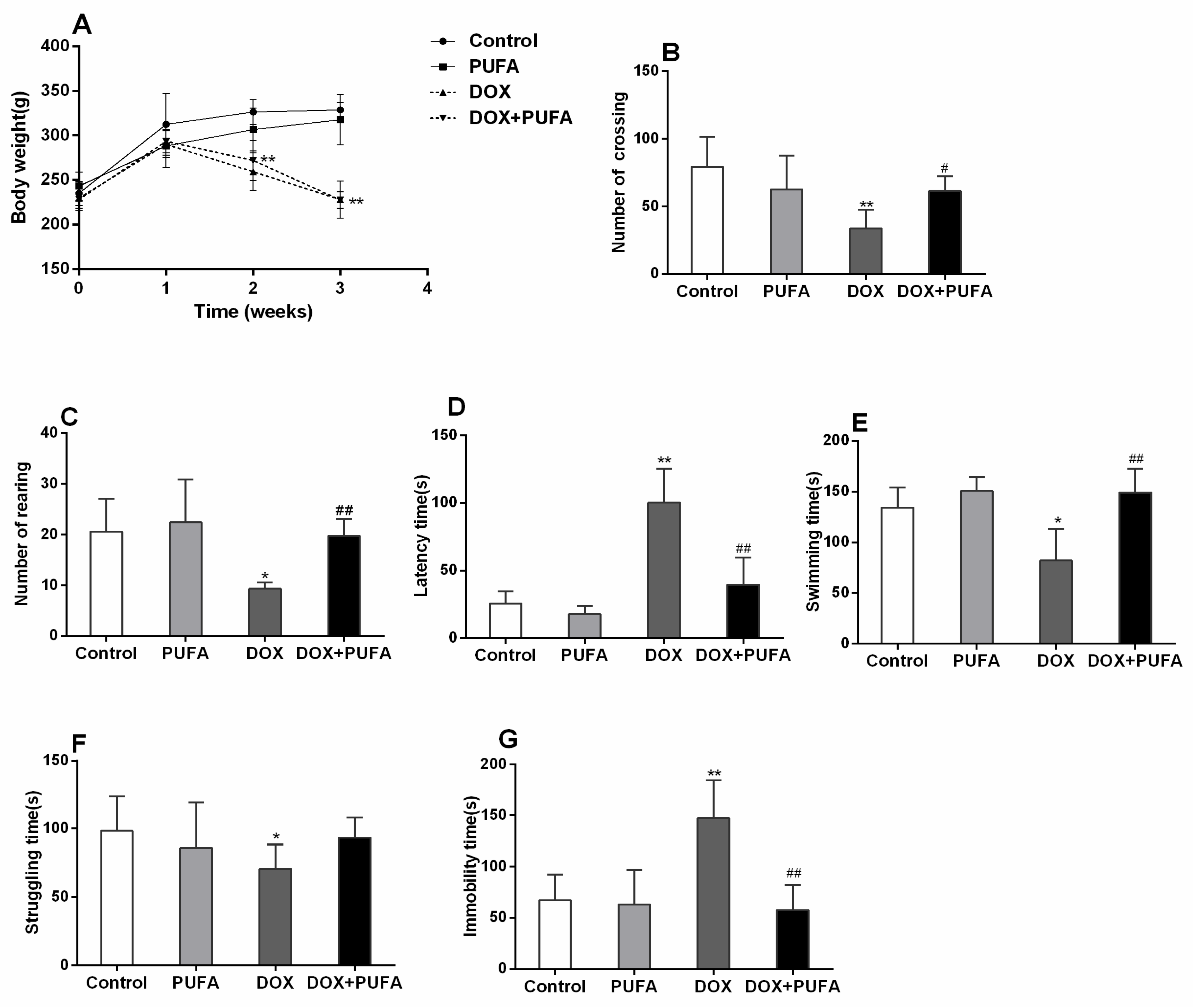
2.3. Behavioral Test
2.3.1. Open-Field Test (OFT)
2.3.2. Forced Swimming Test (FST)
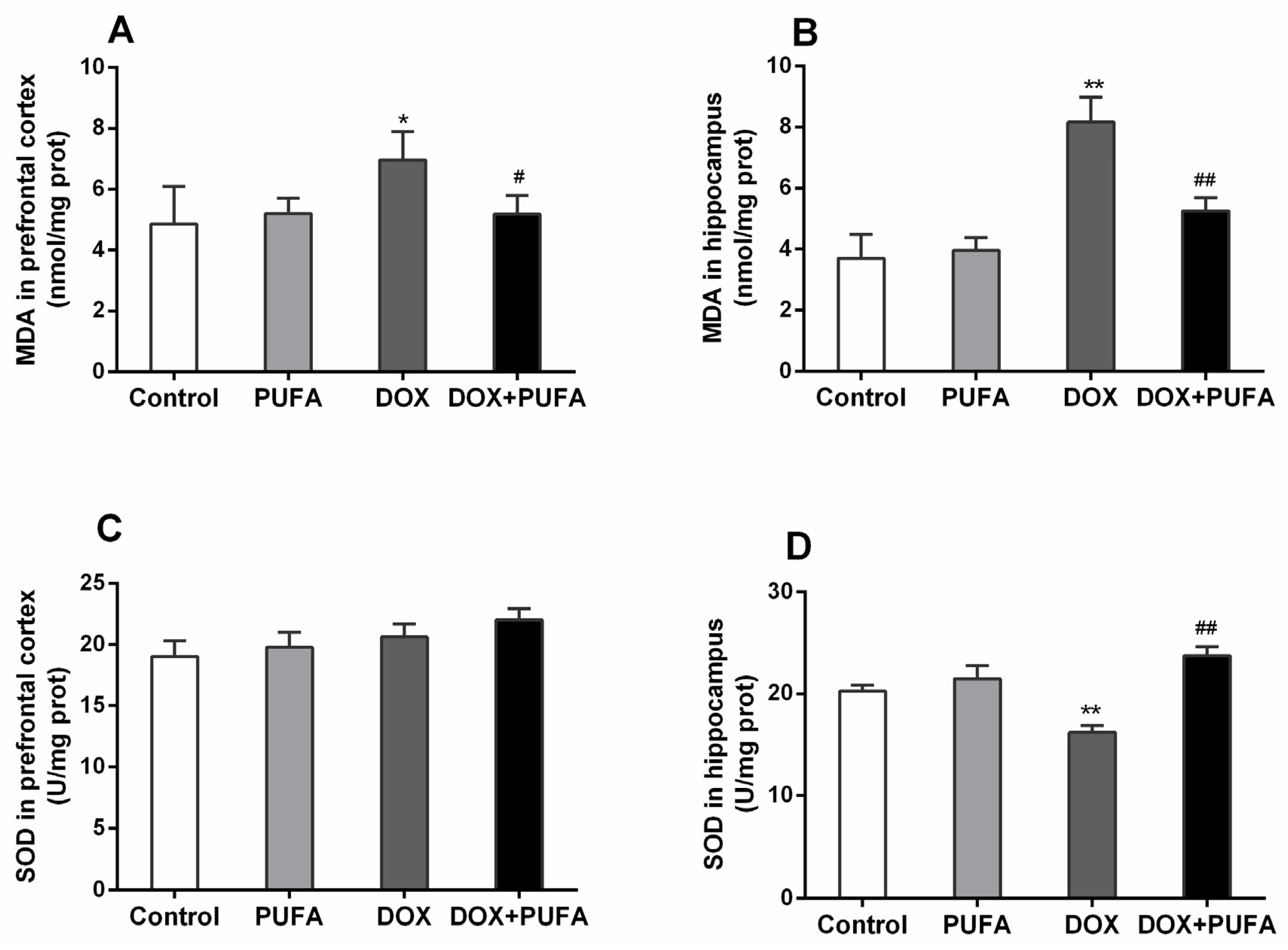
2.4. Measurement of Oxidative Stress
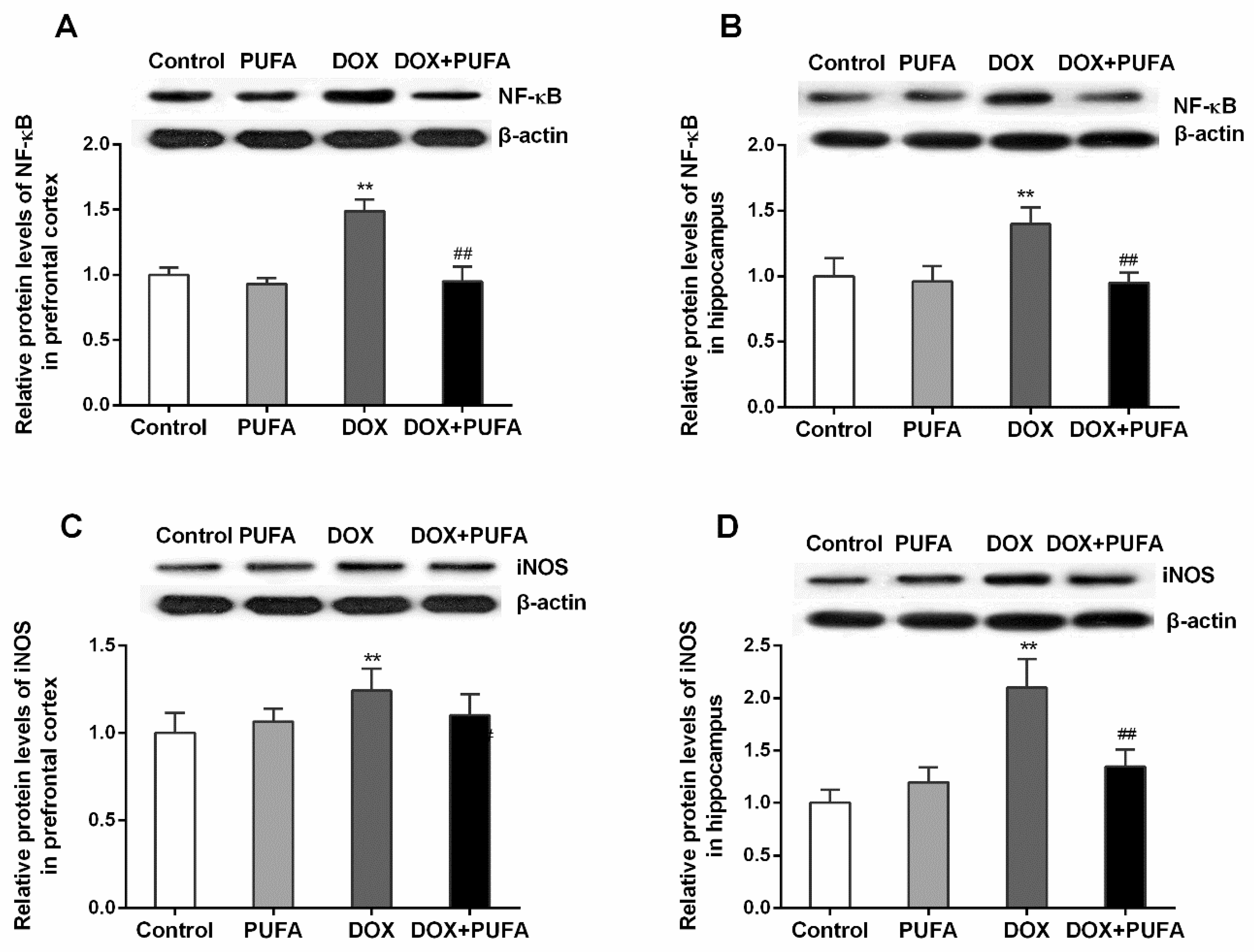
2.5. Western Blot Analysis
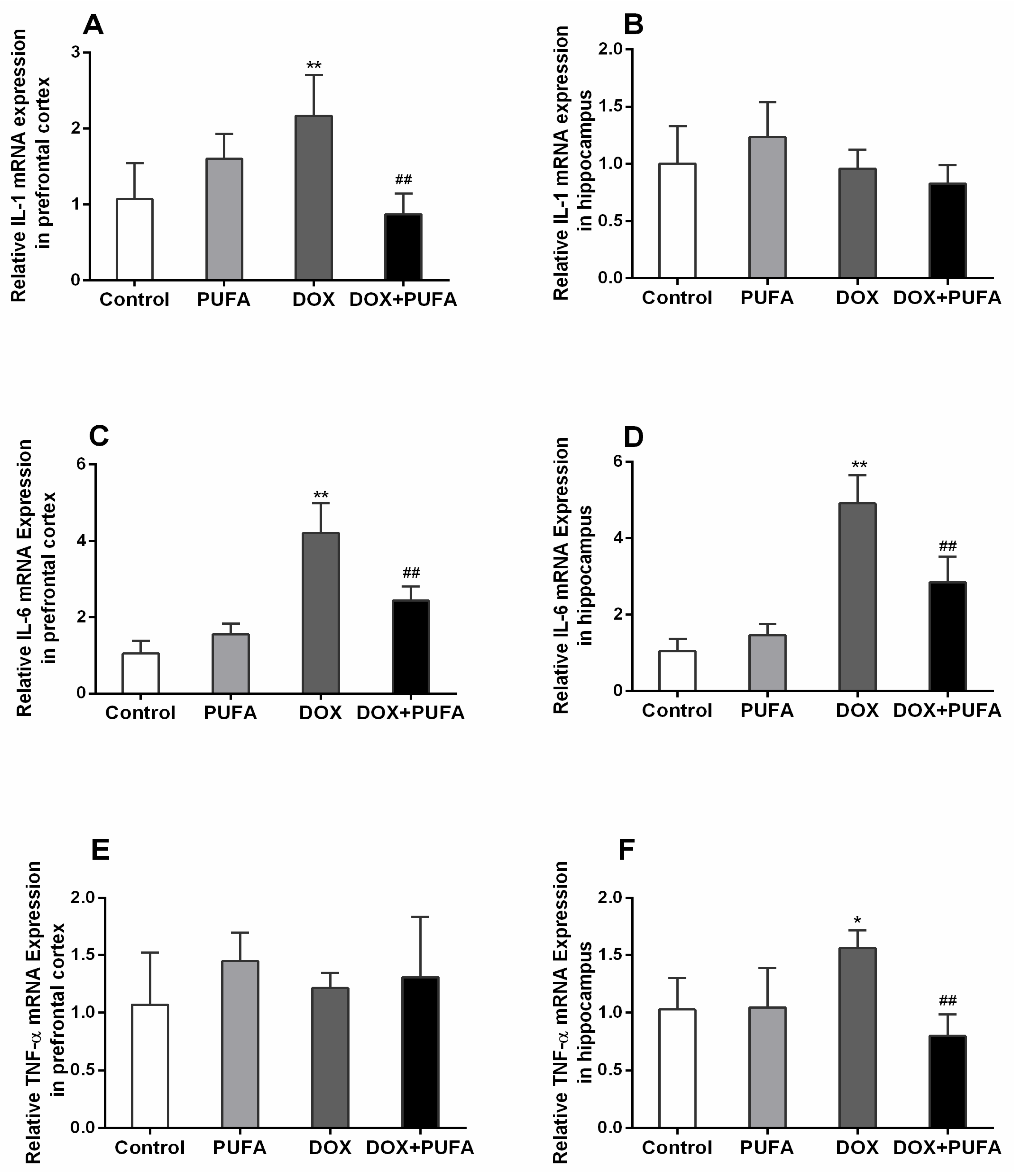
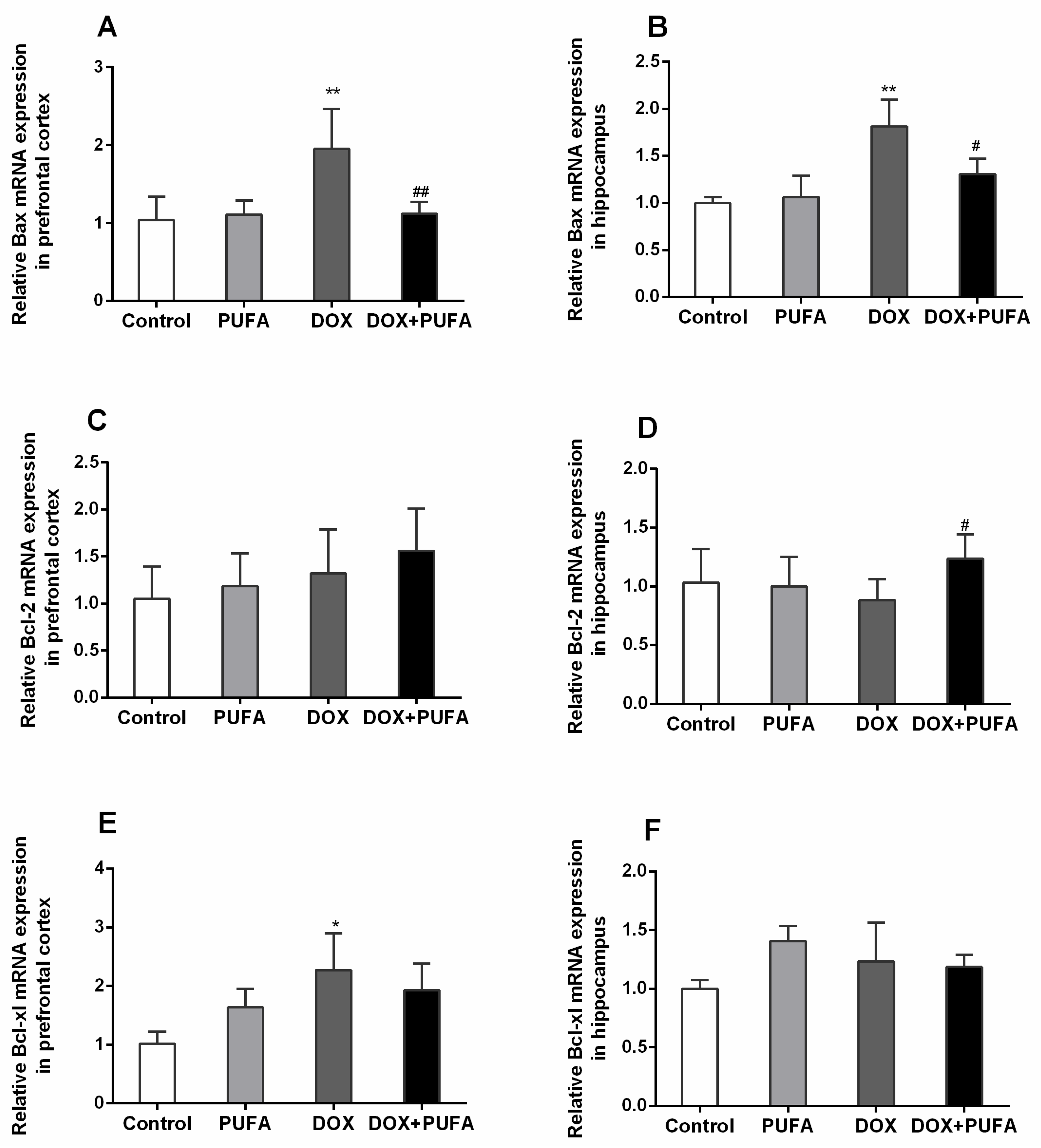
2.6. Real-Time PCR Analysis
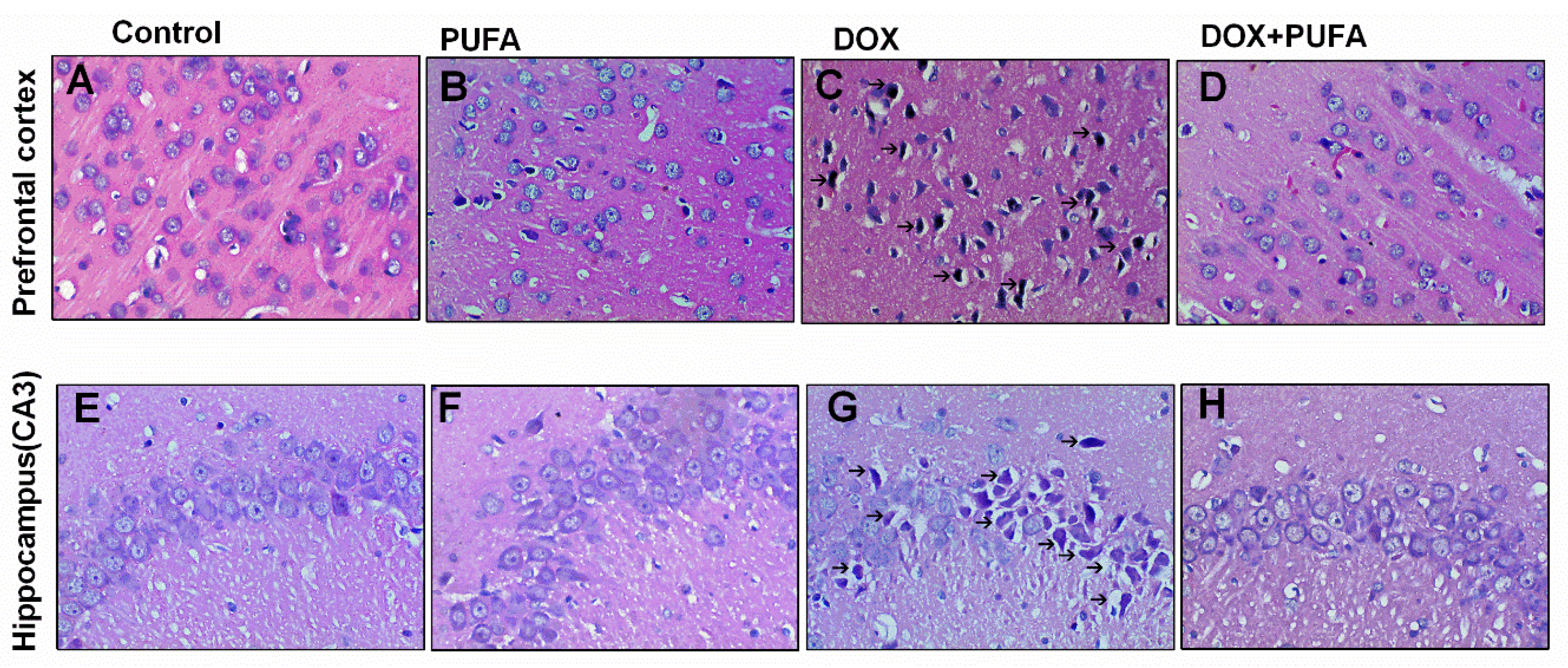
2.7. Histopathological Examination
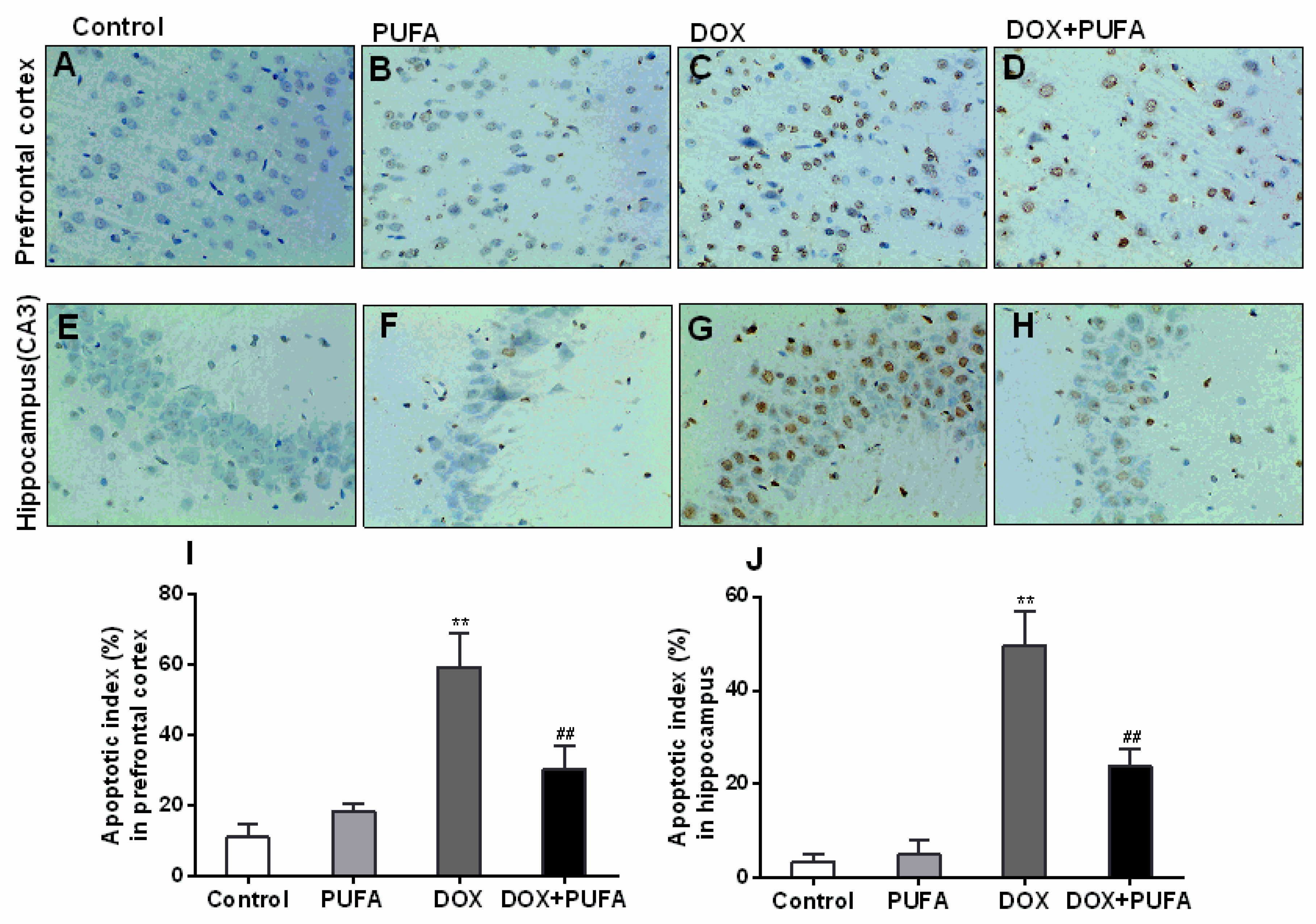
2.8. Immunohistochemical Study
2.9. Statistical Analysis
3. Results
3.1. Effect of DOX and ω-3 PUFAs on Body Weight Gain and Behavioral Changes
3.2. Effects of DOX and ω-3 PUFAs on Oxidative Stress Markers
3.3. Effects of DOX and ω-3 PUFAs on Neuroinflammation Biomarkers
3.4. Effects of DOX and ω-3 PUFAs on Histopathological Changes
3.5. Effects of DOX and ω-3 PUFAs on Neural Apoptotic Markers
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mohamed, R.H.; Karam, R.A.; Amer, M.G. Epicatechin attenuates doxorubicin-induced brain toxicity: Critical role of TNF-alpha, iNOS and NF-kappaB. Brain Res. Bull. 2011, 86, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Merzoug, S.; Toumi, M.L.; Boukhris, N.; Baudin, B.; Tahraoui, A. Adriamycin-related anxiety-like behavior, brain oxidative stress and myelotoxicity in male Wistar rats. Pharmacol. Biochem. Behav. 2011, 99, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Merzoug, S.; Toumi, M.L.; Tahraoui, A. Quercetin mitigates Adriamycin-induced anxiety- and depression-like behaviors, immune dysfunction, and brain oxidative stress in rats. Naunyn Schmiedebergs Arch. Pharmacol. 2014, 387, 921–933. [Google Scholar] [CrossRef] [PubMed]
- Christie, L.A.; Acharya, M.M.; Parihar, V.K.; Nguyen, A.; Martirosian, V.; Limoli, C.L. Impaired Cognitive Function and Hippocampal Neurogenesis following Cancer Chemotherapy. Clin. Cancer Res. 2012, 18, 1954–1965. [Google Scholar] [CrossRef] [PubMed]
- Schlatter, M.C.; Cameron, L.D. Emotional suppression tendencies as predictors of symptoms, mood, and coping appraisals during AC chemotherapy for breast cancer treatment. Ann. Behav. Med. 2010, 40, 15–29. [Google Scholar] [CrossRef] [PubMed]
- Joshi, G.; Aluise, C.D.; Cole, M.P.; Sultana, R.; Pierce, W.M.; Vore, M.; Clair, D.K.S.; Butterfield, D.A. Alterations in brain antioxidant enzymes and redox proteomic identification of oxidized brain proteins induced by the anti-cancer drug adriamycin: Implications for oxidative stress-mediated chemobrain. Neuroscience 2010, 166, 796–807. [Google Scholar] [CrossRef] [PubMed]
- Keeney, J.T.R.; Miriyala, S.; Noel, T.; Moscow, J.A.; Clair, D.K.S.; Butterfield, D.A. Superoxide induces protein oxidation in plasma and TNF-α elevation in macrophage culture: Insights into mechanisms of neurotoxicity following doxorubicin chemotherapy. Cancer Lett. 2015, 367, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Tangpong, J.; Cole, M.P.; Sultana, R.; Joshi, G.; Estus, S.; Vore, M.; St, C.W.; Ratanachaiyavong, S.; St, C.D.; Butterfield, D.A. Adriamycin-induced, TNF-alpha-mediated central nervous system toxicity. Neurobiol. Dis. 2006, 23, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Ahles, T.A.; Saykin, A.J. Candidate mechanisms for chemotherapy-induced cognitive changes. Nat. Rev. Cancer 2007, 7, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Vichaya, E.G.; Chiu, G.S.; Krukowski, K.; Lacourt, T.E.; Kavelaars, A.; Dantzer, R.; Heijnen, C.J.; Walker, A.K. Mechanisms of chemotherapy-induced behavioral toxicities. Front. Neurosci. 2015, 9, 131. [Google Scholar] [CrossRef] [PubMed]
- Venna, V.R.; Deplanque, D.; Allet, C.; Belarbi, K.; Hamdane, M.; Bordet, R. PUFA induce antidepressant-like effects in parallel to structural and molecular changes in the hippocampus. Psychoneuroendocrinology 2009, 34, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Paterniti, I.; Impellizzeri, D.; Di Paola, R.; Esposito, E.; Gladman, S.; Yip, P.; Priestley, J.V.; Michael-Titus, A.T.; Cuzzocrea, S. Docosahexaenoic acid attenuates the early inflammatory response following spinal cord injury in mice: In-vivo and in-vitro studies. J. Neuroinflamm. 2014, 11, 6. [Google Scholar] [CrossRef] [PubMed]
- Ferraz, A.C.; Delattre, A.M.; Almendra, R.G.; Sonagli, M.; Borges, C.; Araujo, P.; Andersen, M.L.; Tufik, S.; Lima, M.M.S. Chronic ω-3 fatty acids supplementation promotes beneficial effects on anxiety, cognitive and depressive-like behaviors in rats subjected to a restraint stress protocol. Behav. Brain Res. 2011, 1, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Rapaport, M.H.; Nierenberg, A.A.; Schettler, P.J.; Kinkead, B.; Cardoos, A.; Walker, R.; Mischoulon, D. Inflammation as a predictive biomarker for response to omega-3 fatty acids in major depressive disorder: A proof-of-concept study. Mol. Psychiatry 2016, 21, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Jiang, P.; Li, H.; Cai, H.; Liu, Y.; Gong, H.; Zhang, L. Antidepressant-like effect of n-3 PUFAs in CUMS rats: Role of tPA/PAI-1 system. Physiol. Behav. 2015, 139, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.N.; Huang, W.; Hall, J.C.; Ward, R.E.; Priestley, J.V.; Michael-Titus, A.T. The acute administration of eicosapentaenoic acid is neuroprotective after spinal cord compression injury in rats. Prostaglandins Leukot. Essent. Fatty Acids 2010, 83, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Uygur, R.; Aktas, C.; Tulubas, F.; Uygur, E.; Kanter, M.; Erboga, M.; Caglar, V.; Topcu, B.; Ozen, O.A. Protective effects of fish omega-3 fatty acids on doxorubicin-induced testicular apoptosis and oxidative damage in rats. Andrologia 2014, 46, 917–926. [Google Scholar] [CrossRef] [PubMed]
- Uygur, R.; Aktas, C.; Tulubas, F.; Alpsoy, S.; Topcu, B.; Ozen, O.A. Cardioprotective effects of fish omega-3 fatty acids on doxorubicin-induced cardiotoxicity in rats. Hum. Exp. Toxicol. 2014, 33, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Appleton, K.M.; Rogers, P.J.; Ness, A.R. Updated systematic review and meta-analysis of the effects of n-3 long-chain polyunsaturated fatty acids on depressed mood. Am. J. Clin. Nutr. 2010, 91, 757–770. [Google Scholar] [CrossRef] [PubMed]
- Pascoe, M.C.; Crewther, S.G.; Carey, L.M.; Crewther, D.P. What you eat is what you are—A role for polyunsaturated fatty acids in neuroinflammation induced depression? Clin. Nutr. 2011, 30, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Dasilva, G.; Pazos, M.; Garcia-Egido, E.; Gallardo, J.M.; Rodriguez, I.; Cela, R.; Medina, I. Healthy effect of different proportions of marine omega-3 PUFAs EPA and DHA supplementation in Wistar rats: Lipidomic biomarkers of oxidative stress and inflammation. J. Nutr. Biochem. 2015, 26, 1385–1392. [Google Scholar] [CrossRef] [PubMed]
- Wuryanti, S.; Andrijono; Susworo; Witjaksono, F. The Effect of High Poly Unsaturated Fatty Acid (PUFA) Dietary Supplementation on Inflammatory Status of Patients with Advanced Cervical Cancer on Radiation Treatment. Acta Med. Indones. 2015, 47, 45–49. [Google Scholar] [PubMed]
- Zararsiz, I.; Kus, I.; Akpolat, N.; Songur, A.; Ogeturk, M.; Sarsilmaz, M. Protective effects of omega-3 essential fatty acids against formaldehyde-induced neuronal damage in prefrontal cortex of rats. Cell Biochem. Funct. 2006, 24, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Ozen, O.A.; Cosar, M.; Sahin, O.; Fidan, H.; Eser, O.; Mollaoglu, H.; Alkoc, O.; Yaman, M.; Songur, A. The protective effect of fish n-3 fatty acids on cerebral ischemia in rat prefrontal cortex. Neurol. Sci. 2008, 29, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Alsina, E.; Macri, E.V.; Lifshitz, F.; Bozzini, C.; Rodriguez, P.N.; Boyer, P.M.; Friedman, S.M. Efficacy of phytosterols and fish-oil supplemented high-oleic-sunflower oil rich diets in hypercholesterolemic growing rats. Int. J. Food Sci. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, A.M.; Francisco, P.C.; Motta, K.; Chagas, T.R.; Dos, S.C.; Rafacho, A.; Nunes, E.A. Fish oil supplementation attenuates changes in plasma lipids caused by dexamethasone treatment in rats. Appl. Physiol. Nutr. Metab. 2016, 41, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Aziriova, S.; Repova, B.K.; Krajcirovicova, K.; Hrenak, J.; Rajkovicova, R.; Arendasova, K.; Kamodyova, N.; Celec, P.; Zorad, S.; Adamcova, M.; et al. Doxorubicin-induced behavioral disturbances in rats: Protective effect of melatonin and captopril. Pharmacol. Biochem. Behav. 2014, 124, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Mischoulon, D.; Best-Popescu, C.; Laposata, M.; Merens, W.; Murakami, J.L.; Wu, S.L.; Papakostas, G.I.; Dording, C.M.; Sonawalla, S.B.; Nierenberg, A.A.; et al. A double-blind dose-finding pilot study of docosahexaenoic acid (DHA) for major depressive disorder. Eur. Neuropsychopharmacol. 2008, 18, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Lesperance, F.; Frasure-Smith, N.; St-Andre, E.; Turecki, G.; Lesperance, P.; Wisniewski, S.R. The efficacy of omega-3 supplementation for major depression: A randomized controlled trial. J. Clin. Psychiatry 2011, 72, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Mischoulon, D.; Nierenberg, A.A.; Schettler, P.J.; Kinkead, B.L.; Fehling, K.; Martinson, M.A.; Hyman, R.M. A double-blind, randomized controlled clinical trial comparing eicosapentaenoic acid versus docosahexaenoic acid for depression. J. Clin. Psychiatry 2015, 76, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Bloch, M.H.; Hannestad, J. Omega-3 fatty acids for the treatment of depression: Systematic review and meta-analysis. Mol. Psychiatry 2011, 17, 1272–1282. [Google Scholar] [CrossRef] [PubMed]
- Lakhwani, L.; Tongia, S.K.; Pal, V.S.; Agrawal, R.P.; Nyati, P.; Phadnis, P. Omega-3 fatty acids have antidepressant activity in forced swimming test in Wistar rats. Acta Pol. Pharm. 2007, 64, 271–276. [Google Scholar] [PubMed]
- Huang, S.Y.; Yang, H.T.; Chiu, C.C.; Pariante, C.M.; Su, K.P. Omega-3 fatty acids on the forced-swimming test. J. Psychiatr. Res. 2008, 42, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Tangpong, J.; Cole, M.P.; Sultana, R.; Estus, S.; Vore, M.; Clair, W.S.; Ratanachaiyavong, S.; Clair, D.K.S.; Butterfield, D.A. Adriamycin-mediated nitration of manganese superoxide dismutase in the central nervous system: Insight into the mechanism of chemobrain. J. Neurochem. 2007, 100, 191–201. [Google Scholar] [CrossRef] [PubMed]
- El-Naga, R.N.; Ahmed, H.I.; Abd, A.H.E. Effects of indole-3-carbinol on clonidine-induced neurotoxicity in rats: Impact on oxidative stress, inflammation, apoptosis and monoamine levels. Neurotoxicology 2014, 44, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Rawdin, B.J.; Mellon, S.H.; Dhabhar, F.S.; Epel, E.S.; Puterman, E.; Su, Y.; Burke, H.M.; Reus, V.I.; Rosser, R.; Hamilton, S.P.; et al. Dysregulated relationship of inflammation and oxidative stress in major depression. Brain Behav. Immun. 2013, 31, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Makhija, K.; Karunakaran, S. The role of inflammatory cytokines on the aetiopathogenesis of depression. Aust. N. Z. J. Psychiatry 2013, 47, 828–839. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, J.D.; Cordero, K.; Baldeosingh, K.; Torrado, A.I.; Walker, R.L.; Miranda, J.D.; De Leon, M. Docosahexaenoic Acid Pretreatment Confers Protection and Functional Improvements after Acute Spinal Cord Injury in Adult Rats. J. Neurotrauma 2012, 29, 551–566. [Google Scholar] [CrossRef] [PubMed]
- De Smedt-Peyrusse, V.; Sargueil, F.; Moranis, A.; Harizi, H.; Mongrand, S.; Layé, S. Docosahexaenoic acid prevents lipopolysaccharide-induced cytokine production in microglial cells by inhibiting lipopolysaccharide receptor presentation but not its membrane subdomain localization. J. Neurochem. 2008, 105, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Kreisel, T.; Frank, M.G.; Licht, T.; Reshef, R.; Ben-Menachem-Zidon, O.; Baratta, M.V.; Maier, S.F.; Yirmiya, R. Dynamic microglial alterations underlie stress-induced depressive-like behavior and suppressed neurogenesis. Mol. Psychiatry 2014, 19, 699–709. [Google Scholar] [CrossRef] [PubMed]
- Lopes, M.A.; Meisel, A.; Dirnagl, U.; Carvalho, F.D.; Bastos, M.L. Doxorubicin induces biphasic neurotoxicity to rat cortical neurons. Neurotoxicology 2008, 29, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.; Heyob, K.M.; Velten, M.; Tipple, T.E.; Rogers, L.K. DHA suppresses chronic apoptosis in the lung caused by perinatal inflammation. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 309, L441–L448. [Google Scholar] [CrossRef] [PubMed]
- Mergenthaler, P.; Dirnagl, U.; Meisel, A. Pathophysiology of stroke: Lessons from animal models. Metab. Brain Dis. 2004, 19, 151–167. [Google Scholar] [CrossRef] [PubMed]
- Skibinski, C.G.; Das, A.; Chen, K.M.; Liao, J.; Manni, A.; Kester, M.; El-Bayoumy, K. A novel biologically active acid stable liposomal formulation of docosahexaenoic acid in human breast cancer cell lines. Chem. Biol. Interact. 2016. [Google Scholar] [CrossRef] [PubMed]
- Eltweri, A.M.; Thomas, A.L.; Metcalfe, M.; Calder, P.C.; Dennison, A.R.; Bowrey, D.J. Potential applications of fish oils rich in omega-3 polyunsaturated fatty acids in the management of gastrointestinal cancer. Clin. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Ulu, A.; Wan, D.; Yang, J.; Hammock, B.D.; Weiss, R.H. Addition of DHA synergistically enhances the efficacy of regorafenib for kidney cancer therapy. Mol. Cancer Ther. 2016. [Google Scholar] [CrossRef] [PubMed]
| Target Gene | Primers Sequences | Size (bp) | |
|---|---|---|---|
| IL-1β | Forward | 5′-AGGTCGTCATCATCCCACGAG-3′ | 119 |
| Reverse | 5′-GCTGTGGCAGCTACCTATGTCTTG-3′ | ||
| IL-6 | Forward | 5′-CACAAGTCCGGAGAGGAGAC-3′ | 167 |
| Reverse | 5′-ACAGTGCATCATCGCTGTTC-3′ | ||
| TNF-α | Forward | 5′-GAGAGATTGGCTGCTGGAAC-3′ | 82 |
| Reverse | 5′-TGGAGACCATGATGACCGTA-3′ | ||
| Bax | Forward | 5′-CCAGGACGCATCCACCAAGAAGC-3′ | 135 |
| Reverse | 5′-TGCCACACGGAAGAAGACCTCTCG-3′ | ||
| Bcl-xl | Forward | 5′-CAGCTTCATATAACCCCAGGGAC-3′ | 207 |
| Reverse | 5′-GCTCTAGGTGGTCATTCAGGTAGG-3′ | ||
| Bcl-2 | Forward | 5′-AGCCCTGTGCCACCTGTGGT-3′ | 93 |
| Reverse | 5′-ACTGGACATCTCTGCAAAGTCGCG-3′ | ||
| β-Actin | Forward | 5′-CATCCTGCGTCTGGACCTGG-3′ | 116 |
| Reverse | 5′-TAATGTCACGCACGATTTCC-3′ |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Y.-Q.; Dang, R.-L.; Tang, M.-M.; Cai, H.-L.; Li, H.-D.; Liao, D.-H.; He, X.; Cao, L.-J.; Xue, Y.; Jiang, P. Long Chain Omega-3 Polyunsaturated Fatty Acid Supplementation Alleviates Doxorubicin-Induced Depressive-Like Behaviors and Neurotoxicity in Rats: Involvement of Oxidative Stress and Neuroinflammation. Nutrients 2016, 8, 243. https://doi.org/10.3390/nu8040243
Wu Y-Q, Dang R-L, Tang M-M, Cai H-L, Li H-D, Liao D-H, He X, Cao L-J, Xue Y, Jiang P. Long Chain Omega-3 Polyunsaturated Fatty Acid Supplementation Alleviates Doxorubicin-Induced Depressive-Like Behaviors and Neurotoxicity in Rats: Involvement of Oxidative Stress and Neuroinflammation. Nutrients. 2016; 8(4):243. https://doi.org/10.3390/nu8040243
Chicago/Turabian StyleWu, Yan-Qin, Rui-Li Dang, Mi-Mi Tang, Hua-Lin Cai, Huan-De Li, De-Hua Liao, Xin He, Ling-Juan Cao, Ying Xue, and Pei Jiang. 2016. "Long Chain Omega-3 Polyunsaturated Fatty Acid Supplementation Alleviates Doxorubicin-Induced Depressive-Like Behaviors and Neurotoxicity in Rats: Involvement of Oxidative Stress and Neuroinflammation" Nutrients 8, no. 4: 243. https://doi.org/10.3390/nu8040243
APA StyleWu, Y.-Q., Dang, R.-L., Tang, M.-M., Cai, H.-L., Li, H.-D., Liao, D.-H., He, X., Cao, L.-J., Xue, Y., & Jiang, P. (2016). Long Chain Omega-3 Polyunsaturated Fatty Acid Supplementation Alleviates Doxorubicin-Induced Depressive-Like Behaviors and Neurotoxicity in Rats: Involvement of Oxidative Stress and Neuroinflammation. Nutrients, 8(4), 243. https://doi.org/10.3390/nu8040243




