High Risk of Metabolic and Adipose Tissue Dysfunctions in Adult Male Progeny, Due to Prenatal and Adulthood Malnutrition Induced by Fructose Rich Diet
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Experimental Designs
2.1.1. Sixty-Day-Old Rats
2.1.2. Eighty-One Day-Old Rats
2.2. RPAT Adipocyte Isolation and Incubation
2.3. RPAT Pad Histology
2.4. RNA Isolation and Real-Time Quantitative PCR
2.5. SVF Cell Composition Analysis by Flow Cytometry (FACS)
2.6. Intravenous Glucose Tolerance Test (i.v. GTT)
2.7. Peripheral Metabolite Measurements
2.8. Statistical Analysis
3. Results
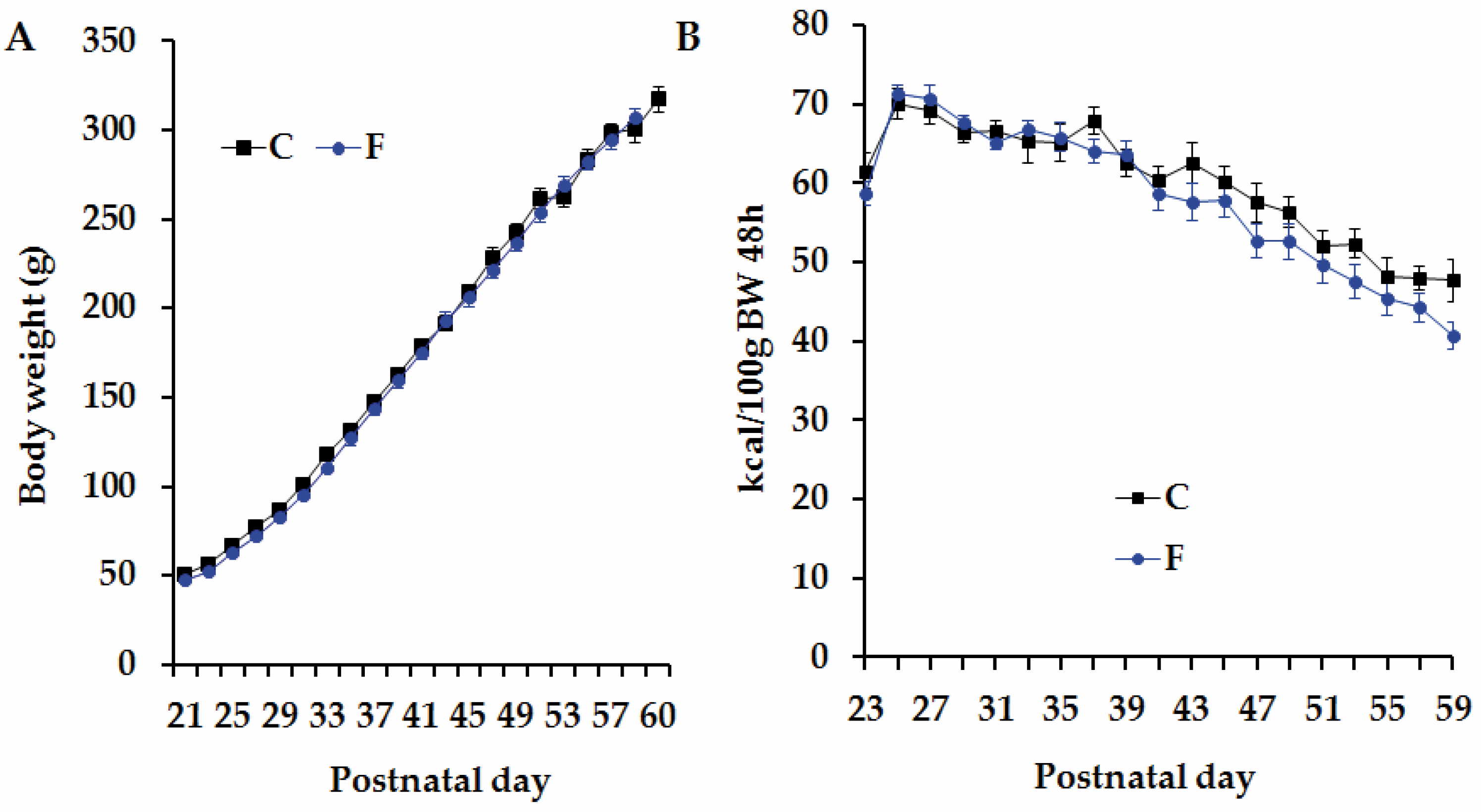
3.1. Pregnant Rats’ Body Weight and Food-Provided Energy Intake
3.2. Body Weight and Caloric Intake in Adult Male Offspring Born to CM and FM
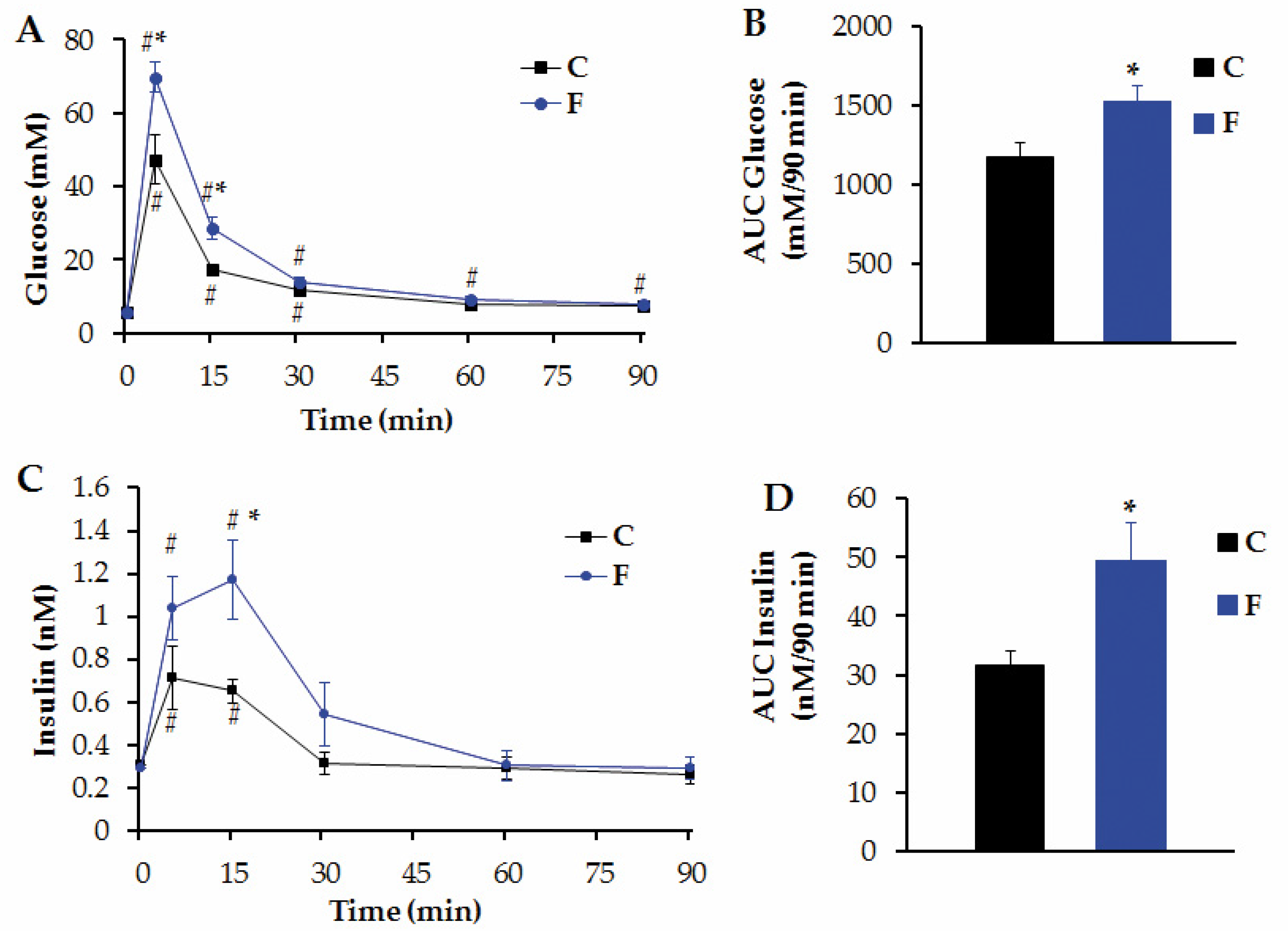
3.3. Peripheral Levels of Several Metabolites and i.v. GTT in Adult Male Offspring
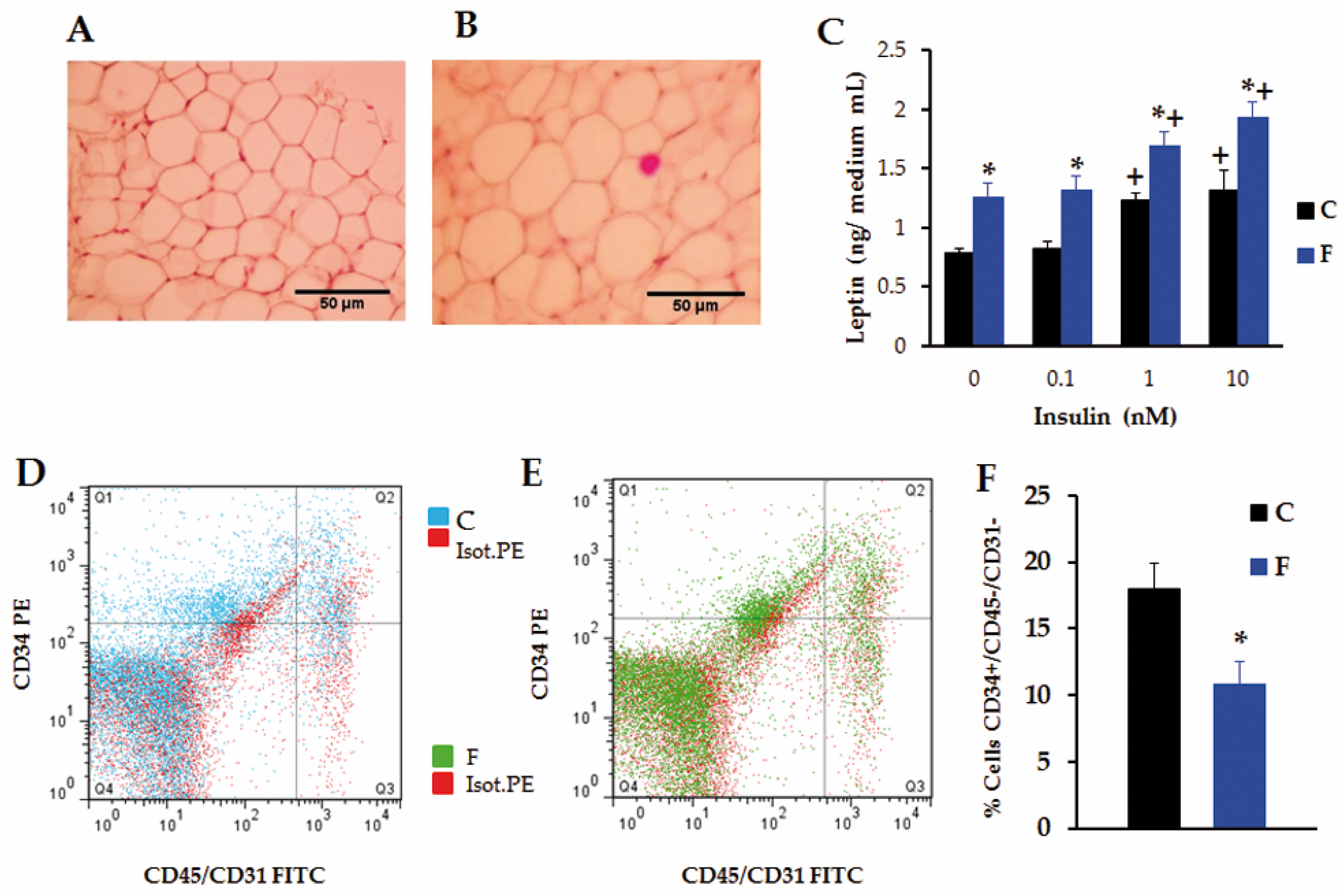
3.4. Retroperitoneal Adipose Tissue Characteristics and Functionality in Adult Male Offspring
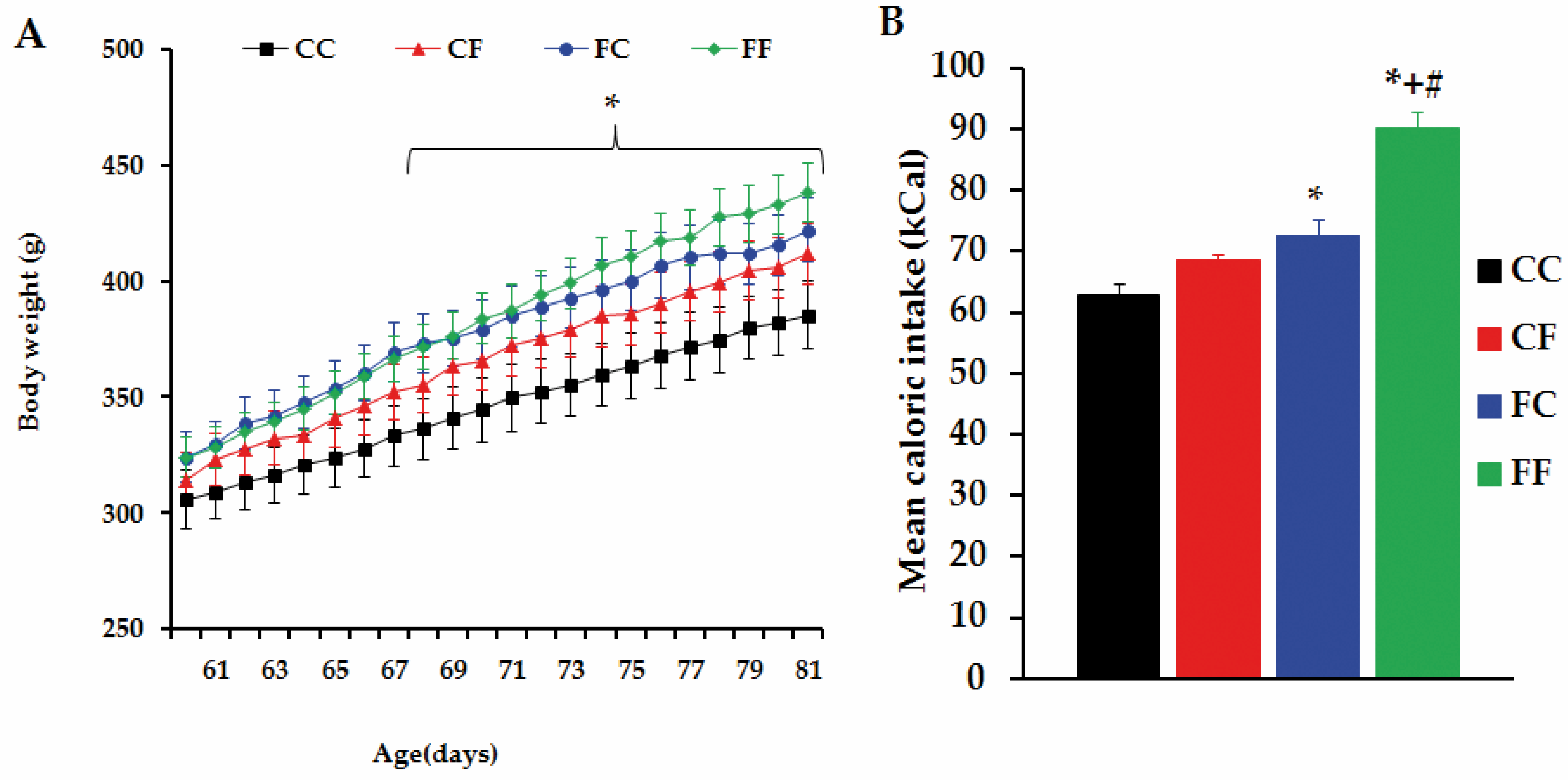
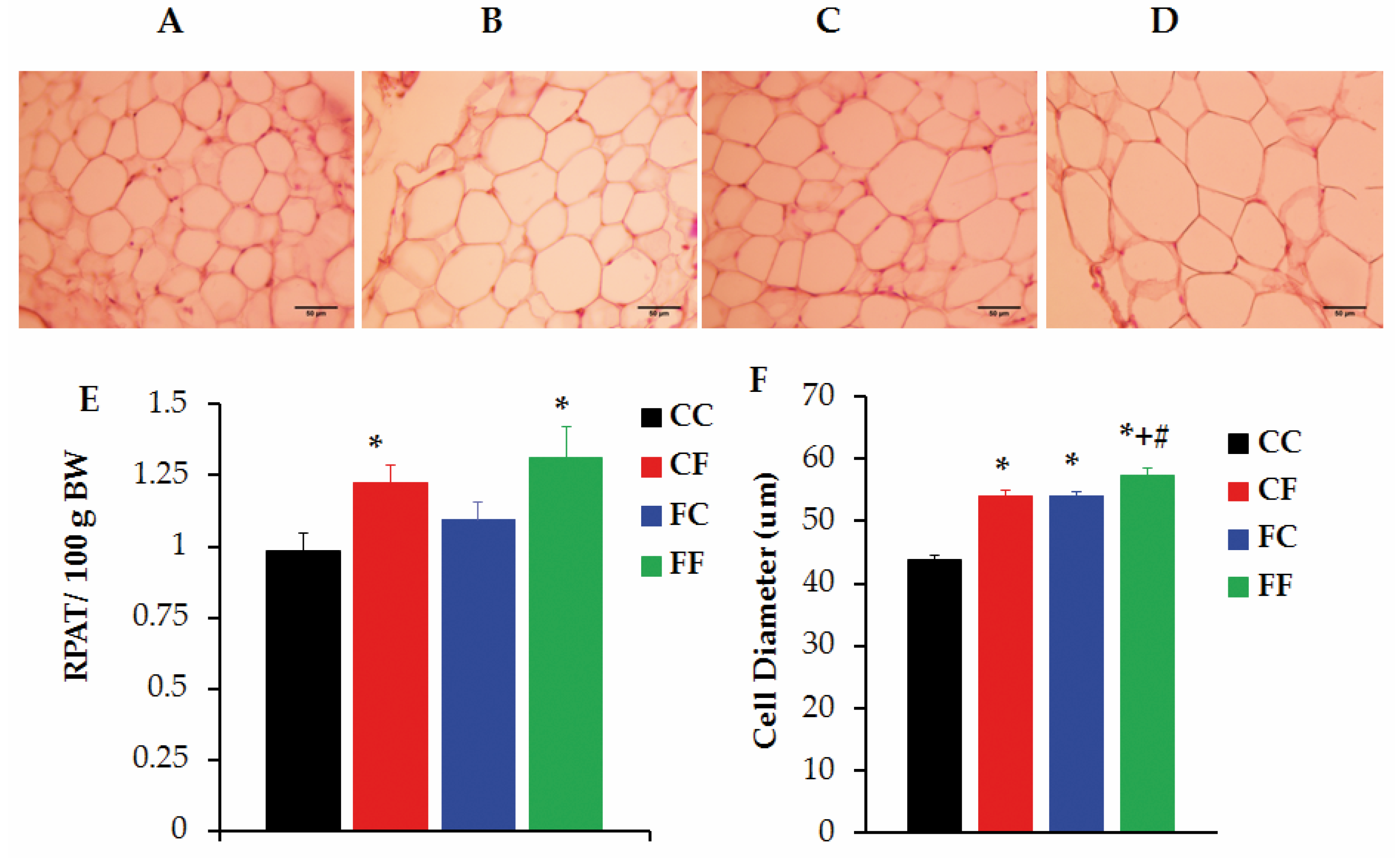
3.5. Challenging Adult Male Offspring with a FRD
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic: Report of a WHO Consultation. Available online: http://www.who.int/nutrition/publications/obesity/WHO_TRS_894/en/ (accessed on 14 March 2016).
- Johnson, R.J.; Segal, M.S.; Sautin, Y.; Nakagawa, T.; Feig, D.I.; Kang, D.-H.; Gersch, M.S.; Benner, S.; Sánchez-Lozada, L.G. Potential role of sugar (fructose) in the epidemic of hypertension, obesity and the metabolic syndrome, diabetes, kidney disease, and cardiovascular disease. Am. J. Clin. Nutr. 2007, 86, 899–906. [Google Scholar] [PubMed]
- Huang, B.-W.; Chiang, M.-T.; Yao, H.-T.; Chiang, W. The effect of high-fat and high-fructose diets on glucose tolerance and plasma lipid and leptin levels in rats. Diabetes Obes. Metab. 2004, 6, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Blakely, S.R.; Hallfrisch, J.; Reiser, S.; Prather, E.S. Long-term effects of moderate fructose feeding on glucose tolerance parameters in rats. J. Nutr. 1981, 111, 307–314. [Google Scholar] [PubMed]
- Nagai, Y.; Nishio, Y.; Nakamura, T.; Maegawa, H.; Kikkawa, R.; Kashiwagi, A. Amelioration of high fructose-induced metabolic derangements by activation of PPARalpha. Am. J. Physiol. Endocrinol. Metab. 2002, 282, E1180–E1190. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J.P. The origins of the developmental origins theory. J. Intern. Med. 2007, 261, 412–417. [Google Scholar] [CrossRef] [PubMed]
- Armitage, J.A.; Taylor, P.D.; Poston, L. Experimental models of developmental programming: Consequences of exposure to an energy rich diet during development. J. Physiol. 2005, 565, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Bruce, K.D.; Hanson, M.A. The developmental origins, mechanisms, and implications of metabolic syndrome. J. Nutr. 2010, 140, 648–652. [Google Scholar] [CrossRef] [PubMed]
- Stocker, C.J.; Arch, J.R.S.; Cawthorne, M.A. Fetal origins of insulin resistance and obesity. Proc. Nutr. Soc. 2007, 64, 143–151. [Google Scholar] [CrossRef]
- Wells, J.C.K. The thrifty phenotype as an adaptive maternal effect. Biol. Rev. Camb. Philos. Soc. 2007, 82, 143–172. [Google Scholar] [CrossRef] [PubMed]
- Rawana, S.; Clark, K.; Zhong, S.; Buison, A.; Chackunkal, S.; Jen, K.L. Low dose fructose ingestion during gestation and lactation affects carbohydrate metabolism in rat dams and their offspring. J. Nutr. 1993, 123, 2158–2165. [Google Scholar] [PubMed]
- Srinivasan, M.; Dodds, C.; Ghanim, H.; Gao, T.; Ross, P.J.; Browne, R.W.; Dandona, P.; Patel, M.S. Maternal obesity and fetal programming: effects of a high-carbohydrate nutritional modification in the immediate postnatal life of female rats. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E895–E903. [Google Scholar] [CrossRef] [PubMed]
- Alzamendi, A.; Castrogiovanni, D.; Gaillard, R.C.; Spinedi, E.; Giovambattista, A. Increased male offspring’s risk of metabolic-neuroendocrine dysfunction and overweight after fructose-rich diet intake by the lactating mother. Endocrinology 2010, 151, 4214–4223. [Google Scholar] [CrossRef] [PubMed]
- Vickers, M.H.; Clayton, Z.E.; Yap, C.; Sloboda, D.M. Maternal fructose intake during pregnancy and lactation alters placental growth and leads to sex-specific changes in fetal and neonatal endocrine function. Endocrinology 2011, 152, 1378–1387. [Google Scholar] [CrossRef] [PubMed]
- Stephan, B.C.M.; Wells, J.C.K.; Brayne, C.; Albanese, E.; Siervo, M. Increased fructose intake as a risk factor for dementia. J. Gerontol. A. Biol. Sci. Med. Sci. 2010, 65, 809–814. [Google Scholar] [CrossRef] [PubMed]
- Fergusson, M.A.; Koski, K.G. Comparison of effects of dietary glucose versus fructose during pregnancy on fetal growth and development in rats. J. Nutr. 1990, 120, 1312–1319. [Google Scholar] [PubMed]
- Mukai, Y.; Kumazawa, M.; Sato, S. Fructose intake during pregnancy up-regulates the expression of maternal and fetal hepatic sterol regulatory element-binding protein-1c in rats. Endocrine 2013, 44, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Zou, M.; Arentson, E.J.; Teegarden, D.; Koser, S.L.; Onyskow, L.; Donkin, S.S. Fructose consumption during pregnancy and lactation induces fatty liver and glucose intolerance in rats. Nutr. Res. 2012, 32, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Jen, K.L.; Rochon, C.; Zhong, S.B.; Whitcomb, L. Fructose and sucrose feeding during pregnancy and lactation in rats changes maternal and pup fuel metabolism. J. Nutr. 1991, 121, 1999–2005. [Google Scholar] [PubMed]
- Chen, C.-Y.O.; Crott, J.; Liu, Z.; Smith, D.E. Fructose and saturated fats predispose hyperinsulinemia in lean male rat offspring. Eur. J. Nutr. 2010, 49, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, L.; Otero, P.; Panadero, M.I.; Rodrigo, S.; Álvarez-Millán, J.J.; Bocos, C. Maternal fructose intake induces insulin resistance and oxidative stress in male, but not female, offspring. J. Nutr. Metab. 2015, 2015, 158091. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, L.; Panadero, M.I.; Roglans, N.; Otero, P.; Rodrigo, S.; Álvarez-Millán, J.J.; Laguna, J.C.; Bocos, C. Fructose only in pregnancy provokes hyperinsulinemia, hypoadiponectinemia, and impaired insulin signaling in adult male, but not female, progeny. Eur. J. Nutr. 2015, 55, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Lineker, C.; Kerr, P.M.; Nguyen, P.; Bloor, I.; Astbury, S.; Patel, N.; Budge, H.; Hemmings, D.G.; Plane, F.; Symonds, M.E.; et al. High fructose consumption in pregnancy alters the perinatal environment without increasing metabolic disease in the offspring. Reprod. Fertil.Dev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Alzamendi, A.; Del Zotto, H.; Castrogiovanni, D.; Romero, J.; Giovambattista, A.; Spinedi, E. Oral Metformin Treatment Prevents Enhanced Insulin Demand and Placental Dysfunction in the Pregnant Rat Fed a Fructose-Rich Diet. ISRN Endocrinol. 2012, 2012, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Giovambattista, A.; Piermaría, J.; Suescun, M.O.; Calandra, R.S.; Gaillard, R.C.; Spinedi, E. Direct effect of ghrelin on leptin production by cultured rat white adipocytes. Obesity 2006, 14, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Perello, M.; Castrogiovanni, D.; Giovambattista, A.; Gaillard, R.C.; Spinedi, E. Impairment in insulin sensitivity after early androgenization in the post-pubertal female rat. Life Sci. 2007, 80, 1792–1798. [Google Scholar] [CrossRef] [PubMed]
- Zubiría, M.G.; Fariña, J.P.; Moreno, G.; Gagliardino, J.J.; Spinedi, E.; Giovambattista, A. Excess fructose intake-induced hypertrophic visceral adipose tissue results from unbalanced precursor cell adipogenic signals. FEBS J. 2013, 280, 5864–5874. [Google Scholar] [CrossRef] [PubMed]
- Perelló, M.; Castrogiovanni, D.; Moreno, G.; Gaillard, R.C.; Spinedi, E. Neonatal hypothalamic androgenization in the female rat induces changes in peripheral insulin sensitivity and adiposity function at adulthood. Neuro Endocrinol. Lett. 2003, 24, 241–248. [Google Scholar] [PubMed]
- Giovambattista, A.; Chisari, A.N.; Gaillard, R.C.; Spinedi, E. Food intake-induced leptin secretion modulates hypothalamo-pituitary-adrenal axis response and hypothalamic Ob-Rb expression to insulin administration. Neuroendocrinology 2000, 72, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Alzamendi, A.; Giovambattista, A.; Raschia, A.; Madrid, V.; Gaillard, R.C.; Rebolledo, O.; Gagliardino, J.J.; Spinedi, E. Fructose-rich diet-induced abdominal adipose tissue endocrine dysfunction in normal male rats. Endocrine 2009, 35, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Langley, S.C.; Jackson, A.A. Increased Systolic Blood Pressure in Adult Rats Induced by Fetal Exposure to Maternal Low Protein Diets. Clin. Sci. 1994, 86, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Cambonie, G.; Comte, B.; Yzydorczyk, C.; Ntimbane, T.; Germain, N.; Lê, N.L.O.; Pladys, P.; Gauthier, C.; Lahaie, I.; Abran, D.; et al. Antenatal antioxidant prevents adult hypertension, vascular dysfunction, and microvascular rarefaction associated with in utero exposure to a low-protein diet. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 292, R1236–R1245. [Google Scholar] [CrossRef] [PubMed]
- Theys, N.; Ahn, M.-T.; Bouckenooghe, T.; Reusens, B.; Remacle, C. Maternal malnutrition programs pancreatic islet mitochondrial dysfunction in the adult offspring. J. Nutr. Biochem. 2011, 22, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Howie, G.J.; Sloboda, D.M.; Kamal, T.; Vickers, M.H. Maternal nutritional history predicts obesity in adult offspring independent of postnatal diet. J. Physiol. 2009, 587, 905–915. [Google Scholar] [CrossRef] [PubMed]
- Ashino, N.G.; Saito, K.N.; Souza, F.D.; Nakutz, F.S.; Roman, E.A.; Velloso, L.A.; Torsoni, A.S.; Torsoni, M.A. Maternal high-fat feeding through pregnancy and lactation predisposes mouse offspring to molecular insulin resistance and fatty liver. J. Nutr. Biochem. 2012, 23, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Ghezzi, A.C.; Cambri, L.T.; Ribeiro, C.; Botezelli, J.D.; Mello, M.A.R. Impact of early fructose intake on metabolic profile and aerobic capacity of rats. Lipids Health Dis. 2011, 10, 3. [Google Scholar] [CrossRef] [PubMed]
- Ching, R.H.H.; Yeung, L.O.Y.; Tse, I.M.Y.; Sit, W.-H.; Li, E.T.S. Supplementation of bitter melon to rats fed a high-fructose diet during gestation and lactation ameliorates fructose-induced dyslipidemia and hepatic oxidative stress in male offspring. J. Nutr. 2011, 141, 1664–1672. [Google Scholar] [CrossRef] [PubMed]
- Clayton, Z.E.; Vickers, M.H.; Bernal, A.; Yap, C.; Sloboda, D.M. Early Life Exposure to Fructose Alters Maternal, Fetal and Neonatal Hepatic Gene Expression and Leads to Sex-Dependent Changes in Lipid Metabolism in Rat Offspring. PLoS ONE 2015, 10, e0141962. [Google Scholar] [CrossRef] [PubMed]
- Skurk, T.; Alberti-Huber, C.; Herder, C.; Hauner, H. Relationship between adipocyte size and adipokine expression and secretion. J. Clin. Endocrinol. Metab. 2007, 92, 1023–1033. [Google Scholar] [CrossRef] [PubMed]
- Franck, N.; Stenkula, K.G.; Ost, A.; Lindström, T.; Strålfors, P.; Nystrom, F.H. Insulin-induced GLUT4 translocation to the plasma membrane is blunted in large compared with small primary fat cells isolated from the same individual. Diabetologia 2007, 50, 1716–1722. [Google Scholar] [CrossRef] [PubMed]
- Wåhlen, K.; Sjölin, E.; Löfgren, P. Role of fat cell size for plasma leptin in a large population based sample. Exp. Clin. Endocrinol. Diabetes 2011, 119, 291–294. [Google Scholar] [CrossRef] [PubMed]
- Walder, K.; Filippis, A.; Clark, S.; Zimmet, P.; Collier, G.R. Leptin inhibits insulin binding in isolated rat adipocytes. J. Endocrinol. 1997, 155, R5–R7. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.A.; Tao, C.; Gupta, R.K.; Scherer, P.E. Tracking adipogenesis during white adipose tissue development, expansion and regeneration. Nat. Med. 2013, 19, 1338–1344. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, J.C.; Lisboa, P.C.; de Moura, E.G.; Barella, L.F.; Miranda, R.A.; Malta, A.; Franco, C.C.; Ribeiro, T.A.; Torrezan, R.; Gravena, C.; et al. Poor pubertal protein nutrition disturbs glucose-induced insulin secretion process in pancreatic islets and programs rats in adulthood to increase fat accumulation. J. Endocrinol. 2013, 216, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Muhlhausler, B.; Smith, S.R. Early-life origins of metabolic dysfunction: Role of the adipocyte. Trends Endocrinol. Metab. 2009, 20, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Maumus, M.; Sengenès, C.; Decaunes, P.; Zakaroff-Girard, A.; Bourlier, V.; Lafontan, M.; Galitzky, J.; Bouloumié, A. Evidence of in situ proliferation of adult adipose tissue-derived progenitor cells: influence of fat mass microenvironment and growth. J. Clin. Endocrinol. Metab. 2008, 93, 4098–4106. [Google Scholar] [CrossRef] [PubMed]
- Musri, M.M.; Párrizas, M. Epigenetic regulation of adipogenesis. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Masuyama, H.; Hiramatsu, Y. Effects of a high-fat diet exposure in utero on the metabolic syndrome-like phenomenon in mouse offspring through epigenetic changes in adipocytokine gene expression. Endocrinology 2012, 153, 2823–2830. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-E.; Ge, K. Transcriptional and epigenetic regulation of PPARγ expression during adipogenesis. Cell Biosci. 2014, 4, 29. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, K.; Kano, F.; Shiota, K.; Murata, M. Expression of the peroxisome proliferator activated receptor gamma gene is repressed by DNA methylation in visceral adipose tissue of mouse models of diabetes. BMC Biol. 2009, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Yokomori, N.; Tawata, M.; Onaya, T. DNA demethylation modulates mouse leptin promoter activity during the differentiation of 3T3-L1 cells. Diabetologia 2002, 45, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Yokomori, N.; Tawata, M.; Onaya, T. DNA demethylation during the differentiation of 3T3-L1 cells affects the expression of the mouse GLUT4 gene. Diabetes 1999, 48, 685–690. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, T.K.; Idelman, G.; Blanco, V.; Blomkalns, A.L.; Piegore, M.G.; Weintraub, D.S.; Kumar, S.; Rajsheker, S.; Manka, D.; Rudich, S.M.; et al. Histone deacetylase 9 is a negative regulator of adipogenic differentiation. J. Biol. Chem. 2011, 286, 27836–27847. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Bhanot, S.; Yao, L.; McNeill, J.H. Vascular insulin resistance in fructose-hypertensive rats. Eur. J. Pharmacol. 1997, 322, R1–R2. [Google Scholar] [CrossRef]
- Dupas, J.; Goanvec, C.; Feray, A.; Guernec, A.; Alain, C.; Guerrero, F.; Mansourati, J. Progressive Induction of Type 2 Diabetes: Effects of a Reality-Like Fructose Enriched Diet in Young Wistar Rats. PLoS ONE 2016, 11, e0146821. [Google Scholar] [CrossRef] [PubMed]
- Chakraborti, C.K. Role of adiponectin and some other factors linking type 2 diabetes mellitus and obesity. World J. Diabetes 2015, 6, 1296–1308. [Google Scholar] [CrossRef] [PubMed]
- Awazawa, M.; Ueki, K.; Inabe, K.; Yamauchi, T.; Kubota, N.; Kaneko, K.; Kobayashi, M.; Iwane, A.; Sasako, T.; Okazaki, Y.; et al. Adiponectin enhances insulin sensitivity by increasing hepatic IRS-2 expression via a macrophage-derived IL-6-dependent pathway. Cell Metab. 2011, 13, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Hajer, G.R.; van Haeften, T.W.; Visseren, F.L.J. Adipose tissue dysfunction in obesity, diabetes, and vascular diseases. Eur. Heart J. 2008, 29, 2959–2971. [Google Scholar] [CrossRef] [PubMed]
- Bastard, J.-P.; Maachi, M.; Lagathu, C.; Kim, M.J.; Caron, M.; Vidal, H.; Capeau, J.; Feve, B. Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur. Cytokine Netw. 2006, 17, 4–12. [Google Scholar] [PubMed]
- Yoon, M.J.; Lee, G.Y.; Chung, J.-J.; Ahn, Y.H.; Hong, S.H.; Kim, J.B. Adiponectin increases fatty acid oxidation in skeletal muscle cells by sequential activation of AMP-activated protein kinase, p38 mitogen-activated protein kinase, and peroxisome proliferator-activated receptor alpha. Diabetes 2006, 55, 2562–2570. [Google Scholar] [CrossRef] [PubMed]

| C | F | |
|---|---|---|
| GLU (mmol/L) | 6.58 ± 0.18 | 6.08 ± 0.24 |
| TG (mmol/L) | 1.44 ± 0.14 | 1.12 ± 0.06 * |
| TC (mmol/L) | 1.56 ± 0.07 | 1.88 ± 0.13 |
| CORT (nmol/L) | 4.67 ± 1.08 | 3.43 ± 1.02 |
| INS (nmol/L) | 0.31 ± 0.02 | 0.26 ± 0.05 |
| LEP (ng/mL) | 2.41 ± 0.35 | 4.10 ± 0.31 * |
| ADIPOQ (μg/mL) | 5.87 ± 0.72 | 5.60 ± 0.44 |
| C | F | |
|---|---|---|
| Pad mass (g per 100g BW) | 0.71 ± 0.07 | 0.54 ± 0.03 * |
| Adipocyte diameter (µm) | 37.21 ± 0.09 | 48.45 ± 0.28 * |
| Adipocyte area (µm2) | 1182.34 ± 10.64 | 2057.79 ± 23.57 * |
| Adipocyte volume (μm3 × 103) | 26.97 ± 1.84 | 59.97 ± 3.47 * |
| LEP mRNA (AU) | 1.09 ± 0.19 | 2.99 ± 0.18 * |
| ADIPOQ mRNA (AU) | 1.28 ± 0.40 | 1.94 ± 0.41 |
| CC | CF | FC | FF | |
|---|---|---|---|---|
| GLU (mmol/L) | 4.96 ± 0.27 | 5.1 ± 0.33 | 5.88 ± 0.28 * | 7.91± 0.56 *,+,# |
| TG (mmol/L) | 1.19 ± 0.16 | 1.89 ± 0.17 * | 1.7 ± 0.12 * | 2.62 ± 0.11 *,+,# |
| TC (mmol/L) | 0.26 ± 0.03 | 0.23 ± 0.01 | 0.23 ± 0.001 | 0.21 ± 0.02 |
| CORT (nmol/L) | 140.81 ± 37.41 | 87.45 ± 24.15 | 84.52 ± 12.99 | 190.98 ± 58.14 |
| INS (nmol/L) | 0.28 ± 0.04 | 0.31 ± 0.04 | 0.37 ± 0.06 | 0.34 ± 0.05 |
| LEP (ng/mL) | 2.87 ± 0.14 | 3.89 ± 0.38 * | 3.89 ± 0.43 * | 5.57 ± 1.4 * |
| ADIPOQ (μg/mL) | 14.82 ± 0.94 | 20.08 ± 2.77 * | 13.61 ± 1.95 | 14.89 ± 2.72 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alzamendi, A.; Zubiría, G.; Moreno, G.; Portales, A.; Spinedi, E.; Giovambattista, A. High Risk of Metabolic and Adipose Tissue Dysfunctions in Adult Male Progeny, Due to Prenatal and Adulthood Malnutrition Induced by Fructose Rich Diet. Nutrients 2016, 8, 178. https://doi.org/10.3390/nu8030178
Alzamendi A, Zubiría G, Moreno G, Portales A, Spinedi E, Giovambattista A. High Risk of Metabolic and Adipose Tissue Dysfunctions in Adult Male Progeny, Due to Prenatal and Adulthood Malnutrition Induced by Fructose Rich Diet. Nutrients. 2016; 8(3):178. https://doi.org/10.3390/nu8030178
Chicago/Turabian StyleAlzamendi, Ana, Guillermina Zubiría, Griselda Moreno, Andrea Portales, Eduardo Spinedi, and Andrés Giovambattista. 2016. "High Risk of Metabolic and Adipose Tissue Dysfunctions in Adult Male Progeny, Due to Prenatal and Adulthood Malnutrition Induced by Fructose Rich Diet" Nutrients 8, no. 3: 178. https://doi.org/10.3390/nu8030178
APA StyleAlzamendi, A., Zubiría, G., Moreno, G., Portales, A., Spinedi, E., & Giovambattista, A. (2016). High Risk of Metabolic and Adipose Tissue Dysfunctions in Adult Male Progeny, Due to Prenatal and Adulthood Malnutrition Induced by Fructose Rich Diet. Nutrients, 8(3), 178. https://doi.org/10.3390/nu8030178




