Health-Care Costs, Glycemic Control and Nutritional Status in Malnourished Older Diabetics Treated with a Hypercaloric Diabetes-Specific Enteral Nutritional Formula
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design
2.2. Recorded Variables
2.3. Cost Per Controlled Patient
2.4. Correlation and Regression Analysis
2.5. Statistical Analysis
2.6. Ethical Statement
3. Results
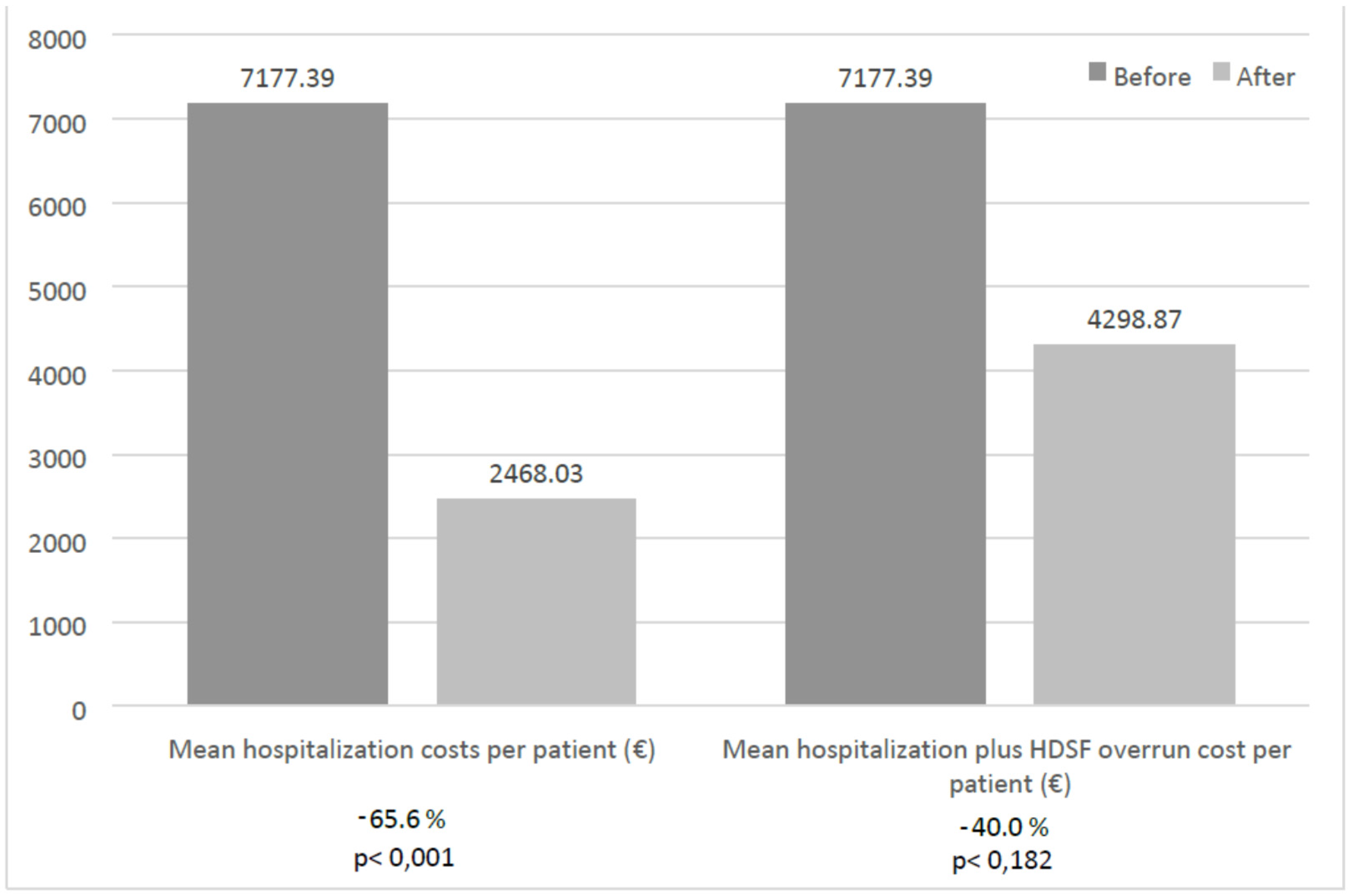
3.1. Use of Health-Care Resources and Health-Care Costs
3.2. Glucose Control Status
3.3. Nutritional Laboratory Parameters
3.4. Cost Per Controlled Patient
3.5. Correlation and Regression Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| HDSF | hypercaloric diabetes-specific formula |
| DSF | diabetes-specific formula |
| T2DM | type-2 diabetes mellitus |
| DM | Diabetes mellitus |
| SD | standard deviation |
| BMI | body mass index |
| DPP4 | Dipeptidyl Peptidase IV |
| HRQoL | Health Related Quality of Life |
| HbA1c | glycosylated hemoglobin |
References
- International Diabetes Federation, Atlas, International Diabetes Federation, 6th ed., 2014. Available online: https://www.idf.org/sites/default/files/EN_6E_Atlas_Full_0.pdf (accessed on 13 January 2016).
- Zhuo, X.; Zhang, P.; Hoerger, T.J. Lifetime direct medical costs of treating type 2 diabetes and diabetic complications. Am. J. Prev. Med. 2013, 45, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Zhang, X.; Brown, J.; Vistisen, D.; Sicree, R.; Shaw, J.; Nichols, G. Global healthcare expenditure on diabetes for 2010 and 2030. Diabetes Res. Clin. Pract. 2010, 87, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Tunceli, K.; Bradley, C.J.; Nerenz, D.; Williams, L.K.; Pladevall, M.; Elston Lafata, J. The impact of diabetes on employment and work productivity. Diabetes Care 2005, 28, 2662–2667. [Google Scholar] [CrossRef] [PubMed]
- Alexander, G.C.; Sehgal, N.L.; Moloney, R.M.; Stafford, R.S. National trends in treatment of type 2 diabetes mellitus, 1994–2007. Arch. Intern. Med. 2008, 168, 2088–2094. [Google Scholar] [CrossRef] [PubMed]
- Hamdy, O.; Ernst, F.R.; Baumer, D.; Mustad, V.; Partridge, J.; Hegazi, R. Differences in resource utilization between patients with diabetes receiving glycemia-targeted specialized nutrition vs. standard nutrition formulas in U.S. hospitals. J. Parenter. Enter. Nutr. 2014, 38, 86S–91S. [Google Scholar] [CrossRef] [PubMed]
- Sanz París, A.; García, J.M.; Gómez-Candela, C.; Burgos, R.; Martín, Á.; Matía, P. Malnutrition prevalence in hospitalized elderly diabetic patients. Nutr. Hosp. 2013, 28, 592–599. [Google Scholar] [PubMed]
- Kelly, I.E.; Tessier, S.; Cahill, A.; Morris, S.E.; Crumley, A.; McLaughlin, D.; McKee, R.F.; Lean, M.E. Still hungry in hospital: Identifying malnutrition in acute admissions. QJM 2000, 93, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Alish, C.J.; Garvey, W.T.; Maki, K.C.; Sacks, G.S.; Hustead, D.S.; Hegazi, R.A.; Mustard, V.A. A diabetes-specific enteral formula improves glycemic variability in patients with type 2 diabetes. Diabetes Technol. Ther. 2010, 12, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Ojo, O.; Brooke, J. Evaluation of the role of enteral nutrition in managing patients with diabetes: A systematic review. Nutrients 2014, 6, 5142–5152. [Google Scholar] [CrossRef] [PubMed]
- Elia, M.; Ceriello, A.; Laube, H.; Sinclair, A.J.; Engfer, M.; Stratton, R.J. Enteral nutrition support and use of diabetes-specific formulas for patients with diabetes: A systematic review and meta-analysis. Diabetes Care 2005, 28, 2267–2279. [Google Scholar] [CrossRef] [PubMed]
- McMahon, M.M.; Nystrom, E.; Braunschweig, C.; Miles, J.; Compher, C. ASPEN clinical guidelines: Nutrition support of adult patients with hyperglycemia. J. Parenter. Enter. Nutr. 2013, 37, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Philipson, T.; Linthicum, M.T.; Thornton, J. Tutorial on Health economics and Outcomes research in Nutrition. J. Parenter. Enter. Nutr. 2014, 38, 5S–16S. [Google Scholar] [CrossRef] [PubMed]
- CCEMG-EPPI-Centre Cost Converter (v.1.4 Last Update: 27 January 2014). Available online: http://eppi.ioe.ac.uk/costconversion/default.aspx (accessed on 13 January 2016).
- Agencia Estatal Boletín Oficial del Estado. Real Decreto 1205/2010, de 24 de Septiembre, por el que se Fijan las Bases Para la Inclusión de los Alimentos Dietéticos Para Usos Médicos Especiales en la Prestación con Productos Dietéticos del Sistema Nacional de Salud y Para el Establecimiento de sus Importes Máximos de Financiación. Available online: https://www.boe.es/diario_boe/txt.php?id=BOE-A-2010-14660 (accessed on 13 January 2016). (In Spanish)
- Inzucchi, S.E.; Berfenstal, R.M.; Buse, J.B.; Diamant, M.; Ferrannini, E.; Nauck, M.; Peters, A.L.; Tsapas, A.; Wender, R.; Matthews, D.R. Management of Hyperglycemia in type 2 diabetes: A patient centered approach: Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2012, 35, 1364–1379. [Google Scholar] [CrossRef] [PubMed]
- Caberizo, S.; Cuadras, D.; Gomex-Busto, F.; Artaza-Artabe, I.; Martin-Ciancas, F.; Malafarina, V. Serum albumin and health in older people: Review and meta-analysis. Maturitas 2015, 81, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Langer, J.; Hunt, B.; Valentine, W.J. Evaluating the short-term cost effectiveness of liraglutide versus sitagliptine in patients with type 2 diabetes failing metformin monotherapy in the United States. J. Manag. Care Pharm. 2013, 19, 237–246. [Google Scholar] [PubMed]
- Scalone, L.; Cesana, G.; Furneri, G.; Ciampichini, R.; Beck-Peccoz, P.; Chiodini, V.; Mangioni, S.; Orsi, E.; Fornari, C.; Mantovani, L.G. Burden of diabetes mellitus estimated with a longitudinal population based study using administrative databases. PLoS ONE 2014, 9, e113741. [Google Scholar] [CrossRef] [PubMed]
- Umpierrez, G.E.; Hellman, R.; Korytkowski, M.T.; Kosiborod, M.; Maynard, G.A.; Montori, V.M.; Seley, J.J.; Van den Berghe, G. Management of hyperglycemia in hospitalized patients in non-critical care setting: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2012, 97, 16–38. [Google Scholar] [CrossRef] [PubMed]
- Lansink, M.; van Laere, K.M.; Vendrig, L.; Rutten, G.E. Lower postprandial glucose responses at baseline and after 4 weeks use of a diabetes-specific formula in diabetes type 2 patients. Diabetes Res. Clin. Pract. 2011, 93, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Vaisman, N.; Lansink, M.; Rouws, C.H.; van Laere, K.M.; Segal, R.; Niv, E.; Bowling, T.E.; Waitzberg, D.L.; Morley, J.E. Tube feeding with a diabetes-specific feed for 12 weeks improves glycaemic control in type 2 diabetes patients. Clin. Nutr. 2009, 28, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Cook, C.B.; Elias, B.; Kongable, G.L.; Potter, D.J.; Shepherd, K.M.; McMahon, D. Diabetes and hyperglycemia quality improvement efforts in hospitals in the United States: Current status, practice variation, and barriers to implementation. Endocr. Pract. 2010, 16, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Simmons, D.; Wenzel, H. Diabetes inpatients: A case of lose, lose, lose. Is it time to use a ‘diabetes-attributable hospitalization cost’ to assess the impact of diabetes? Diabet. Med. 2011, 28, 1123–1130. [Google Scholar] [CrossRef] [PubMed]
- Ordoñez, A.M.; Schieferdecker, M.E.M.; Cestonaro, T.; Cardoso Neto, J.; Ligocki Campos, A.C. Nutritional status influences the length of stay and clinical outcomes in patients hospitalized in internal medicine wards. Nutr. Hosp. 2013, 28, 1313–1320. [Google Scholar] [PubMed]
- Blanco-Moreno, A.; Urbanos-Garrido, R.M.; Thuissard-Vasallo, I. Public healthcare expenditure in Spain: Measuring the impact of driving factors. Health Policy 2013, 111, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Hernández, J.; León Sanz, M.; Planas Vilá, M.; Araujo, K.; García de Lorenzo, A.; Celaya Pérez, S. Prevalence and costs of malnutrition in hospitalized patients; the PREDyCES Study. Nutr. Hosp. 2012, 27, 1049–1059. [Google Scholar] [PubMed]
- Covinsky, K.E.; Covinsky, M.H.; Palmer, R.M.; Sehgal, A.R. Serum albumin concentration and clinical assessment of nutritional status in hospitalized older perple: Different sides of different coins? JAGS 2002, 50, 631–637. [Google Scholar] [CrossRef]
- Röhrig, G.; Becker, I.; Polidori, M.C.; Schulz, R.J.; Noreik, M. Association of anemia and hypoalbuminemia in German geriatric inpatients. Relationship to nutritional status and comprehensive geriatric assessment. Z. Gerontol. Geriatr. 2015, 48, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Ferrill, M.J.; Brown, D.A.; Kyle, J.A. Clinical versus statistical significance: Interpreting p values and confidence intervals related to measures of association to guide decision making. J. Pharm. Pract. 2010, 23, 344–351. [Google Scholar] [CrossRef] [PubMed]
- McCabe, C.; Claxton, K.; Culyer, A.J. The NICE cost-effectiveness threshold: What it is and what that means. Pharmacoeconomics 2008, 26, 733–744. [Google Scholar] [CrossRef] [PubMed]
- Kubota, K.; Kadomura, T.; Ohta, K.; Koyama, K.; Okuda, H.; Kobayashi, M.; Ishii, C.; Fujiwara, Y.; Nishiora, T.; Ohmae, Y.; et al. Analyses of laboratory data and establishment of reference values and intervals for healthy elderly people. J. Nutr. Health Aging 2012, 16, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Higgins, P.A.; Daly, B.J.; Lipson, A.R.; Guo, S.E. Assessing nutritional status in chronically critically ill adult patients. Am. J. Crit. Care 2006, 15, 166–176. [Google Scholar] [PubMed]
- Leandro-Merhi, V.A.; de Aquino, J.L.; Sales Chagas, J.F. Nutrition status and risk factors associated with length of hospital stay for surgical patients. J. Parenter. Enter. Nutr. 2011, 35, 241–248. [Google Scholar] [CrossRef] [PubMed]

| (A) Sociodemographic and Clinical Variables | Patients |
| Mean age (SD) | 84.9 (10.8) |
| Mean BMI (SD) | 23.55 (3.16) |
| Mean time since the diagnosis of T2DM (SD) | 7.6 (4.8) |
| Mean Charlson index (SD) | 4.8 (1.6) |
| Males | 48.4% |
| Living at home | 75.3% |
| Living at nursing homes | 22.6% |
| (B) Pharmacological Treatment | |
| Metformin | 26% |
| Dietetic treatment | 23% |
| Insulin glargine | 19% |
| Sulfonylureas | 17% |
| Mixed insulin | 8% |
| DPP4 inhibitors | 7% |
| (C) Comorbidities | |
| Hypertension | 80% |
| Dyslipidemia | 63% |
| Heart Disease | 51% |
| Brain disease | 40% |
| Neuropathy | 22% |
| Nephropathy | 18% |
| Retinopathy | 18% |
| Variable | Before | After | Dif. (%) | p-Value | ||||
|---|---|---|---|---|---|---|---|---|
| N | Mean | SD | N | Mean | SD | |||
| Glucose (g/dL) | 93 | 128.98 | 60.87 | 93 | 122.78 | 56.65 | −4.8 | 0.428 |
| HbA1c (%) | 84 | 6.62 | 1.44 | 84 | 6.45 | 0.99 | −0.17 | 0.143 |
| Albumin (g/dL) | 93 | 3.12 | 0.34 | 93 | 3.45 | 0.46 | 10.6 | 0.000 |
| Creatinine (mg/dL) | 92 | 1.05 | 0.69 | 78 | 1.21 | 1.18 | 15.2 | 0.307 |
| Cholesterol (mg/dL) | 89 | 168.43 | 62.80 | 76 | 172.30 | 72.95 | 2.3 | 0.462 |
| LDL (mg/dL) | 62 | 101.08 | 30.07 | 53 | 98.70 | 33.43 | −2.4 | 0.492 |
| HDL (mg/dL) | 67 | 42.82 | 16.59 | 54 | 43.85 | 15.39 | 2.4 | 0.451 |
| Triglycerides (mg/dL) | 69 | 144.19 | 93.75 | 54 | 131.33 | 55.45 | −8.9 | 0.163 |
| Iron (μg/dL) | 60 | 52.70 | 35.63 | 57 | 47.60 | 24.51 | −9.7 | 0.503 |
| Ferritin (ng/dL) | 59 | 267.94 | 356.97 | 55 | 172.24 | 185.75 | −35.7 | 0.322 |
| Hemoglobin (g/dL) | 64 | 11.35 | 2.17 | 59 | 12.08 | 1.80 | 6.4 | 0.026 |
| Lymphocytes (%) | 64 | 21.85 | 10.89 | 57 | 23.54 | 9.48 | 9.5 | 0.326 |
| Vitamin B9 (pg/mL) | 48 | 9.74 | 5.87 | 40 | 11.00 | 7.13 | 12.9 | 0.073 |
| Vitamin B12 (pg/mL) | 47 | 347.26 | 230.51 | 40 | 441.28 | 399.28 | 27.1 | 0.412 |
| Before and After Variation | Hospitalization Length | Emergency Visits | Hospitalization Costs | Hospitalization + HDSF Overrun Costs |
|---|---|---|---|---|
| HbA1c | 0.273 | 0.271 | 0.268 | |
| Glucose | - | 0.233 | - | - |
| Hemoglobin | −0.369 | - | −0.364 | −0.364 |
| Lymphocytes | −0.350 | - | −0.349 | −0.349 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanz-Paris, A.; Boj-Carceller, D.; Lardies-Sanchez, B.; Perez-Fernandez, L.; Cruz-Jentoft, A.J. Health-Care Costs, Glycemic Control and Nutritional Status in Malnourished Older Diabetics Treated with a Hypercaloric Diabetes-Specific Enteral Nutritional Formula. Nutrients 2016, 8, 153. https://doi.org/10.3390/nu8030153
Sanz-Paris A, Boj-Carceller D, Lardies-Sanchez B, Perez-Fernandez L, Cruz-Jentoft AJ. Health-Care Costs, Glycemic Control and Nutritional Status in Malnourished Older Diabetics Treated with a Hypercaloric Diabetes-Specific Enteral Nutritional Formula. Nutrients. 2016; 8(3):153. https://doi.org/10.3390/nu8030153
Chicago/Turabian StyleSanz-Paris, Alejandro, Diana Boj-Carceller, Beatriz Lardies-Sanchez, Leticia Perez-Fernandez, and Alfonso J. Cruz-Jentoft. 2016. "Health-Care Costs, Glycemic Control and Nutritional Status in Malnourished Older Diabetics Treated with a Hypercaloric Diabetes-Specific Enteral Nutritional Formula" Nutrients 8, no. 3: 153. https://doi.org/10.3390/nu8030153
APA StyleSanz-Paris, A., Boj-Carceller, D., Lardies-Sanchez, B., Perez-Fernandez, L., & Cruz-Jentoft, A. J. (2016). Health-Care Costs, Glycemic Control and Nutritional Status in Malnourished Older Diabetics Treated with a Hypercaloric Diabetes-Specific Enteral Nutritional Formula. Nutrients, 8(3), 153. https://doi.org/10.3390/nu8030153




