An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity
Abstract
:1. Introduction
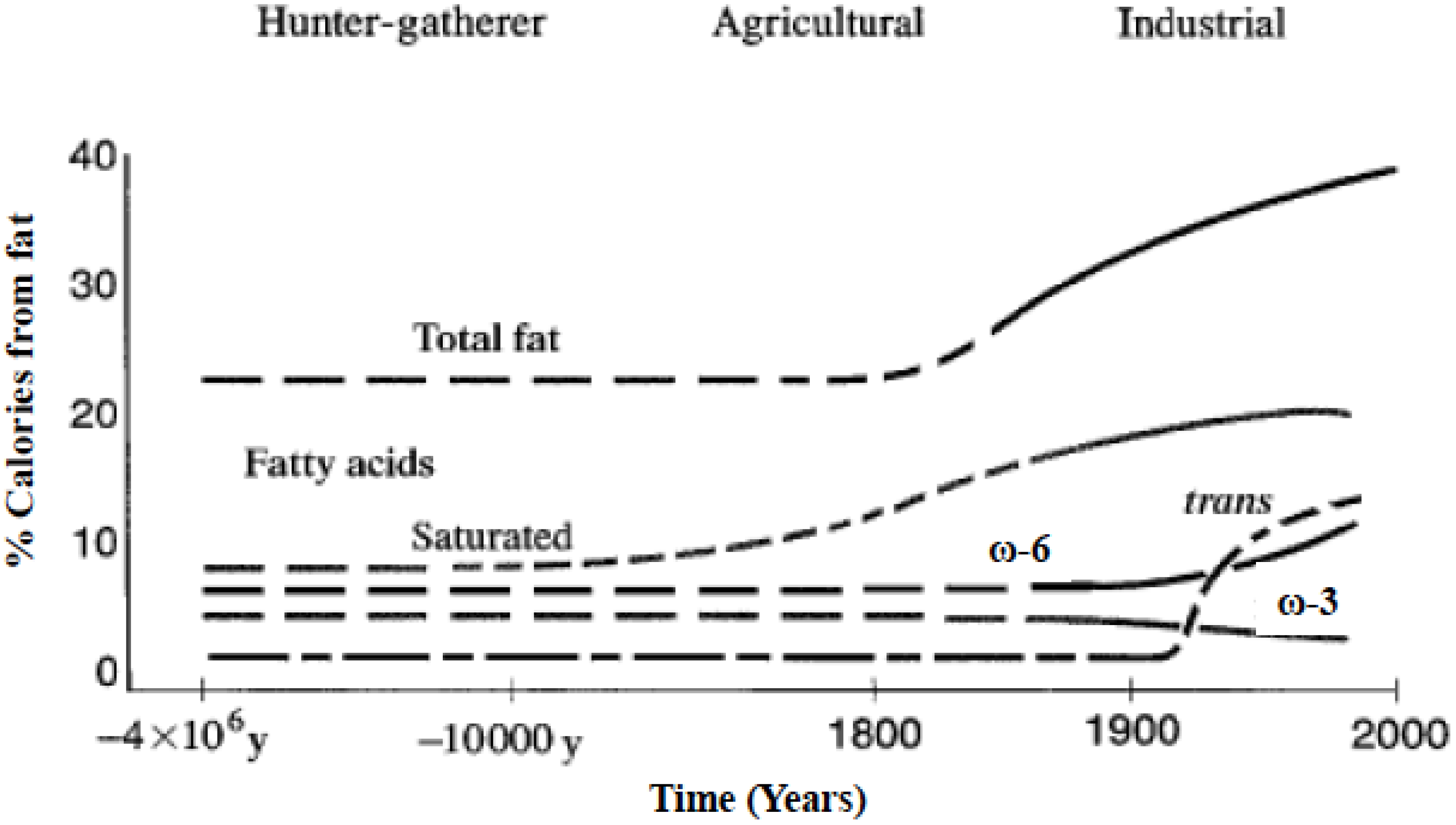
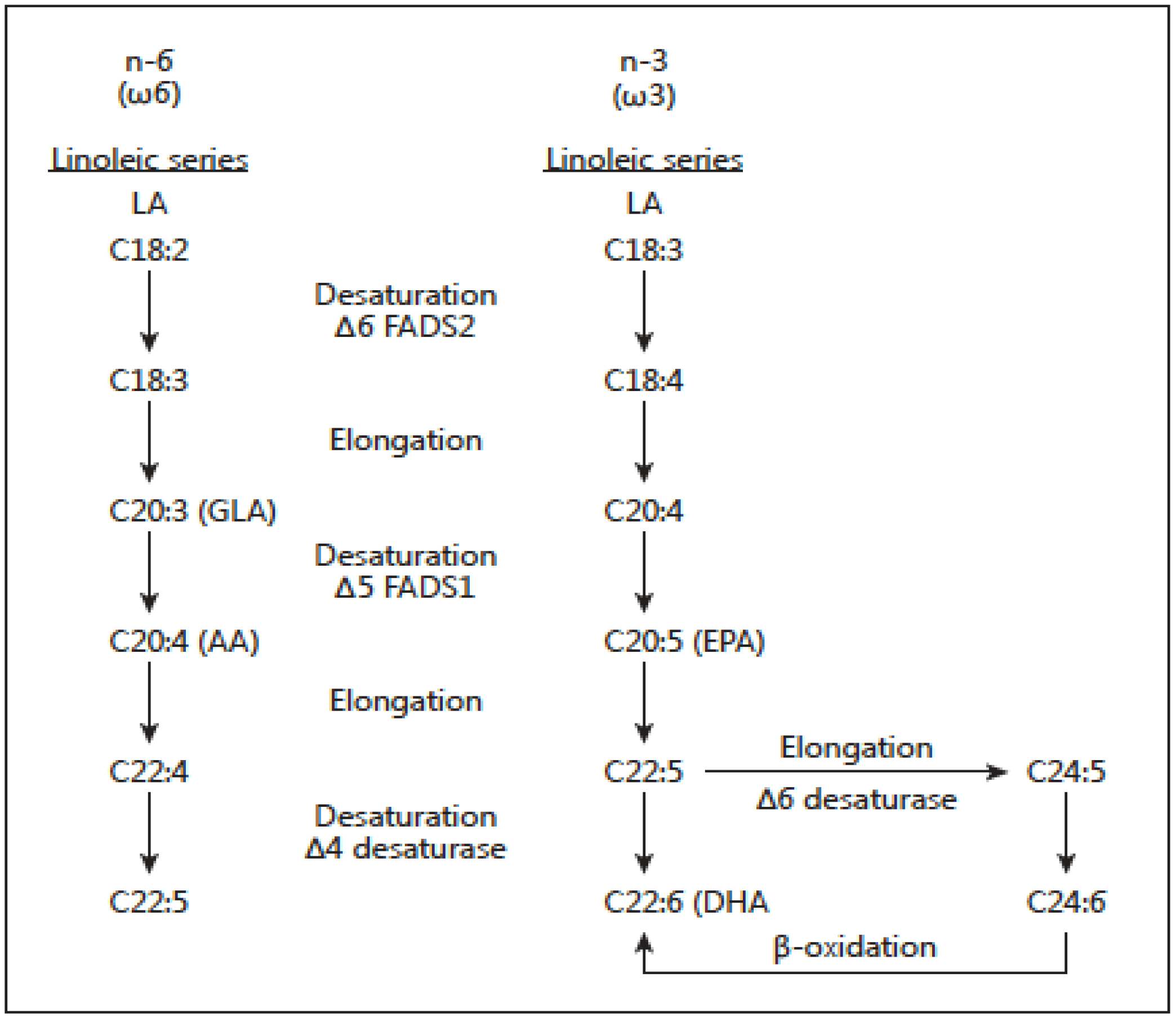
2. The Importance of the Omega-6/Omega-3 Fatty Acid Ratio: Metabolic, Physiological and Evolutionary Aspects
3. Effects of Omega-6 and Omega-3 Fatty Acids and their Ratio on Obesity
3.1. The Fat-1 Transgenic Mouse Model
3.2. Human Studies
4. Genetics: The Fat Mass and Obesity-Associated Gene
5. Omega-6/Omega-3 Fatty Acid Ratio: Endocannabinoid System
6. Conclusions and Recommendations
- Human beings evolved on a diet that was balanced in the omega-6 and omega-3 essential fatty acids.
- A high omega-6 fatty acid intake and a high omega-6/omega-3 ratio are associated with weight gain in both animal and human studies, whereas a high omega-3 fatty acid intake decreases the risk for weight gain. Lowering the LA/ALA ratio in animals prevents overweight and obesity.
- Omega-6/omega-3 fatty acids compete for their biosynthetic enzymes and because they have distinct physiological and metabolic properties, their balanced omega-6/omega-3 ratio is a critical factor for health throughout the life cycle.
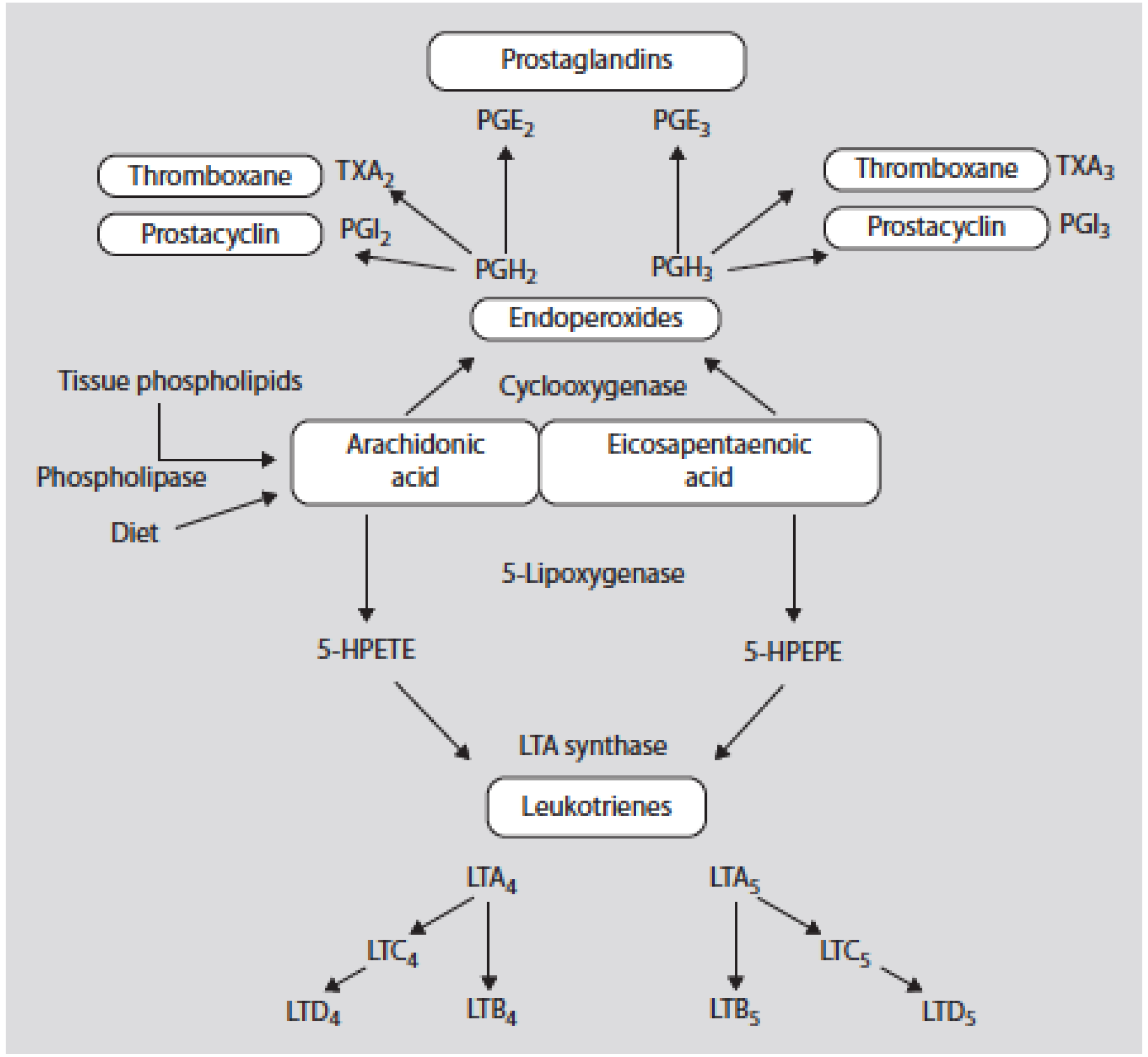
- Adipose tissue is the main peripheral target organ handling fatty acids, and AA is required for adipocyte differentiation (adipogenesis). The increased LA and AA content of foods has been accompanied by a significant increase in the AA/EPA + DHA ratio within adipose tissue, leading to increased production in AA metabolites, PGI2 which stimulates white adipogenesis and PGF2α which inhibits the browning process, whereas increased consumption of EPA and DHA leads to adipose tissue homeostasis through adipose tissue loss and increased mitochondrial biogenesis.
- High omega-6 fatty acid intake leads to hyperactivity of endocannabinoid system, whereas omega-3 fatty acids lead to normal homeostasis (decrease hyperactivity).
- High omega-6 fatty acids increase leptin resistance and insulin resistance, whereas omega-3 fatty acids lead to homeostasis and weight loss.
- Because a high omega-6/omega-3 ratio is associated with overweight/obesity, whereas a balanced ratio decreases obesity and weight gain, it is essential that every effort is made to decrease the omega-6 fatty acids in the diet, while increasing the omega-3 fatty acid intake. This can be accomplished by (1) changing dietary vegetable oils high in omega-6 fatty acids (corn oil, sunflower, safflower, cottonseed, and soybean oils) to oils high in omega-3s (flax, perilla, chia, rapeseed), and high in monounsaturated oils such as olive oil, macadamia nut oil, hazelnut oil, or the new high monounsaturated sunflower oil; and (2) increasing fish intake to 2–3 times per week, while decreasing meat intake.
- In clinical investigations and intervention trials it is essential that the background diet is precisely defined in terms of the omega-6 and omega-3 fatty acid content. Because the final concentrations of omega-6 and omega-3 fatty acids are determined by both dietary intake and endogenous metabolism, it is essential that in all clinical investigations and intervention trials the omega-6 and omega-3 fatty acids are precisely determined in the red blood cell membrane phospholipids. In severe obesity drugs and bariatric surgery have been part of treatment.
- The risk allele rs 1421085 T to C SNV in intron 1 and 2 in the FTO gene functioned similarly to AA metabolites, PGI2 and PGF2a increasing proliferation of white adipose tissue and decreasing its browning respectively, whereas the knockdown of IRX3 and IRX5 genes functioned similarly to omega-3 fatty acid metabolites increasing the browning of white adipose tissue, mitochondrial biogenesis, and thermogenesis. Therefore, further research should include studies on the effects of omega-3 fatty acids in blocking the effects of the risk allele (rs 1421085), which appears to be responsible for the association between the first intron of FTO gene and obesity in humans.
- In the future studies on genetic variants from GWAS will provide opportunities to precisely treat and prevent obesity by both nutritional and pharmaceutical interventions.
Acknowledgments
Conflicts of Interest
References
- Simopoulos, A.P. Evolutionary aspects of diet and essential fatty acids. In Fatty Acids and Lipids—New Findings; Hamazaki, T., Okuyama, H., Eds.; Karger: Basel, Switzerland, 2001; Volume 88, pp. 18–27. [Google Scholar]
- Kang, J.X. The importance of omega-6/omega-3 fatty acid ratio in cell function. The gene transfer of omega-3 fatty acid desaturase. In Omega-6/Omega-3 Essential Fatty Acid Ratio: The Scientific Evidence; Simopoulos, A.P., Cleland, L.G., Eds.; Karger: Basel, Switzerland, 2003; Volume 92, pp. 23–36. [Google Scholar]
- Simopoulos, A.P. The importance of the omega-6/omega-3 Fatty Acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Dietary Omega-3 Fatty Acid Deficiency and High Fructose Intake in the Development of Metabolic Syndrome, Brain Metabolic Abnormalities, and Non-Alcoholic Fatty Liver Disease. Nutrients 2013, 5, 2901–2923. [Google Scholar] [CrossRef] [PubMed]
- Donahue, S.M.; Rifas-Shiman, S.L.; Gold, D.R.; Jouni, Z.E.; Gillman, M.W.; Oken, E. Prenatal fatty acid status and child adiposity at age 3 years: Results from a US pregnancy cohort. Am. J. Clin. Nutr. 2011, 93, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Kromhout, D.; de Goede, J. Update on cardiometabolic health effects of ω-3 fatty acids. Curr. Opin. Lipidol. 2014, 25, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Kromann, N.; Green, A. Epidemiological studies in the Upernavik district, Greenland. Incidence of some chronic diseases 1950–1974. Acta Med. Scand. 1980, 208, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Adler, A.I.; Boyko, E.J.; Schraer, C.D.; Murphy, N.J. Lower prevalence of impaired glucose tolerance and diabetes associated with daily seal oil or salmon consumption among Alaska Natives. Diabetes Care 1994, 17, 1498–1501. [Google Scholar] [CrossRef] [PubMed]
- Schraer, C.D.; Risica, P.M.; Ebbesson, S.O.; Go, O.T.; Howard, B.V.; Mayer, A.M. Low fasting insulin levels in Eskimos compared to American Indians: are Eskimos less insulin resistant? Int. J. Circumpolar Health 1999, 58, 272–280. [Google Scholar] [PubMed]
- Nettleton, J.A.; Katz, R. n-3 long-chain polyunsaturated fatty acids in type 2 diabetes: A review. J. Am. Diet. Assoc. 2005, 105, 428–440. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Rimm, E.B. Fish intake, contaminants, and human health: Evaluating the risks and the benefits. JAMA 2006, 296, 1885–1899. [Google Scholar] [CrossRef] [PubMed]
- Birch, E.E.; Hoffman, D.R.; Castañeda, Y.S.; Fawcett, S.L.; Birch, D.G.; Uauy, R.D. A randomized controlled trial of long-chain polyunsaturated fatty acid supplementation of formula in term infants after weaning at 6 wk of age. Am. J. Clin. Nutr. 2002, 75, 570–580. [Google Scholar] [PubMed]
- Guesnet, P.; Pugo-Gunsam, P.; Maurage, C.; Pinault, M.; Giraudeau, B.; Alessandri, J.M.; Durand, G.; Antoine, J.M.; Couet, C. Blood lipid concenttrations of docosahexaenoic and arachidonic acids at birth determine their relative postnatal changes in term infants fed breast milk or formula. Am. J. Clin. Nutr. 1999, 70, 292–298. [Google Scholar] [PubMed]
- Couet, C.; Delarue, J.; Ritz, P.; Antoine, J.M.; Lamisse, F. Effect of dietary fish oil on body fat mass and basal fat oxidation in healthy adults. Int. J. Obes. Relat. Metab. Disord. 1997, 21, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Fontani, G.; Corradeschi, F.; Felici, A.; Alfatti, F.; Bugarini, R.; Fiaschi, A.I.; Cerretani, D.; Montorfano, G.; Rizzo, A.M.; Berra, B. Blood profiles, body fat and mood state in healthy subjects on different diets supplemented with omega-3 polyunsaturated fatty acids. Eur. J. Clin. Investig. 2005, 35, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.M.; Buckley, J.D.; Murphy, K.J.; Howe, P.R. Combining fish-oil supplements with regular aerobic exercise improves body composition and cardiovascular disease risk factors. Am. J. Clin. Nutr. 2007, 85, 1267–1274. [Google Scholar] [PubMed]
- Belury, M.A.; Mahon, A.; Banni, S. The conjugated linoleic acid (CLA) isomer, t10c12-CLA, is inversely associated with changes in body weight and serum leptin in subjects with type 2 diabetes mellitus. J. Nutr. 2003, 133, 257S–260S. [Google Scholar] [PubMed]
- Chan, D.C.; Watts, G.F.; Nguyen, M.N.; Barrett, P.H. Factorial study of the effect of n-3 fatty acid supplementation and atorvastatin on the kinetics of HDL apolipoproteins A-I and A-II in men with abdominal obesity. Am. J. Clin. Nutr. 2006, 84, 37–43. [Google Scholar] [PubMed]
- Simopoulos, A.P. The Impact of the Bellagio Report on Healthy Agriculture, Healthy Nutrition, Healthy People: Scientific and Policy Aspects and the International Network of Centers for Genetics, Nutrition and Fitness for Health. J. Nutrigenet. Nutrigenom. 2015, 7, 189–209. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. ω-3 fatty acids in health and disease and in growth and development. Am. J. Clin. Nutr. 1991, 54, 438–463. [Google Scholar] [PubMed]
- De Gomez Dumm, I.N.T.; Brenner, R.R. Oxidative desaturation of alphalinolenic, linoleic, and stearic acids by human liver microsomes. Lipids 1975, 10, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Emken, E.A.; Adlof, R.O.; Rakoff, H.; Rohwedder, W.K. Metabolism of deuterium-labeled linolenic, linoleic, oleic, stearic and palmitic acid in human subjects. In Synthesis and Application of Isotopically Labeled Compounds 1988; Baillie, T.A., Jones, J.R., Eds.; Elsevier Science Publishers: Amsterdam, The Netherlands, 1989; pp. 713–716. [Google Scholar]
- Hague, T.A.; Christoffersen, B.O. Effect of dietary fats on arachidonic acid and eicosapentaenoic acid biosynthesis and conversion to C22 fatty acids in isolated liver cells. Biochim. Biophys. Acta 1984, 796, 205–217. [Google Scholar]
- Hague, T.A.; Christoffersen, B.O. Evidence for peroxisomal retroconversion of adrenic acid (22, 4n6) and docosahexaenoic acid (22, 6n3) in isolated liver cells. Biochim. Biophys. Acta 1986, 875, 165–173. [Google Scholar]
- Indu, M.; Ghafoorunissa, P. N-3 fatty acids in Indian diets—Comparison of the effects of precursor (alpha-linolenic acid) vs. product (long chain n-3 polyunsaturated fatty acids). Nutr. Res. 1992, 12, 569–582. [Google Scholar]
- Ameur, A.; Enroth, S.; Johansson, A.; Zaboli, G.; Igl, W.; Johansson, A.C.; Rivas, M.A.; Daly, M.J.; Schmitz, G.; Hicks, A.A.; et al. Genetic Adaptation of Fatty-Acid Metabolism: A Human-Specific Haplotype Increasing the Biosynthesis of Long-Chain ω-3 and ω-6 Fatty Acids. Am. J. Hum. Genet. 2012, 90, 809–820. [Google Scholar] [PubMed]
- Sergeant, S.; Hugenschmidt, C.E.; Rudock, M.E.; Ziegler, J.T.; Ivester, P.; Ainsworth, H.C.; Vaidya, D.; Case, L.D.; Langefeld, C.D.; Freedman, B.I.; et al. Differences in arachidonic acid levels and fatty acid desaturase (FADS) gene variants in African Americans and European Americans with diabetes or the metabolic syndrome. Br. J. Nutr. 2012, 107, 547–555. [Google Scholar] [PubMed]
- Mathias, R.A.; Sergeant, S.; Ruczinski, I.; Torgerson, D.G.; Hugenschmidt, C.E.; Kubala, M.; Vaidya, D.; Suktitipat, B.; Ziegler, J.T.; Ivester, P.; et al. The impact of FADS genetic variants on ω6 polyunsaturated fatty acid metabolism in African Americans. BMC Genet. 2011, 12, 50. [Google Scholar]
- Carlson, S.E.; Rhodes, P.G.; Ferguson, M.G. Docosahexaenoic acid status of preterm infants at birth and following feeding with human milk or formula. Am. J. Clin. Nutr. 1986, 44, 798–804. [Google Scholar] [PubMed]
- Singer, P.; Jaeger, W.; Voigt, S.; Theil, H. Defective desaturation and elongation of n-6 and n-3 fatty acids in hypertensive patients. Prostaglandins Leukot. Med. 1984, 15, 159–165. [Google Scholar] [CrossRef]
- Honigmann, G.; Schimke, E.; Beitz, J.; Mest, H.J.; Schliack, V. Influence of a diet rich in linolenic acid on lipids, thrombocyte aggregation and prostaglandins in type I (insulin-dependent) diabetes. Diabetologia 1982, 23, 175. [Google Scholar]
- Simopoulos, A.P. Essential fatty acids in health and chronic disease. Am. J. Clin. Nutr. 1999, 70 (Suppl.), 560S–569S. [Google Scholar] [PubMed]
- Simopoulos, A.P. The importance of the ω-6/ω-3 Balance in Health and Disease: Evolutionary Aspects of Diet. In Healthy Agriculture, Healthy Nutrition, Healthy People; Simopoulos, A.P., Ed.; Karger: Basel, Switzerland, 2011; Volume 102, pp. 10–21. [Google Scholar]
- Eaton, S.B.; Konner, M. Paleolithic nutrition. A consideration of its nature and current implications. N. Engl. J. Med. 1985, 312, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Overview of evolutionary aspects of ω3 fatty acids in the diet. In The Return of ω-3 Fatty Acids into the Food Supply. I. Land-Based Animal Food Products and Their Health Effects; Simopoulos, A.P., Ed.; Karger: Basel, Switzerland, 1998; Volume 83, pp. 1–11. [Google Scholar]
- Crawford, M.A. Fatty acid ratios in free-living and domestic animals. Lancet 1968, 1, 1329–1333. [Google Scholar] [CrossRef]
- Wo, C.K.; Draper, H.H. Vitamin E status of Alaskan Eskimos. Am. J. Clin. Nutr. 1975, 28, 808–813. [Google Scholar]
- Crawford, M.A.; Gale, M.M.; Woodford, M.H. Linoleic acid and linoleic acid elongation products in muscle tissue of Syncerus caffer and other ruminant species. Biochem. J. 1969, 115, 25–27. [Google Scholar] [CrossRef] [PubMed]
- Raper, N.R.; Cronin, F.J.; Exler, J. ω-3 fatty acid content of the US food supply. J. Am. College Nutr. 1992, 11, 304. [Google Scholar] [CrossRef]
- Simopoulos, A.P.; Salem, N., Jr. Purslane: A terrestrial source of ω-3 fatty acids. N. Engl. J. Med. 1986, 315, 833. [Google Scholar] [PubMed]
- Van Vliet, T.; Katan, M.B. Lower ratio of n-3 to n-6 fatty acids in cultured than in wild fish. Am. J. Clin. Nutr. 1990, 51, l–2. [Google Scholar]
- Simopoulos, A.P.; Salem, N., Jr. Egg yolk as a source of long-chain polyunsaturated fatty acids in infant feeding. Am. J. Clin. Nutr. 1992, 55, 411–414. [Google Scholar] [PubMed]
- Simopoulos, A.P.; Norman, H.A.; Gillapsy, J.E.; Duke, J.A. Common purslane: A source of ω-3 fatty acids and antioxidants. J. Am. Coll. Nutr. 1992, 11, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Nutrition and Fitness. JAMA 1989, 261, 2862–2863. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P.; Salem, N., Jr. N-3 Fatty acid in eggs from range-fed Greek chickens. N. Engl. J. Med. 1989, 321, 1412. [Google Scholar] [PubMed]
- Simopoulos, A.P.; Norman, H.A.; Gillapsy, J.E. Purlsane in human nutrition and its potential for world agriculture. In Plants in Human Nutrition; Simopoulos, A.P., Ed.; Karger: Basel, Switzerland, 1995; Volume 77, pp. 47–74. [Google Scholar]
- Simopoulos, A.P. Trans-fatty acids. In Handbook of Lipids in Human Nutrition; Spiller, G.A., Ed.; CRC Press: Boca Raton, FL, USA, 1995; pp. 91–99. [Google Scholar]
- Dupont, J.; White, P.J.; Feldman, E.B. Saturated and hydrogenated fats in food in relation to health. J. Am. Coll. Nutr. 1991, 10, 577–592. [Google Scholar] [CrossRef]
- Litin, L.; Sacks, F. Trans-fatty acid content of common foods. N. Engl. J. Med. 1993, 329, 1969–1970. [Google Scholar] [CrossRef] [PubMed]
- Guil, J.L.; Torija, M.E.; Gimenez, J.J.; Rodríguez, I. Identification of fatty acids in edible wild plants by gas chromatography. J. Chromatogr. A 1996, 719, 229–235. [Google Scholar] [CrossRef]
- Cordain, L.; Martin, C.; Florant, G.; Watkins, B.A. The fatty acid composition of muscle, brain, marrow and adipose tissue in elk: Evolutionary implications for human dietary lipid requirements. World Rev. Nutr. Diet. 1998, 83, 225–226. [Google Scholar]
- Sinclair, A.J.; Slattery, W.J.; O’Dea, K. The analysis of polyunsaturated fatty acids in meat by capillary gas-liquid chromatography. J. Food Sci. Agri. 1982, 33, 771–776. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The role of fatty acids in gene expression: Health implications. Ann. Nutr. Metab. 1996, 40, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Elton, S. Environments, Adaptation, and Evolutionary Medicine: Should We Be Eating a Stone Age Diet? In Medicine and Evolution: Current Applications, Future Prospects; Elton, S., O’Higgins, P., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 9–34. [Google Scholar]
- Eaton, S.B.; Konner, M.; Shostak, M. Stone agers in the fast lane: Chronic degenerative diseases in evolutionary perspective. Am. J. Med. 1988, 84, 739–749. [Google Scholar] [CrossRef]
- Kuipers, R.S.; Luxwolda, M.F.; Dijck-Brouwer, D.A.; Eaton, S.B.; Crawford, M.A.; Cordain, L.; Muskiet, F.A. Estimated macronutrient and fatty acid intakes from an East African Paleolithic diet. Br. J. Nutr. 2010, 104, 1666–1687. [Google Scholar] [CrossRef] [PubMed]
- Guil-Guerrero, J.L.; Tikhonov, A.; Rodríguez-García, I.; Protopopov, A.; Grigoriev, S.; Ramos-Bueno, R.P. The fat from frozen mammals reveals sources of essential fatty acids suitable for Palaeolithic and Neolithic humans. PLoS ONE 2014, 9, e84480. [Google Scholar]
- Eaton, S.B.; Eaton, S.B., III; Sinclair, A.J.; Cordain, L.; Mann, N.J. Dietary intake of long-chain polyunsaturated fatty acids during the Paleolithic. In The Return of w-3 Fatty Acids into the Food Supply. I. Land-Based Animal Food Products and Their Health Effects; Simopoulos, A.P., Ed.; Karger: Basel, Switzerland, 1998; Volume 83, pp. 12–23. [Google Scholar]
- Simopoulos, A.P. Importance of the ratio of ω-6/ω-3 essential fatty acids: Evolutionary aspects. In Omega-6/Omega-3 Essential Fatty Acid Ratio: The Scientific Evidence; Simopoulos, A.P., Cleland, L.G., Eds.; Karger: Basel, Switzerland, 2003; Volume 92, pp. 1–22. [Google Scholar]
- Amri, E.Z.; Ailhaud, G.; Grimaldi, P.A. Fatty acids as signal transducing molecules: Involvement in the differentiation of preadipose to adipose cells. J. Lipid Res. 1994, 35, 930–937. [Google Scholar] [PubMed]
- Jump, D.B.; Clarke, S.D.; Thelen, A.; Liimatta, M. Coordinate regulation of glycolytic and lipogenic gene expression by polyunsaturated fatty acids. J. Lipid Res. 1994, 35, 1076–1084. [Google Scholar] [PubMed]
- Clarke, S.D.; Jump, D. Polyunsaturated fatty acids regulate lipogenic and peroxisomal gene expression by independent mechanisms. Prostaglandins Leukot. Essent. Fatty Acids 1997, 57, 65–69. [Google Scholar] [CrossRef]
- Schwinkendorf, D.R.; Tsatsos, N.G.; Gosnell, B.A.; Mashek, D.G. Effects of central administration of distinct fatty acids on hypothalamic neuropeptide expression and energy metabolism. Int. J. Obes. 2011, 35, 336–344. [Google Scholar] [CrossRef] [PubMed]
- James, M.J.; Gibson, R.A.; Cleland, L.G. Dietary polyunsaturated fatty acids and inflammatory mediator production. Am. J. Clin. Nutr. 2000, 71, 343S–348S. [Google Scholar] [PubMed]
- Gaillard, D.; Negrel, R.; Lagarde, M.; Ailhaud, G. Requirement and role of arachidonic acid in the differentiation of preadipose cells. Biochem. J. 1989, 257, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Corey, E.J.; Shih, C.; Cashman, J.R. Docosahexaenoic acid is a strong inhibitor of prostaglandin but not leukotriene biosynthesis. Proc. Natl. Acad. Sci. USA 1983, 80, 3581–3584. [Google Scholar] [CrossRef] [PubMed]
- Massaro, M.; Habib, A.; Lubrano, L.; Del Turco, S.; Lazzerini, G.; Bourcier, T.; Weksler, B.B.; De Caterina, R. The omega-3 fatty acid docosahexaenoate attenuates endothelial cyclooxygenase-2 induction through both NADP(H) oxidase and PKC epsilon inhibition. Proc. Natl. Acad. Sci. USA 2006, 103, 15184–15189. [Google Scholar] [CrossRef] [PubMed]
- Ringbom, T.; Huss, U.; Stenholm, A.; Flock, S.; Skattebol, L.; Perera, P.; Bohlin, L. Cox-2 inhibitory effects of naturally occurring and modified fatty acids. J. Nat. Prod. 2001, 64, 745–749. [Google Scholar] [CrossRef] [PubMed]
- Mirnikjoo, B.; Brown, S.E.; Kim, H.F.; Marangell, L.B.; Sweatt, J.D.; Weeber, E.J. Protein kinase inhibition by omega-3 fatty acids. J. Biol. Chem. 2001, 276, 10888–10896. [Google Scholar] [CrossRef] [PubMed]
- Hennig, B.; Watkins, B.A. Linoleic acid and linolenic acid: Effect on permeability properties of cultured endothelial cell monolayers. Am. J. Clin. Nutr. 1989, 49, 301–305. [Google Scholar] [PubMed]
- Ukropec, J.; Reseland, J.E.; Gasperikova, D.; Demcakova, E.; Madsen, L.; Berge, R.K.; Rustan, A.C.; Klimes, I.; Drevon, C.A.; Sebokova, E. The hypotriglyceridemic effect of dietary n-3 FA is associated with increased beta-oxidation and reduced leptin expression. Lipids 2003, 38, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Lepperdinger, G. Inflammation and mesenchymal stem cell aging. Curr. Opin. Immunol. 2011, 23, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Massiera, F.; Barbry, P.; Guesnet, P.; Joly, A.; Luquet, S.; Moreilhon-Brest, C.; Mohsen-Kanson, T.; Amri, E.Z.; Ailhaud, G. A Western-like fat diet is sufficient to induce a gradual enhancement in fat mass over generations. J. Lipid Res. 2010, 51, 2352–2561. [Google Scholar] [CrossRef] [PubMed]
- Pisani, D.F.; Amri, E.Z.; Ailhaud, G. Disequilibrium of polyunsaturated fatty acids status and its dual effect in modulating adipose tissue development and functions. OCL 2015, 22, D405. [Google Scholar] [CrossRef]
- Baillie, R.A.; Takada, R.; Nakamura, M.; Clarke, S.D. Coordinate induction of peroxisomal acyl-CoA oxidase and UCP-3 by dietary fish oil: A mechanism for decreased body fat deposition. Prostaglandins Leukot. Essent. Fatty Acids 1999, 60, 351–356. [Google Scholar] [CrossRef]
- Phillips, C.M.; Goumidi, L.; Bertrais, S.; Field, M.R.; Ordovas, J.M.; Cupples, L.A.; Defoort, C.; Lovegrove, J.A.; Drevon, C.A.; Blaak, E.E.; et al. Leptin receptor polymorphisms interact with polyunsaturated fatty acids to augment risk of insulin resistance and metabolic syndrome in adults. J. Nutr. 2010, 140, 238–244. [Google Scholar] [PubMed]
- Cheng, L.; Yu, Y.; Zhang, Q.; Szabo, A.; Wang, H.; Huang, X.F. Arachidonic acid impairs hypothalamic leptin signaling and hepatic energy homeostasis in mice. Mol. Cell. Endocrinol. 2015, 5, 412, 12–18. [Google Scholar] [CrossRef]
- Perez-Matute, P.; Perez-Echarri, N.; Martinez, J.A.; Marti, A.; Moreno-Aliaga, M.J. Eicosapentaenoic acid actions on adiposity and insulin resistance in control and high-fat-fed rats: Role of apoptosis, adiponectin and tumour necrosis factor-alpha. Br. J. Nutr. 2007, 97, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Hassanali, Z.; Ametaj, B.N.; Field, C.J.; Proctor, S.D.; Vine, D.F. Dietary supplementation of n-3 PUFA reduces weight gain and improves postprandial lipaemia and the associated inflammatory response in the obese JCR: LA-cp rat. Diabetes Obes. Metab. 2010, 12, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Alvheim, A.R.; Torstensen, B.E.; Lin, Y.H.; Lillefosse, H.H.; Lock, E.J.; Madsen, L.; Frøyland, L.; Hibbeln, J.R.; Malde, M.K. Dietary linoleic acid elevates the endocannabinoids 2-AG and anandamide and promotes weight gain in mice fed a low fat diet. Lipids 2014, 49, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Banni, S.; Di Marzo, V. Effect of dietary fat on endocannabinoids and related mediators: consequences on energy homeostasis, inflammation and mood. Mol. Nutr. Food Res. 2010, 54, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Matias, I.; Di Marzo, V. Endocannabinoids and the control of energy balance. Trends Endocrinol. Metab. 2007, 18, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Buckley, J.D.; Howe, P.R. Anti-obesity effects of long-chain omega-3 polyunsaturated fatty acids. Obes. Rev. 2009, 10, 648–659. [Google Scholar] [CrossRef] [PubMed]
- Hainault, I.; Carolotti, M.; Hajduch, E.; Guichard, C.; Lavau, M. Fish oil in a high lard diet prevents obesity, hyperlipemia, and adipocyte insulin resistance in rats. Ann. N. Y. Acad. Sci. 1993, 683, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Belzung, F.; Raclot, T.; Groscolas, R. Fish oil n-3 fatty acids selectively limit the hypertrophy of abdominal fat depots in growing rats fed high-fat diets. Am. J. Physiol. 1993, 264, R1111–R1118. [Google Scholar] [PubMed]
- Parrish, C.C.; Pathy, D.A.; Angel, A. Dietary fish oils limit adipose tissue hypertrophy in rats. Metabolism 1990, 39, 217–219. [Google Scholar] [CrossRef]
- Ruzickova, J.; Rossmeisl, M.; Prazak, T.; Flachs, P.; Sponarova, J.; Veck, M.; Tvrzicka, E.; Bryhn, M.; Kopecky, J. Omega-3 PUFA of marine origin limit diet-induced obesity in mice by reducing cellularity of adipose tissue. Lipids 2004, 39, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Nuernberg, K.; Breier, B.H.; Jayasinghe, S.N.; Bergmann, H.; Thompson, N.; Nuernberg, G.; Dannenberger, D.; Schneider, F.; Renne, U.; Langhammer, M.; et al. Metabolic responses to high-fat diets rich in n-3 or n-6 long-chain polyunsaturated fatty acids in mice selected for either high body weight or leanness explain different health outcomes. Nutr. Metab. 2011, 8, 56. [Google Scholar] [PubMed]
- Kang, J.X.; Wang, J.; Wu, L.; Kang, Z.B. Transgenic mice: Fat-1 mice convert n-6 to n-3 fatty acids. Nature 2004, 427, 504. [Google Scholar] [CrossRef] [PubMed]
- Jump, D.B.; Clarke, S.D. Regulation of gene expression by dietary fat. Annu. Rev. Nutr. 1999, 19, 63–90. [Google Scholar] [CrossRef] [PubMed]
- Jump, D.B. Dietary polyunsaturated fatty acids and regulation of gene transcription. Curr. Opin. Lipidol. 2002, 13, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Suzukawa, M.; Abbey, M.; Howe, P.R.; Nestel, P.J. Effects of fish oil fatty acids on low-density lipoprotein size, oxidizability, and uptake by macrophages. J. Lipid Res. 1995, 36, 473–484. [Google Scholar] [PubMed]
- Li, J.; Li, F.R.; Wei, D.; Jia, W.; Kang, J.X.; Stefanovic-Racic, M.; Dai, Y.; Zhao, A.Z. Endogenous ω-3 Polyunsaturated Fatty Acid Production Confers Resistance to Obesity, Dyslipidemia, and Diabetes in Mice. Mol. Endocrinol. 2014, 28, 1316–1328. [Google Scholar] [CrossRef] [PubMed]
- Kabir, M.; Skurnik, G.; Naour, N.; Pechtner, V.; Meugnier, E.; Rome, S.; Quignard-Boulangé, A.; Vidal, H.; Slama, G.; Clément, K.; et al. Treatment for 2 mo with n 3 polyunsaturated fatty acids reduces adiposity and some atherogenic factors but does not improve insulin sensitivity in women with type 2 diabetes: A randomized controlled study. Am. J. Clin. Nutr. 2007, 86, 1670–1679. [Google Scholar]
- Thorsdottir, I.; Tomasson, H.; Gunnarsdottir, I.; Gisladottir, E.; Kiely, M.; Parra, M.D.; Bandarra, N.M.; Schaafsma, G.; Martinéz, J.A. Randomized trial of weight-loss-diets for young adults varying in fish and fish oil content. Int. J. Obes. 2007, 31, 1560–1566. [Google Scholar] [CrossRef] [PubMed]
- Parra, D.; Ramel, A.; Bandarra, N.; Kiely, M.; Martinez, J.A.; Thorsdottir, I. A diet rich in long chain omega-3 fatty acids modulates satiety in overweight and obese volunteers during weight loss. Appetite 2008, 51, 676–680. [Google Scholar] [CrossRef] [PubMed]
- Krebs, J.D.; Browning, L.M.; McLean, N.K.; Rothwell, J.L.; Mishra, G.D.; Moore, C.S.; Jebb, S.A. Additive benefits of long-chain n-3 polyunsaturated fatty acids and weight-loss in the management of cardiovascular disease risk in overweight hyperinsulinaemic women. Int. J. Obes. 2006, 30, 1535–1544. [Google Scholar] [CrossRef] [PubMed]
- Kunesova, M.; Braunerová, R.; Hlavatý, P.; Tvrzická, E.; Stanková, B.; Skrha, J.; Hilgertová, J.; Hill, M.; Kopecký, J.; Wagenknecht, M.; et al. The influence of n-3 polyunsaturated fatty acids and very low calorie diet during a short-term weight reducing regimen on weight loss and serum fatty acid composition in severely obese women. Physiol. Res. 2006, 55, 63–72. [Google Scholar] [PubMed]
- Simopoulos, A.P. Genetic variants in the metabolism of omega-6 and omega-3 fatty acids: their role in the determination of nutritional requirements and chronic disease risk. Exp. Biol. Med. 2010, 235, 785–795. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Manson, J.E.; Rautiainen, S.; Gaziano, J.M.; Buring, J.E.; Tsai, M.Y.; Sesso, H.D. A prospective study of erythrocyte polyunsaturated fatty acid, weight gain, and risk of becoming overweight or obese in middle‑aged and older women. Eur. J. Nutr. 2015. [Google Scholar] [CrossRef] [PubMed]
- Claussnitzer, M.; Dankel, S.N.; Kim, K.H.; Quon, G.; Meuleman, W.; Haugen, C.; Glunk, V.; Sousa, I.S.; Beaudry, J.L.; Puviindran, V.; et al. FTO Obesity Variant Circuitry and Adipocyte Browning in Humans. N. Engl. J. Med. 2015, 373, 895–907. [Google Scholar] [PubMed]
- Savva, S.C.; Chadjigeorgiou, C.; Hatzis, C.; Kyriakakis, M.; Tsimbinos, G.; Tornaritis, M.; Kafatos, A. Association of adipose tissue arachidonic acid content with BMI and overweight status in children from Cyprus and Crete. Br. J. Nutr. 2004, 91, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Jensen, C.L.; Prager, T.C.; Fraley, J.K.; Chen, H.; Anderson, R.E.; Heird, W.C. Effect of dietary linoleic/alpha-linolenic acid ratio on growth and visual function of term infants. J. Pediatr. 1997, 131, 200–209. [Google Scholar] [CrossRef]
- Ahima, R.S.; Antwi, D.A. Brain regulation of appetite and satiety. Endocrinol. Metab. Clin. N. Am. 2008, 37, 811–823. [Google Scholar] [CrossRef] [PubMed]
- Artmann, A.; Petersen, G.; Hellgren, L.; Boberg, J.; Skonberg, C.; Nellemann, C.; Hansen, S.H.; Hansen, H.S. Influence of dietary fatty acids on endocannabinoid and N-acylethanolamine levels in rat brain, liver and small intestine. Biochim. Biophys. Acta 2008, 1781, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, R.; Ohkuri, T.; Jyotaki, M.; Yasuo, T.; Horio, N.; Yasumatsu, K.; Sanematsu, K.; Shigemura, N.; Yamamoto, T.; Margolskee, R.F.; et al. Endocannabinoids selectively enhance sweet taste. Proc. Natl. Acad. Sci. USA 2010, 107, 935–939. [Google Scholar] [CrossRef] [PubMed]
- Van Gaal, L.; Pi-Sunyer, X.; Despres, J.P.; McCarthy, C.; Scheen, A. Efficacy and safety of rimonabant for improvement of multiple cardiometabolic risk factors in overweight/obese patients: Pooled 1-year data from the Rimonabant in Obesity (RIO) program. Diabetes Care 2008, 31, S229–S240. [Google Scholar] [CrossRef] [PubMed]
- Christensen, R.; Kristensen, P.K.; Bartels, E.M.; Bliddal, H.; Astrup, A. Efficacy and safety of the weight-loss drug rimonabant: a meta-analysis of randomised trials. Lancet 2007, 370, 1706–1713. [Google Scholar] [CrossRef]
|
|
|
|
|
|
|
| - Determination of the composition of the background diet in terms of omega-6 and omega-3 fatty acids and inflammatory markers i.e., US, UK and Northern European countries have the highest amount of LA + AA in their diets, which competes with omega-3 PUFAs; they also have the lowest amount of vegetable and fruit intake, which are needed for optimal absorption of omega-3 PUFA from supplements |
| - Background inflammation |
| - Some studies are using fish and others omega-3 supplements; studies show that a continuous daily intake of omega-3 supplements leads to higher concentrations in the blood than eating fish two times/week |
| - Variation in the dose of omega-3 fatty acids |
| - Variation in the number of subjects |
| - Variation in the severity of disease |
| - Variation in the pharmacologic treatment |
| - Genetic variants predisposing to Obesity |
| - Dietary intake by means of questionnaires instead of actual measurements of omega-3 PUFAs in the red blood cell membrane phospholipids or plasma is a major problem that leads to conflicting results |
| - Length of intervention |
| - Genetic variants in the metabolism of omega-6 and omega-3 fatty acids |
| Plants | |
| LA | 4.28 |
| ALA | 11.40 |
| Animals | |
| LA | 4.56 |
| ALA | 1.21 |
| Total | |
| LA | 8.84 |
| ALA | 12.60 |
| Animal | |
| AA (ω6) | 1.81 |
| EPA (ω3) | 0.39 |
| DTA (ω6) | 0.12 |
| DPA (ω3) | 0.42 |
| DHA (ω3) | 0.27 |
| Ratios of ω6/ω3 | |
| LA/ALA | 0.70 |
| AA + DTA/EPA + DPA + DHA | 1.79 |
| Total ω6/ω3 | 0.79 b |
| Population | ω-6/ω-3 |
|---|---|
| Paleolithic | 0.79 |
| Greece prior to 1960 | 1.00–2.00 |
| Current Japan | 4.00 |
| Current India, rural | 5–6.1 |
| Current UK and northern Europe | 15.00 |
| Current US | 16.74 |
| Current India, urban | 38–50 |
| Conditions | Omega-6 | Omega-3 |
|---|---|---|
| Adipogenesis (Pre-adipocyte-Adipocyte) | High AA via the PI2 receptor activates the cAMP protein kinase A, signaling pathway leads to proliferation and differentiation of WAT, prevention of its browning through inhibition of PPARy target genes including UCPI, decrease mitochondrial biogenesis [74], increasing triglycerides [73], insulin resistance, leptin resistance, decreased adiponectin levels, decreased fatty acid oxidation and hepatic steatosis [4]. | High EPA and DHA partially inhibit cAMP signaling pathways triggered by AA at levels upstream of PKA [71] block COX-2 metabolites PGI2 and PGEF2a that stimulate white adipogenesis and inhibit the browning process respectively, prevent increased triglycerides and adipose tissue proliferation through UCP-I and PPARy activation, increased mitochondrial biogenesis, increased fatty acid oxidation, and apoptosis [71,75]. |
| Inflammation | AA metabolites prostaglandin 2 thromboxane 2 and leukotriene 4 are prothrombotic and proinflammatory leading to increased production of IL-1, IL-6, NFKB and TNF and inflammation [1,64]. | High dietary intake of EPA and DHA blocks the metabolites of AA and prevents inflammation, which is the hallmark of obesity [1,64,67]. |
| Insulin Resistance Leptin Resistance Adiponectin | AA leads to insulin resistance, leptin resistance, lower adiponectin and hepatic steatosis. AA blunts PI3-Akt pathway leading to leptin resistance in the brain and deregulation of food intake [19,76,77]. | EPA and DHA regulate glucose utilization, insulin sensitivity (Akt phosphorylation) in part mediated by PPARy and AMPK activation [19]. EPA and DHA regulate the secretion of adipokines involved in energy homeostasis and intermediate metabolism and in glucose and lipid metabolism. DHA restores insulin sensitivity in skeletal muscle by preventing lipotoxicity and inflammation [78,79]. |
| Cannabinoids | AA increases the concentration of (2-AG) and (AEA) leading to excessive endocannabinoid signaling, and dysregulation of the cannabinoid system, weight gain, larger adipocytes and more macrophages in adipose tissue [80], inflammation and a metabolic profile associated with obesity [81,82]. | EPA and DHA decrease 2-AG and AA in the brain while increasing DHA, decreasing the dysregulation of the cannabinoid system, improving insulin sensitivity and decreasing central body fat. |
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simopoulos, A.P. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 2016, 8, 128. https://doi.org/10.3390/nu8030128
Simopoulos AP. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients. 2016; 8(3):128. https://doi.org/10.3390/nu8030128
Chicago/Turabian StyleSimopoulos, Artemis P. 2016. "An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity" Nutrients 8, no. 3: 128. https://doi.org/10.3390/nu8030128
APA StyleSimopoulos, A. P. (2016). An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients, 8(3), 128. https://doi.org/10.3390/nu8030128




