Phlorizin Supplementation Attenuates Obesity, Inflammation, and Hyperglycemia in Diet-Induced Obese Mice Fed a High-Fat Diet
Abstract
:1. Introduction
2. Experimental Section
2.1. Animals and Diets
2.2. Plasma Biomarkers and Hepatic Lipids
2.3. Hepatic Enzymes Activities
2.4. Histopathological Analysis
2.5. Statistical Analysis
3. Results
3.1. Body and Fat Weights, Adipocyte Size and Food Intake

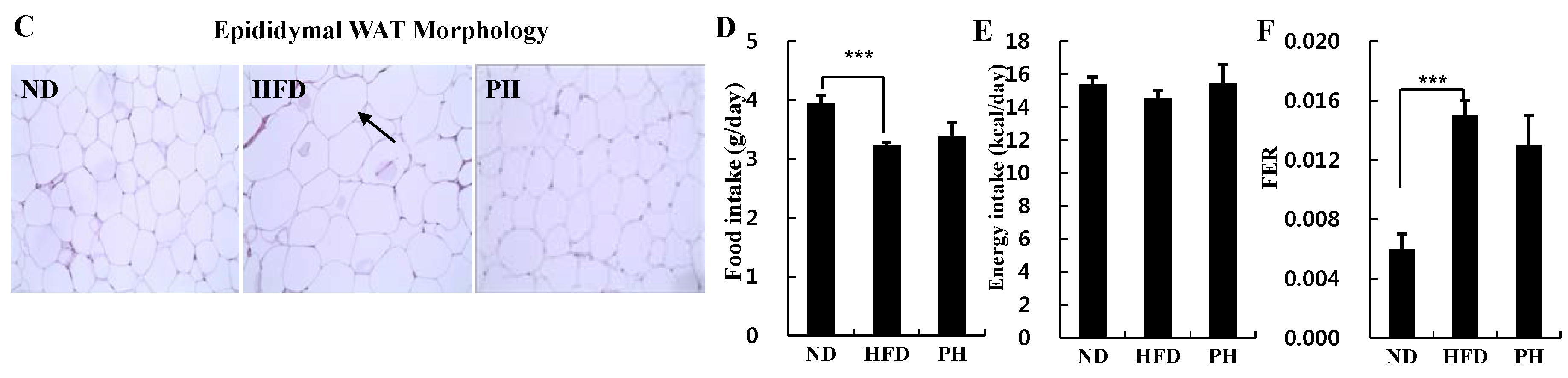
3.2. Plasma Lipids Levels
| ND | HFD | PH | |
|---|---|---|---|
| Total cholesterol (mmol/L) | 2.93 ± 0.12 | 5.96 ± 0.38 *** | 5.51 ± 0.29 |
| Triglycerides (mmol/L) | 0.89 ± 0.03 | 1.48 ± 0.07 *** | 1.42 ± 0.06 |
| Free fatty acid (mmol/L) | 1.07 ± 0.07 | 1.29 ± 0.02 * | 1.16 ± 0.02 †† |
| HDL-cholesterol (mmol/L) | 1.05 ± 0.07 | 1.99 ± 0.10 *** | 2.45 ± 0.12 †† |
| Non HDL-cholesterol (mmol/L) | 1.92 ± 0.10 | 4.01 ± 0.38 *** | 3.07 ± 0.21 † |
| Apolipoprotein-AI (mg/dL) | 38.46 ± 0.34 | 38.59 ± 0.36 | 48.07 ± 0.51 ††† |
| Apolipoprotein B (mg/dL) | 4.47 ± 0.40 | 9.65 ± 0.53 *** | 9.28 ± 0.51 |
| Apo A-I/Apo B | 8.97 ± 0.46 | 4.04 ± 0.18 *** | 5.24 ± 0.37 † |
| HTR (%) | 35.69 ± 2.16 | 32.64 ± 1.24 | 43.49 ± 1.60 ††† |
| AI | 1.75 ± 0.13 | 2.34 ± 0.21 * | 1.21 ± 0.06 †† |
3.3. Hepatic Lipids Levels and Enzyme Activity
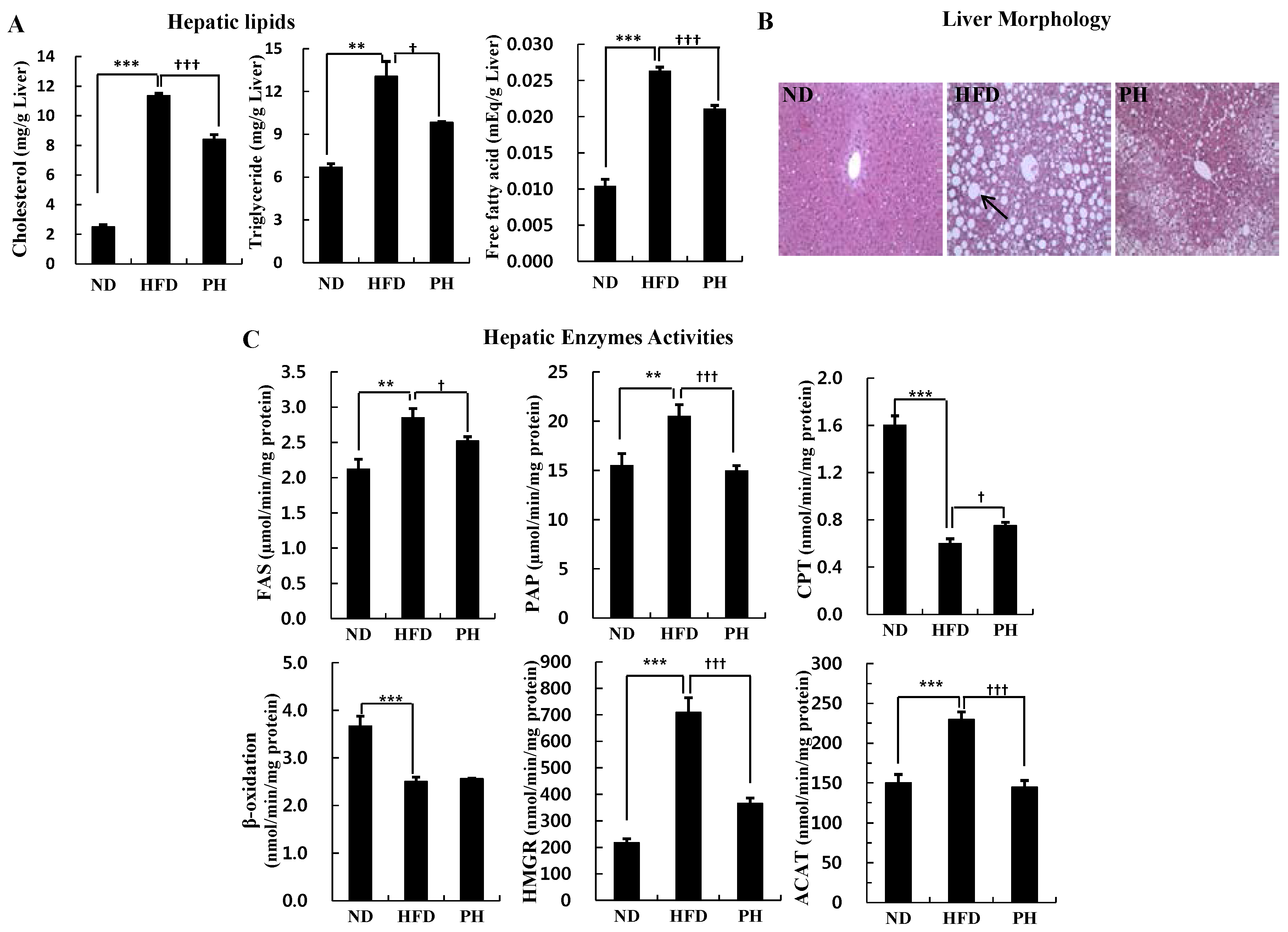
3.4. Plasma Glycemia Markers
| ND | HFD | PH | |
|---|---|---|---|
| Glucose (mmol/L) | 9.71 ± 0.61 | 15.88 ± 0.41 *** | 12.92 ± 0.19 ††† |
| Insulin (ng/mL) | 0.41 ± 0.10 | 1.80 ± 0.21 *** | 1.07 ± 0.14 † |
| Glucagon (pg/mL) | 57.04 ± 2.85 | 95.38 ± 4.30 *** | 68.32 ± 5.29 †† |
| Insulin/glucagon | 7.28 ± 1.39 | 18.83 ± 2.75 ** | 15.39 ± 3.18 |
| HOMA-IR | 1.72 ± 0.34 | 15.07 ± 1.62 *** | 5.24 ± 0.57 ††† |
3.5. Plasma Adipokine Levels and Tissue Fibrosis
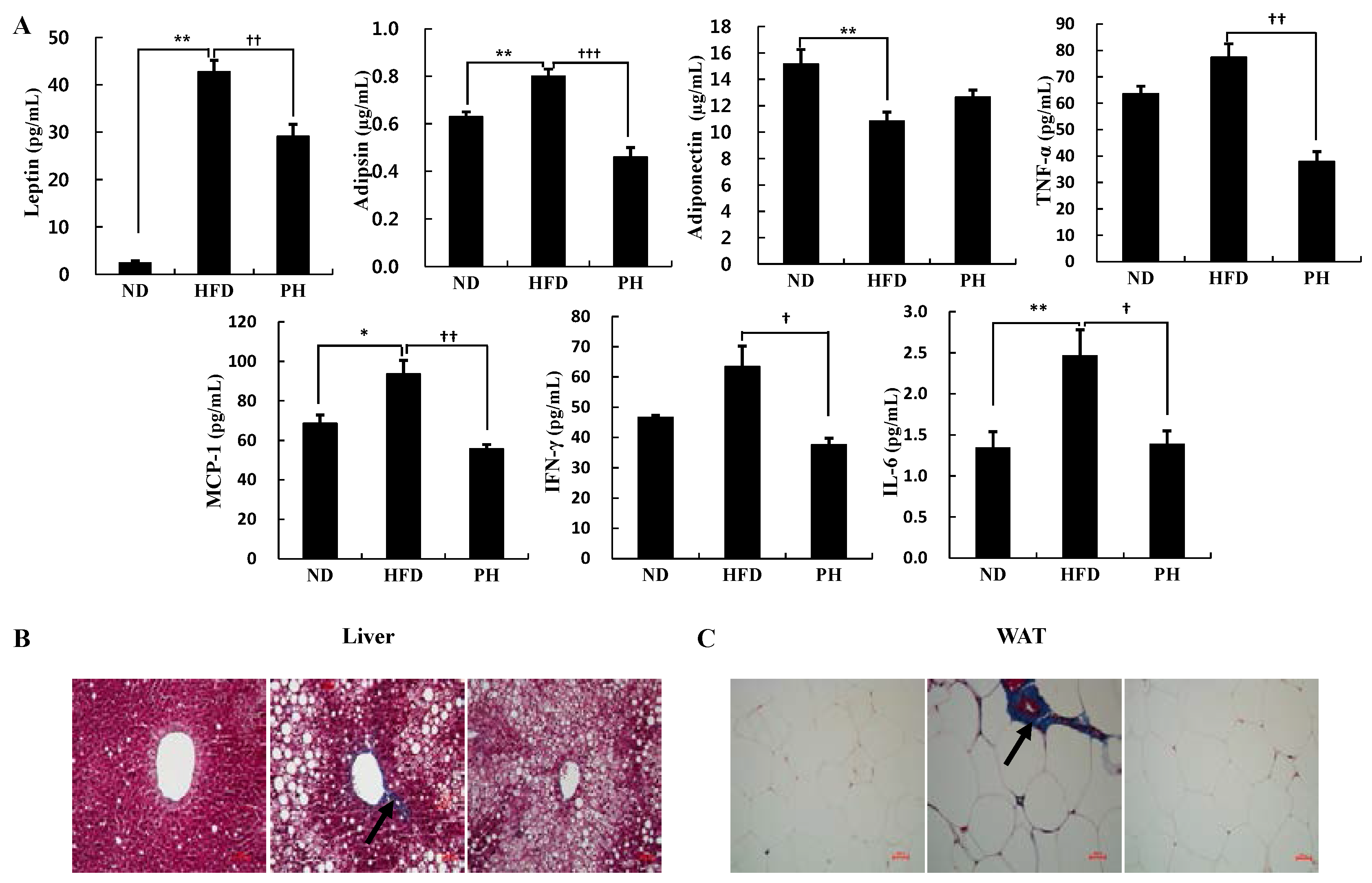
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Petersen, C. Analyse des Phloridzins. Annal. Pharm. 1835, 15. [Google Scholar] [CrossRef]
- Gosch, C.; Halbwirth, H.; Stich, K. Phloridzin: Biosynthesis, distribution and physiological relevance in plants. Phytochemistry 2010, 71, 838–843. [Google Scholar] [CrossRef] [PubMed]
- Ehrenkranz, J.R.; Lewis, N.G.; Kahn, C.R.; Roth, J. Phlorizin: A review. Diabetes Metab. Res. Rev. 2005, 21, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Rossetti, L.; Smith, D.; Shulman, G.I.; Papachristou, D.; DeFronzo, R.A. Correction of hyperglycemia with phlorizin normalizes tissue sensitivity to insulin in diabetic rats. J. Clin. Investig. 1987, 79, 1510–1515. [Google Scholar] [CrossRef] [PubMed]
- Rossetti, L.; Shulman, G.I.; Zawalich, W.; DeFronzo, R.A. Effect of chronic hyperglycemia on in vivo insulin secretion in partially pancreatectomized rats. J. Clin. Investig. 1987, 80, 1037–1044. [Google Scholar] [CrossRef] [PubMed]
- Takii, H.; Matsumoto, K.; Kometani, T.; Okada, S.; Fushiki, T. Lowering effect of phenolic glycosides on the rise in postprandial glucose in mice. Biosci. Biotechnol. Biochem. 1997, 61, 1531–1535. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Prentice, A.M. The emerging epidemic of obesity in developing countries. Int. J. Epidemiol. 2006, 35, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Collins, S.; Martin, T.L.; Surwit, R.S.; Robidoux, J. Genetic vulnerability to diet-induced obesity in the C57BL/6J mouse: Physiological and molecular characteristics. Physiol. Behav. 2004, 81, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Buettner, R.; Schölmerich, J.; Bollheimer, L.C. High-fat diets: Modeling the metabolic disorders of human obesity in rodents. Obesity (Silver Spring) 2007, 15, 798–808. [Google Scholar] [CrossRef] [PubMed]
- Prpic, V.; Watson, P.M.; Frampton, I.C.; Sabol, M.A.; Jezek, G.E.; Gettys, T.W. Differential mechanisms and development of leptin resistance in A/J vs. C57BL/6J mice during diet-induced obesity. Endocrinology 2003, 144, 1155–1163. [Google Scholar] [CrossRef] [PubMed]
- Surwit, R.S.; Kuhn, C.M.; Cochrane, C.; McCubbin, J.A.; Feinglos, M.N. Diet-induced type II diabetes in C57BL/6J mice. Diabetes 1988, 37, 1163–1167. [Google Scholar] [CrossRef] [PubMed]
- Tsunoda, N.; Ikemoto, S.; Takahashi, M.; Maruyama, K.; Watanabe, H.; Goto, N.; Ezaki, O. High-monounsaturated fat diet-induced obesity and diabetes in C57BL/6J mice. Metabolism 1998, 47, 724–730. [Google Scholar] [CrossRef]
- Fantuzzi, G. Adipose tissue, adipokines, and inflammation. J. Allergy Clin. Immunol. 2005, 115, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Hulcher, F.H.; Oleson, W.H. Simplified spectrophotometric assay for microsomal 3-hydroxy-3-methylglutaryl CoA reductase by measurement of coenzyme A. J. Lipid Res. 1973, 14, 625–631. [Google Scholar] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Nepokroeff, C.M.; Lakshmanan, M.R.; Porter, J.W. Fatty-acid synthase from rat liver. Methods Enzymol. 1975, 35, 37–44. [Google Scholar] [PubMed]
- Walton, P.A.; Possmayer, F. Mg2-dependent phosphatidate phosphohydrolase of rat lung: Development of an assay employing a defined chemical substrate which reflects the phosphohydrolase activity measured using membrane-bound substrate. Anal. Biochem. 1985, 151, 479–486. [Google Scholar] [PubMed]
- Markwell, M.A.; McGroarty, E.J.; Bieber, L.L.; Tolbert, N.E. The subcellular distribution of carnitine acyltransferases in mammalian liver and kidney. A new peroxisomal enzyme. J. Biol. Chem. 1973, 248, 3426–3432. [Google Scholar] [PubMed]
- Lazarow, P.B. Assay of peroxisomal beta-oxidation of fatty acids. Methods Enzymol. 1981, 72, 315–319. [Google Scholar] [PubMed]
- Shapiro, D.J.; Nordstrom, J.L.; Mitschelen, J.J.; Rodwell, V.W.; Schimke, R.T. Micro assay for 3-hdyroxy-3-methylglutaryl-CoA reductase in rat liver and in L-cell fibroblasts. Biochim. Biophys. Acta 1974, 370, 369–377. [Google Scholar] [CrossRef]
- Gillies, P.J.; Rathgeb, K.A.; Perri, M.A.; Robinson, C.S. Regulation of acyl-CoA: Cholesterol acyltransferase activity in normal and atherosclerotic rabbit aortas: Role of a cholesterol substrate pool. Exp. Mol. Pathol. 1986, 44, 329–339. [Google Scholar] [CrossRef]
- Rebuffé-Scrive, M.; Surwit, R.; Feinglos, M.; Kuhn, C.; Rodin, J. Regional fat distribution and metabolism in a new mouse model (C57BL/6J) of non-insulin-dependent diabetes mellitus. Metabolism 1993, 42, 1405–1409. [Google Scholar] [CrossRef]
- Surwit, R.S.; Feinglos, M.N.; Rodin, J.; Sutherland, A.; Petro, A.E.; Opara, E.C.; Kuhn, C.M.; Rebuffé-Scrive, M. Differential effects of fat and sucrose on the development of obesity and diabetes in C57BL/6J and A/J mice. Metabolism 1995, 44, 645–651. [Google Scholar] [CrossRef]
- Smith, S.R.; Lovejoy, J.C.; Greenway, F.; Ryan, D.; de Jonge, L.; de la Bretonne, J.; Volafova, J.; Bray, G.A. Contributions of total body fat, abdominal subcutaneous adipose tissue compartments, and visceral adipose tissue to the metabolic complications of obesity. Metabolism 2001, 50, 425–435. [Google Scholar] [PubMed]
- Assmann, G.; Gotto, A.M., Jr. HDL cholesterol and protective factors in atherosclerosis. Circulation 2004, 15, III8–III14. [Google Scholar]
- Berg, A.H.; Scherer, P.E. Adipose tissue, inflammation, and cardiovascular disease. Circ. Res. 2005, 13, 939–949. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, S.P.; McCann, D.; Desai, M.; Rosenbaum, M.; Leibel, R.L.; Ferrante, A.W. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Investig. 2003, 112, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Fantuzzi, G.; Faggioni, R. Leptin in the regulation of immunity, inflammation, and hematopoiesis. J. Leukoc. Biol. 2000, 68, 437–446. [Google Scholar]
- Fernández-Sánchez, A.; Madrigal-Santillán, E.; Bautista, M.; Esquivel-Soto, J.; Morales-González, A.; Esquivel-Chirino, C.; Durante-Montiel, I.; Sánchez-Rivera, G.; Valadez-Vega, C.; Morales-González, J.A. Inflammation, oxidative stress, and obesity. Int. J. Mol. Sci. 2011, 12, 3117–3132. [Google Scholar] [CrossRef] [PubMed]
- Baas, T. Adipsin meets β cells. SciBX 2014, 7. [Google Scholar] [CrossRef]
- Bertolani, C.; Marra, F. The role of adipokines in liver fibrosis. Pathophysiology 2008, 15, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Friedman, S.L. Hepatic fibrosis-overview. Toxicology 2008, 254, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Santra, A.; Lahiri, S.; Guha Mazumder, D.N. Implications of oxidative stress and hepatic cytokine (TNF-alpha and IL-6) response in the pathogenesis of hepatic collagenesis in chronic arsenic toxicity. Toxicol. Appl. Pharmacol. 2005, 204, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Charlton, M.; Krishnan, A.; Viker, K.; Sanderson, S.; Cazanave, S.; McConico, A.; Masuoko, H.; Gores, G. Fast food diet mouse: Novel small animal model of NASH with ballooning, progressive fibrosis, and high physiological fidelity to the human condition. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 301, G825–G834. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shin, S.-K.; Cho, S.-J.; Jung, U.J.; Ryu, R.; Choi, M.-S. Phlorizin Supplementation Attenuates Obesity, Inflammation, and Hyperglycemia in Diet-Induced Obese Mice Fed a High-Fat Diet. Nutrients 2016, 8, 92. https://doi.org/10.3390/nu8020092
Shin S-K, Cho S-J, Jung UJ, Ryu R, Choi M-S. Phlorizin Supplementation Attenuates Obesity, Inflammation, and Hyperglycemia in Diet-Induced Obese Mice Fed a High-Fat Diet. Nutrients. 2016; 8(2):92. https://doi.org/10.3390/nu8020092
Chicago/Turabian StyleShin, Su-Kyung, Su-Jung Cho, Un Ju Jung, Ri Ryu, and Myung-Sook Choi. 2016. "Phlorizin Supplementation Attenuates Obesity, Inflammation, and Hyperglycemia in Diet-Induced Obese Mice Fed a High-Fat Diet" Nutrients 8, no. 2: 92. https://doi.org/10.3390/nu8020092
APA StyleShin, S.-K., Cho, S.-J., Jung, U. J., Ryu, R., & Choi, M.-S. (2016). Phlorizin Supplementation Attenuates Obesity, Inflammation, and Hyperglycemia in Diet-Induced Obese Mice Fed a High-Fat Diet. Nutrients, 8(2), 92. https://doi.org/10.3390/nu8020092





