Cardiometabolic Health in Submariners Returning from a 3-Month Patrol
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects
2.2. Anthropometrics
2.3. Dietary Intake
2.4. Physical Activity
2.5. Blood Collection and Biochemical Assessment
2.6. Metabolic Syndrome Assessment
2.7. Statistical Analysis
3. Results
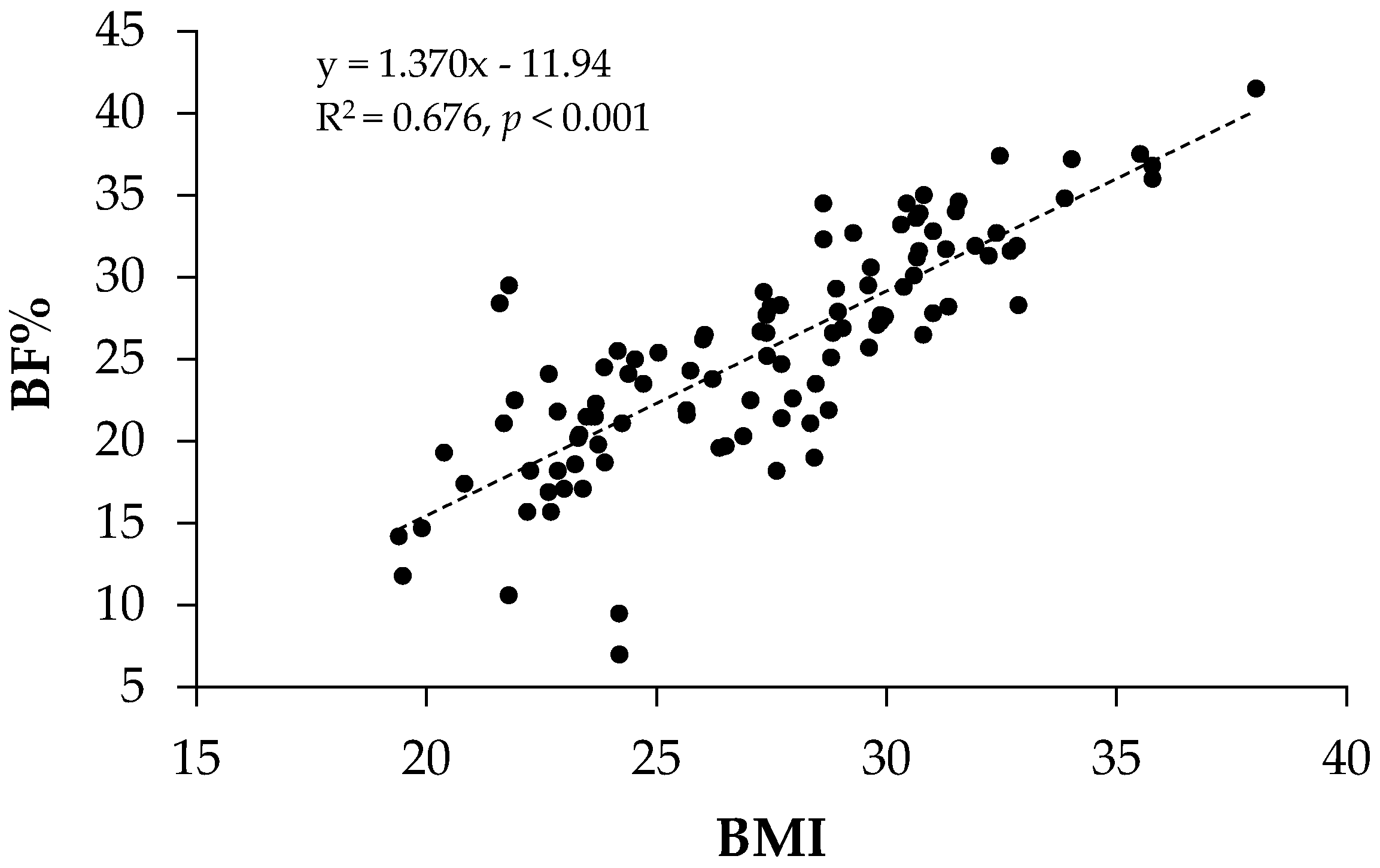
3.1. Anthropometrics
| Time | BMI Classification | ||
|---|---|---|---|
| Normal (18.5–24.9 kg/m2) | Overweight (25–29.9 kg/m2) | Obese (≥30 kg/m2) | |
| Pre- | 20.5 ± 4.6 (32) | 26.5 ± 3.8 (32) | 33.3 ± 3.4 (36) |
| Post- | 18.2 ± 5.3 (34) | 24.8 ± 3.7 (47) | 32.4 ± 3.5 (19) |
3.2. Clinical and Health Related Characteristics
3.3. Biomarkers of Glucose Metabolism, Lipid Profiles, Appetite and Energy Homeostasis and Inflammation
3.4. Metabolic Syndrome
| Variable | BF < 25% (n = 20) | BF ≥ 25% (n = 33) | p Value | ||||
|---|---|---|---|---|---|---|---|
| Pre- | Post- | Pre- | Post- | Group | Time | Int. | |
| Anthropometrics | |||||||
| Height, cm | 178 ± 8 | - | 178 ± 6 | - | |||
| Weight, kg | 75 ± 8 a | 75 ± 9 b | 96 ± 12 a,c | 91 ± 11 b,c | <0.001 | <0.001 | <0.001 |
| BMI, kg/m2 | 24 ± 2 a | 24 ± 3 b | 30 ± 3 a,c | 29 ± 3 b,c | <0.001 | <0.001 | <0.001 |
| Waist circumference, cm | 86 ± 6 a | 86 ± 6 b | 102 ± 6 a,c | 97 ± 8 b,c | <0.001 | <0.001 | <0.001 |
| Fat mass, kg | 17 ± 5 a | 16 ± 6 b | 30 ± 5 a,c | 27 ± 6 b,c | <0.001 | <0.001 | <0.001 |
| BF% | 20 ± 4 a | 19 ± 5 b | 31 ± 4 a,c | 27 ± 5 b,c | <0.001 | <0.001 | <0.001 |
| Fat-free mass, kg | 60 ± 7 | 61 ± 7 | 65 ± 7 | 65 ± 7 | 0.021 | 0.067 | 0.183 |
| Blood pressure and pulse | |||||||
| Systolic, mmHg | 126 ± 8 | 129 ± 15 | 133 ± 12 | 133 ± 12 | 0.082 | 0.348 | 0.500 |
| Diastolic, mmHg | 80 ± 5 | 78 ± 9 | 82 ± 8 | 80 ± 9 | 0.192 | 0.059 | 0.856 |
| Heart rate, beats/min | 66 ± 8 | 65 ± 11 | 72 ± 11 | 69 ± 21 | 0.094 | 0.603 | 0.695 |
| Daily dietary intake | |||||||
| Energy, kJ | 9556 ± 2824 a | 9881 ± 3813 | 11673 ± 3935 a,c | 9624 ± 3766 c | 0.309 | 0.074 | 0.017 |
| Carbohydrate, g | 254 ± 77 | 260 ± 111 | 310 ± 121 c | 235 ± 111 c | 0.515 | 0.029 | 0.012 |
| Protein, g | 98 ± 34 | 105 ± 45 | 117 ± 42 | 109 ± 44 | 0.268 | 0.895 | 0.210 |
| Fat, g | 91 ± 28 | 104 ± 38 | 107 ± 39 | 106 ± 45 | 0.316 | 0.362 | 0.210 |
| Saturated fat, g | 29 ± 9 | 35 ± 13 | 37 ± 14 | 36 ± 17 | 0.215 | 0.243 | 0.149 |
| Cholesterol, mg | 380 ± 159 | 408 ± 149 | 441 ± 206 | 450 ± 261 | 0.303 | 0.624 | 0.769 |
| Sodium, mg | 3785 ± 1239 | 4049 ± 1498 | 4269 ± 1473 | 4056 ± 1385 | 0.455 | 0.995 | 0.247 |
| Fruits & vegetables, servings | 3 ± 1 | 2 ± 2 | 4 ± 3 | 2 ± 2 | 0.463 | 0.011 | 0.323 |
| Fiber, g | 20 ± 7 | 19 ± 7 | 23 ± 9 | 19 ± 6 | 0.439 | 0.045 | 0.086 |
| Alcohol, servings | 1 ± 1 | 0 | 2 ± 3 | 0 | 0.386 | 0.001 | 0.386 |
| Activity and sleep | |||||||
| Daily PAEE, kJ | 3989 ± 2262 | 3900 ± 1989 | 5860 ± 2252 | 4794 ± 2436 | 0.028 | 0.216 | 0.295 |
| Daily sleep, h/day | 7 ± 1 | 6 ± 2 | 6 ± 2 | 5 ± 2 | 0.012 | 0.001 | 0.343 |
| Variable | BF < 25% (n = 20) | BF ≥ 25% (n = 33) | p Value | ||||
|---|---|---|---|---|---|---|---|
| Pre- | Post- | Pre- | Post- | Group | Time | Int. | |
| Glucose, mg/dL | 84 ± 6 | 88 ± 5 | 89 ± 8 | 89 ± 7 | 0.064 | 0.157 | 0.081 |
| Insulin, mU/L | 4.6 ± 1.9 | 5.2 ± 2.4 | 9.4 ± 5.0 | 9.8 ± 5.9 | <0.001 | 0.430 | 0.747 |
| HOMA-IR | 1.0 ± 0.4 | 1.1 ± 0.5 | 2.1 ± 1.2 | 2.2 ± 1.4 | <0.001 | 0.349 | 0.484 |
| Adiponectin, µg/mL | 6.3 ± 2.6 | 5.7 ± 2.6 | 5.0 ± 3.0 | 4.4 ± 2.4 | 0.066 | 0.075 | 0.085 |
| Total cholesterol, mg/dL | 180 ± 41 | 170 ± 37 | 184 ± 27 | 174 ± 29 | 0.644 | <0.001 | 0.951 |
| Triglycerides | 89 ± 43 | 75 ± 21 | 162 ± 120 | 131 ± 76 | 0.001 | 0.098 | 0.612 |
| LDL-C, mg/dL | 114 ± 34 | 106 ± 33 | 112 ± 24 | 103 ± 29 | 0.804 | 0.003 | 0.706 |
| HDL-C, mg/dL | 49 ± 10 | 48 ± 9 | 44 ± 9 | 44 ± 8 | 0.061 | 0.745 | 0.785 |
| Total cholesterol/HDL-C | 3.8 ± 0.9 | 3.6 ± 0.8 | 4.4 ± 1.3 | 4.1 ± 1.0 | 0.048 | 0.001 | 0.396 |
| Ghrelin, pg/mL | 985 ± 335 | 1042 ± 273 | 848 ± 241 | 915 ± 277 | 0.065 | 0.053 | 0.795 |
| Leptin, ng/mL | 3.9 ± 2.1 a | 4.2 ± 2.9 b | 11.7 ± 5.9 a,c | 8.7 ± 5.1 b,c | <0.001 | 0.100 | 0.017 |
| Leptin/Adiponectin | 0.8 ± 0.6 | 0.9 ± 0.7 | 3.4 ± 2.7 | 2.6 ± 2.2 | <0.001 | 0.461 | 0.246 |
| GRO, pg/mL | 427 ± 154 | 429 ± 153 | 541 ± 221 | 539 ± 208 | 0.038 | 0.964 | 0.959 |
| IP-10, pg/mL | 311 ± 178 | 292 ± 156 | 298 ± 96 | 249 ± 83 | 0.412 | 0.007 | 0.228 |
| MCP-1, pg/mL | 554 ± 191 | 471 ± 197 | 623 ± 274 | 556 ± 281 | 0.263 | 0.022 | 0.692 |
| MDC, pg/mL | 929 ± 241 | 931 ± 315 | 1079 ± 319 | 1136 ± 335 | 0.044 | 0.235 | 0.251 |
| RANTES, ng/mL | 208 ± 127 | 188 ± 99 | 284 ± 164 | 248 ± 172 | 0.086 | 0.182 | 0.727 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Disclaimer
References
- An, R. Prevalence and trends of adult obesity in the US, 1999–2012. ISRN. Obesity 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.J.; Marriott, B.P.; Dotson, L.; Bathalon, G.P.; Funderburk, L.; White, A.; Hadden, L.; Young, A.J. Overweight and obesity in military personnel: Sociodemographic predictors. Obesity 2012, 20, 1534–1538. [Google Scholar] [CrossRef] [PubMed]
- Haslam, D.W.; James, W.P. Obesity. Lancet 2005, 366, 1197–1209. [Google Scholar] [CrossRef]
- Finkelstein, E.A.; Trogdon, J.G.; Cohen, J.W.; Dietz, W. Annual medical spending attributable to obesity: Payer-and service-specific estimates. Health Aff. 2009, 28, w822–w831. [Google Scholar] [CrossRef] [PubMed]
- Dall, T.M.; Zhang, Y.; Chen, Y.J.; Wagner, R.C.; Hogan, P.F.; Fagan, N.K.; Olaiya, S.T.; Tornberg, D.N. Cost associated with being overweight and with obesity, high alcohol consumption, and tobacco use within the military health system’s tricare prime-enrolled population. Am. J. Health Promot. 2007, 22, 120–139. [Google Scholar] [CrossRef] [PubMed]
- Tanofsky-Kraff, M.; Sbrocco, T.; Theim, K.R.; Cohen, L.A.; Mackey, E.R.; Stice, E.; Henderson, J.L.; McCreight, S.J.; Bryant, E.J.; Stephens, M.B. Obesity and the US military family. Obesity 2013, 21, 2205–2220. [Google Scholar] [CrossRef] [PubMed]
- Kelly, T.L.; Neri, D.F.; Grill, J.T.; Ryman, D.; Hunt, P.D.; Dijk, D.J.; Shanahan, T.L.; Czeisler, C.A. Nonentrained circadian rhythms of melatonin in submariners scheduled to an 18-hour day. J. Biol. Rhythms. 1999, 14, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Castanon-Cervantes, O.; Wu, M.; Ehlen, J.C.; Paul, K.; Gamble, K.L.; Johnson, R.L.; Besing, R.C.; Menaker, M.; Gewirtz, A.T.; Davidson, A.J. Dysregulation of inflammatory responses by chronic circadian disruption. J. Immunol. 2010, 185, 5796–5805. [Google Scholar] [CrossRef] [PubMed]
- Lund, J.; Arendt, J.; Hampton, S.M.; English, J.; Morgan, L.M. Postprandial hormone and metabolic responses amongst shift workers in Antarctica. J. Endocrinol. 2001, 171, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Scheer, F.A.; Hilton, M.F.; Mantzoros, C.S.; Shea, S.A. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc. Natl. Acad. Sci. USA 2009, 106, 4453–4458. [Google Scholar] [CrossRef] [PubMed]
- Shea, S.A.; Hilton, M.F.; Orlova, C.; Ayers, R.T.; Mantzoros, C.S. Independent circadian and sleep/wake regulation of adipokines and glucose in humans. J. Clin. Endocrinol. Metab. 2005, 90, 2537–2544. [Google Scholar] [CrossRef] [PubMed]
- Sookoian, S.; Gemma, C.; Fernandez Gianotti, T.; Burgueno, A.; Alvarez, A.; Gonzalez, C.D.; Pirola, C.J. Effects of rotating shift work on biomarkers of metabolic syndrome and inflammation. J. Intern. Med. 2007, 261, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Tenkanen, L.; Sjoblom, T.; Kalimo, R.; Alikoski, T.; Harma, M. Shift work, occupation and coronary heart disease over 6 years of follow-up in the Helsinki Heart Study. Scand. J. Work Environ. Health 1997, 23, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Gasier, H.G.; Gaffney-Stomberg, E.; Young, C.R.; McAdams, D.C.; Lutz, L.J.; McClung, J.P. The efficacy of vitamin D supplementation during a prolonged submarine patrol. Calcif. Tissue Int. 2014, 95, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Fields, D.A.; Goran, M.I.; McCrory, M.A. Body-composition assessment via air-displacement plethysmography in adults and children: A review. Am. J. Clin. Nutr. 2002, 75, 453–467. [Google Scholar] [PubMed]
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic; Report of a WHO Consultation; World Health Organization: Geneva, Switzerland, 2000; pp. 1–253. [Google Scholar]
- Bray, G.A. Fat distribution and body weight. Obes. Res. 1993, 1, 203–205. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, A.; Bianchi, A.; Maroni, P.; Iannarelli, A.; di Daniele, N.; Iacopino, L.; di Renzo, L. Adiposity rather than BMI determines metabolic risk. Int. J. Cardiol. 2011, 166, 111–117. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, A.; Deurenberg, P.; Pietrantuono, M.; di Daniele, N.; Cervelli, V.; Andreoli, A. How fat is obese? Acta Diabetol. 2003, 40, S254–S257. [Google Scholar] [CrossRef] [PubMed]
- Deurenberg, P. Universal cut-off BMI points for obesity are not appropriate. Br. J. Nutr. 2001, 85, 135–136. [Google Scholar] [CrossRef] [PubMed]
- Romero-Corral, A.; Somers, V.K.; Sierra-Johnson, J.; Thomas, R.J.; Collazo-Clavell, M.L.; Korinek, J.; Allison, T.G.; Batsis, J.A.; Sert-Kuniyoshi, F.H.; Lopez-Jimenez, F. Accuracy of body mass index in diagnosing obesity in the adult general population. Int. J. Obesity. (Lond.) 2008, 32, 959–966. [Google Scholar] [CrossRef] [PubMed]
- Kolar, A.S.; Patterson, R.E.; White, E.; Neuhouser, M.L.; Frank, L.L.; Standley, J.; Potter, J.D.; Kristal, A.R. A practical method for collecting 3-day food records in a large cohort. Epidemiology 2005, 16, 579–583. [Google Scholar] [CrossRef] [PubMed]
- Newby, P.K.; Maras, J.; Bakun, P.; Muller, D.; Ferrucci, L.; Tucker, K.L. Intake of whole grains, refined grains, and cereal fiber measured with 7-d diet records and associations with risk factors for chronic disease. Am. J. Clin. Nutr. 2007, 86, 1745–1753. [Google Scholar] [PubMed]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett, D.R., Jr.; Tudor-Locke, C.; Greer, J.L.; Vezina, J.; Whitt-Glover, M.C.; Leon, A.S. 2011 compendium of physical activities: A second update of codes and met values. Med. Sci. Sports. Exerc. 2011, 43, 1575–1581. [Google Scholar] [CrossRef]
- Staten, L.K.; Taren, D.L.; Howell, W.H.; Tobar, M.; Poehlman, E.T.; Hill, A.; Reid, P.M.; Ritenbaugh, C. Validation of the Arizona activity frequency questionnaire using doubly labeled water. Med. Sci. Sports Exerc. 2001, 33, 1959–1967. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.H.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith, S.C., Jr.; et al. Diagnosis and management of the metabolic syndrome: An american heart association/national heart, lung, and blood institute scientific statement. Circulation 2005, 112, 2735–2752. [Google Scholar] [CrossRef] [PubMed]
- Gregg, M.A., 2nd; Jankosky, C.J. Physical readiness and obesity among male U.S. navy personnel with limited exercise availability while at sea. Mil. Med. 2012, 177, 1302–1307. [Google Scholar] [CrossRef] [PubMed]
- Gasier, H.G.; Hughes, L.M.; Young, C.R.; Richardson, A.M. Comparison of body composition assessed by dual-energy X-ray absorptiometry and BMI in current and former U.S. navy service members. PLoS. ONE 2015, 10, e0132157. [Google Scholar]
- Peltz, G.; Aguirre, M.T.; Sanderson, M.; Fadden, M.K. The role of fat mass index in determining obesity. Am. J. Hum. Biol. 2010, 22, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.R.; Braverman, E.R. Measuring adiposity in patients: The utility of body mass index (BMI), percent body fat, and leptin. PLoS. ONE 2012, 7, e33308. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Li, C.; Zhao, G. Prevalence and correlates of metabolic syndrome based on a harmonious definition among adults in the US. J. Diabetes 2010, 2, 180–193. [Google Scholar] [CrossRef] [PubMed]
- Sumner, A.E.; Cowie, C.C. Ethnic differences in the ability of triglyceride levels to identify insulin resistance. Atherosclerosis 2008, 196, 696–703. [Google Scholar] [CrossRef]
- Kadowaki, T.; Yamauchi, T.; Kubota, N.; Hara, K.; Ueki, K.; Tobe, K. Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J. Clin. Invest. 2006, 116, 1784–1792. [Google Scholar] [CrossRef] [PubMed]
- Esser, N.; Legrand-Poels, S.; Piette, J.; Scheen, A.J.; Paquot, N. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes. Res. Clin. Pract. 2014, 105, 141–150. [Google Scholar] [CrossRef]
- Baturcam, E.; Abubaker, J.; Tiss, A.; Abu-Farha, M.; Khadir, A.; Al-Ghimlas, F.; Al-Khairi, I.; Cherian, P.; Elkum, N.; Hammad, M.; et al. Physical exercise reduces the expression of rantes and its CCR5 receptor in the adipose tissue of obese humans. Mediat. Inflamm. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Maury, E.; Brichard, S.M.; Pataky, Z.; Carpentier, A.; Golay, A.; Bobbioni-Harsch, E. Effect of obesity on growth-related oncogene factor-alpha, thrombopoietin, and tissue inhibitor metalloproteinase-1 serum levels. Obesity 2010, 18, 1503–1509. [Google Scholar] [CrossRef] [PubMed]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Nyce, M.R.; Ohannesian, J.P.; Marco, C.C.; McKee, L.J.; Bauer, T.L.; et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med. 1996, 334, 292–295. [Google Scholar] [CrossRef]
- Pan, H.; Guo, J.; Su, Z. Advances in understanding the interrelations between leptin resistance and obesity. Physiol. Behav. 2014, 130, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Jaramillo, P.; Gomez-Arbelaez, D.; Lopez-Lopez, J.; Lopez-Lopez, C.; Martinez-Ortega, J.; Gomez-Rodriguez, A.; Triana-Cubillos, S. The role of leptin/adiponectin ratio in metabolic syndrome and diabetes. Horm. Mol. Biol. Clin. Investig. 2014, 18, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C.M.; Tierney, A.C.; Perez-Martinez, P.; Defoort, C.; Blaak, E.E.; Gjelstad, I.M.; Lopez-Miranda, J.; Kiec-Klimczak, M.; Malczewska-Malec, M.; Drevon, C.A.; et al. Obesity and body fat classification in the metabolic syndrome: Impact on cardiometabolic risk metabotype. Obesity. 2013, 21, E154–E161. [Google Scholar] [CrossRef] [PubMed]
- Martynowicz, H.; Janus, A.; Nowacki, D.; Mazur, G. The role of chemokines in hypertension. Adv. Clin. Exp. Med. 2014, 23, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Aikawa, M.; Sugiyama, S.; Hill, C.C.; Voglic, S.J.; Rabkin, E.; Fukumoto, Y.; Schoen, F.J.; Witztum, J.L.; Libby, P. Lipid lowering reduces oxidative stress and endothelial cell activation in rabbit atheroma. Circulation 2002, 106, 1390–1396. [Google Scholar] [CrossRef] [PubMed]
- Webber, B.J.; Seguin, P.G.; Burnett, D.G.; Clark, L.L.; Otto, J.L. Prevalence of and risk factors for autopsy-determined atherosclerosis among US service members, 2001–2011. JAMA 2012, 308, 2577–2583. [Google Scholar] [CrossRef]
- Strohacker, K.; McCaffery, J.M.; MacLean, P.S.; Wing, R.R. Adaptations of leptin, ghrelin or insulin during weight loss as predictors of weight regain: A review of current literature. Int. J. Obesity. (Lond.) 2014, 38, 388–396. [Google Scholar] [CrossRef]
- Sumithran, P.; Proietto, J. The defence of body weight: A physiological basis for weight regain after weight loss. Clin. Sci. (Lond.) 2013, 124, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.W.; D’Agostino, R.B.; Parise, H.; Sullivan, L.; Meigs, J.B. Metabolic syndrome as a precursor of cardiovascular disease and type 2 diabetes mellitus. Circulation 2005, 112, 3066–3072. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gasier, H.G.; Young, C.R.; Gaffney-Stomberg, E.; McAdams, D.C.; Lutz, L.J.; McClung, J.P. Cardiometabolic Health in Submariners Returning from a 3-Month Patrol. Nutrients 2016, 8, 85. https://doi.org/10.3390/nu8020085
Gasier HG, Young CR, Gaffney-Stomberg E, McAdams DC, Lutz LJ, McClung JP. Cardiometabolic Health in Submariners Returning from a 3-Month Patrol. Nutrients. 2016; 8(2):85. https://doi.org/10.3390/nu8020085
Chicago/Turabian StyleGasier, Heath G., Colin R. Young, Erin Gaffney-Stomberg, Douglas C. McAdams, Laura J. Lutz, and James P. McClung. 2016. "Cardiometabolic Health in Submariners Returning from a 3-Month Patrol" Nutrients 8, no. 2: 85. https://doi.org/10.3390/nu8020085
APA StyleGasier, H. G., Young, C. R., Gaffney-Stomberg, E., McAdams, D. C., Lutz, L. J., & McClung, J. P. (2016). Cardiometabolic Health in Submariners Returning from a 3-Month Patrol. Nutrients, 8(2), 85. https://doi.org/10.3390/nu8020085




