Resveratrol Supplementation in Schizophrenia Patients: A Randomized Clinical Trial Evaluating Serum Glucose and Cardiovascular Risk Factors
Abstract
:1. Introduction
2. Methods and Materials
2.1. Anthropometric Measurements
2.2. Plasma Biochemistry
2.3. Psychopathology Severity Assessment
2.4. Statistical Analysis
3. Results
| Resveratrol (n = 10) | Placebo (n = 9) | p Value * | |
|---|---|---|---|
| Age (years) | 46.40 ± 11.18 | 41.00 ± 7.87 | 0.245 |
| Education (in years) | 9.90 ± 3.95 | 10.22 ± 2.10 | 0.830 |
| Smoking (n(%)) | 4 (40) | 3(33) | 1.000 |
| Number of cigarette/day | 15 (6–20) | 30 (20–40) | 0.114 |
| Age of onset of the disease (years) | 23.90 ± 5.58 | 26.44 ± 9.67 | 0.486 |
| Length of illness (years) | 22.50 ± 10.00 | 14.56 ± 7.92 | 0.074 |
| Clozapine dose (mg/day) | 485 ± 213.50 | 600 ± 180.27 | 0.225 |
| Practice exercise (n(%)) | 8(80) | 7(77.8) | 1.000 |
| Time of exercise (min/week) | 135 (0–450) | 60 (0–300) | 0.152 |
| Anthropometric measurements | |||
| Body weight (kg) | 80.96 ± 13.99 | 90.45 ± 22.30 | 0.277 |
| BMI (kg/m2) | 26.88 ± 3.94 | 28.43 ± 5.85 | 0.505 |
| Waist circumference (cm) | 101.80 ± 12.30 | 105.78 ± 17.18 | 0.566 |
| Waist to hip ratio | 0.98 ± 0.06 | 0.98 ± 0.07 | 0.947 |
| Conicity index | 1.36 ± 0.07 | 1.36 ± 0.07 | 0.922 |
| Body fat percentage (%) | 26.82 ± 6.23 | 28.51 ± 5.99 | 0.568 |
| Plasma biomarkers | |||
| Serum glucose (mg/dL) | 98.90 ± 13.82 | 94.89 ± 12.43 | 0.517 |
| Total cholesterol (mg/dL) | 186.10 ± 26.96 | 185.56 ± 33.23 | 0.969 |
| LDL-cholesterol (mg/dL) | 110.26 ± 25.49 | 107.24 ± 26.98 | 0.805 |
| HDL-cholesterol (mg/dL) | 36.30 ± 2.946 | 39.56 ± 7.58 | 0.254 |
| TG (mg/dL) | 197.70 ± 66.73 | 194.33 ± 85.23 | 0.924 |
| 24-h recall | |||
| Energy 24 h intake (kcal) | 2057 ± 463 | 2119 ± 769 | 0.832 |
| Carbohydrate 24 h intake (%) | 51.8 ± 9.9 | 49.4 ± 9.1 | 0.596 |
| Protein 24 h intake (%) | 21.6 ± 6.2 | 19.3 ± 4.0 | 0.351 |
| Total lipids 24 h intake (%) | 26.6 ± 6.8 | 31.1 ± 7.3 | 0.186 |
| Cholesterol 24 h intake (mg/day) | 284 (81.3–473) | 217 (97–637) | 0.780 |
| Fiber 24 h intake (g/day) | 27.6 ± 6.4 | 25.2 ± 11.6 | 0.572 |
| Assessment of Symptoms | |||
| BPRS total score | 10.5 (2–27) | 13 (5–21) | 0.720 |
| BPRS positive symptoms | 0.5 (0–5) | 0 (0–9) | 0.968 |
| BPRS negative symptoms | 2 (0–14) | 3 (0–8) | 0.968 |
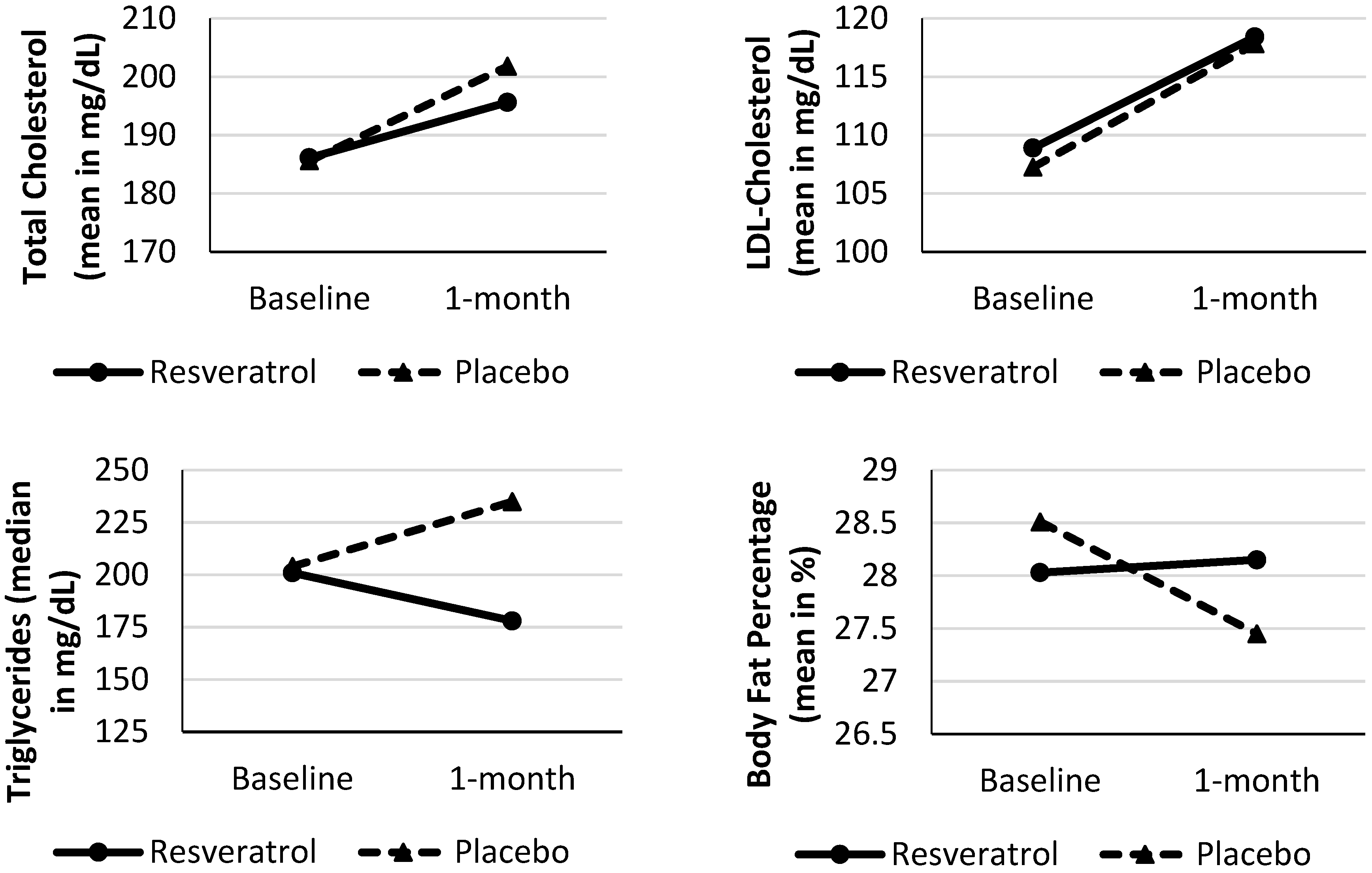
| Resveratrol | Placebo | |||||
|---|---|---|---|---|---|---|
| Characteristics | Day 1 (Baseline) | Day 30 | p Value * | Day 1 (Baseline) | Day 30 | p Value * |
| Exercise (min/week) | 135 (0–450) | 140 (0–450) | 0.463 | 60 (0–300) | 80 (0–200) | 0.461 |
| Weight (kg) | 80.96 ± 13.99 | 81.08 ± 13.93 | 0.845 | 90.45 ± 22.30 | 90.16 ± 23.02 | 0.354 |
| BMI (kg/m2) | 26.88 ± 3.94 | 26.83 ± 3.90 | 0.739 | 28.43 ± 5.85 | 28.29 ± 6.08 | 0.209 |
| Waist circumference (cm) | 101.80 ± 12.30 | 101.40 ± 12.03 | 0.583 | 105.78 ± 17.18 | 105.17 ± 17.35 | 0.202 |
| Waist to hip ratio | 0.98 ± 0.06 | 0.98 ± 0.07 | 0.581 | 0.98 ± 0.07 | 0.98 ± 0.07 | 0.232 |
| Conicity index | 1.36 ± 0.07 | 1.36 ± 0.07 | 0.454 | 1.36 ± 0.07 | 1.36 ± 0.07 | 0.394 |
| Body fat percentage (%) | 28.03 ± 5.20 | 28.15 ± 5.77 | 0.824 | 28.51 ± 5.99 | 27.45 ± 6.29 | 0.038 * |
| Serum glucose (mg/dL) | 98.90 ± 13.82 | 98.80 ± 11.81 | 0.957 | 94.89 ± 12.43 | 92.67 ± 7.73 | 0.568 |
| Total cholesterol (mg/dL) | 186.10 ± 26.96 | 195.60 ± 37.64 | 0.149 | 185.56 ± 33.23 | 201.78 ± 40.12 | 0.024 * |
| LDL-cholesterol (mg/dL) | 108.88 ± 26.64 | 118.40 ± 28.18 | 0.110 | 107.24 ± 26.98 | 117.80 ± 26.73 | 0.002 * |
| HDL-cholesterol (mg/dL) | 36.30 ± 2.94 | 36.80 ± 3.29 | 0.586 | 39.56 ± 7.58 | 40.33 ± 7.00 | 0.718 |
| TG (mg/dL) | 201 (66–297) | 178 (76–574) | 0.508 | 204 (87–294) | 235 (60–415) | 0.236 |
| Energy 24 h intake (kcal) | 2057 ± 463 | 2095 ± 551 | 0.790 | 2119 ± 769 | 1842 ± 472 | 0.219 |
| Carbohydrate 24 h intake (%) | 51.8 ± 9.9 | 53.0 ± 6.9 | 0.529 | 49.4 ± 9.1 | 55.6 ± 10.3 | 0.255 |
| Protein 24 h intake (%) | 21.6 ± 6.2 | 20.4 ± 5.3 | 0.312 | 19.3 ± 4.0 | 19.0 ± 5.6 | 0.888 |
| Total lipids 24 h intake (%) | 26.6 ± 6.8 | 26.6 ± 5.4 | 0.992 | 31.1 ± 7.3 | 25.5 ± 6.1 | 0.154 |
| Cholesterol 24 h intake (mg/day) | 284 (185–446) | 284 (126–434) | 0.753 | 217 (152–377) | 191 (135–298) | 0.260 |
| Fiber 24 h intake (g/day) | 27.6 ± 6.4 | 27.2 ± 7.8 | 0.851 | 25.2 ± 11.6 | 22.2 ± 7.7 | 0.396 |
| BPRS total score | 10.5 (2.8–21.3) | 16 (3–25.3) | 0.475 | 13 (8–18.5) | 14 (4–17.5) | 0.917 |
| BPRS positive symptoms | 0.5 (0–3.3) | 0.5 (0–7) | 0.245 | 0 (0–4) | 0.5 (0–2.8) | 0.480 |
| BPRS negative symptoms | 2 (1.5–8) | 3 (0–8.3) | 0.933 | 3 (1–6) | 6 (1.5–7.8) | 0.197 |
| Non-adherence (pill returned) | - | 5 (0–12) | - | - | 9 (2–18) | 0.447 |
| Resveratrol | Placebo | |
|---|---|---|
| Characteristics | Difference 1–30 (%) | Difference 1–30 (%) |
| Body fat percentage (%) | 1.35 (−9.42 to 11.68) | −2.74 (−12.56 to 1.36) |
| Serum glucose (mg/dL) | 0.57 (−8.27 to 6.80) | −2.12 (−20 to 17.57) |
| Total cholesterol (mg/dL) | 4.55 (−13.90 to 22.22) | 9.22 (−4.84 to 24) |
| LDL-cholesterol (mg/dL) | 4.95 (−14.13 to 51.65) | 9.14 (−2.56 to 36.19) |
| HDL-cholesterol (mg/dL) | 0 (−7.14 to 13.89) | −4.88 (−19.57 to 29.27) |
| TG (mg/dL) | −7.64 (−57.78 to 103.55) | 9.19 (−31.03 to 43.10) |
| Energy 24 h intake (kcal) | 0 (−39.62 to 29.07) | −4.49 (−48.76 to 34.28) |
| Carbohydrate 24 h intake (%) | 0 (−15.89 to 31.99) | 12.11 (−25.05 to 86.04) |
| Protein 24 h intake (%) | 0 (−38.7 to 13.22) | 9.98 (−54.23 to 44.15) |
| Total lipids 24 h intake (%) | 0 (−23.75 to 51.47) | −19.33 (−60.83 to 33.16) |
| Cholesterol 24 h intake (mg/day) | 0 (−41.21 to 23.78) | −18.84 (−90.67 to 78.69) |
| Correlation Coefficient | ||
|---|---|---|
| Associations | Resveratrol | Placebo |
| Education (years of study) vs. BMI | r = 0.747 (p = 0.013) | r = 0.361 (p = 0.340) |
| Number of cigarette/day vs. ∆ Body fat percentage (%) | rs = 0.683 (p = 0.042) | rs = 0.164 (p = 0.699) |
| Age (years) vs. ∆ Body weight (kg) | r = −0.697 (p = 0.025) | r = −0.795 (p = 0.010) |
| Length of illness (years) vs. ∆ Body weight (kg) | r = −0.749 (p = 0.013) | r = −0.596 (p = 0.090) |
| Length of illness (years) vs. ∆ HDL-cholesterol (mg/dL) | rs = 0.602 (p = 0.066) | rs = −0.625 (p = 0.072) |
| Non-adherence (pill returned) vs. ∆ Waist circumference (cm) | rs = 0.816 (p = 0.004) | rs = −0.366 (p = 0.333) |
| Non-adherence (pill returned) vs. ∆ Conicity index | rs = 0.597 (p = 0.068) | rs = −0.412 (p = 0.271) |
| Non-adherence (pill returned) vs. ∆ Body weight (kg) | rs = 0.596 (p = 0.069) | rs = 0.055 (p = 0.889) |
| Clozapine dose (mg/day) vs. ∆ TG | r = −0.734 (p = 0.016) | r = −0.077 (p = 0.804) |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hjorth, P.; Davidsen, A.S.; Kilian, R.; Skrubbeltrang, C. A systematic review of controlled interventions to reduce overweight and obesity in people with schizophrenia. Acta Psychiatr. Scand. 2014, 130, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Henderson, D.C.; Nguyen, D.D.; Copeland, P.M.; Hayden, D.L.; Borba, C.P.; Louie, P.M.; Freudenreich, O.; Evins, A.E.; Cather, C.; Goff, D.C. Clozapine, diabetes mellitus, hyperlipidemia and cardiovascular risks and mortality: Results of a 10-year naturalistic study. J. Clin. Psychiatry 2005, 66, 1116–1121. [Google Scholar] [CrossRef] [PubMed]
- Elkis, H.; Gama, C.S.; Suplicy, H.; Tambascia, M.; Bressan, R.; Lyra, R.; Cavalcante, S.; Minicucci, W. Brazilian Consensus on second-generation antipsychotics and metabolic disorders. Rev. Bras. Psiquiatr. 2008, 30, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Bresee, L.C.; Majumdar, S.R.; Patten, S.B.; Johnson, J.A. Diabetes, cardiovascular disease, and health care use in people with and without schizophrenia. Eur. Psychiatry 2011, 26, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Zortea, K.; Bock, P.M.; Moreno, D.B.; Belmonte-de-Abreu, P.S. Anthropometric and biochemical assessment of schizophrenic patients taking clozapine. Rev. Nutr. Camp. 2009, 22, 697–705. [Google Scholar]
- Bakker, G.C.; van Erk, M.J.; Pellis, L.; Wopereis, S.; Rubingh, C.M.; Cnubben, N.H.; Kooistra, T.; van Ommen, B.; Hendriks, H.F. An antiinflammatory dietary mix modulates inflammation and oxidative and metabolic stress in overweight men: A nutrigenomics approach. Am. J. Clin. Nutr. 2010, 91, 1044–1059. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, R.K. Emerging risk biomarkers in cardiovascular diseases and disorders. J. Lipids 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Ma, L.; Ruan, L.; Kong, Y.; Mou, H.; Zhang, Z.; Wang, Z.; Wang, J.M.; Le, Y. Resveratrol differentially modulates inflammatory responses of microglia and astrocytes. J. Neuroinflamm. 2010, 7, 46. [Google Scholar] [CrossRef] [PubMed]
- Catalgol, B.; Batirel, S.; Taga, Y.; Ozer, N.K. Resveratrol: French paradox revisited. Front. Pharmacol. 2012, 3, 141. [Google Scholar] [CrossRef] [PubMed]
- Csiszar, A.; Smith, K.; Labinskyy, N.; Orosz, Z.; Rivera, A.; Ungvari, Z. Resveratrol attenuates TNF-alfa-induced activation of coronary arterial endothelial cells: Role of NF-kB inhibition. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, H1694–H1699. [Google Scholar] [CrossRef] [PubMed]
- Di Castelnuovo, A.; Rotondo, S.; Iacoviello, L.; Donati, M.B.; de Gaetano, G. Meta-analysis of wine and beer consumption in relation to vascular risk. Circulation 2002, 105, 2836–2844. [Google Scholar] [CrossRef] [PubMed]
- Sahebkar, A.; Serban, C.; Ursoniu, S.; Wong, N.D.; Muntner, P.; Graham, I.M.; Mikhailidis, D.P.; Rizzo, M.; Rysz, J.; Sperling, L.S.; et al. Lack of efficacy of resveratrol on C-reactive protein and selected cardiovascular risk factors—Results from a systematic review and meta-analysis of randomized controlled trials. Int. J. Cardiol. 2015, 189, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Valdez, R. A simple model-based index of abdominal adiposity. J. Clin. Epidemiol. 1991, 44, 955–956. [Google Scholar] [CrossRef]
- Romano, F.; Elkis, H. Translation and adaptation of the Brief Psychiatric Rating Scale-anchored version (BPRS-A). J. Bras. Psiquiatr. 1996, 45, 43–49. [Google Scholar]
- De Ligt, M.; Timmers, S.; Schrauwen, P. Resveratrol and obesity: Can resveratrol relieve metabolic disturbances? Biochim. Biophys. Acta 2015, 1852, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Larrosa, M.; González-Sarrías, A.; Tomás-Barberán, F.A.; García-Conesa, M.T.; Espín, J.C. Resveratrol and clinical trials: The crossroad from in vitro studies to human evidence. Curr. Pharm. Des. 2013, 19, 6064–6093. [Google Scholar] [CrossRef] [PubMed]
- Sahebkar, A. Effects of resveratrol supplementation on plasma lipids: A systematic review and meta-analysis of randomized controlled trials. Nutr. Rev. 2013, 71, 822–835. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.A.; Khan, D.A.; Mahjabeen, W.; Papasian, C.J.; Qureshi, N. Nutritional Supplement-5 with a Combination of Proteasome Inhibitors (Resveratrol, Quercetin, δ- Tocotrienol) Modulate Age-Associated Biomarkers and Cardiovascular Lipid Parameters in Human Subjects. J. Clin. Exp. Cardiol. 2013, 4, 238. [Google Scholar] [CrossRef] [PubMed]
- Kjær, T.N.; Poulsen, M.M.; Ornstrup, M.J.; Jørgensen, H.S.; Jørgensen, J.O.; Richelsen, B.; Pedersen, S.B. The antiinflammatory effects of resveratrol: A randomized controlled trial. In Proceedings of the Resveratrol 2014, the 3rd International Conference on Resveratrol and Health, Waikoloa, Hawaii Island, HI, USA, 2014; p. 10. Available online: https://www.dropbox.com/s/1frl61xq472iztg/12.12.14-Program_Web.pdf?dl=0 (accessed on 15 January 2015).
- Liu, K.; Zhou, R.; Wang, B.; Mi, M.T. Effect of resveratrol on glucose control and insulin sensitivity: A meta-analysis of 11 randomized controlled trials. Am. J. Clin. Nutr. 2014, 99, 1510–1519. [Google Scholar] [CrossRef] [PubMed]
- Gurmu, A.E.; Abdela, E.; Allele, B.; Cheru, E.; Amogne, B. Rate of Nonadherence to Antipsychotic Medications and Factors Leading to Nonadherence among Psychiatric Patients in Gondar University Hospital, Northwest Ethiopia. Adv. Psychiatry 2014. [Google Scholar] [CrossRef]
- Oksanen, J. Supporting adherence to drug therapy in psychiatry. Duodecim 2015, 131, 1437–1442. [Google Scholar] [PubMed]
- Zullig, L.L.; Gellad, W.F.; Moaddeb, J.; Crowley, M.J.; Shrank, W.; Granger, B.B.; Granger, C.B.; Trygstad, T.; Liu, L.Z.; Bosworth, H.B. Improving diabetes medication adherence: Successful, scalable interventions. Patient Prefer. Adherence 2015, 9, 139–149. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zortea, K.; Franco, V.C.; Francesconi, L.P.; Cereser, K.M.M.; Lobato, M.I.R.; Belmonte-de-Abreu, P.S. Resveratrol Supplementation in Schizophrenia Patients: A Randomized Clinical Trial Evaluating Serum Glucose and Cardiovascular Risk Factors. Nutrients 2016, 8, 73. https://doi.org/10.3390/nu8020073
Zortea K, Franco VC, Francesconi LP, Cereser KMM, Lobato MIR, Belmonte-de-Abreu PS. Resveratrol Supplementation in Schizophrenia Patients: A Randomized Clinical Trial Evaluating Serum Glucose and Cardiovascular Risk Factors. Nutrients. 2016; 8(2):73. https://doi.org/10.3390/nu8020073
Chicago/Turabian StyleZortea, Karine, Viviane C. Franco, Lenise P. Francesconi, Keila M. M. Cereser, Maria Inês R. Lobato, and Paulo S. Belmonte-de-Abreu. 2016. "Resveratrol Supplementation in Schizophrenia Patients: A Randomized Clinical Trial Evaluating Serum Glucose and Cardiovascular Risk Factors" Nutrients 8, no. 2: 73. https://doi.org/10.3390/nu8020073
APA StyleZortea, K., Franco, V. C., Francesconi, L. P., Cereser, K. M. M., Lobato, M. I. R., & Belmonte-de-Abreu, P. S. (2016). Resveratrol Supplementation in Schizophrenia Patients: A Randomized Clinical Trial Evaluating Serum Glucose and Cardiovascular Risk Factors. Nutrients, 8(2), 73. https://doi.org/10.3390/nu8020073





