Effect of Tea Polyphenol Compounds on Anticancer Drugs in Terms of Anti-Tumor Activity, Toxicology, and Pharmacokinetics
Abstract
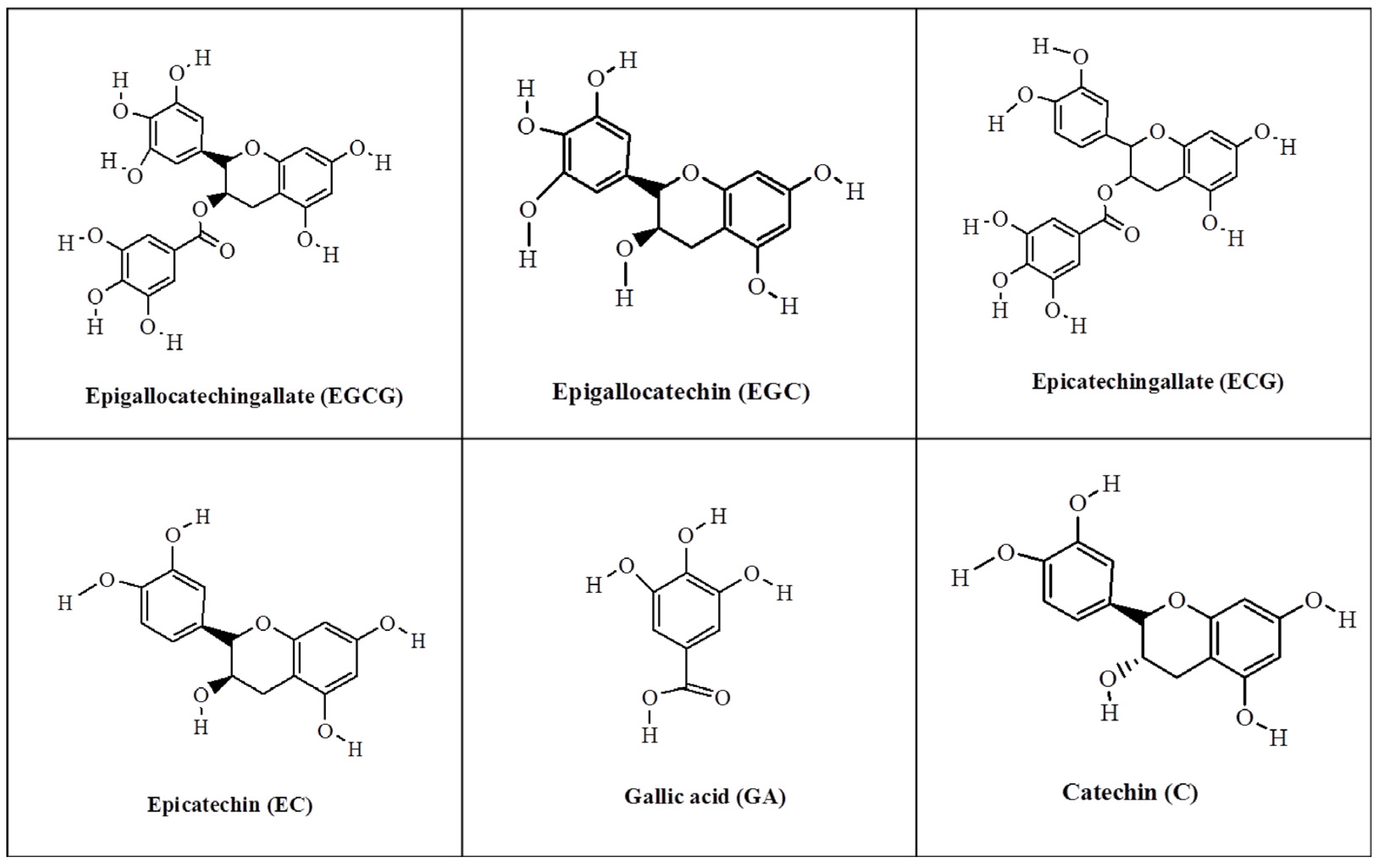
:1. Introduction
2. Synergistic Anticancer Activity of Tea Polyphenols and Chemotherapeutic Agents
2.1. Combination of Tea Polyphenols and Bleomycin
2.2. Combination of Tea Polyphenols and Cisplatin
2.3. Combination of Tea Polyphenols and Ibuprofen
2.4. Combination of Tea Polyphenols and Tamoxifen
2.5. Combination of Tea Polyphenols and Bortezomib
2.6. Combination of Tea Polyphenols and Other Anticancer Drugs
2.7. Combination of Caffeine and Anticancer Drugs
3. Ameliorating Toxicity Induced by Chemotherapeutic Agents
4. Pharmacokinetic Effect on Chemotherapeutic Agents
5. Human Trials
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Qiao, J.; Kong, X.; Kong, A.; Han, M. Pharmacokinetics and biotransformation of tea polyphenols. Curr. Drug Metab. 2014, 15, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, H.; Suganuma, M.; Imai, K.; Nakachi, K. Green tea: Cancer preventive beverage and/or drug. Cancer Lett. 2002, 188, 9–13. [Google Scholar] [CrossRef]
- Afzal, M.; Safer, A.M.; Menon, M. Green tea polyphenols and their potential role in health and disease. Inflammopharmacology 2015, 23, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Hara, Y. Tea catechins and their applications as supplements and pharmaceutics. Pharmacol. Res. 2011, 64, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, H. Green tea: Health benefits as cancer preventive for humans. Chem. Rec. 2005, 5, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.D.; Yang, C.S. Mechanisms of cancer prevention by tea constituents. J. Nutr. 2003, 133, 3262S–3267S. [Google Scholar] [PubMed]
- Higdon, J.V.; Frei, B. Tea catechins and polyphenols: Health effects, metabolism, and antioxidant functions. Crit. Rev. Food Sci. Nutr. 2003, 43, 89–143. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Maliakal, P.; Meng, X. Inhibition of carcinogenesis by tea. Annu. Rev. Pharmacol. Toxicol. 2002, 42, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, H.; Imai, K.; Nakachi, K.; Shimizu, M.; Moriwaki, H.; Suganuma, M. Challenging the effectiveness of green tea in primary and tertiary cancer prevention. J. Cancer Res. Clin. Oncol. 2012, 138, 1259–1270. [Google Scholar] [CrossRef] [PubMed]
- Alshatwi, A.A.; Periasamy, V.S.; Athinarayanan, J.; Elango, R. Synergistic anticancer activity of dietary tea polyphenols and bleomycin hydrochloride in human cervicalcancer cell: Caspase-dependent and independent apoptotic pathways. Chem. Biol. Interact. 2016, 247, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Suganuma, M.; Saha, A.; Fujiki, H. New cancer treatment strategy using combination of green tea catechins and anticancer drugs. Cancer Sci. 2011, 102, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Morre, D.J.; Morre, D.M.; Sun, H.; Cooper, R.; Chang, J.; Janle, E.M. Tea catechin synergies in inhibition of cancer cell proliferation and of a cancer specific cell surface oxidase (ECTO-NOX). Pharmacol. Toxicol. 2003, 92, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, H.; Sueoka, E.; Watanabe, T.; Suganuma, M. Synergistic enhancement of anticancer effects on numerous human cancer cell lines treated with thecombination of EGCG, other green tea catechins, and anticancer compounds. J. Cancer Res. Clin. Oncol. 2015, 141, 1511–1522. [Google Scholar] [CrossRef] [PubMed]
- Adhami, V.M.; Malik, A.; Zaman, N.; Sarfaraz, S.; Siddiqui, I.A.; Syed, D.N.; Afaq, F.; Pasha, F.S.; Saleem, M.; Mukhtar, H. Combined inhibitory effects of green tea polyphenols and selective cyclooxygenase-2 inhibitors on the growth of human prostate cancer cells both in vitro and in vivo. Clin. Cancer Res. 2007, 13, 1611–1619. [Google Scholar] [CrossRef] [PubMed]
- Qiao, J.; Gu, C.; Shang, W.; Du, J.; Yin, W.; Zhu, M.; Wang, W.; Han, M.; Lu, W. Effect of green tea on pharmacokinetics of 5-fluorouracil in rats and pharmacodynamics in human cell lines in vitro. Food Chem. Toxicol. 2011, 49, 1410–1415. [Google Scholar] [CrossRef] [PubMed]
- Sriram, N.; Kalayarasan, S.; Sudhandiran, G. Epigallocatechin-3-gallate exhibits anti-fibrotic effect by attenuating bleomycin-induced glycoconjugates, lysosomal hydrolases and ultrastructural changes in rat model pulmonary fibrosis. Chem. Biol. Interact. 2009, 180, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Periasamy, V.S.; Alshatwi, A.A. Tea polyphenols modulate antioxidant redox system on cisplatin-induced reactive oxygen species generation in a human breast cancer cell. Basic Clin. Pharmacol. Toxicol. 2013, 112, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.Z.; Zhen, Y.S. Molecular targets of tea polyphenols and its roles of anticancer drugs in experimental therapy. Yao Xue Xue Bao 2013, 48, 1–7. [Google Scholar] [PubMed]
- Ahmad, K.A.; Harris, N.H.; Johnson, A.D.; Lindvall, H.C.; Wang, G.; Ahmed, K. Protein kinase CK2 modulates apoptosis induced by resveratrol and epigallocatechin-3-gallate in prostate cancer cells. Mol. Cancer Ther. 2007, 6, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Mazumder, M.E.; Beale, P.; Chan, C.; Yu, J.Q.; Huq, F. Epigallocatechin gallate acts synergistically in combination with cisplatin and designed trans-palladiums in ovarian cancer cells. Anticancer Res. 2012, 32, 4851–4860. [Google Scholar] [PubMed]
- Chan, M.M.; Soprano, K.J.; Einstein, K.; Fong, D. Epigallocatechin-3-gallate delivers hydrogen peroxide to induce death of ovarian cancer cells and enhancestheir cisplatin susceptibility. J. Cell Physiol. 2006, 207, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.M.; Fong, D.; Soprano, K.J.; Holmes, W.F.; Heverling, H. Inhibition of growth and sensitization to cisplatin-mediated killing of ovarian cancer cells by polyphenolic chemopreventive agents. J. Cell Physiol. 2003, 194, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Chung, J. Synergistic cell death by EGCG and ibuprofen in DU-145 prostate cancer cell line. Anticancer Res. 2007, 27, 3947–3956. [Google Scholar] [PubMed]
- Farabegoli, F.; Papi, A.; Orlandi, M. (−)-Epigallocatechin-3-gallate down-regulates EGFR, MMP-2, MMP-9 and EMMPRIN and inhibits the invasion of MCF-7 tamoxifen-resistant cells. Biosci. Rep. 2011, 31, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Scandlyn, M.J.; Stuart, E.C.; Somers-Edgar, T.J.; Menzies, A.R.; Rosengren, R.J. A new role for tamoxifen in oestrogen receptor-negative breast cancer when it is combined with epigallocatechin gallate. Br. J. Cancer 2008, 99, 1056–1063. [Google Scholar] [CrossRef] [PubMed]
- Chisholm, K.; Bray, B.J.; Rosengren, R.J. Tamoxifen and epigallocatechin gallate are synergistically cytotoxic to MDA-MB-231 human breast cancer cells. Anticancer Drugs 2004, 15, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Suganuma, M.; Okabe, S.; Kai, Y.; Sueoka, N.; Sueoka, E.; Fujiki, H. Synergistic effects of (−)-epigallocatechin gallate with (−)-epicatechin, sulindac, or tamoxifen on cancer-preventive activity in the human lung cancer cell line PC-9. Cancer Res. 1999, 59, 44–47. [Google Scholar] [PubMed]
- Fujiki, H.; Suganuma, M.; Kurusu, M.; Okabe, S.; Imayoshi, Y.; Taniguchi, S.; Yoshida, T. New TNF-α releasing inhibitors as cancer preventive agents from traditional herbal medicine and combination cancer prevention study with EGCG and sulindac or tamoxifen. Mutat. Res. 2003, 524, 119–125. [Google Scholar] [CrossRef]
- Bannerman, B.; Xu, L.; Jones, M.; Tsu, C.; Yu, J.; Hales, P.; Monbaliu, J.; Fleming, P.; Dick, L.; Manfredi, M.; et al. Preclinical evaluation of the antitumor activity of bortezomib in combination with vitamin C or with epigallocatechin gallate, a component of green tea. Cancer Chemother. Pharmacol. 2011, 68, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Golden, E.B.; Lam, P.Y.; Kardosh, A.; Kardosh, A.; Gaffney, K.J.; Cadenas, E.; Louie, S.G.; Petasis, N.A.; Chen, T.C.; Schönthal, A.H. Green tea polyphenols block the anticancer effects of bortezomib and other boronic acid-based proteasome inhibitors. Blood 2009, 113, 5927–5937. [Google Scholar] [CrossRef] [PubMed]
- Glynn, S.J.; Gaffney, K.J.; Sainz, M.A.; Louie, S.G.; Petasis, N.A. Molecular characterization of the boron adducts of the proteasome inhibitor bortezomib with epigallocatechin-3-gallate and related polyphenols. Org. Biomol. Chem. 2015, 13, 3887–3899. [Google Scholar] [CrossRef] [PubMed]
- Suganuma, M.; Kurusu, M.; Suzuki, K.; Tasaki, E.; Fujiki, H. Green tea polyphenol stimulates cancer preventive effects of celecoxib in human lung cancer cells by upregulation of GADD153 gene. Int. J. Cancer 2006, 119, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Amin, A.R.; Wang, D.; Zhang, H.; Peng, S.; Shin, H.J.; Brandes, J.C.; Tighiouart, M.; Khuri, F.R.; Chen, Z.G.; Shin, D.M. Enhanced anti-tumor activity by the combination of the natural compounds (−)-epigallocatechin-3-gallate and luteolin: Potential role of p53. J. Biol. Chem. 2010, 285, 34557–34565. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Landen, C.N.; Li, Y.; Alvarez, R.D.; Tollefsbol, T.O. Epigallocatechin gallate and sulforaphane combination treatment induce apoptosis in paclitaxel-resistant ovarian cancer cells through hTERT and Bcl-2 down-regulation. Exp. Cell Res. 2013, 319, 697–706. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.W.; Wang, X.L.; Cao, L.Q.; Jiang, X.F.; Peng, H.P.; Lin, S.M.; Xue, P.; Chen, D. Green tea polyphenol epigallocatechin-3-gallate enhances 5-fluorouracil-induced cell growth inhibition of hepatocellular carcinoma cells. Hepatol. Res. 2012, 42, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Saha, A.; Kuzuhara, T.; Echigo, N.; Suganuma, M.; Fujiki, H. New role of (−)-epicatechin in enhancing the induction of growth inhibition and apoptosis in human lung cancer cells by curcumin. Cancer Prev. Res. 2010, 3, 953–962. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.K.; Kay, N.E.; Secreto, C.R.; Shanafelt, T.D. Curcumin inhibits prosurvival pathways in chronic lymphocytic leukemia B cells and may overcome their stromal protection in combination with EGCG. Clin. Cancer Res. 2009, 15, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Somers-Edgar, T.J.; Scandlyn, M.J.; Stuart, E.C.; Le Nedelec, M.J.; Valentine, S.P.; Rosengren, R.J. The combination of epigallocatechin gallate and curcumin suppresses ER alpha-breast cancer cell growth in vitro and in vivo. Int. J. Cancer 2008, 122, 1966–1971. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.N.; Singh, C.; Nall, D.; Meeker, D.; Shankar, S.; Srivastava, R.K. The dietary bioflavonoid quercetin synergizes with epigallocatechin gallate (EGCG) to inhibit prostate cancer stem cell characteristics, invasion, migration and epithelial-mesenchymal transition. J. Mol. Signal. 2010, 5, 14. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, T.C.; Wu, J.M. Targeting CWR22Rv1 prostate cancer cell proliferation and gene expression by combinations of the phytochemicals EGCG, genistein and quercetin. Anticancer Res. 2009, 29, 4025–4032. [Google Scholar] [PubMed]
- Stearns, M.E.; Wang, M. Synergistic effects of the green tea extract epigallocatechin-3-gallate and taxane in eradication of malignant human prostate tumors. Transl. Oncol. 2011, 4, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Liang, G.; Tang, A.; Lin, X.; Li, L.; Zhang, S.; Huang, Z.; Tang, H.; Li, Q.Q. Green tea catechins augment the antitumor activity of doxorubicin in an in vivo mouse model for chemoresistant liver cancer. Int. J. Oncol. 2010, 37, 111–123. [Google Scholar] [PubMed]
- Hsieh, T.C.; Wu, J.M. Suppression of cell proliferation and gene expression by combinatorial synergy of EGCG, resveratrol and gamma-tocotrienol in estrogen receptor-positive MCF-7 breast cancer cells. Int. J. Oncol. 2008, 33, 851–859. [Google Scholar] [PubMed]
- Nair, S.; Hebbar, V.; Shen, G.; Gopalakrishnan, A.; Khor, T.O.; Yu, S.; Xu, C.; Kong, A.N. Synergistic effects of a combination of dietary factors sulforaphane and (−)-epigallocatechin-3-gallate in HT-29 AP-1 human colon carcinoma cells. Pharm. Res. 2008, 25, 387–399. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Bhoopalan, V.; Wang, D.; Wang, L.; Xu, X. The effect of caffeine on cisplatin-induced apoptosis of lung cancer cells. Exp. Hematol. Oncol. 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Mori, Y.; Ueda, Y.; Okada, G.; Tomita, K. Sensitization and caffeine potentiation of cisplatin cytotoxicity resulting from introduction of wild-type p53 gene in human osteosarcoma. Anticancer Res. 2000, 20, 235–242. [Google Scholar] [PubMed]
- Karita, M.; Tsuchiya, H.; Kawahara, M.; Kasaoka, S.; Tomita, K. The antitumor effect of liposome-encapsulated cisplatin on rat osteosarcoma and its enhancement by caffeine. Anticancer Res. 2008, 28, 1449–1457. [Google Scholar] [PubMed]
- Geriyol, P.; Basavanneppa, H.B.; Dhananjaya, B.L. Protecting effect of caffeine against vinblastine (an anticancer drug) induced genotoxicity in mice. Drug Chem. Toxicol. 2015, 38, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Dudka, J.; Gieroba, R.; Korga, A.; Burdan, F.; Matysiak, W.; Jodlowska-Jedrych, B.; Mandziuk, S.; Korobowicz, E.; Murias, M. Different effects of resveratrol on dose-related doxorubicin-induced heart and liver toxicity. Evid.-Based Complement. Altern. Med. 2012, 2012, 606183. [Google Scholar] [CrossRef] [PubMed]
- Chari, N.S.; Pinaire, N.L.; Thorpe, L.; Medeiros, L.J.; Routbort, M.J.; McDonnell, T.J. The p53 tumor suppressor network in cancer and the therapeutic modulation of cell death. Apoptosis 2009, 14, 336–347. [Google Scholar] [CrossRef] [PubMed]
- Weijl, N.I.; Elsendoorn, T.J.; Lentjes, E.G.; Hopman, G.D.; Wipkink-Bakker, A.; Zwinderman, A.H. Supplementation with antioxidant micronutrients and chemotherapy-induced toxicity in cancer patients treated with cisplatin-based chemotherapy: A randomised, double-blind, placebo-controlled study. Eur. J. Cancer 2004, 40, 1713–1723. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Nie, S.; Xie, M.; Chen, Y.; Li, C.; Zhang, H. A major green tea component, (−)-epigallocatechin-3-gallate ameliorates doxorubicin-mediated cardiotoxicity in cardiomyocytes of neonatal rats. J. Agric. Food Chem. 2010, 58, 8877. [Google Scholar] [CrossRef] [PubMed]
- Khan, G.; Haque, S.E.; Anwer, T.; Ahsan, M.N.; Safhi, M.M.; Alam, M.F. Cardioprotective effect of green tea extract on doxorubicin-induced cardiotoxicity in rats. Acta Pol. Pharm. 2014, 71, 861–867. [Google Scholar] [PubMed]
- Saeed, N.M.; El-Naga, R.N.; El-Bakly, W.M.; Abdel-Rahman, H.M.; Salah El-Din, R.A.; El-Demerdash, E. Epigallocatechin-3-gallate pretreatment attenuates doxorubicin-induced cardiotoxicity in rats: A mechanistic study. Biochem. Pharmacol. 2015, 95, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Fleisher, B.; Unum, J.; Shao, J.; An, G. Ingredients in fruit juices interact with dasatinib through inhibition of BCRP: A new mechanism of beverage-drug interaction. J. Pharm. Sci. 2015, 104, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Knop, J.; Misaka, S.; Singer, K.; Hoier, E.; Müller, F.; Glaeser, H.; König, J.; Fromm, M.F. Inhibitory Effects of Green Tea and (−)-Epigallocatechin Gallate on Transport by OATP1B1, OATP1B3, OCT1, OCT2, MATE1, MATE2-K and P-Glycoprotein. PLoS ONE 2015, 10, e0139370. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.; Lu, W.; Han, M.; Qiao, J. The interactions of anticancer agents with tea catechins: Current evidence from preclinical studies. Anticancer Agents Med. Chem. 2014, 14, 1343–1450. [Google Scholar] [CrossRef] [PubMed]
- Mirkov, S.; Komoroski, B.J.; Ramírez, J.; Graber, A.Y.; Ratain, M.J.; Strom, S.C.; Innocenti, F. Effects of green tea compounds on irinotecan metabolism. Drug Metab. Dispos. 2007, 35, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Tang, J.; Du, Y.; Ding, J.; Liu, J.Y. The green tea polyphenol EGCG potentiates the antiproliferative activity of sunitinib in human cancer cells. Tumor Biol. 2016, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials gov. Available online: https://clinicaltrials.gov/ (accessed on 26 September 2016).
- Jonsson Comprehensive Cancer Center. Available online: https://clinicaltrials.gov/ct2/show/NCT00685516 (accessed on 22 May 2008).
- Henning, S.M.; Wang, P.; Said, J.W.; Huang, M.; Grogan, T.; Elashoff, D.; Carpenter, C.L.; Heber, D.; Aronson, W.J. Randomized clinical trial of brewed green and black tea in men with prostate cancer prior to prostatectomy. Prostate 2015, 75, 550–559. [Google Scholar] [CrossRef] [PubMed]
| Anticancer Drugs | Experiment | Effects | Reference |
|---|---|---|---|
| Bleomycin | SiHa cervical cancer cells or uterine cervical cancer cells were treated with tea polyphenol and bleomycin; poly-caspase activity, early apoptosis, and the expression of caspase-3, caspase-8, caspase-9, Bcl-2, and p53 were assessed. | Synergistic increase in antitumor effects. | [10] |
| 5-Fluorouracil (5-FU) | Some cancer cells—such as human SW480, BIU-87, BGC823, and Hep3B—were treated with green tea and 5-FU; the cytotoxicity, cell apoptosis, and proliferation were studied. | Increase in cell apoptosis; synergistic inhibition of cell proliferation; no reduction in antitumor activity. | [15,35] |
| Cisplatin | Cancer cells YCU-N861, YCU-H891, Hep3B, SW480, BIU-87, BGC823, et al. were coadministered cisplatin with tea polypnenols; the cell apoptosis and proliferation were studied. | Synergistic inhibition of cell proliferation; induction of apoptosis. | [20,21,22] |
| Ibuprofen | DU-145 cells were treated with EGCG and ibuprofen; cell death analysis, immunoblotting, RT-PCR analysis, and caspase activity assay were used. | Synergistic effect on the anti-proliferative and pro-apoptotic action. | [23] |
| Tamoxifen | Cancer cells PC-9, MCF-7, and MDA-MB-231were treated with tea polyphenols and tamoxifen; some factors such as EGFR, MMP-2, MMP-9, and EMMPRIN were assessed. | Induction of apoptosis; enhanced expression of apoptotic genes; synergistic increase in antitumor effects. | [24,25,26] |
| Sulindac | PC-9 cancer cells were treated with sulindac and tea polyphenols; gene expression was assessed. | Induction of apoptosis; enhanced expression of apoptotic genes. | [27,28] |
| Bortezomib | Cancer cells 26S and CWR22 were treated with bortezomib and tea polyphenols; cell apoptosis and proliferation were assessed. | Antagonized antitumor activity. | [29,30,31] |
| Celecoxib | A549 and MCF-7 cancer cells were treated with celecoxib and tea polyphenols; the cell activity and gene expression were assessed. | Increased cell apoptosis; enhanced expression of GADD153 gene | [32] |
| Luteolin | Cancer cells H292, A549, H460, and Tu212 were treated with luteolin and EGCG; phosphorylation of p53 was studied. | Induction of caspase-8 and caspase-3 cleavage; increase in cell apoptosis. | [33] |
| Docetaxel | PC-3ML cancer cells were treated with docetaxel and tea polyphenols; hTERT and Bcl-2 were studied. | Increase in the expression of apoptotic genes; reduction in growth rate of cancer cells. | [34] |
| Curcumin | Cancer cells PC-9, A549, NCI-H460, and ER alpha-breast cancer cells were treated with curcumin and tea polyphenols; the cell activity and cell cycle were assessed. | Induction of apoptosis; enhancement of cell cycle arrest at G1 and S/G2 phases. | [36,37,38] |
| Quercetin | Cancer cells PC-3, LNCaP, and CWR22Rv1 were treated with quercetin and tea polyphenols; the cell growth and gene expression were assessed. | Synergistic expression of androgen receptor; inhibition of cancer cell growth. | [39,40] |
| Paclitaxel | PC-3ML cancer cells were treated with paclitaxel and tea polyphenols; the cell growth and apoptotic gene expression were assessed. | Increase in the expression of apoptotic genes; reduction in growth rate of cancer cells. | [34,41] |
| Doxorubicin | Cancer cells BEL-7404/DOX, PC-3ML, IBC-10a, and PCa-20a were treated with doxorubicin and tea; the cell proliferation and apoptosis were assessed. | Enhanced sensitivity to doxorubicin; synergistic increase in antitumor effects. | [42] |
| Resveratrol | Cancer cells ALVA-41, PC-3, and MCF-7 were treated with resveratrol and green tea; the cell growth and apoptosis were assessed. | Inhibition of cell growth; induction of apoptosis | [19,43] |
| Sulforaphane | Cancer cells PC-3 AP-1, HT-29, SKOV-ip1, SKOVTR-ip2 were treated with sulforaphane and EGCG; the cell activity and gene expression were assessed. | Diminished induction of cancer cell activity; inhibition of cell viability; increase in apoptosis. | [34,44] |
| Anticancer Drugs | Experiment | Effects | Reference |
|---|---|---|---|
| Doxorubicin | Wistar albino rats with cardiotoxicity induced by doxorubicin were treated with green tea. AST, CK, LDH, LPO, cytochrome P450, blood glutathione, tissue glutathione, and enzymatic and non-enzymatic antioxidants were evaluated along with histopathological studies. | Oral administration of green tea prevented doxorubicin-induced cardiotoxicity by accelerating heart antioxidant defense mechanisms and downregulating the LPO levels to the normal levels. | [52] |
| Doxorubicin (DOX) | Neonatal Rats with cardiotoxicity induced by doxorubicin were treated with EGCG; LDH, MnSOD, catalase, and glutathione peroxidase were detected. | EGCG could protect cardiomyocytes from DOX-induced oxidative stress by attenuating ROS production and apoptosis, and increasing activities and protein expression of endogenous antioxidant enzymes. | [53] |
| Doxorubicin | Rats were treated with doxorubicin and different doses of EGCG. Cardiac enzymes (creatine kinase isoenzyme-MB and lactate dehydrogenase) and histopathological changes were studied. | EGCG possesses cardioprotective action against doxorubicin-induced cardiotoxicity by suppressing oxidative stress, inflammation, and apoptotic signals, as well as the activation of pro-survival pathways. | [54] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, J.; Han, J.; Xiao, H.; Qiao, J.; Han, M. Effect of Tea Polyphenol Compounds on Anticancer Drugs in Terms of Anti-Tumor Activity, Toxicology, and Pharmacokinetics. Nutrients 2016, 8, 762. https://doi.org/10.3390/nu8120762
Cao J, Han J, Xiao H, Qiao J, Han M. Effect of Tea Polyphenol Compounds on Anticancer Drugs in Terms of Anti-Tumor Activity, Toxicology, and Pharmacokinetics. Nutrients. 2016; 8(12):762. https://doi.org/10.3390/nu8120762
Chicago/Turabian StyleCao, Jianhua, Jie Han, Hao Xiao, Jinping Qiao, and Mei Han. 2016. "Effect of Tea Polyphenol Compounds on Anticancer Drugs in Terms of Anti-Tumor Activity, Toxicology, and Pharmacokinetics" Nutrients 8, no. 12: 762. https://doi.org/10.3390/nu8120762
APA StyleCao, J., Han, J., Xiao, H., Qiao, J., & Han, M. (2016). Effect of Tea Polyphenol Compounds on Anticancer Drugs in Terms of Anti-Tumor Activity, Toxicology, and Pharmacokinetics. Nutrients, 8(12), 762. https://doi.org/10.3390/nu8120762




