Liver Fatty Acid Composition and Inflammation in Mice Fed with High-Carbohydrate Diet or High-Fat Diet
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Treatments
2.2. Measurements of Diet and Liver Fatty Acid Composition
2.3. Estimation of Enzyme Activities (SCD-1, D6D, Elongase) and DNL in the Liver
2.4. Determination of Nitric Oxide (NO) Production in the Liver
2.5. Determination of Myeloperoxidase Activity in the Liver
2.6. Expressions of the De Novo Lipogenesis Synthesis Enzyme Acetyl-CoA Carboxylase 1 (ACC1) and Inflammatory Genes and Estimation of the Inflammatory Marker Index (IMI) in the Liver
2.7. Statistical Analysis
3. Results
3.1. Fatty Acid Composition of the Livers
3.2. Estimated Activities of SCD-1, D6D, Elongase and DNL in the Liver
3.3. Inflammatory Parameters
3.3.1. Liver Myeloperoxidase Activity and Nitric Oxide Levels
3.3.2. mRNA Expressions of F4/80, Type I Collagen, IL-6, IL-1β, IL-10, and TNF-α in the Liver
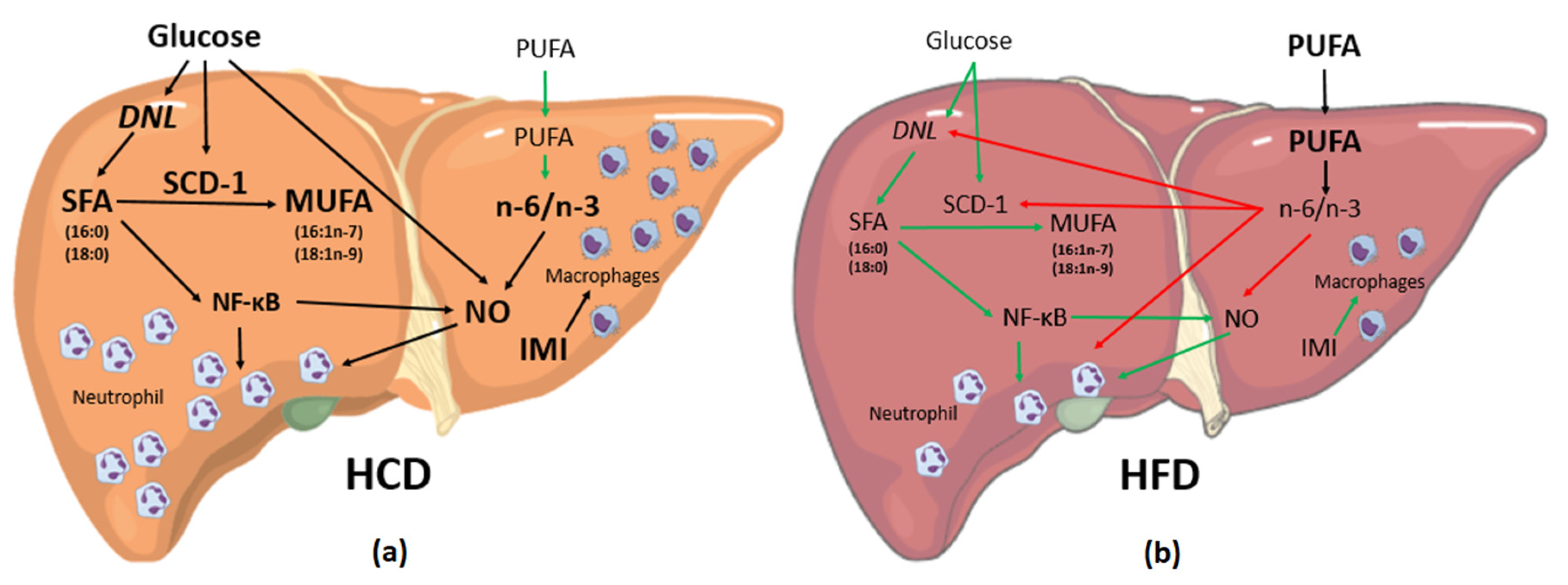
4. Discussion
4.1. Liver FA Accumulation
4.2. Activities of Elongase, Desaturase and SCD-1
4.3. Inflammation Associated with Liver FA Accumulation
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Loomba, R.; Sanyal, A.J. The global NAFLD epidemic. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 686–690. [Google Scholar] [CrossRef] [PubMed]
- Orci, L.A.; Gariani, K.; Oldani, G.; Delaune, V.; Morel, P.; Toso, C. Exercise-based interventions for non-alcoholic fatty liver disease: A meta-analysis and meta-regression. Clin. Gastroenterol. Hepatol. 2016, 14, 1398–1411. [Google Scholar] [CrossRef] [PubMed]
- Yki-Järvinen, H. Nutritional modulation of non-alcoholic fatty liver disease and insulin resistance. Nutrients 2015, 7, 9127–9138. [Google Scholar] [CrossRef] [PubMed]
- Sayiner, M.; Koenig, A.; Henry, L.; Younossi, Z.M. Epidemiology of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis in the United States and the rest of the world. Clin. Liver Dis. 2016, 20, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Bazotte, R.B.; Silva, L.G.; Schiavon, F.P.M. Insulin resistance in the liver: Deficiency or excess of insulin? Cell Cycle 2014, 13, 2494–2500. [Google Scholar] [CrossRef] [PubMed]
- Zelber-Sagi, S.; Salomone, F.; Yeshua, H.; Lotan, R.; Webb, M.; Halpern, Z.; Santo, E.; Oren, R.; Shibolet, O. Non-high-density lipoprotein cholesterol independently predicts new onset of non-alcoholic fatty liver disease. Liver Int. 2014, 34, e128–e135. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Byrne, C.D.; Lonardo, A.; Zoppini, G.; Barbui, C. Nonalcoholic fatty liver disease and risk of incident cardiovascular disease: A meta-analysis of observational studies. J. Hepatol. 2016, 65, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.B.; Gunn, P.J.; Fielding, B.A. The role of dietary sugars and de novo lipogenesis in non-alcoholic fatty liver disease. Nutrients 2014, 6, 5679–5703. [Google Scholar] [CrossRef] [PubMed]
- Araújo, E.P.; Souza, C.T.; Ueno, M.; Cintra, D.E.; Bertolo, M.B.; Carvalheira, J.B.; Saad, M.J.; Velloso, L.A. Infliximab restores glucose homeostasis in an animal model of diet-induced obesity and diabetes. Endocrinology 2007, 148, 5991–5997. [Google Scholar] [CrossRef] [PubMed]
- Fernandes-lima, F.; Luís, T.; Gomes, R. Short exposure to a high-sucrose diet and the first “hit” of nonalcoholic fatty liver disease in mice. Cells Tissues Organs 2016, 201, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Masi, L.N.; Rabello, A.; Roque, A.; Mancini, J.; Lira, C.; Curi, R.; Hirabara, S.M. Inflammatory state of periaortic adipose tissue in mice under obesogenic dietary regimens. J. Nutr. Intermed. Metab. 2016, 6, 1–7. [Google Scholar] [CrossRef]
- West, D.B.; Boozer, C.N.; Moody, D.L.; Atkinson, R.L. Dietary obesity in nine inbred mouse strains. Am. J. Physiol. 1992, 262, R1025–R1032. [Google Scholar] [PubMed]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C. AIN-93 purified diets for laboratory rodents: Final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [PubMed]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Santos, O.O.; Montanher, P.F.; Bonafé, E.G.; Prado, I.N.; Maruyama, S.A.; Matsushita, M.; Visentainer, J.V. A simple, fast and efficient method for transesterification of fatty acids in foods assisted by ultrasound energy. J. Braz. Chem. Soc. 2014, 25, 1712–1719. [Google Scholar] [CrossRef]
- Saleh, T.S.F.; Calixto, J.B.; Medeiros, Y.S. Effects of anti-inflammatory drugs upon nitrate and myeloperoxidase levels in the mouse pleurisy induced by carrageenan. Peptides 1999, 20, 949–956. [Google Scholar] [CrossRef]
- Liu, W.; Saint, D. A new quantitative method of real time reverse transcription polymerase chain reaction assay based on simulation of polymerase chain reaction kinetics. Anal. Biochem. 2002, 302, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Mizukoshi, E.; Sunagozaka, H.; Arai, K.; Yamashita, T.; Takeshita, Y.; Misu, H.; Takamura, T.; Kitamura, S.; Zen, Y.; et al. Characteristics of hepatic fatty acid compositions in patients with nonalcoholic steatohepatitis. Liver Int. 2015, 35, 1773. [Google Scholar] [CrossRef] [PubMed]
- Lambertucci, R.H.; Leandro, C.G.; Vinolo, M.A.; Nachbar, R.T.; Silveira, L.R.; Hirabara, S.M.; Curi, R.; Pithon-Curi, T.C. The effects of palmitic acid on nitric oxide production by rat skeletal muscle: Mechanism via superoxide and iNOS activation. Cell. Physiol. Biochem. 2012, 30, 1169–1180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hodson, L.; Fielding, B.A. Stearoyl-CoA desaturase: Rogue or innocent bystander? Prog. Lipid Res. 2013, 52, 15–42. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Viscarra, J.; Kim, S.-J.; Sul, H.S. Transcriptional regulation of hepatic lipogenesis. Nat. Rev. Mol. Cell Biol. 2015, 16, 678–689. [Google Scholar] [CrossRef] [PubMed]
- Softic, S.; Cohen, D.E.; Kahn, C.R. Role of dietary fructose and hepatic de novo lipogenesis in fatty liver disease. Dig. Dis. Sci. 2016, 61, 1282–1293. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, N.; Zhang, X.; Sugiyama, E.; Kono, H.; Horiuchi, A.; Nakajima, T.; Kanbe, H.; Tanaka, E.; Gonzalez, F.J.; Aoyama, T. Eicosapentaenoic acid improves hepatic steatosis independent of PPAR alpha SREBP-1. Biochem. Pharmacol. 2010, 80, 1601–1612. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. An Increase in the omega-6/omega-3 fatty acid ratio increases the risk for obesity. Nutrients 2016, 8, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, E.J. Lipoproteins, nutrition, and heart disease. Am. J. Clin. Nutr. 2002, 75, 191–212. [Google Scholar] [PubMed]
- Shapiro, H.; Tehilla, M.; Attal-Singer, J.; Bruck, R.; Luzzatti, R.; Singer, P. The therapeutic potential of long-chain omega-3 fatty acids in nonalcoholic fatty liver disease. Clin. Nutr. 2011, 30, 6–19. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y. Tissue-specific, nutritional, and developmental regulation of rat fatty acid elongases. J. Lipid Res. 2005, 46, 706–715. [Google Scholar] [CrossRef] [PubMed]
- Jakobsson, A.; Westerberg, R.; Jacobsson, A. Fatty acid elongases in mammals: Their regulation and roles in metabolism. Prog. Lipid Res. 2006, 45, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Moon, Y.-A.; Ochoa, C.R.; Mitsche, M.A.; Hammer, R.E.; Horton, J.D. Deletion of ELOVL6 blocks the synthesis of oleic acid but does not prevent the development of fatty liver or insulin resistance. J. Lipid Res. 2014, 55, 2597–2605. [Google Scholar] [CrossRef] [PubMed]
- Vessby, B.; Gustafsson, I.-B.B.; Tengblad, S.; Berglund, L. Indices of fatty acid desaturase activity in healthy human subjects: Effects of different types of dietary fat. Br. J. Nutr 2013, 110, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Guillou, H.; Zadravec, D.; Martin, P.G.P.; Jacobsson, A. The key roles of elongases and desaturases in mammalian fatty acid metabolism: Insights from transgenic mice. Prog. Lipid Res. 2010, 49, 186–199. [Google Scholar] [CrossRef] [PubMed]
- Ntambi, J.M.; Miyazaki, M. Recent insights into stearoyl-CoA desaturase-1. Curr. Opin. Lipidol. 2003, 14, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Mauvoisin, D.; Mounier, C. Hormonal and nutritional regulation of SCD1 gene expression. Biochimie 2011, 93, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Sampath, H.; Ntambi, J.M. Polyunsaturated fatty acid regulation of genes of lipid metabolism. Annu. Rev. Nutr. 2005, 25, 317–340. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Li, Z.; Liu, F.; Ellsworth, K.; Dallas-yang, Q.; Wu, M.; Ronan, J.; Esau, C.; Murphy, C.; Szalkowski, D.; et al. Prevention of obesity in mice by antisense oligonucleotide inhibitors of stearoyl-CoA desaturase—1. J. Clin. Investig. 2005, 115, 1030–1038. [Google Scholar] [CrossRef] [PubMed]
- Warensjo, E.; Sundstrom, J.; Vessby, B.; Cederholm, T.; Risérus, U. Markers of dietary fat quality and fatty acid desaturation as predictors of total and cardiovascular mortality: A population-based prospective study. Am. J. Clin. Nutr. 2008, 88, 203–209. [Google Scholar] [PubMed]
- Paillard, F.; Catheline, D.; Le Duff, F.; Bouriel, M.; Deugnier, Y.; Pouchard, M.; Daubert, J.C.; Legrand, P. Plasma palmitoleic acid, a product of stearoyl-coA desaturase activity, is an independent marker of triglyceridemia and abdominal adiposity. Nutr. Metab. Cardiovasc. Dis. 2008, 18, 436–440. [Google Scholar] [CrossRef] [PubMed]
- Warensjö, E.; Risérus, U.; Vessby, B. Fatty acid composition of serum lipids predicts the development of the metabolic syndrome in men. Diabetologia 2005, 48, 1999–2005. [Google Scholar] [CrossRef] [PubMed]
- Risérus, U.; Ärnlöv, J.; Berglund, L. Long-term predictors of insulin resistance. Diabetes Care 2007, 30, 2928–2933. [Google Scholar] [CrossRef] [PubMed]
- Vessby, B.; Gustafsson, I.; Tengblad, S.; Boberg, M.; Andersson, A. Desaturation and elongation of fatty acids and insulin action. Ann. N. Y. Acad. Sci. 2002, 967, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. n-3 Polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am. J. Clin. Nutr. 2006, 83, 1505S–1519S. [Google Scholar] [PubMed]
- Martin, S.A.; Brash, A.R.; Murphy, R.C. The discovery and early structural studies of arachidonic Acid. J. Lipid Res. 2016, 1, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Galland, L. Diet and inflammation. Nutr. Clin. Pract. 2010, 25, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Etherton, T.D.; Martin, K.R.; Vanden Heuvel, J.P.; Gillies, P.J.; West, S.G.; Kris-Etherton, P.M. Anti-inflammatory effects of polyunsaturated fatty acids in THP-1 cells. Biochem. Biophys. Res. Commun. 2005, 336, 909–917. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Ruan, X.Z.; Powis, S.H.; Fernando, R.; Mon, W.Y.; Wheeler, D.C.; Moorhead, J.F.; Varghese, Z. EPA and DHA reduce LPS-induced inflammation responses in HK-2 cells: Evidence for a PPAR-gamma-dependent mechanism. Kidney Int. 2005, 67, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Ajuwon, K.M.; Spurlock, M.E. Palmitate activates the NF-kappa B transcription factor and induces IL-6 and TNF alpha expression in 3T3-L1 adipocytes. J. Nutr. 2005, 135, 1841–1846. [Google Scholar] [PubMed]
- Winterbourn, C.C.; Kettle, A.J. Biomarkers of myeloperoxidase-derived hypochlorous acid. Free Radic. Biol. Med. 2000, 29, 403–409. [Google Scholar] [CrossRef]
- Chen, T.; Zamora, R.; Zuckerbraun, B.; Billiar, T.R. Role of nitric oxide in liver injury. Curr. Mol. Med. 2003, 3, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Lima, T.M.; Lima, L.S.; Scavone, C.; Curi, R. Fatty acid control of nitric oxide production by macrophages. FEBS Lett. 2006, 580, 3287–3295. [Google Scholar] [CrossRef] [PubMed]
- Cacicedo, J.M.; Benjachareowong, S.; Chou, E.; Ruderman, N.B.; Ido, Y. Palmitate-induced apoptosis in cultured bovine retinal pericytes: Roles of NAD(P)H oxidase, oxidant stress, and ceramide. Diabetes 2005, 54, 1838–1845. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-H.; Faunce, D.E.; Stacey, M.; Terajewicz, A.; Nakamura, T.; Zhang-Hoover, J.; Kerley, M.; Mucenski, M.L.; Gordon, S.; Stein-Streilein, J. The macrophage F4/80 receptor is required for the induction of antigen-specific efferent regulatory t cells in peripheral tolerance. J. Exp. Med. 2005, 201, 1615–1625. [Google Scholar] [CrossRef] [PubMed]
- Ceriello, A. Postprandial hyperglycemia and diabetes complications: Is it time to treat? Diabetes 2005, 54, 1–7. [Google Scholar] [CrossRef] [PubMed]
| Fatty Acids | Diets | |
|---|---|---|
| HCD | HFD | |
| Myristic Acid (14:0) | 88.5 ± 0.9 | 386.9 ± 6.4 * |
| Palmitic Acid (16:0) | 1141.8 ± 12.8 | 6530.4 ± 58.6 * |
| Stearic Acid (18:0) | 555.0 ± 7.0 | 3169.1 ± 19.9 * |
| Hipogeic Acid (16:1n-9) | 17.2 ± 0.4 | 104.1 ± 4.9 * |
| Palmitoleic Acid (16:1n-7) | 87.7 ± 0.5 | 520.1 ± 6.3 * |
| Oleic Acid (18:1n-9) | 1793.2 ± 12.1 | 10,678.8 ± 74.9 * |
| Vaccenic Acid (18:1n-7) | 133.1 ± 1.5 | 811.15 ± 22.9 * |
| Linoleic Acid (18:2n-6) | 1155.9 ± 9.7 | 6653.5 ± 38.7 * |
| α-Linolenic Acid (18:3n-3) | 76.8 ± 0.5 | 403.5 ± 19.2 * |
| SFAs | 1785.3 ± 14.6 | 10,086.5 ± 62.2 a |
| MUFAs | 2031.2 ± 12.2 | 12,114.2 ± 78.8 * |
| PUFAs | 1232.8 ± 9.7 | 7057.0 ± 43.2 * |
| Total n-6 PUFA | 1155.9 ± 9.7 | 6653.5 ± 38.7 * |
| Total n-3 PUFA | 76.8 ± 0.5 | 403.47 ± 19.2 * |
| Fatty Acids | 0 (Day) | 7 (Day) | 14 (Day) | 28 (Day) | 56 (Day) | |
|---|---|---|---|---|---|---|
| Myristic Acid (14:0) | HCD | 26.4 ± 2.0 | 31.5 ± 1.1 | 38.8 ± 1.1 a | 26.6 ± 1.4 c | 58.6 ± 4.3 a,b,c,d |
| HFD | 19.8 ± 1.4 a,* | 15.1 ± 0.9 a,b,* | 15.1 ± 0.7 a,b,* | 14.3 ± 0.9 a,b,* | ||
| Palmitic Acid (16:0) | HCD | 1084.9 ± 22 | 1387.5 ± 12.5 a | 1299.0 ± 23.8 a,b | 1063.3 ± 22.9 b,c | 1916.8 ± 29.6 a,b,c,d |
| HFD | 1219.5 ± 10.4 a,* | 1154.1 ± 6.1 a,b,* | 1288.6 ± 19.9 a,b,c,* | 1314.7 ± 0.3 a,b,c,* | ||
| Stearic Acid (18:0) | HCD | 381.0 ± 28.3 | 415.3 ± 3.6 | 400.6 ± 23.2 | 311.9 ± 26.8 a,b,c | 415.4 ± 30.2 d |
| HFD | 503.2 ± 5.5 a,* | 611.1 ± 8.2 a,b,* | 633.3 ± 39.3 a,b,* | 525.8 ± 13.1 a,c,d,* | ||
| Heneicosanoic Acid (21:0) | HCD | 27.3 ± 2.2 | 48.5 ± 2.1 a | 45.6 ± 2.8 a | 44.5 ± 4.3 a | 69.1 ± 1.6 a,b,c,d |
| HFD | 32.4 ± 1.3 a,* | 33.0 ± 1.8 a,* | 44.5 ± 2.5 a,b,c | 61.5 ± 0.9 a,b,c,d* | ||
| Tetracosanoic Acid (24:0) | HCD | 26.2 ± 0.5 | 20.2 ± 0.2 a | 17.2 ± 1.2 a,b | 7.2 ± 0.3 a,b,c | 9.1 ± 0.9 a,b,c,d |
| HFD | 26.8 ± 0.9 * | 28.5 ± 2.0 * | 24.2 ± 0.8 c,* | 20.9 ± 0.6 a,b,c,d,* | ||
| 7-hexadecanoic Acid (16:1n-9) | HCD | 23.8 ± 0.4 | 29.8 ± 0.5 a | 39.7 ± 0.4 a,b | 34.0 ± 2.2 a,b,c | 64.1 ± 0.9 a,b,c,d |
| HFD | 21.9 ± 0.6 * | 18.1 ± 1.1 a,b,* | 17.1 ± 1.0 a,b,* | 32.3 ± 1.7 a,b,c,d,* | ||
| Palmitoleic Acid (16:1n-7) | HCD | 82.6 ± 3.0 | 191.0 ± 7.8 a | 250.3 ± 13.0 a,b | 209.1 ± 19.4 a | 524.3 ± 25.0 a,b,c,d |
| HFD | 57.0 ± 3.7 a,* | 43.8 ± 0.8 a,b,* | 46.2 ± 1.4 a,b,* | 53.0 ± 2.3 a,c,d,* | ||
| Oleic Acid (18:1n-9) | HCD | 710.7 ± 46.4 | 1294.1 ± 16.9 a | 1613.4 ± 25.9 a,b | 1479.0 ± 104.8 a | 3077.9 ± 125.4 a,b,c,d |
| HFD | 888.0 ± 7.8 a,* | 852.6 ± 20.9 a,* | 937.3 ± 41.9 a,* | 1152.7 ± 46.4 a,b,c,d,* | ||
| Vaccenic Acid (18:1n-7) | HCD | 76.6 ± 3.7 | 133.5 ± 4.4 a | 159.9 ± 3.1 a | 212.4 ± 19.2 a,b,c | 488.7 ± 13.4 a,b,c,d |
| HFD | 88.2 ± 1.2 a,* | 87.7 ± 2.8 a,* | 97.6 ± 1.2 a,b,c,* | 114.0 ± 2.3 a,b,c,d,* | ||
| Linoleic Acid (18:2n-6) | HCD | 1472.8 ± 21.9 | 1125.3 ± 36.0 a | 1040.2 ± 12.9 a | 621.3 ± 52.7 a,b,c,d | 685.0 ± 24.3 a,b,c,d |
| HFD | 1307.0 ± 19.0 a,* | 1274.9 ± 29.1 a,* | 1176.0 ± 37.7 a,b,c,* | 1066.1 ± 4.4 a,b,c,d,* | ||
| γ-linolenic Acid (18:3n-6) | HCD | 51.6 ± 0.6 | 48.6 ± 1.4 | 43.7 ± 2.9 a,b | 27.0 ± 1.7 a,b,c | 41.1 ± 1.4 a,b,d |
| HFD | 36.3 ± 2.2 a,* | 40.0 ± 1.9 a | 25.0 ± 1.5 a,b,c | 20.3 ± 0.6 a,b,c,d,* | ||
| Arachidonic Acid (20:4n-6) | HCD | 453.4 ± 31.0 | 541.4 ± 10.9 | 508.6 ± 19.7 | 443.8 ± 44.2 b | 606.1 ± 40.7 a,d |
| HFD | 585.5 ± 1.0 a,* | 679.7 ± 4.2 a,b,* | 778.0 ± 28.6 a,b,c,* | 700.5 ± 24.7 a,b,d,* | ||
| Docosatetraenoic Acid (22:4n-6) | HCD | 18.5 ± 1.5 | 20.7 ± 0.6 | 19.7 ± 1.3 | 10.9 ± 0.7 a,b,c | 16.1 ± 1.6 b,d |
| HFD | 24.1 ± 0.6 a,* | 27.3 ± 1.7 a,b,* | 26.8 ± 0.5 a,* | 27.5 ± 1.1 a,b,* | ||
| Docosapentaenoic Acid (22:5n-6) | HCD | 17.8 ± 1.4 | 41.2 ± 1.1 a | 47.2 ± 4.5 a | 31.2 ± 3.0 a,b,c | 65.5 ± 4.4 a,b,c,d |
| HFD | 29.0 ± 1.2 a,* | 37.7 ± 3.0 a,b | 31.6 ± 1.2 a,c | 30.5 ± 1.0 a,c,* | ||
| α-Linolenic Acid (18:3n-3) | HCD | 60.1 ± 4.4 | 29.5 ± 0.8 a | 24.2 ± 0.4 a | 7.9 ± 0.4 a,b,c | 9.8 ± 0.4 a,b,c |
| HFD | 35.8 ± 0.9 a,* | 26.6 ± 0.6 a,b,* | 20.1 ± 0.9 a,b,c,* | 18.1 ± 0.2 a,b,c,* | ||
| Eicosapentaenoic Acid (20:5n-3) | HCD | 8.1 ± 0.5 | 6.5 ± 0.2 a | 5.0 ± 0.4 a,b | 4.1 ± 0.2 a,b | 3.5 ± 0.1 a,b,c |
| HFD | 8.2 ± 0.2 * | 9.2 ± 0.6 a,* | 7.1 ± 0.3 b,c,* | 6.7 ± 0.1 a,b,c,* | ||
| Docosahexaenoic Acid (22:6n-3) | HCD | 346.4 ± 25.7 | 394.9 ± 9.7 a | 281.1 ± 11.8 a,b | 213.4 ± 17.7 a,b | 244.7 ± 12.4 a,b,c |
| HFD | 460.3 ± 7.6 a,* | 490.9 ± 9.9 a,* | 547.8 ± 24.7 a,b,c,* | 464.7 ± 15.3 a,d,* |
| Fatty Acids | 0 (Day) | 7 (Day) | 14 (Day) | 28 (Day) | 56 (Day) | |
|---|---|---|---|---|---|---|
| SFA | HCD | 1545.9 ± 35.7 | 1903.0 ± 13.3 a | 1801.3 ± 33.4 a,b | 1453.6 ± 35.6 a,b,c | 2469.0 ± 42.5 a,b,c,d |
| HFD | 1801.7 ± 12.0 a,* | 1841.9 ± 10.6 a,* | 2005.7 ± 44.1 a,b,c,* | 1937.2 ± 13.2 a,b,c,d,* | ||
| MUFA | HCD | 893.7 ± 46.6 | 1648.3 ± 19.1 a | 2063.4 ± 29.1 a,b | 1934.6 ± 108.3 a,b | 4155.0 ± 128.6 a,b,c,d |
| HFD | 1055.0 ± 8.7 a,* | 1002.2 ± 21.1 * | 1098.2 ± 42.0 a,* | 1351.9 ± 46.5 a,b,c,d,* | ||
| PUFA | HCD | 2428.8 ± 46.1 | 2208.2 ± 38.9 a | 1969.7 ± 26.9 a,b | 1359.7 ± 71.2 a,b,c | 1671.8 ± 49.2 a,b,c,d |
| HFD | 2486.3 ± 20.7 * | 2586.5 ± 31.3 a,b,* | 2612.4 ± 53.5 a,b,* | 2334.4 ± 29.4 a,b,c,d,* | ||
| n-6 | HCD | 2014.2 ± 38.0 | 1777.3 ± 37.7 a | 1659.5 ± 24.2 a | 1134.2 ± 68.9 a,b,c | 1413.7 ± 47.6 a,b,c,d |
| HFD | 1981.9 ± 19.2 * | 2059.7 ± 29.6 b,* | 2037.5 ± 47.4 * | 1844.9 ± 25.1 a,b,c,d,* | ||
| n-3 | HCD | 414.6 ± 26.1 | 430.9 ± 9.7 | 310.2 ± 11.8 a,b | 225.5 ± 17.8 a,b,c | 258.0 ± 12.3 a,b,c |
| HFD | 504.4 ± 7.7 a,* | 526.8 ± 10.0 a,* | 574.9 ± 24.7 a,b,c,* | 489.5 ± 15.3 a,d,* | ||
| PUFA/SFA | HCD | 1.6 ± 0.03 | 1.2 ± 0.02 a | 1.1 ± 0.02 a | 0.9 ± 0.06 a,b,c | 0.7 ± 0.03 a,b,c,d |
| HFD | 1.4 ± 0.01 a,* | 1.4 ± 0.01 * | 1.3 ± 0.03 a,b,c,* | 1.2 ± 0.01 a,b,c,d,* | ||
| MUFA/SFA | HCD | 0.6 ± 0.05 | 0.9 ± 0.02 a | 1.1 ± 0.02 a,b | 1.3 ± 0.06 a,b,c | 1.7 ± 0.04 a,b,c,d |
| HFD | 0.6 ± 0.01 * | 0.5 ± 0.02 * | 0.5 ± 0.04 * | 0.7 ± 0.04 a,b,c,d,* | ||
| n-6/n-3 | HCD | 4.8 ± 0.07 | 4.1 ± 0.03 a | 5.3 ± 0.04 b | 5.0 ± 0.10 b | 5.5 ± 0.06 a,b |
| HFD | 3.9 ± 0.02 a,* | 3.9 ± 0.02 a,* | 3.5 ± 0.05 a,* | 3.8 ± 0.03 a,* | ||
| SUM | HCD | 4868.4 | 5759.6 | 5834.3 | 4747.8 | 8295.8 |
| HFD | 5343.0 | 5430.5 | 5716.3 | 5623.6 |
| 0 (Day) | 7 (Day) | 14 (Day) | 28 (Day) | 56 (Day) | ||
|---|---|---|---|---|---|---|
| SCD-1 (16:1n-7/16:0) | HCD | 0.076 ± 0.003 | 0.138 ± 0.004 a | 0.193 ± 0.006 a,b | 0.197 ± 0.021 a,b | 0.274 ± 0.016 a,b,c,d |
| HFD | 0.047 ± 0.003 a,* | 0.038 ± 0.001 a,b,* | 0.036 ± 0.0005 a,b,* | 0.040 ± 0.002 a,b,* | ||
| SCD-1 (18:1n-9/18:0) | HCD | 1.877 ± 0.252 | 3.116 ± 0.051 a | 4.036 ± 0.298 a | 4.786 ± 0.763 a,b | 7.449 ± 0.824 a,b,c,d |
| HFD | 1.765 ± 0.015 * | 1.395 ± 0.043 * | 1.48 ± 0.159 * | 2.195 ± 0.141 * | ||
| D6D (18:3n-6/18:2n-6) | HCD | 0.035 ± 0.001 | 0.043 ± 0.0002 a | 0.042 ± 0.002 a | 0.044 ± 0.002 a | 0.060 ± 0.001 a,b,c,d |
| HFD | 0.028 ± 0.001 a,* | 0.031 ± 0.001 a,b,* | 0.021 ± 0.0007 a,b,c,* | 0.019 ± 0.001 a,b,c,* | ||
| Elongase (18:0/16:0) | HCD | 0.352 ± 0.031 | 0.299 ± 0.002 | 0.309 ± 0.024 | 0.294 ± 0.031 | 0.217 ± 0.012 a,b,c,d |
| HFD | 0.413 ± 0.001 a,* | 0.529 ± 0.007 a,b,* | 0.492 ± 0.031 a,b,* | 0.400 ± 0.010 c,d,* | ||
| DNL (16:0/18:2n-6) | HCD | 0.74 ± 0.06 | 1.23 ± 0.03 a | 1.25 ± 0.32 a | 1.72 ± 0.09 a,b,c | 2.80 ± 0.06 a,b,c,d |
| HFD | 0.93 ± 0.02 a,* | 0.91 ± 0.02 a,* | 1.10 ± 0.05 a,b,c,* | 1.23 ± 0.03 a,b,c,d,* |
| HCD | HFD | |
|---|---|---|
| MPO activity (DO 460 nm) | 0.450 ± 0.106 | 0.302 ± 0.073 * |
| NO (Total Nitrite μM) | 190.60 ± 54.55 | 112.11 ± 51.15 * |
| Gene | HCD | HFD |
|---|---|---|
| F4/80 | 1.02 ± 0.2 | 1.01 ± 0.4 |
| Type I collagen | 1.02 ± 0.2 | 0.87 ± 0.1 |
| IL-6 | 1.24 ± 0.9 | 1.40 ± 0.7 |
| IL-1β | 1.10 ± 0.5 | 0.91 ± 0.3 |
| TNF-α | 1.20 ± 0.7 | 1.20 ± 0.6 |
| IL-10 | 1.10 ± 0.6 | 1.80 ± 0.7 |
| Inflammatory marker index (IMI) | 25.06 ± 0.5 | 17.87 ± 0.5 * |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da Silva-Santi, L.G.; Antunes, M.M.; Caparroz-Assef, S.M.; Carbonera, F.; Masi, L.N.; Curi, R.; Visentainer, J.V.; Bazotte, R.B. Liver Fatty Acid Composition and Inflammation in Mice Fed with High-Carbohydrate Diet or High-Fat Diet. Nutrients 2016, 8, 682. https://doi.org/10.3390/nu8110682
Da Silva-Santi LG, Antunes MM, Caparroz-Assef SM, Carbonera F, Masi LN, Curi R, Visentainer JV, Bazotte RB. Liver Fatty Acid Composition and Inflammation in Mice Fed with High-Carbohydrate Diet or High-Fat Diet. Nutrients. 2016; 8(11):682. https://doi.org/10.3390/nu8110682
Chicago/Turabian StyleDa Silva-Santi, Lorena Gimenez, Marina Masetto Antunes, Silvana Martins Caparroz-Assef, Fabiana Carbonera, Laureane Nunes Masi, Rui Curi, Jesuí Vergílio Visentainer, and Roberto Barbosa Bazotte. 2016. "Liver Fatty Acid Composition and Inflammation in Mice Fed with High-Carbohydrate Diet or High-Fat Diet" Nutrients 8, no. 11: 682. https://doi.org/10.3390/nu8110682
APA StyleDa Silva-Santi, L. G., Antunes, M. M., Caparroz-Assef, S. M., Carbonera, F., Masi, L. N., Curi, R., Visentainer, J. V., & Bazotte, R. B. (2016). Liver Fatty Acid Composition and Inflammation in Mice Fed with High-Carbohydrate Diet or High-Fat Diet. Nutrients, 8(11), 682. https://doi.org/10.3390/nu8110682





