Probiotic Streptococcus thermophilus FP4 and Bifidobacterium breve BR03 Supplementation Attenuates Performance and Range-of-Motion Decrements Following Muscle Damaging Exercise
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Experimental Protocol
2.3. Upper Arm Circumference
2.4. Range of Motion
2.5. Soreness
2.6. Isometric Peak
2.7. Eccentric Exercise Protocol
2.8. Blood Analyses
2.9. Statistical Analysis
2.9.1. Data Presentation and Transformation
2.9.2. Statistical Inference
3. Results
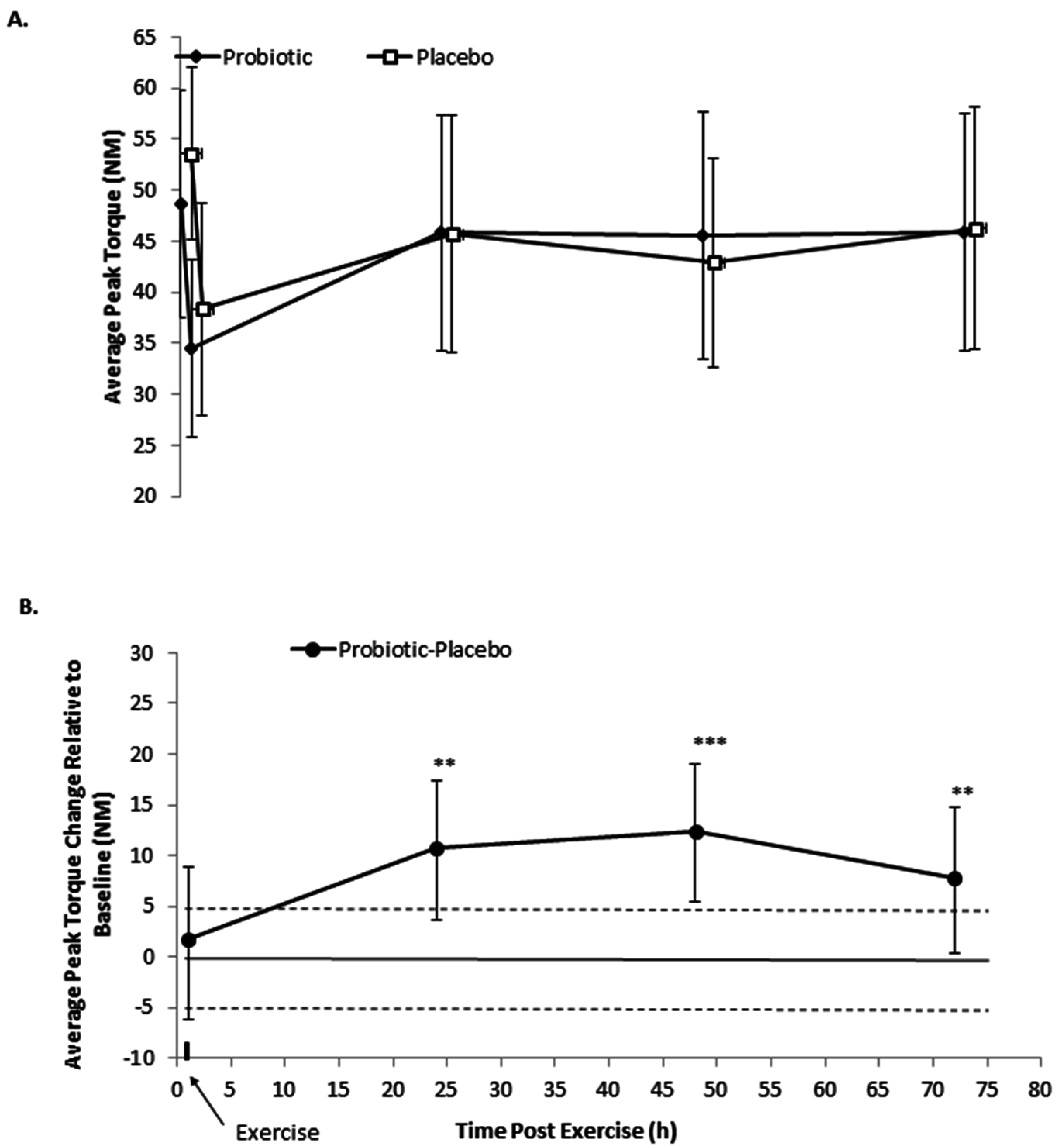
3.1. Isometric Peak Torque
3.2. Soreness, Range of Motion, and Upper Arm Circumference
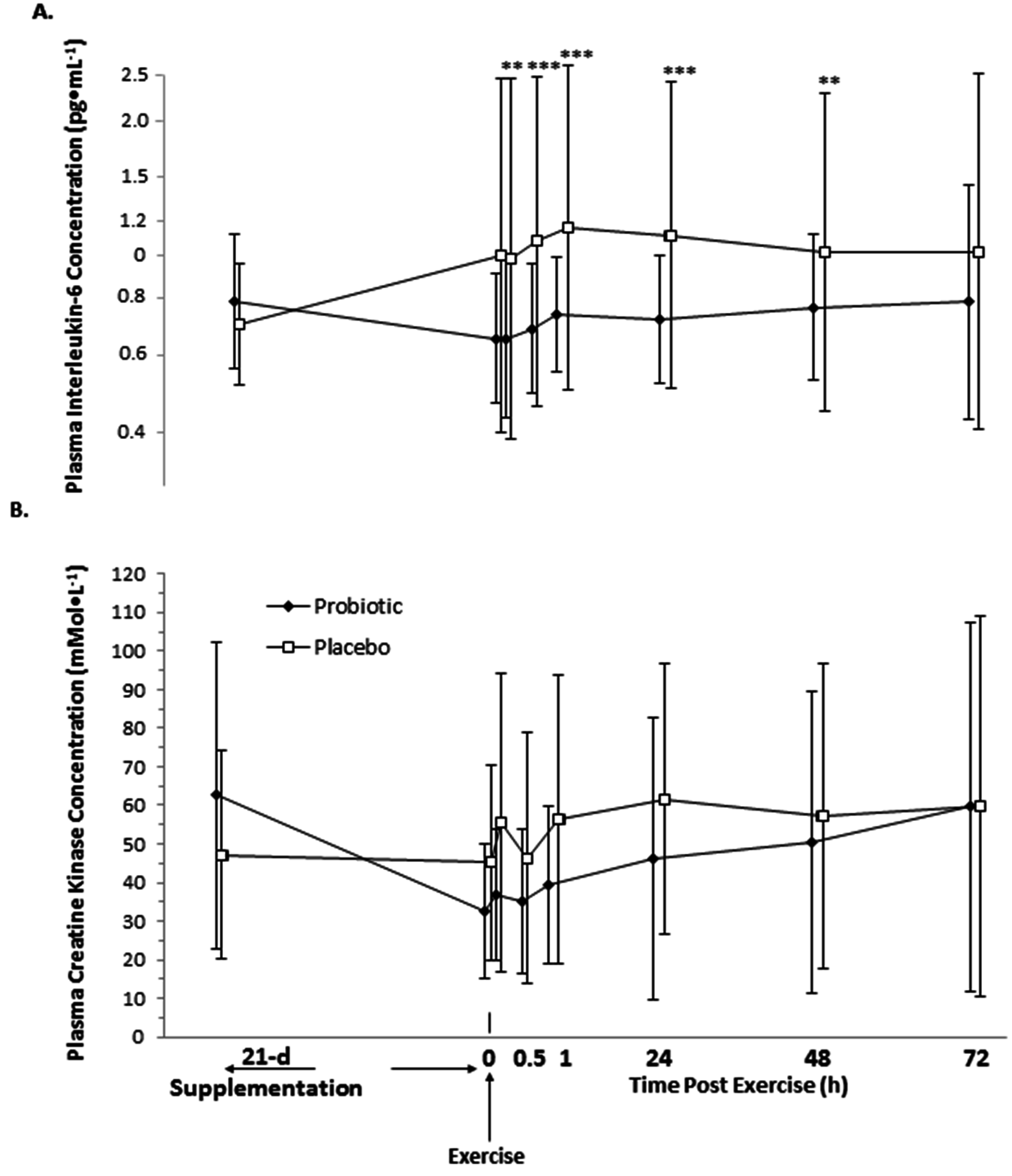
3.3. Interleukin-6 and Creatine Kinase
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Smits, H.H.; Engering, A.; van der Kleij, D.; de Jong, E.C.; Schipper, K.; van Capel, T.M.; Zaat, B.A.; Yazdanbakhsh, M.; Wierenga, E.A.; van Kooyk, Y.; et al. Selective probiotic bacteria induce IL-10-producing regulatory T cells in vitro by modulating dendritic cell function through dendritic cell-specific intercellular adhesion molecule 3-grabbing nonintegrin. J. Allergy Clin. Immunol. 2005, 115, 1260–1267. [Google Scholar] [CrossRef] [PubMed]
- Loguercio, C.; Federico, A.; Tuccillo, C.; Terracciano, F.; D’Auria, M.V.; De Simone, C.; Del Vecchio Blanco, C. Beneficial effects of a probiotic VSL#3 on parameters of liver dysfunction in chronic liver diseases. J. Clin. Gastroenterol. 2005, 39, 540–543. [Google Scholar] [PubMed]
- Bruunsgaard, H.; Galbo, H.; Halkjaer-Kristensen, J.; Johansen, T.L.; MacLean, D.A.; Pedersen, B.K. Exercise-induced increase in serum interleukin-6 in humans is related to muscle damage. J. Physiol. 1997, 499, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, M.; Bishop, N.; Oliveira, M.; McCauley, T.; Tauler, P.; Lawrence, C. Effects of a lactobacillus salivarius probiotic intervention on infection, cold symptom duration and severity, and mucosal immunity in endurance athletes. Int. J. Sport Nutr. Exerc. 2012, 22, 235–242. [Google Scholar] [CrossRef]
- Pyne, D.B.; West, N.P.; Cox, A.J.; Cripps, A.W. Probiotics supplementation for athletes-clinical and physiological effects. Eur. J. Sport Sci. 2015, 15, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Jäger, R.; Shields, K.A.; Lowery, R.P.; De Souza, E.O.; Partl, J.M.; Hollmer, C.; Purpura, M.; Wilson, J.M. Probiotic bacillus coagulans GBI-30, 6086 reduces exercise-induced muscle damage and increases recovery. PeerJ 2016, 4, e2276. [Google Scholar] [CrossRef] [PubMed]
- Nosaka, K.; Sakamoto, K. Effect of elbow joint angle on the magnitude of muscle damage to the elbow flexors. Med. Sci. Sports Exerc. 2001, 33, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Proske, U.; Morgan, D.L. Muscle damage from eccentric exercise: Mechanism, mechanical signs, adaptation and clinical applications. J. Physiol. 2001, 537, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, G.; Mikkelsen, U.R.; Raastad, T.; Peake, J.M. Leucocytes, cytokines and satellite cells: What role do they play in muscle damage and regeneration following eccentric exercise. Exerc. Immunol. Rev. 2012, 18, 42–97. [Google Scholar] [PubMed]
- Tidball, J.G.; Villalta, S.A. Regulatory interactions between muscle and the immune system during muscle regeneration. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R1173–R1187. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.J. A framework for understanding the training process leading to elite performance. Sports Med. 2003, 33, 1103–1126. [Google Scholar] [CrossRef] [PubMed]
- Klemenak, M.; Dolinsek, J.; Langerholc, T.; Di Gioia, D.; Micetic-Turk, D. Administration of Bifidobacterium breve decreases the production of TNF-alpha in children with celiac disease. Dig. Dis. Sci. 2015, 60, 3386–3392. [Google Scholar] [CrossRef] [PubMed]
- Nicola, S.; Mogna, L.; Allesina, S.; Barba, M.; Deidda, F.; Lorenzini, P.; Raiteri, E.; Strozzi, G.P.; Mogna, G. Interaction between probiotics and human immune cells. The prospective anti-inflammatory activity of Bifidobacterium breve BRO3. Agro Food Ind. Hi-Tech 2010, 21, 9–13. [Google Scholar]
- Saggioro, A. Probiotics in the treatment of irritable bowel syndrome. J. Clin. Gastroenterol. 2004, 38, S104–S106. [Google Scholar] [CrossRef] [PubMed]
- Correa, N.B.; Peret Filho, L.A.; Penna, F.J.; Lima, F.M.; Nicoli, J.R. A randomized formula controlled trial of Bifidobacterium lactis and Streptococcus thermophilus for prevention of antibiotic-associated diarrhea in infants. J. Clin. Gastroenterol. 2005, 39, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Del Piano, M.; Carmagnola, S.; Andorno, S.; Pagliarulo, M.; Tari, R.; Mogna, L.; Strozzi, G.P.; Sforza, F.; Capurso, L. Evaluation of the intestinal colonization by microencapsulated probiotic bacteria in comparison with the same uncoated strains. J. Clin. Gastroenterol. 2010, 44, S42–S46. [Google Scholar] [CrossRef] [PubMed]
- Saavedra, J.M.; Bauman, N.A.; Oung, I.; Perman, J.A.; Yolken, R.H. Feeding of Bifidobacterium bifidum and Streptococcus thermophilus to infants in hospital for prevention of diarrhoea and shedding of rotavirus. Lancet 1994, 344, 1046–1049. [Google Scholar] [CrossRef]
- Thibault, H.; Aubert-Jacquin, C.; Goulet, O. Effects of long-term consumption of a fermented infant formula (with Bifidobacterium breve c50 and Streptococcus thermophilus 065) on acute diarrhea in healthy infants. J. Pediatr. Gastroenterol. Nutr. 2004, 39, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, P.M.; Hubal, M.J. Exercise-induced muscle damage in humans. Am. J. Phys. Med. Rehabil. 2002, 81, S52–S69. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Gonzalo, R.; Lundberg, T.R.; Alvarez-Alvarez, L.; de Paz, J.A. Muscle damage responses and adaptations to eccentric-overload resistance exercise in men and women. Eur. J. Appl. Physiol. 2014, 114, 1075–1084. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, N.; Eston, R.; Marginson, V.; McHugh, M. Effects of prior concentric training on eccentric exercise induced muscle damage. Br. J. Sports Med. 2003, 37, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Chapman, D.; Newton, M.; Sacco, P.; Nosaka, K. Greater muscle damage induced by fast versus slow velocity eccentric exercise. Int. J. Sports Med. 2006, 27, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Hirose, L.; Nosaka, K.; Newton, M.; Laveder, A.; Kano, M.; Peake, J.; Suzuki, K. Changes in inflammatory mediators following eccentric exercise of the elbow flexors. Exerc. Immunol. Rev. 2004, 10, 20. [Google Scholar]
- Hopkins, W.; Marshall, S.; Batterham, A.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. Sports Exerc. 2009, 41, 3. [Google Scholar] [CrossRef] [PubMed]
- Rowlands, D.S.; Nelson, A.R.; Phillips, S.M.; Faulkner, J.A.; Clarke, J.; Burd, N.A.; Moore, D.; Stellingwerff, T. Protein–leucine fed dose effects on muscle protein synthesis after endurance exercise. Med. Sci. Sports Exerc. 2014, 47, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Rowlands, D.S.; Rossler, K.; Thorp, R.M.; Graham, D.F.; Timmons, B.W.; Stannard, S.R.; Tarnopolsky, M.A. Effect of dietary protein content during recovery from high-intensity cycling on subsequent performance and markers of stress, inflammation, and muscle damage in well-trained men. Appl. Physiol. Nutr. Metab. 2008, 33, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Nosaka, K.; Lavender, A.; Newton, M.; Sacco, P. Muscle damage in resistance training. Int. J. Sport Health Sci. 2003, 1, 1–8. [Google Scholar] [CrossRef]
- Neurath, M.F.; Finotto, S. IL-6 signaling in autoimmunity, chronic inflammation and inflammation-associated cancer. Cytokine Growth Factor Rev. 2011, 22, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Warren, G.L.; Lowe, D.A.; Armstrong, R.B. Measurement tools used in the study of eccentric contraction-induced injury. Sports Med. 1999, 27, 43–59. [Google Scholar] [CrossRef] [PubMed]
- Kiesel, K.; Plisky, P.J.; Voight, M.L. Can serious injury in professional football be predicted by a preseason functional movement screen? N. Am. J. Sports Phys. Ther. 2007, 2, 147–158. [Google Scholar] [PubMed]
- Chapman, R.F.; Laymon, A.S.; Arnold, T. Functional movement scores and longitudinal performance outcomes in elite track and field athletes. Int. J. Sports Physiol. Perform. 2014, 9, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.K.; Cochrane, T. Mobility impairment, muscle imbalance, muscle weakness, scapular asymmetry and shoulder injury in elite volleyball athletes. J. Sports Med. Phys. Fitness 2001, 41, 403–410. [Google Scholar] [PubMed]
- Hirano, T. Interleukin 6 and its receptor: Ten years later. Int. Rev. Immunol. 1998, 16, 249–284. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.; Anwar, A.; Fragen, M.; Rananto, C.; Johnson, R.; Holbert, D. Cytokines and cell adhesion molecules associated with high-intensity eccentric exercise. Eur. J. Appl. Physiol. 2000, 82, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Willoughby, D.; McFarlin, B.; Bois, C. Interleukin-6 expression after repeated bouts of eccentric exercise. Int. J. Sports Med. 2003, 24, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Fischer, C.P. Interleukin-6 in acute exercise and training: What is the biological relevance. Exerc. Immunol. Rev. 2006, 12, 41. [Google Scholar]
- McKay, B.R.; De Lisio, M.; Johnston, A.P.; O’Reilly, C.E.; Phillips, S.M.; Tarnopolsky, M.A.; Parise, G. Association of interleukin-6 signalling with the muscle stem cell response following muscle-lengthening contractions in humans. PLoS ONE 2009, 4, e6027. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.J.; Churchward-Venne, T.A.; Bellamy, L.; Parise, G.; Baker, S.K.; Phillips, S.M. Muscular and systemic correlates of resistance training-induced muscle hypertrophy. PLoS ONE 2013, 8, e78636. [Google Scholar] [CrossRef] [PubMed]

| Contrast | Probiotic–Placebo Effect (%) a | 90% Confidence Limits (%) | Threshold Smallest Standardized Change | Qualitative Inference b | p-Value | |
|---|---|---|---|---|---|---|
| Upper | Lower | |||||
| Range of Motion | ||||||
| Relaxed arm angle | ||||||
| Pre-exercise | −1.0 | 0.4 | −2.4 | 0.6 | Small decrease likely | 0.257 |
| Post–Pre | 0.4 | 2.4 | −1.6 | 0.6 | Unclear | 0.718 |
| 24 h Pre | 2.4 | 4.4 | 0.4 | 0.6 | Moderate increase likely | 0.053 |
| 48 h Pre | 1.9 | 3.9 | −0.1 | 0.6 | Moderate increase likely | 0.116 |
| 72 h Pre | 0.1 | 2.1 | −2 | 0.6 | Unclear | 0.952 |
| Flexed arm angle | ||||||
| Pre-exercise | 2.2 | 8.3 | −4.3 | 2.1 | Unclear | 0.571 |
| Post–Pre | −0.3 | 8.4 | −9.9 | 2.1 | Unclear | 0.952 |
| 24 h Pre | −3.3 | 5.9 | −13.4 | 2.1 | Unclear | 0.565 |
| 48 h Pre | −1.6 | 7.5 | −11.5 | 2.1 | Unclear | 0.783 |
| 72 h Pre | −5.6 | 3.7 | −15.8 | 2.1 | Unclear | 0.329 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jäger, R.; Purpura, M.; Stone, J.D.; Turner, S.M.; Anzalone, A.J.; Eimerbrink, M.J.; Pane, M.; Amoruso, A.; Rowlands, D.S.; Oliver, J.M. Probiotic Streptococcus thermophilus FP4 and Bifidobacterium breve BR03 Supplementation Attenuates Performance and Range-of-Motion Decrements Following Muscle Damaging Exercise. Nutrients 2016, 8, 642. https://doi.org/10.3390/nu8100642
Jäger R, Purpura M, Stone JD, Turner SM, Anzalone AJ, Eimerbrink MJ, Pane M, Amoruso A, Rowlands DS, Oliver JM. Probiotic Streptococcus thermophilus FP4 and Bifidobacterium breve BR03 Supplementation Attenuates Performance and Range-of-Motion Decrements Following Muscle Damaging Exercise. Nutrients. 2016; 8(10):642. https://doi.org/10.3390/nu8100642
Chicago/Turabian StyleJäger, Ralf, Martin Purpura, Jason D. Stone, Stephanie M. Turner, Anthony J. Anzalone, Micah J. Eimerbrink, Marco Pane, Angela Amoruso, David S. Rowlands, and Jonathan M. Oliver. 2016. "Probiotic Streptococcus thermophilus FP4 and Bifidobacterium breve BR03 Supplementation Attenuates Performance and Range-of-Motion Decrements Following Muscle Damaging Exercise" Nutrients 8, no. 10: 642. https://doi.org/10.3390/nu8100642
APA StyleJäger, R., Purpura, M., Stone, J. D., Turner, S. M., Anzalone, A. J., Eimerbrink, M. J., Pane, M., Amoruso, A., Rowlands, D. S., & Oliver, J. M. (2016). Probiotic Streptococcus thermophilus FP4 and Bifidobacterium breve BR03 Supplementation Attenuates Performance and Range-of-Motion Decrements Following Muscle Damaging Exercise. Nutrients, 8(10), 642. https://doi.org/10.3390/nu8100642





