High Fat Diet Administration during Specific Periods of Pregnancy Alters Maternal Fatty Acid Profiles in the Near-Term Rat
Abstract
:1. Introduction
2. Experimental Section
2.1. Experimental Design
| Groups | Gestational Diet | ||
|---|---|---|---|
| Week 1 | Week 2 | Week 3 | |
| Control | Control | Control | Control |
| HF1 | HFD | Control | Control |
| HF2 | Control | HFD | Control |
| HF3 | Control | Control | HFD |
| HFG | HFD | HFD | HFD |
2.2. Sample Collection
2.3. Determination of Blood Glucose and Serum Insulin Concentrations and Lipid Profiles
2.4. Statistical Analysis
3. Results
3.1. Anthropometry
3.2. Glycemia, Insulinemia and HOMA-Insulin Resistance
3.3. Fatty Acid Profile in the Diets

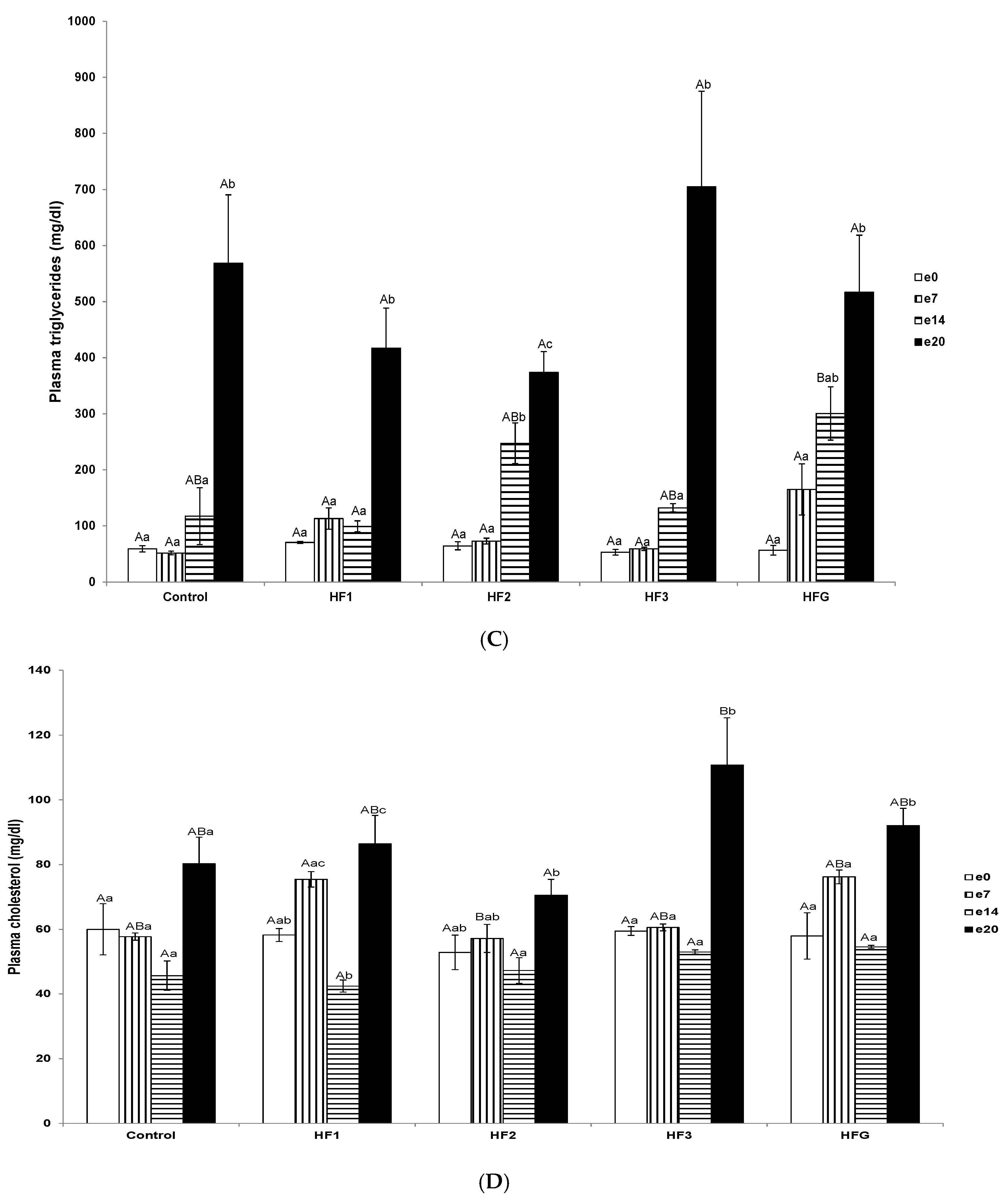
3.4. Plasma Total Non-Esterified Fatty Acid, Triglyceride, and Cholesterol Concentrations and Individual Fatty Acid Profiles

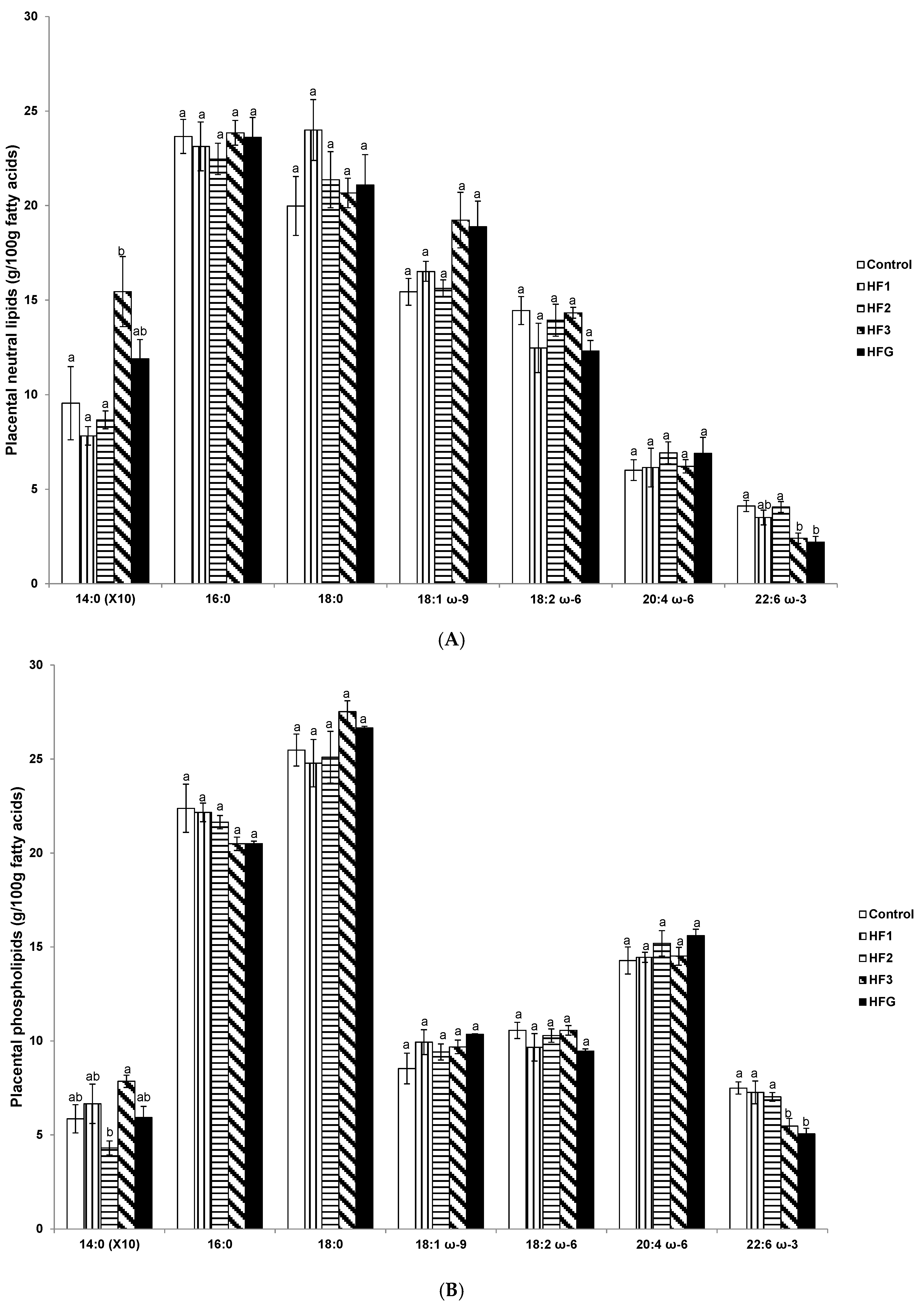
3.5. Placental Neutral Lipids and Phospholipid Fatty Acids Near Term (e20)
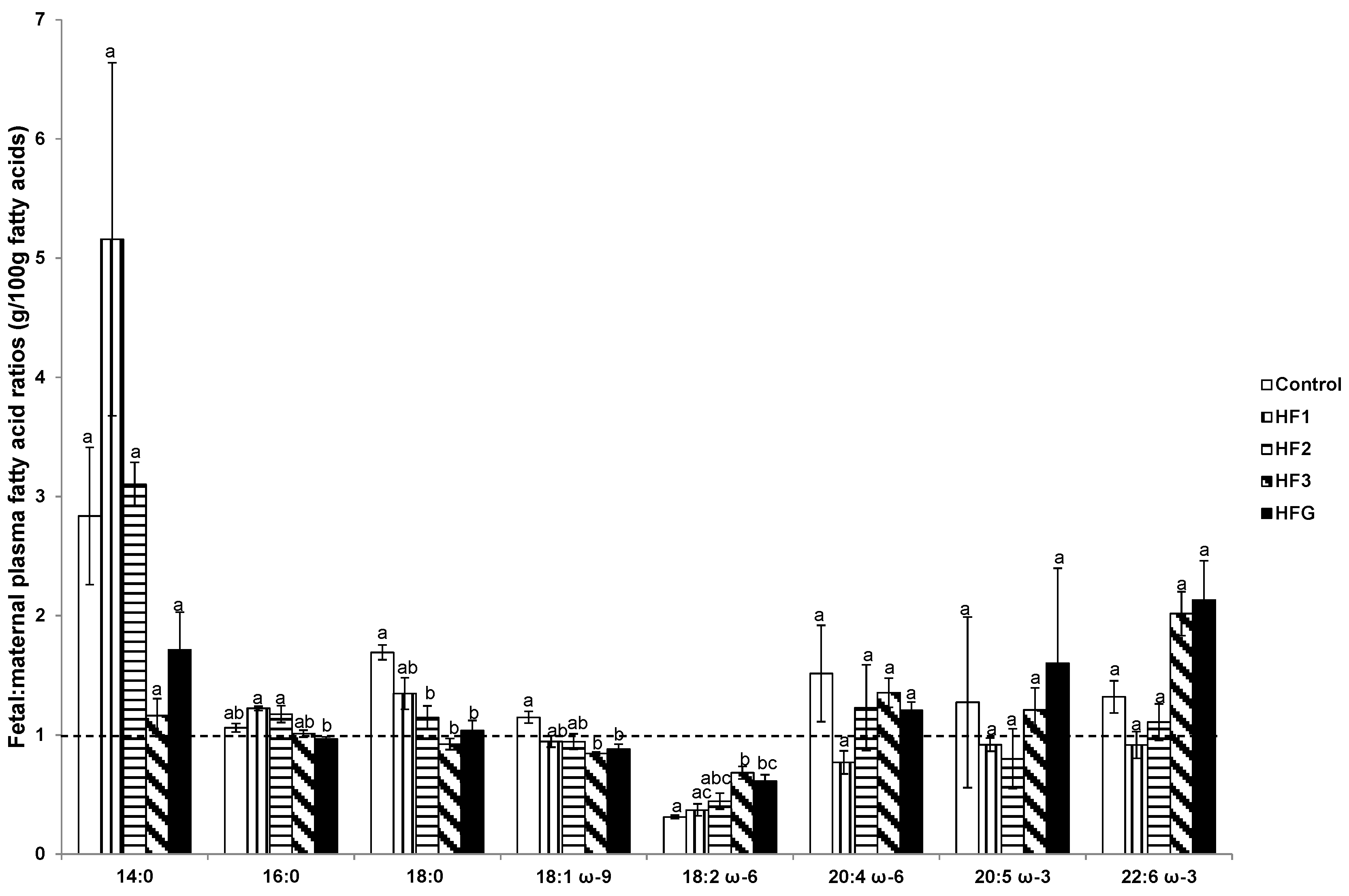
3.6. Plasma Fetal: Maternal Fatty Acids and Fetal and Maternal AA:LA Ratios
3.7. Hepatic Fatty Acids
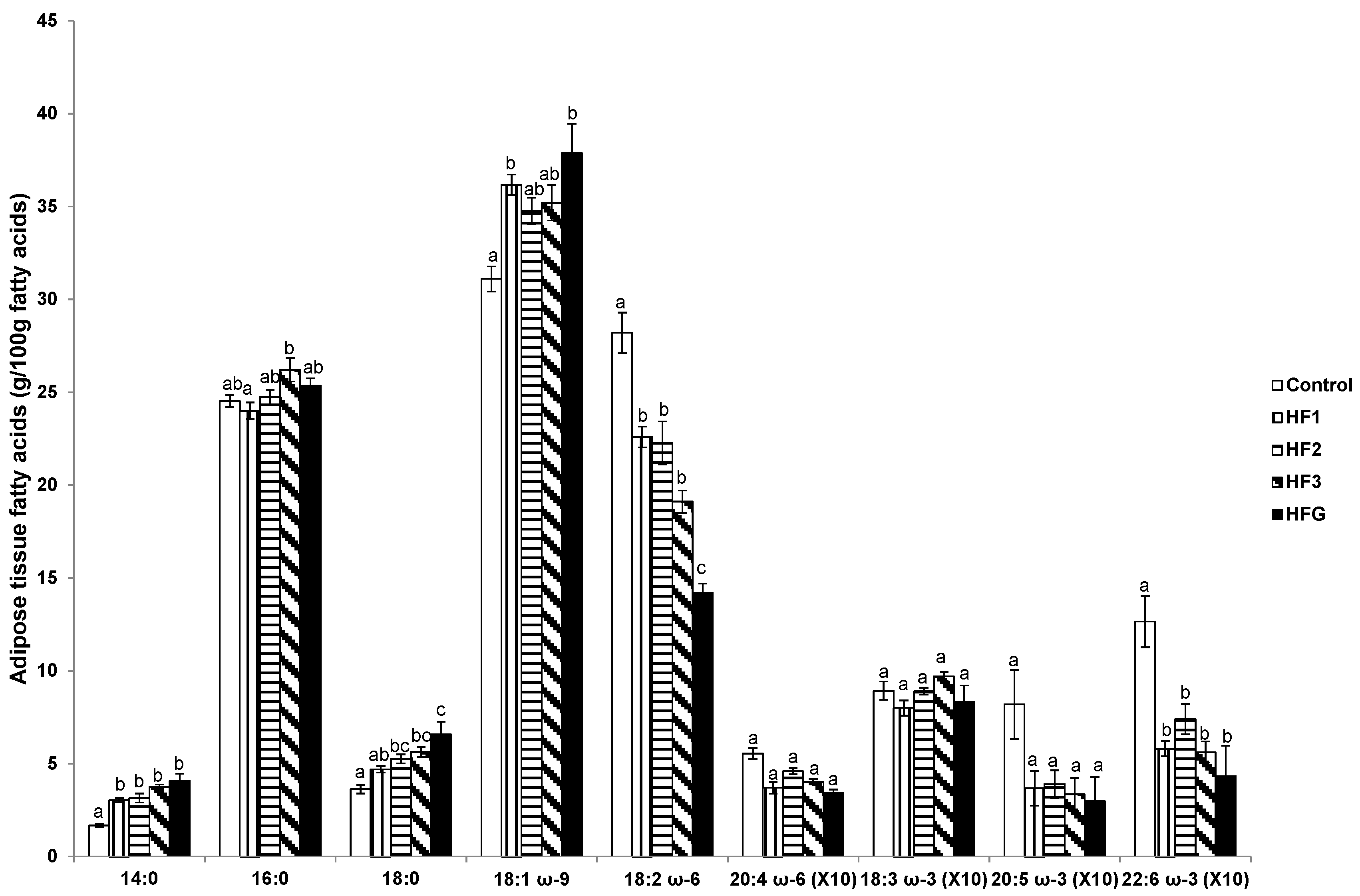
3.8. Adipose Tissue Fatty Acids

4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lorente-Cebrian, S.; Costa, A.G.; Navas-Carretero, S.; Zabala, M.; Alfredo Martinez, J.; Moreno-Aliaga, J. Role of omega-3 fatty acids in obesity, metabolic syndrome, and cardiovascular diseases: A review of the evidence. J. Physiol. Biochem. 2013, 69, 633–651. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.L. Dietary n-6 and n-3 polyunsaturated fatty acids: From biochemistry to clinical implications in cardiovascular prevention. Biochem. Pharmacol. 2009, 77, 937–946. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, M.; Kubota, M. Plasma fatty acid profiles in 37 pairs of maternal and umbilical cord blood samples. Environ. Health Prev. Med. 2004, 9, 67–69. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Le, H.D.; Meisel, J.A.; de Meijer, V.E.; Gura, K.M.; Puder, M. The essentiality of arachidonic acid and docosahexaenoic acid. Prostaglandins Leukot. Essent. Fatty Acids 2009, 81, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M. Essential fatty acids in growth and development. Prog. Lipid Res. 1991, 30, 39–103. [Google Scholar] [CrossRef]
- Uauy, R.; Treen, M.; Hoffman, D.R. Essential fatty acid metabolism and requirements during development. Semin. Perinatol. 1989, 13, 118–130. [Google Scholar] [PubMed]
- Burdge, G.C.; Lillycrop, K.A. Fatty acids and epigenetics. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Casas-Agustench, P.; Fernandes, F.S.; Tavares do Carmo, M.G.; Visioli, F.; Herrera, E.; Davalos, A. Consumption of distinct dietary lipids during early pregnancy differentially modulates the expression of microRNAs in mothers and offspring. PLoS ONE 2015, 10, e0117858. [Google Scholar] [CrossRef] [PubMed]
- Herrera, E.; Ortega-Senovilla, H. Maternal lipid metabolism during normal pregnancy and its implications to fetal development. Clin. Lipidol. 2010, 5, 899–911. [Google Scholar] [CrossRef]
- Hytten, F.E.; Leitch, I. The gross composition of the components of weight gain. In The Physiology of Human Pregnancy; Hytten, F.E., Leitch, I., Eds.; Blackwell: Oxford, UK, 1971; pp. 370–387. [Google Scholar]
- Herrera, E.; Lasunción, M.A.; Gomez Coronado, D.; Aranda, P.; López-Luna, P.; Maier, I. Role of lipoprotein lipase activity on lipoprotein metabolism and the fate of circulating triglycerides in pregnancy. Am. J. Obstet. Gynecol. 1988, 158, 1575–1583. [Google Scholar] [CrossRef]
- Herrera, E.; Ortega-Senovilla, H. Lipid metabolism during pregnancy and its implications for fetal growth. Curr. Pharm. Biotechnol. 2014, 15, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Jones, H.N.; Woollett, L.A.; Barbour, N.; Prasad, P.D.; Powell, T.L.; Jansson, T. High-fat diet before and during pregnancy causes marked up-regulation of placental nutrient transport and fetal overgrowth in C57/BL6 mice. FASEB J. 2009, 23, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Amusquivar, E.; Schiffner, S.; Herrera, E. Evaluation of two methods for plasma fatty acid analysis by GC. Eur. J. Lipid Sci. Technol. 2011, 113, 711–716. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Cerf, M.E.; Louw, J.; Herrera, E. High fat diet exposure during late fetal life enhances hepatic omega 6 fatty acid profiles in fetal Wistar rats. Nutrients 2015, 7, 7231–7241. [Google Scholar] [CrossRef] [PubMed]
- Mampel, T.; Villarroya, F.; Herrera, E. Hepatectomy-nephrectomy effects in the pregnant rat and fetus. Biochem. Biophys. Res. Commun. 1985, 131, 1219–1225. [Google Scholar] [CrossRef]
- Mampel, T.; Camprodon, R.; Solsona, J.; Juncá, V.; Herrera, E. Changes in circulating glycerol, free fatty acids and glucose levels following liver transplant in the pig. Arch. Int. Physiol. Biochim. 1981, 89, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Wasfi, I.; Weinstein, I.; Heimberg, M. Increased formation of triglyceride from oleate in perfused livers from pregnant rats. Endocrinology 1980, 107, 584–590. [Google Scholar] [CrossRef] [PubMed]
- Wasfi, I.; Weinstein, I.; Heimberg, M. Hepatic metabolism of [1-14C] oleate in pregnancy. Biochim. Biophys. Acta 1980, 619, 471–481. [Google Scholar] [CrossRef]
- Amusquivar, E.; Herrera, E. Influence of changes in dietary fatty acids during pregnancy on placental and fetal fatty acid profile in the rat. Biol. Neonate 2003, 83, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Oosterveer, M.H.; van Dijk, T.H.; Tietge, U.J.; Boer, T.; Havinga, R.; Stellaard, F.; Groen, A.K.; Kuipers, F.; Renjngound, D.J. High fat feeding induces hepatic fatty acid elongation in mice. PLoS ONE 2009, 4, e6066. [Google Scholar] [CrossRef] [PubMed]
- Ghebremeskel, K.; Bitsanis, D.; Koukkou, E.; Lowy, C.; Poston, L.; Crawford, M.A. Maternal diet high in fat reduces docosahexaenoic acid in liver lipids of newborn and sucking rat pups. Br. J. Nutr. 1999, 81, 395–404. [Google Scholar] [PubMed]
- Buettner, R.; Parhofer, K.G.; Woenckhaus, M.; Wrede, C.E.; Kunz-Schughart, L.A.; Schölmerich, J.; Bollheimer, L.C. Defining high-fat-diet rat models: Metabolic and molecular effects of different fat types. J. Mol. Endocrinol. 2006, 36, 485–501. [Google Scholar] [CrossRef] [PubMed]
- Gabory, A.; Ferry, L.; Fajardy, I.; Vige, A.; Mayeur, S.; Attig, L.; Lesage, J.; Vieau, D.; Jais, J.P.; Junien, C. Maternal diets trigger sex-specific divergent trajectories of gene expression and epigenetic systems in mouse placenta. PLoS ONE 2012, 7, e47986. [Google Scholar] [CrossRef] [PubMed]
- Tarrade, A.; Rousseau-Ralliard, D.; Aubriere, M.C.; Peynot, N.; Dahirel, M.; Bertrand-Michel, J.; Aguirre-Lavin, T.; Morel, O.; Beaujean, N.; Duranthon, V.; et al. Sexual dimorphism of the feto-placental phenotype in response to a high fat and control maternal diets in a rabbit model. PLoS ONE 2013, 8, e83458. [Google Scholar] [CrossRef] [PubMed]
- Diamant, Y.Z.; Shafrir, E. Placental enzymes of glycolysis, gluconeogenesis and lipogenesis in the diabetic rat and in starvation. Diabetologia 1978, 15, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Hummel, L.; Zimmermann, T.; Schirrmeister, W.; Wagner, H. Synthesis, turnover and compartment analysis of the free fatty acids in the placenta of rats. Acta Biol. Med. Ger. 1976, 35, 1311–1316. [Google Scholar] [PubMed]
- Vileisis, R.A.; Oh, W. Enhanced fatty acid synthesis in hyperinsulinemic rat fetuses. J. Nutr. 1983, 113, 246–252. [Google Scholar] [PubMed]
- Dutta-Roy, A.K. Insulin mediated processes in platelets, erythrocytes and monocytes/macrophages: Effects of essential fatty acid metabolism. Prostaglandins Leukot. Essent. Fatty Acids 1994, 51, 385–399. [Google Scholar] [CrossRef]
- Haggarty, P. Fatty acid supply to the human fetus. Annu. Rev. Nutr. 2010, 30, 237–255. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, F.S.; Tavares do Carmo, M.; Herrera, E. Influence of maternal diet during early pregnancy on the fatty acid profile in the fetus at late pregnancy in rats. Lipids 2012, 47, 505–517. [Google Scholar] [CrossRef] [PubMed]
- Benyshek, D.C.; Kachinski, J.J.; Jin, H. F0 prenatal/lactation diets varying in saturated fat and long-chain polyunsaturated fatty acids alters the insulin sensitivity of F1 rats fed a high fat Western diet postweaning. Open J. Endocr. Metab. Dis. 2014, 4, 245–252. [Google Scholar] [CrossRef]
- Korotkova, M.; Ohlsson, C.; Hanson, L.A.; Strandvik, B. Dietary n-6:n-3 fatty acid ratio in the perinatal period affects bone parameters in adult female rats. Br. J. Nutr. 2004, 92, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Korotkova, M.; Gabrielsson, B.G.; Holmang, A.; Larsson, B.-M.; Hanson, L.A.; Strandvik, B. Gender-related long-term effects in adult rats by perinatal dietary ratio of n-6/n-3 fatty acids. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, R575–R579. [Google Scholar] [CrossRef] [PubMed]
- Korotkova, M.; Gabrielsson, B.; Lonn, M.; Hanson, L.-A.; Strandvik, B. Leptin levels in rat offspring are modified by the ratio of linoleic to alpha-linolenic acid in the maternal diet. J. Lipid Res. 2002, 43, 1743–1749. [Google Scholar] [CrossRef] [PubMed]
- Sardinha, F.L.; Fernandes, F.S.; Tavares do Carmo, M.G.; Herrera, E. Sex-dependent nutritional programming: Fish oil intake during early pregnancy in rats reduces age-dependent insulin resistance in male, but not female, offspring. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, R313–R320. [Google Scholar] [CrossRef] [PubMed]
- Courville, A.B.; Harel, O.; Lammi-Keefe, C.J. Consumption of a DHA-containing functional food during pregnancy is associated with lower infant ponderal index and cord plasma insulin concentration. Br. J. Nutr. 2011, 106, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.P.; Levy, E.; Fraser, W.D.; Julien, P.; Delvin, E.; Montouids, A.; Spahis, S.; Garofalo, C.; Nuyt, A.M.; Luo, Z.C. Circulating docosahexaenoic acid levels are associated with fetal insulin sensitivity. PLoS ONE 2014, 9, e85054. [Google Scholar] [PubMed]
- Uauy, R.; Peirano, P.; Hoffman, D.; Mena, P.; Birch, D.; Birch, E. Role of essential fatty acids in the function of the developing nervous system. Lipids 1996, 31, S167–S176. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.L.; Mark, P.J.; Waddell, B.J. Maternal dietary omega-3 fatty acids and placental function. Reproduction 2014, 147, R143–R152. [Google Scholar] [CrossRef] [PubMed]
- Sanders, T.A.; Naismith, D.J. The metabolism of alpha-linolenic acid by the foetal rat. Br. J. Nutr. 1980, 44, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Sanders, T.A.; Rana, S.K. Comparison of the metabolism of linoleic and linolenic acids in the fetal rat. Ann. Nutr. Metab. 1987, 31, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.P.; Nakamura, M.; Clarke, S.D. Cloning, expression, and fatty acid regulation of the human delta-5 desaturase. J. Biol. Chem. 1999, 274, 37335–37339. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.P.; Nakamura, M.T.; Clarke, S.D. Cloning, expression, and nutritional regulation of the mammalian delta-6 desaturase. J. Biol. Chem. 1999, 274, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Botolin, D.; Christian, B.; Busik, J.; Xu, J.; Jump, D.B. Tissue-specific, nutritional, and developmental regulation of rat fatty acid elongases. J. Lipid Res. 2005, 46, 706–715. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Tocher, D.R.; Dickson, C.A.; Bell, J.G.; Teale, A.J. Highly unsaturated fatty acid synthesis in vertebrates: New insights with the cloning and characterization of a delta-6-desaturase of Atlantic salmon. Lipids 2005, 40, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Hofacer, R.; Jandacek, R.; Rider, T.; Tso, P.; Magrisso, J.; Benoit, S.C.; McNamara, R.K. Omega-3 fatty acid deficiency selectively up-regulates delta6-desaturase expression and activity indices in rat liver: Prevention by normalization of omega-3 fatty acid status. Nutr. Res. 2011, 31, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.A.; Neumann, M.A.; Lien, E.L.; Boyd, K.A.; Tu, W.C. Docosahexaenoic acid synthesis from alpha-linolenic acid is inhibited by diets high in polyunsaturated fatty acids. Prostaglandins Leukot. Essent. Fatty Acids 2013, 88, 139–146. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cerf, M.E.; Herrera, E. High Fat Diet Administration during Specific Periods of Pregnancy Alters Maternal Fatty Acid Profiles in the Near-Term Rat. Nutrients 2016, 8, 25. https://doi.org/10.3390/nu8010025
Cerf ME, Herrera E. High Fat Diet Administration during Specific Periods of Pregnancy Alters Maternal Fatty Acid Profiles in the Near-Term Rat. Nutrients. 2016; 8(1):25. https://doi.org/10.3390/nu8010025
Chicago/Turabian StyleCerf, Marlon E., and Emilio Herrera. 2016. "High Fat Diet Administration during Specific Periods of Pregnancy Alters Maternal Fatty Acid Profiles in the Near-Term Rat" Nutrients 8, no. 1: 25. https://doi.org/10.3390/nu8010025
APA StyleCerf, M. E., & Herrera, E. (2016). High Fat Diet Administration during Specific Periods of Pregnancy Alters Maternal Fatty Acid Profiles in the Near-Term Rat. Nutrients, 8(1), 25. https://doi.org/10.3390/nu8010025




