Increased Intake of Foods with High Nutrient Density Can Help to Break the Intergenerational Cycle of Malnutrition and Obesity
Abstract
:1. Introduction
2. Current Nutritional Situation
3. Critical Windows throughout the Life Course
3.1. Programming in Early Life
3.2. Adulthood and Old Age
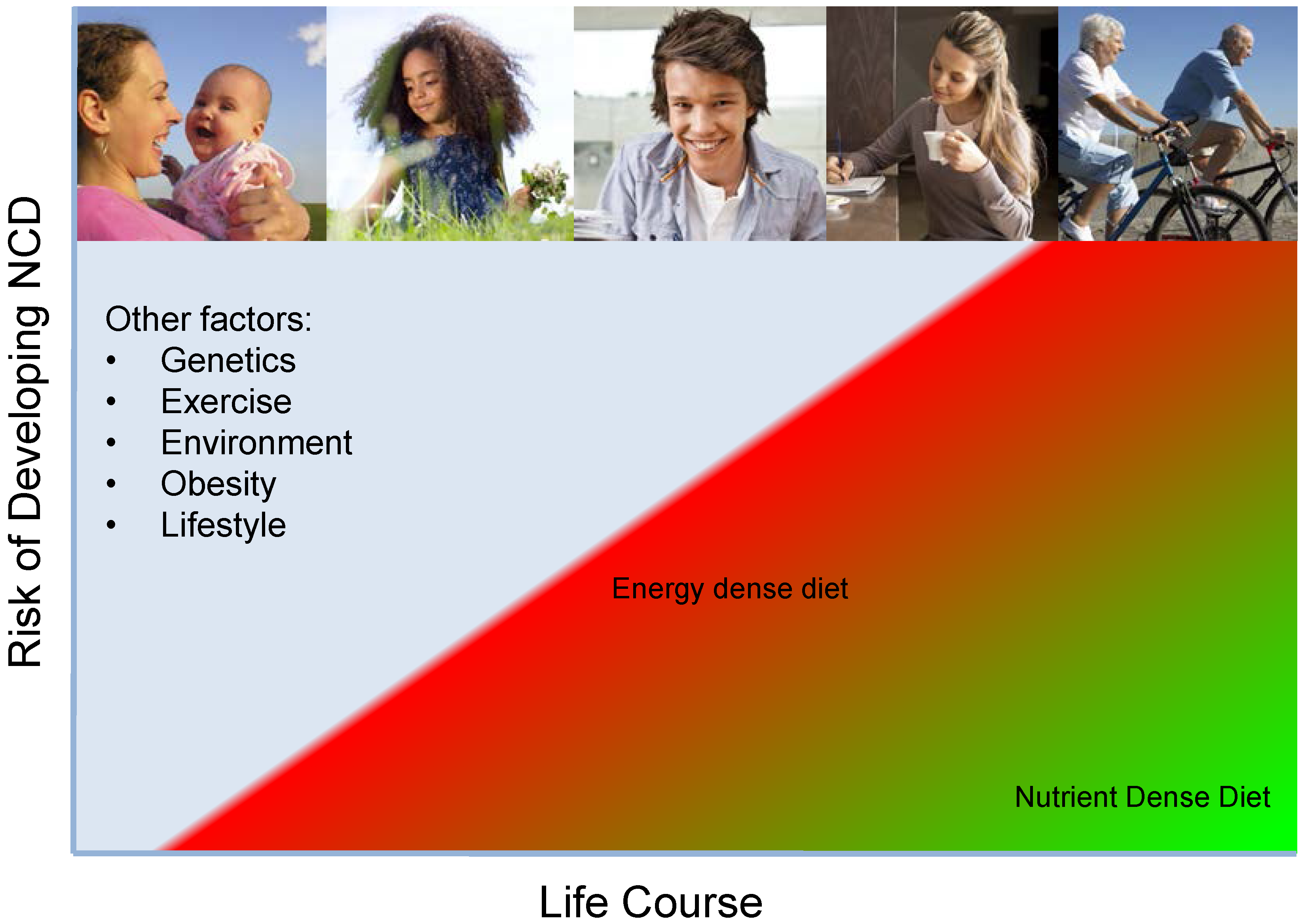
4. Improving Nutrition along the Life Course
Improving Dietary Quality through Micronutrient Density
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kirkwood, T.B.L. A systematic look at an old problem. Nature 2008, 451, 644–647. [Google Scholar] [CrossRef] [PubMed]
- UNICEF. Child Mortalit Decline between 1990 and 2006. Available online: http://www.unicef.org/progressforchildren/2007n6/index_41802.htm (accessed on 24 April 2014).
- World Health Organization. World Health Statistics; World Health Organization Press: Geneva, Switzerland, 2014. [Google Scholar]
- Kinsella, K.; He, W. An Aging World: 2008 International Population Reports; U.S. Department of Health and Human Services, National Institute of Health, National Institute of Aging: Washington, DC, USA, 2009.
- Lunenfeld, B. An aging world—Demographics and challenges. Gynecol. Endocrinol. 2008, 24, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Beaglehole, R.; Bonita, R.; Horton, R.; Adams, C.; Alleyne, G.; Asaria, P.; Baugh, V.; Bekedam, H.; Billo, N.; Casswell, S.; et al. Priority actions for the non-communicable disease crisis. Lancet 2011, 377, 1438–1447. [Google Scholar] [CrossRef]
- Adeyi, O.; Smith, O.; Robles, S. Public Policy and the Challenge of Chronic Noncommunicable Diseases. Available online: http://siteresources.worldbank.org/INTPH/Resources/PublicPolicyandNCDsWorldBank2007FullReport.pdf (accessed on 2 June 2014).
- World Health Organization. Who Global Status Report on Noncommunicable Diseases 2010; World Health Organization Press: Geneva, Switzerland, 2010. [Google Scholar]
- Troesch, B.; Hoeft, B.; McBurney, M.; Eggersdorfer, M.; Weber, P. Dietary surveys indicate vitamin intakes below recommendations are common in representative Western countries. Br. J. Nutr. 2012, 108, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Krebs-Smith, S.M.; Guenther, P.M.; Subar, A.F.; Kirkpatrick, S.I.; Dodd, K.W. Americans do not meet federal dietary recommendations. J. Nutr. 2010, 140, 1832–1838. [Google Scholar] [CrossRef] [PubMed]
- Deutsche Gesellschaft für Ernährung e. V. Verzehrsstudie zur ermittlung der lebensmittelaufnahme von säuglingen und kleinkindern (vels). In Ernährungsbericht 2008; Deutsche Gesellschaft für Ernährung e. V.: Bonn, Germany, 2008; pp. 53–60. [Google Scholar]
- Decarli, B.; Cavadini, C.; Michaud, P.A. Habitudes alimentaires et ingestats de nutriments d'un groupe d'adolescents vaudois de 9 à 19 ans. In Fünfter Schweizer Ernährungsbericht; Eichholzer, M., Camenzind-Frey, E., Matzke, A., Amadò, R., Ballmer, P.E., et al., Eds.; Bundesamt für Gesundheit: Bern, Switzerland, 2005; pp. 133–150. [Google Scholar]
- Elia, M.; Stratton, R.J.; Russell, C.; Green, C.; Pang, F. The Cost of Disease-Related Malnutrition in the UK and Economic Considerations for the Use of Oral Nutritional Supplements (ONS) in Adults; British Association for Parenteral and Enteral Nutrition: Redditch, UK, 2006. [Google Scholar]
- Marriott, B.P.; Olsho, L.; Hadden, L.; Connor, P. Intake of added sugars and selected nutrients in the United States, national health and nutrition examination survey (NHANES) 2003—2006. Crit. Rev. Food Sci. Nutr. 2010, 50, 228–258. [Google Scholar] [CrossRef] [PubMed]
- Deutsche Gesellschaft für Ernährung e. V. Ernährung älterer menschen in stationären einrichtungen (ernstes-studies). In Ernährungsbericht 2008; Deutsche Gesellschaft für Ernährung e. V.: Bonn, Germany, 2008; pp. 157–204. [Google Scholar]
- World Health Organization. Fact Sheet N°311: Obesity and Overweight. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/ (accessed on 2 May 2014).
- Damms-Machado, A.; Weser, G.; Bischoff, S. Micronutrient deficiency in obese subjects undergoing low calorie diet. Nutr. J. 2012, 11, 34. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Reider, C.; Brooks, J.R.; Fulgoni, V.L. Comparison of prevalence of inadequate nutrient intake based on body weight status of adults in the United States: An analysis of NHANES 2001–2008. J. Am. College Nutr. 2015, 34, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Cepeda-Lopez, A.C.; Aeberli, I.; Zimmermann, M.B. Does obesity increase risk for iron deficiency? A review of the literature and the potential mechanisms. Int. J. Vitam. Nutr. Res. 2010, 80, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Samuel, L.; Borrell, L.N. The effect of body mass index on optimal vitamin D status in U.S. adults: The national health and nutrition examination survey 2001–2006. Ann. Epidemiol. 2013, 23, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Wortsman, J.; Matsuoka, L.Y.; Chen, T.C.; Lu, Z.; Holick, M.F. Decreased bioavailability of vitamin D in obesity. Am. J. Clin. Nutr. 2000, 72, 690–693. [Google Scholar] [PubMed]
- Heaney, R.P. Nutrition, chronic disease, and the problem of proof. Am. J. Clin. Nutr. 2006, 84, 471–472. [Google Scholar] [PubMed]
- Black, R.E.; Allen, L.H.; Bhutta, Z.A.; Caulfield, L.E.; de Onis, M.; Ezzati, M.; Mathers, C.; Rivera, J. Maternal and child undernutrition: Global and regional exposures and health consequences. Lancet 2008, 371, 243–260. [Google Scholar] [CrossRef]
- Plagemann, A.; Harder, T.; Schellong, K.; Schulz, S.; Stupin, J.H. Early postnatal life as a critical time window for determination of long-term metabolic health. Best Pract. Res. Clin. Endocrinol. Metab. 2012, 26, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Räikkönen, K.; Pesonen, A.-K.; Roseboom, T.J.; Eriksson, J.G. Early determinants of mental health. Best Pract. Res. Clin. Endocrinol. Metab. 2012, 26, 599–611. [Google Scholar] [CrossRef] [PubMed]
- United Nations. Political Declaration of the High-level Meeting of the General Assembly on the Prevention and Control of Non-communicable Diseases. Available online: http://www.who.int/nmh/events/un_ncd_summit2011/political_declaration_en.pdf (accessed on 30 May 2014).
- UNICEF. Improving Child Nutrition: The Achievable Imperative for Global Progress; UNICEF: New York, NY, USA, 2013. [Google Scholar]
- Pampel, F.C.; Denney, J.T.; Krueger, P.M. Obesity, SES, and economic development: A test of the reversal hypothesis. Soc. Sci. Med. 2012, 74, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J.; Osmond, C. Infant mortality, childhood nutrition, and ischaemic heart disease in england and wales. Lancet 1986, 1, 1077–1081. [Google Scholar] [CrossRef]
- Barker, D.J.P. Mothers, Babies and Health in Later Life; Churchill Livingstone: New York, NY, USA, 1998. [Google Scholar]
- Gluckman, P.D.; Hanson, M.A.; Cooper, C.; Thornburg, K.L. Effect of in utero and early-life conditions on adult health and disease. N. Engl. J. Med. 2008, 359, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Painter, R.C.; Roseboom, T.J.; Bleker, O.P. Prenatal exposure to the dutch famine and disease in later life: An overview. Reprod. Toxicol. 2005, 20, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Warner, M.J.; Ozanne, S.E. Mechanisms involved in the developmental programming of adulthood disease. Biochem. J. 2010, 427, 333–347. [Google Scholar] [CrossRef] [PubMed]
- Stunkard, A.J.; Sørensen, T.I.A.; Hanis, C.; Teasdale, T.W.; Chakraborty, R.; Schull, W.J.; Schulsinger, F. An adoption study of human obesity. N. Engl. J. Med. 1986, 314, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, C.; Tremblay, A.; Després, J.-P.; Nadeau, A.; Lupien, P.J.; Thériault, G.; Dussault, J.; Moorjani, S.; Pinault, S.; Fournier, G. The response to long-term overfeeding in identical twins. N. Engl. J. Med. 1990, 322, 1477–1482. [Google Scholar] [CrossRef] [PubMed]
- Lillycrop, K.A.; Burdge, G.C. Epigenetic mechanisms linking early nutrition to long term health. Best Pract. Res. Clin. Endocrinol. Metab. 2012, 26, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Khulan, B.; Cooper, W.N.; Skinner, B.M.; Bauer, J.; Owens, S.; Prentice, A.M.; Belteki, G.; Constancia, M.; Dunger, D.; Affara, N.A. Periconceptional maternal micronutrient supplementation is associated with widespread gender related changes in the epigenome: A study of a unique resource in the gambia. Hum. Mol. Genet. 2012, 21, 2086–2101. [Google Scholar] [CrossRef] [PubMed]
- Harder, T.; Schellong, K.; Stupin, J.; Dudenhausen, J.W.; Plagemann, A. Where is the evidence that low birthweight leads to obesity? Lancet 2007, 369, 1859. [Google Scholar] [CrossRef]
- Boney, C.M.; Verma, A.; Tucker, R.; Vohr, B.R. Metabolic syndrome in childhood: Association with birth weight, maternal obesity, and gestational diabetes mellitus. Pediatrics 2005, 115, e290–e296. [Google Scholar] [CrossRef] [PubMed]
- Catalano, P.M. Obesity and pregnancy—The propagation of a viscous cycle? J. Clin. Endocrinol. Metab. 2003, 88, 3505–3506. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.T.; Phelan, S.T. Nutrition during pregnancy. Obstet. Gynecol. Clin. N. Am. 2008, 35, 369–383. [Google Scholar] [CrossRef] [PubMed]
- Hilson, J.A.; Rasmussen, K.M.; Kjolhede, C.L. Excessive weight gain during pregnancy is associated with earlier termination of breast-feeding among white women. J. Nutr. 2006, 136, 140–146. [Google Scholar] [PubMed]
- Horta, B.L.; Bahl, R.; Martines, J.C.; Victora, C.G. Evidence on the Long-Term Effects of Breastfeeding: Systematic Review and Meta-Analyses; World Health Organization Press: Geneva, Switzerland, 2007. [Google Scholar]
- World Health Organization. Exclusive Breastfeeding. Available online: http://www.who.int/elena/titles/exclusive_breastfeeding/en/ (accessed on 23 June 2014).
- Durmuş, B.; Heppe, D.H.; Gishti, O.; Manniesing, R.; Abrahamse-Berkeveld, M.; van der Beek, E.M.; Hofman, A.; Duijts, L.; Gaillard, R.; Jaddoe, V.W. General and abdominal fat outcomes in school-age children associated with infant breastfeeding patterns. Am. J. Clin. Nutr. 2014, 99, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Frei, B.; Ames, B.N.; Blumberg, J.B.; Willett, W.C. Enough is enough. Ann. Intern. Med. 2014, 160, 807. [Google Scholar] [CrossRef] [PubMed]
- Petherick, A. Development: Mother’s milk: A rich opportunity. Nature 2010, 468, S5–S7. [Google Scholar] [CrossRef] [PubMed]
- Palmer, C.; Bik, E.M.; DiGiulio, D.B.; Relman, D.A.; Brown, P.O. Development of the human infant intestinal microbiota. PLoS. Biol. 2007, 5, 1556–1573. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.K.; Estaki, M.; Gibson, D.L. Clinical consequences of diet-induced dysbiosis. Ann. Nutr. Metab. 2013, 63, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Kootte, R.S.; Vrieze, A.; Holleman, F.; Dallinga-Thie, G.M.; Zoetendal, E.G.; de Vos, W.M.; Groen, A.K.; Hoekstra, J.B.L.; Stroes, E.S.; Nieuwdorp, M. The therapeutic potential of manipulating gut microbiota in obesity and type 2 diabetes mellitus. Diabetes Obes. Metab. 2012, 14, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; DuGar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.-M.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-Rubio, R.; Collado, M.C.; Laitinen, K.; Salminen, S.; Isolauri, E.; Mira, A. The human milk microbiome changes over lactation and is shaped by maternal weight and mode of delivery. Am. J. Clin. Nutr. 2012, 96, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Owen, C.G.; Whincup, P.H.; Odoki, K.; Gilg, J.A.; Cook, D.G. Infant feeding and blood cholesterol: A study in adolescents and a systematic review. Pediatrics 2002, 110, 597–608. [Google Scholar] [CrossRef] [PubMed]
- Owen, C.G.; Whincup, P.H.; Kaye, S.J.; Martin, R.M.; Davey Smith, G.; Cook, D.G.; Bergstrom, E.; Black, S.; Wadsworth, M.E.; Fall, C.H.; et al. Does initial breastfeeding lead to lower blood cholesterol in adult life? A quantitative review of the evidence. Am. J. Clin. Nutr. 2008, 88, 305–314. [Google Scholar] [PubMed]
- Fall, C.H.; Barker, D.J.; Osmond, C.; Winter, P.D.; Clark, P.M.; Hales, C.N. Relation of infant feeding to adult serum cholesterol concentration and death from ischaemic heart disease. BMJ 1992, 304, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Bayley, T.M.; Alasmi, M.; Thorkelson, T.; Krug-Wispe, S.; Jones, P.J.H.; Bulani, J.L.; Tsang, R.C. Influence of formula versus breast milk on cholesterol synthesis rates in four-month-old infants. Pediatr. Res. 1998, 44, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Demmers, T.A.; Jones, P.J.H.; Wang, Y.; Krug, S.; Creutzinger, V.; Heubi, J.E. Effects of early cholesterol intake on cholesterol biosynthesis and plasma lipids among infants until 18 months of age. Pediatrics 2005, 115, 1594–1601. [Google Scholar] [CrossRef] [PubMed]
- Bode, L.; Jantscher-Krenn, E. Structure-function relationships of human milk oligosaccharides. Adv. Nutr. Int. Rev. J. 2012, 3, 383S–391S. [Google Scholar] [CrossRef] [PubMed]
- Allen, L.H. B vitamins in breast milk: Relative importance of maternal status and intake, and effects on infant status and function. Adv. Nutr. Int. Rev. J. 2012, 3, 362–369. [Google Scholar] [CrossRef] [PubMed]
- De-Regil, L.M.; Palacios, C.; Ansary, A.; Kulier, R.; Pena-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2012, 2, CD008873. [Google Scholar] [PubMed]
- Bode, L. Human milk oligosaccharides: Every baby needs a sugar mama. Glycobiology 2012, 22, 1147–1162. [Google Scholar] [CrossRef] [PubMed]
- Lauritzen, L.; Carlson, S.E. Maternal fatty acid status during pregnancy and lactation and relation to newborn and infant status. Matern. Child. Nutr. 2011, 7, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Hoddinott, J.; Behrman, J.R.; Maluccio, J.A.; Melgar, P.; Quisumbing, A.R.; Ramirez-Zea, M.; Stein, A.D.; Yount, K.M.; Martorell, R. Adult consequences of growth failure in early childhood. Am. J. Clin. Nutr. 2013, 98, 1170–1178. [Google Scholar] [CrossRef] [PubMed]
- Schulz, C.; Engel, U.; Kreienberg, R.; Biesalski, H.K. Vitamin A and β-carotene supply of women with gemini or short birth intervals: A pilot study. Eur J. Nutr. 2007, 46, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Al, M.D.M.; van Houwelingen, A.C.; Hornstra, G. Relation between birth order and the maternal and neonatal docosahexaenoic acid status. Eur. J. Clin. Nutr. 1997, 51, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Crowther, N.J.; Cameron, N.; Trusler, J.; Gray, I.P. Association between poor glucose tolerance and rapid post natal weight gain in seven-year-old children. Diabetologia 1998, 41, 1163–1167. [Google Scholar] [CrossRef] [PubMed]
- Forsén, T.; Eriksson, J.; Tuomilehto, J.; Reunanen, A.; Osmond, C.; Barker, D. The fetal and childhood growth of persons who develop type 2 diabetes. Ann. Intern. Med. 2000, 133, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Yajnik, C. Interactions of perturbations in intrauterine growth and growth during childhood on the risk of adult-onset disease. Proc. Nutr. Soc. 2000, 59, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Singhal, A.; Cole, T.J.; Fewtrell, M.; Kennedy, K.; Stephenson, T.; Elias-Jones, A.; Lucas, A. Promotion of faster weight gain in infants born small for gestational age: Is there an adverse effect on later blood pressure? Circulation 2007, 115, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, J.G.; Forsén, T.; Tuomilehto, J.; Winter, P.D.; Osmond, C.; Barker, D.J.P. Catch-up growth in childhood and death from coronary heart disease: Longitudinal study. BMJ 1999, 318, 427–431. [Google Scholar] [CrossRef] [PubMed]
- Cheung, Y.B.; Low, L.; Osmond, C.; Barker, D.; Karlberg, J. Fetal growth and early postnatal growth are related to blood pressure in adults. Hypertension 2000, 36, 795–800. [Google Scholar] [CrossRef] [PubMed]
- Victora, C.G.; Adair, L.; Fall, C.; Hallal, P.C.; Martorell, R.; Richter, L.; Sachdev, H.S. Maternal and child undernutrition: Consequences for adult health and human capital. Lancet 2008, 371, 340–357. [Google Scholar] [CrossRef]
- Golden, M.H. Proposed recommended nutrient densities for moderately malnourished children. Food Nutr. Bull. 2009, 30, 267–342. [Google Scholar]
- Ames, B.N. Low micronutrient intake may accelerate the degenerative diseases of aging through allocation of scarce micronutrients by triage. Proc. Natl. Acad. Sci. USA 2006, 103, 17589–17594. [Google Scholar] [CrossRef] [PubMed]
- Gaziano, J.; Sesso, H.D.; Christen, W.G.; Bubes, V.; Smith, J.P.; MacFadyen, J.; Schvartz, M.; Manson, J.E.; Glynn, R.J.; Buring, J.E. Multivitamins in the prevention of cancer in men: The physicians’ health study ii randomized controlled trial. JAMA 2012, 308, 1871–1880. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Pencina, M.J.; Booth, S.L.; Jacques, P.F.; Ingelsson, E.; Lanier, K.; Benjamin, E.J.; D’Agostino, R.B.; Wolf, M.; Vasan, R.S. Vitamin D deficiency and risk of cardiovascular disease. Circulation 2008, 117, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Dobnig, H.; Pilz, S.; Scharnagl, H.; Renner, W.; Seelhorst, U.; Wellnitz, B.; Kinkeldei, J.; Boehm, B.O.; Weihrauch, G.; Maerz, W. Independent association of low serum 25-hydroxy vitamin D and 1,25-dihydroxy vitamin D levels with all-cause and cardiovascular mortality. Arch. Intern. Med. 2008, 168, 1340–1349. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, C.; Llewellyn, D.J.; Beauchet, O. Low serum vitamin D concentrations in alzheimer’s disease: A systematic review and meta-analysis. J. Alzheimers Dis. 2013, 33, 659–674. [Google Scholar] [PubMed]
- Annweiler, C.; Rolland, Y.; Schott, A.M.; Blain, H.; Vellas, B.; Beauchet, O. Serum vitamin D deficiency as a predictor of incident non-alzheimer dementias: A 7-year longitudinal study. Dement. Geriatr. Cogn. Disord. 2011, 32, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, C.; Rolland, Y.; Schott, A.M.; Blain, H.; Vellas, B.; Herrmann, F.R.; Beauchet, O. Higher vitamin D dietary intake is associated with lower risk of alzheimer’s disease: A 7-year follow-up. J. Gerontol. Ser. A: Biol. Sci. Med. Sci. 2012, 67, 1205–1211. [Google Scholar] [CrossRef] [PubMed]
- Buell, J.S.; Dawson-Hughes, B. Vitamin D and neurocognitive dysfunction: Preventing decline? Mol. Aspects Med. 2008, 29, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.D. The worldwide challenge of the dementias: A role for B vitamins and homocysteine? Food Nutr. Bull. 2008, 29, S143–S172. [Google Scholar] [PubMed]
- Smith, A.D.; Smith, S.M.; de Jager, C.A.; Whitbread, P.; Johnston, C.; Agacinski, G.; Oulhaj, A.; Bradley, K.M.; Jacoby, R.; Refsum, H. Homocysteine-Lowering by B vitamins slows the rate of accelerated brain atrophy in mild cognitive impairment: A randomized controlled trial. PLoS ONE 2010, 5, e12244. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, P.; Polidori, M.C.; Metastasio, A.; Mariani, E.; Mattioli, P.; Cherubini, A.; Catani, M.; Cecchetti, R.; Senin, U.; Mecocci, P. Plasma antioxidants are similarly depleted in mild cognitive impairment and in alzheimer’s disease. Neurobiol. Aging 2003, 24, 915–919. [Google Scholar] [CrossRef]
- Harris, E.; Kirk, J.; Rowsell, R.; Vitetta, L.; Sali, A.; Scholey, A.B.; Pipingas, A. The effect of multivitamin supplementation on mood and stress in healthy older men. Hum. Psychopharmacol. 2011, 26, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Venkatram, S.; Chilimuri, S.; Adrish, M.; Salako, A.; Patel, M.; Diaz-Fuentes, G. Vitamin D deficiency is associated with mortality in the medical intensive care unit. Crit. Care 2011, 15, R292. [Google Scholar] [CrossRef] [PubMed]
- Matthews, L.R.; Ahmed, Y.; Wilson, K.L.; Griggs, D.D.; Danner, O.K. Worsening severity of vitamin D deficiency is associated with increased length of stay, surgical intensive care unit cost, and mortality rate in surgical intensive care unit patients. Am. J. Surg. 2012, 204, 37–43. [Google Scholar] [CrossRef] [PubMed]
- De Castro, J.M.; Stroebele, N. Food intake in the real world: Implications for nutrition and aging. Clin. Geriatr. Med. 2002, 18, 685–697. [Google Scholar] [CrossRef]
- Johnson, K.A.; Bernard, M.A.; Funderburg, K. Vitamin nutrition in older adults. Clin. Geriatr. Med. 2002, 18, 773–799. [Google Scholar] [CrossRef]
- Labadarios, D.; Steyn, N.P.; Nel, J. How diverse is the diet of adult South Africans? Nutr. J. 2011, 10, 33. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.M.; Rasmussen, H. The impact of nutritional needs of older adults on recommended food intakes. Nutr. Clin. Care 1999, 2, 164–176. [Google Scholar] [CrossRef]
- D’Souza, A.L. Ageing and the gut. Postgrad. Med. J. 2007, 83, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Buffa, R.; Floris, G.U.; Putzu, P.F.; Marini, E. Body composition variations in ageing. Coll. Antropol. 2011, 35, 259–265. [Google Scholar] [PubMed]
- Institute of Medicine. Dietary Reference Intakes of Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; The National Academic Press: Washington, DC, USA, 2001. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes of Vitamin C, Vitamin E, Selenium, and Carotenoids; The National Academic Press: Washington, DC, USA, 2000. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin and Cholin; The National Academic Press: Washington, DC, USA, 1998. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Foote, J.A.; Giuliano, A.R.; Harris, R.B. Older adults need guidance to meet nutritional recommendations. J. Am. Coll. Nutr. 2000, 19, 628–640. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, J. Nutritional needs of seniors. J. Am. Coll. Nutr. 1997, 16, 517–523. [Google Scholar] [PubMed]
- Steyn, N.; Nel, J.; Nantel, G.; Kennedy, G.; Labadarios, D. Food variety and dietary diversity scores in children: Are they good indicators of dietary adequacy? Public Health Nutr. 2006, 9, 644–650. [Google Scholar] [CrossRef] [PubMed]
- Assmann, K.E.; Lassale, C.; Galan, P.; Hercberg, S.; Kesse-Guyot, E. Dietary quality and 6-year anthropometric changes in a sample of French middle-aged overweight and obese adults. PLoS ONE 2014, 9, e87083. [Google Scholar] [CrossRef] [PubMed]
- US Department of Agriculture. Dietary Guidelines for Americans, 7th ed.U.S. Government Printing Office: Washington, DC, USA, 2010.
- Murphy, M.M.; Barraj, L.M.; Spungen, J.H.; Herman, D.R.; Randolph, R.K. Global assessment of select phytonutrient intakes by level of fruit and vegetable consumption. Br. J. Nutr. 2014, 112, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Prentice, A.M.; Jebb, S.A. Fast foods, energy density and obesity: A possible mechanistic link. Obes. Rev. 2003, 4, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Poppitt, S.D. Energy density of diets and obesity. Int. J. Obes. Relat. Metab. Disorders. 1995, 19, S20–S26. [Google Scholar]
- Astrup, A.; Grunwald, G.K.; Melanson, E.L.; Saris, W.H.; Hill, J.O. The role of low-fat diets in body weight control: A meta-analysis of ad libitum dietary intervention studies. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Brownell, K.D.; Farley, T.; Willett, W.C.; Popkin, B.M.; Chaloupka, F.J.; Thompson, J.W.; Ludwig, D.S. The public health and economic benefits of taxing sugar-sweetened beverages. N. Engl. J. Med. 2009, 361, 1599–1605. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R.D. Fluid energy—Where’s the problem? J. Am. Diet. Assoc. 2006, 106, 1956–1961. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B. Resolved: There is sufficient scientific evidence that decreasing sugar-sweetened beverage consumption will reduce the prevalence of obesity and obesity-related diseases. Obes. Rev. 2013, 14, 606–619. [Google Scholar] [CrossRef] [PubMed]
- Trumbo, P.R.; Rivers, C.R. Systematic review of the evidence for an association between sugar-sweetened beverage consumption and risk of obesity. Nutr. Rev. 2014. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Malik, V.; Rexrode, K.M.; Manson, J.E.; Willett, W.C.; Hu, F.B. Sweetened beverage consumption and risk of coronary heart disease in women. Am. J. Clin. Nutr. 2009, 89, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Kell, K.P.; Cardel, M.I.; Bohan Brown, M.M.; Fernández, J.R. Added sugars in the diet are positively associated with diastolic blood pressure and triglycerides in children. Am. J. Clin. Nutr. 2014, 100, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Dhingra, R.; Sullivan, L.; Jacques, P.F.; Wang, T.J.; Fox, C.S.; Meigs, J.B.; D’Agostino, R.B.; Gaziano, J.M.; Vasan, R.S. Soft drink consumption and risk of developing cardiometabolic risk factors and the metabolic syndrome in middle-aged adults in the community. Circulation 2007, 116, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Lutsey, P.L.; Steffen, L.M.; Stevens, J. Dietary intake and the development of the metabolic syndrome: The atherosclerosis risk in communities study. Circulation 2008, 117, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.; Nicklas, T.; Baranowski, T.; Zakeri, I.F.; Yang, S.-J.; Srinivasan, S.R.; Berenson, G.S. Comparison of dietary intakes associated with metabolic syndrome risk factors in young adults: The bogalusa heart study. Am. J. Clin. Nutr. 2004, 80, 841–848. [Google Scholar] [PubMed]
- Malik, V.S.; Popkin, B.M.; Bray, G.A.; Després, J.-P.; Willett, W.C.; Hu, F.B. Sugar-Sweetened beverages and risk of metabolic syndrome and type 2 diabetes: A meta-analysis. Diabetes Care 2010, 33, 2477–2483. [Google Scholar] [CrossRef] [PubMed]
- Lana, A.; Rodríguez-Artalejo, F.; Lopez-Garcia, E. Consumption of sugar-sweetened beverages is positively related to insulin resistance and higher plasma leptin concentrations in men and nonoverweight women. J. Nutr. 2014, 144, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Vartanian, L.R.; Schwartz, M.B.; Brownell, K.D. Effects of soft drink consumption on nutrition and health: A systematic review and meta-analysis. Am. J. Public Health 2007, 97, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Ledikwe, J.H.; Blanck, H.M.; Khan, L.K.; Serdula, M.K.; Seymour, J.D.; Tohill, B.C.; Rolls, B.J. Dietary energy density determined by eight calculation methods in a nationally representative united states population. J. Nutr. 2005, 135, 273–278. [Google Scholar] [PubMed]
- Giskes, K.; van Lenthe, F.; Avendano-Pabon, M.; Brug, J. A systematic review of environmental factors and obesogenic dietary intakes among adults: Are we getting closer to understanding obesogenic environments? Obes. Rev. 2011, 12, e95–e106. [Google Scholar] [CrossRef] [PubMed]
- Chaput, J.P. Short sleep duration as a cause of obesity: Myth or reality? Obes. Rev. 2011, 12, e2–e3. [Google Scholar] [PubMed]
- Tremblay, A.; Arguin, H. Healthy eating at school to compensate for the activity-related obesigenic lifestyle in children and adolescents: The quebec experience. Adv. Nutr. Int. Rev. J. 2011, 2, 167S–170S. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A. Concept of a nutritious food: Toward a nutrient density score. Am. J. Clin. Nutr. 2005, 82, 721–732. [Google Scholar] [PubMed]
- Chiuve, S.E.; Sampson, L.; Willett, W.C. The association between a nutritional quality index and risk of chronic disease. Am. J. Prev. Med. 2011, 40, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A. Defining nutrient density: Development and validation of the nutrient rich foods index. J. Am. Coll. Nutr. 2009, 28, 421S–426S. [Google Scholar] [CrossRef] [PubMed]
- Streppel, M.T.; Sluik, D.; van Yperen, J.F.; Geelen, A.; Hofman, A.; Franco, O.H.; Witteman, J.C.M.; Feskens, E.J.M. Nutrient-rich foods, cardiovascular diseases and all-cause mortality: The rotterdam study. Eur. J. Clin. Nutr. 2014, 68, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A.; Darmon, N. The economics of obesity: Dietary energy density and energy cost. Am. J. Clin. Nutr. 2005, 82, 265S–273S. [Google Scholar] [PubMed]
- Drewnowski, A. The economics of food choice behavior: Why poverty and obesity are linked. Nestle Nutr. Inst. Workshop Ser. 2012, 73, 95–112. [Google Scholar] [PubMed]
- Aggarwal, A.; Monsivais, P.; Drewnowski, A. Nutrient intakes linked to better health outcomes are associated with higher diet costs in the us. PLoS ONE 2012, 7, e37533. [Google Scholar] [CrossRef] [PubMed]
- Lallukka, T.; Laaksonen, M.; Rahkonen, O.; Roos, E.; Lahelma, E. Multiple socio-economic circumstances and healthy food habits. Eur. J. Clin. Nutr. 2006, 61, 701–710. [Google Scholar] [CrossRef] [PubMed]
- Satheannoppakao, W.; Aekplakorn, W.; Pradipasen, M. Fruit and vegetable consumption and its recommended intake associated with sociodemographic factors: Thailand national health examination survey III. Public Health Nutr. 2009, 12, 2192–2198. [Google Scholar] [CrossRef] [PubMed]
- Darmon, N.; Ferguson, E.L.; Briend, A. A cost constraint alone has adverse effects on food selection and nutrient density: An analysis of human diets by linear programming. J. Nutr. 2002, 132, 3764–3771. [Google Scholar] [PubMed]
- Monsivais, P.; Aggarwal, A.; Drewnowski, A. Are socio-economic disparities in diet quality explained by diet cost? J. Epidemiol. Commun. Health 2010, 66, 530–535. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. G8 urged to act on food crisis and health. Bull. World Health Organ. 2008, 86, 503–504. [Google Scholar]
- Waters, H.; Saadah, F.; Pradhan, M. The impact of the 1997–98 East Asian economic crisis on health and health care in Indonesia. Health Policy Plan. 2003, 18, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Karanikolos, M.; Mladovsky, P.; Cylus, J.; Thomson, S.; Basu, S.; Stuckler, D.; Mackenbach, J.P.; McKee, M. Financial crisis, austerity, and health in Europe. Lancet 2013, 381, 1323–1331. [Google Scholar] [CrossRef]
- Coleman-Jensen, A.; Nord, M.; Singh, A. Household Food Security in the United States in 2012. Available online: http://www.ers.usda.gov/media/1183208/err-155.pdf (accessed on 8 December 2014).
- Brinkman, H.-J.; de Pee, S.; Sanogo, I.; Subran, L.; Bloem, M.W. High food prices and the global financial crisis have reduced access to nutritious food and worsened nutritional status and health. J. Nutr. 2010, 140, 153S–161S. [Google Scholar] [CrossRef] [PubMed]
- Larson, N.I.; Story, M.T. Food insecurity and weight status among U.S. Children and families: A review of the literature. Am. J. Prev. Med. 2011, 40, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Martin-Fernandez, J.; Caillavet, F.; Lhuissier, A.; Chauvin, P. Food insecurity, a determinant of obesity? An analysis from a population-based survey in the Paris metropolitan area, 2010. Obes. Facts 2014, 7, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A.; Fulgoni, V.L. Nutrient density: Principles and evaluation tools. Am. J. Clin. Nutr. 2014, 99, 1223S–1228S. [Google Scholar] [CrossRef] [PubMed]
- Maillot, M.; Ferguson, E.L.; Drewnowski, A.; Darmon, N. Nutrient profiling can help identify foods of good nutritional quality for their price: A validation study with linear programming. J. Nutr. 2008, 138, 1107–1113. [Google Scholar] [PubMed]
- Darmon, N.; Darmon, M.; Maillot, M.; Drewnowski, A. A nutrient density standard for vegetables and fruits: Nutrients per calorie and nutrients per unit cost. J. Am. Diet. Assoc. 2005, 105, 1881–1887. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A. The contribution of milk and milk products to micronutrient density and affordability of the U.S. Diet. J. Am. Coll. Nutr. 2011, 30, 422S–428S. [Google Scholar] [CrossRef] [PubMed]
- Alexy, U.; Schwager, V.; Kersting, M. Diet quality and diet costs in german children and adolescents. Eur. J. Clin. Nutr. 2014, 68, 1175–1176. [Google Scholar] [CrossRef] [PubMed]
- Marty, L.; Dubois, C.; Gaubard, M.S.; Maidon, A.; Lesturgeon, A.; Gaigi, H.; Darmon, N. Higher nutritional quality at no additional cost among low-income households: Insights from food purchases of “positive deviants”. Am. J. Clin. Nutr. 2015, 102, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A.; Rehm, C.D. Vegetable cost metrics show that potatoes and beans provide most nutrients per penny. PLoS ONE 2013, 8, e63277. [Google Scholar] [CrossRef] [PubMed]
- Dror, D.K.; Allen, L.H. The importance of milk and other animal-source foods for children in low-income countries. Food Nutr. Bull. 2011, 32, 227–243. [Google Scholar] [PubMed]
- Eichler, K.; Wieser, S.; Ruthemann, I.; Brugger, U. Effects of micronutrient fortified milk and cereal food for infants and children: A systematic review. BMC Public Health 2012, 12, 506. [Google Scholar] [CrossRef] [PubMed]
- Yeomans, M.R.; Blundell, J.E.; Leshem, M. Palatability: Response to nutritional need or need-free stimulation of appetite? Br. J. Nutr. 2004, 92, S3–S14. [Google Scholar] [CrossRef] [PubMed]
- Poortvliet, P.C.; Bérubé-Parent, S.; Drapeau, V.; Lamarche, B.; Blundell, J.E.; Tremblay, A. Effects of a healthy meal course on spontaneous energy intake, satiety and palatability. Br. J. Nutr. 2007, 97, 584–590. [Google Scholar] [CrossRef] [PubMed]
- Daniels, S.R. The consequences of childhood overweight and obesity. Future Child. 2006, 16, 47–67. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Troesch, B.; Biesalski, H.K.; Bos, R.; Buskens, E.; Calder, P.C.; Saris, W.H.M.; Spieldenner, J.; Verkade, H.J.; Weber, P.; Eggersdorfer, M. Increased Intake of Foods with High Nutrient Density Can Help to Break the Intergenerational Cycle of Malnutrition and Obesity. Nutrients 2015, 7, 6016-6037. https://doi.org/10.3390/nu7075266
Troesch B, Biesalski HK, Bos R, Buskens E, Calder PC, Saris WHM, Spieldenner J, Verkade HJ, Weber P, Eggersdorfer M. Increased Intake of Foods with High Nutrient Density Can Help to Break the Intergenerational Cycle of Malnutrition and Obesity. Nutrients. 2015; 7(7):6016-6037. https://doi.org/10.3390/nu7075266
Chicago/Turabian StyleTroesch, Barbara, Hans K. Biesalski, Rolf Bos, Erik Buskens, Philip C. Calder, Wim H. M. Saris, Jörg Spieldenner, Henkjan J. Verkade, Peter Weber, and Manfred Eggersdorfer. 2015. "Increased Intake of Foods with High Nutrient Density Can Help to Break the Intergenerational Cycle of Malnutrition and Obesity" Nutrients 7, no. 7: 6016-6037. https://doi.org/10.3390/nu7075266
APA StyleTroesch, B., Biesalski, H. K., Bos, R., Buskens, E., Calder, P. C., Saris, W. H. M., Spieldenner, J., Verkade, H. J., Weber, P., & Eggersdorfer, M. (2015). Increased Intake of Foods with High Nutrient Density Can Help to Break the Intergenerational Cycle of Malnutrition and Obesity. Nutrients, 7(7), 6016-6037. https://doi.org/10.3390/nu7075266





