Postprandial Responses to Lipid and Carbohydrate Ingestion in Repeated Subcutaneous Adipose Tissue Biopsies in Healthy Adults
Abstract
:1. Introduction
2. Experimental Section
2.1. Participants
2.2. Experimental Design
2.3. Beverage Preparation (Table S1)
2.4. Serum Insulin and Adipokine Analysis
2.5. Metabolomics Analysis
2.6. RNA Extraction
2.7. Reverse Transcription and Real-Time-PCR
2.8. Statistical Analysis
3. Results
3.1. Participants’ Baseline Characteristics
| Total | Placebo | Carbohydrate | Lipid | p-value | |
|---|---|---|---|---|---|
| Male | 12 | 3 | 4 | 5 | |
| Female | 21 | 8 | 7 | 6 | |
| Age (year) | 24.5 ± 3.3 | 24.8 ± 3.1 | 23.9 ± 2.9 | 24.6 ± 3.9 | |
| Height (cm) | 171.2 ± 12.1 | 170.7 ± 10.3 | 168.3 ± 9.2 | 174.6 ± 15.6 | 0.802 |
| Weight (cm) | 71.0 ± 12.2 | 70.4 ± 11.5 | 67.8 ± 11.5 | 74.9 ± 12.9 | 0.476 |
| BMI (kg/m2) | 24.1 ± 2.7 | 24.0 ± 1.8 | 23.8 ± 3.0 | 24.6 ± 3.2 | 0.390 |
| Waist (cm) | 80.3 ± 7.9 | 79.7 ± 6.3 | 79.7 ± 10.2 | 81.4 ± 7.1 | 0.802 |
| Hip (cm) | 95.0 ± 7.5 | 95.8 ± 6.3 | 93.4 ± 7.3 | 95.6 ± 9.0 | 0.859 |
| WHR | 0.85 ± 0.06 | 0.83 ± 0.05 | 0.85 ± 0.08 | 0.85 ± 0.06 | 0.715 |
| Serum Adipokines | |||||
| Leptin (ng/mL) | 11.1 ± 10.6 | 9.8 ± 2.5 | 12.6 ± 4.5 | 12.4 ± 4.4 | 0.854 |
| Adiponectin (µg/mL) | 7.1 ± 3.5 | 8.4 ± 4.0 | 5.4 ± 2.6 | 7.6 ± 3.4 | 0.203 |
| TNF-α (pg/mL) | 2.7 ± 1.1 | 2.3 ± 1.3 | 3.0 ± 0.9 | 2.7 ± 1.2 | 0.514 |
3.2. Serum Analytes Respond Differently to Beverages Differing in Macronutrient Content
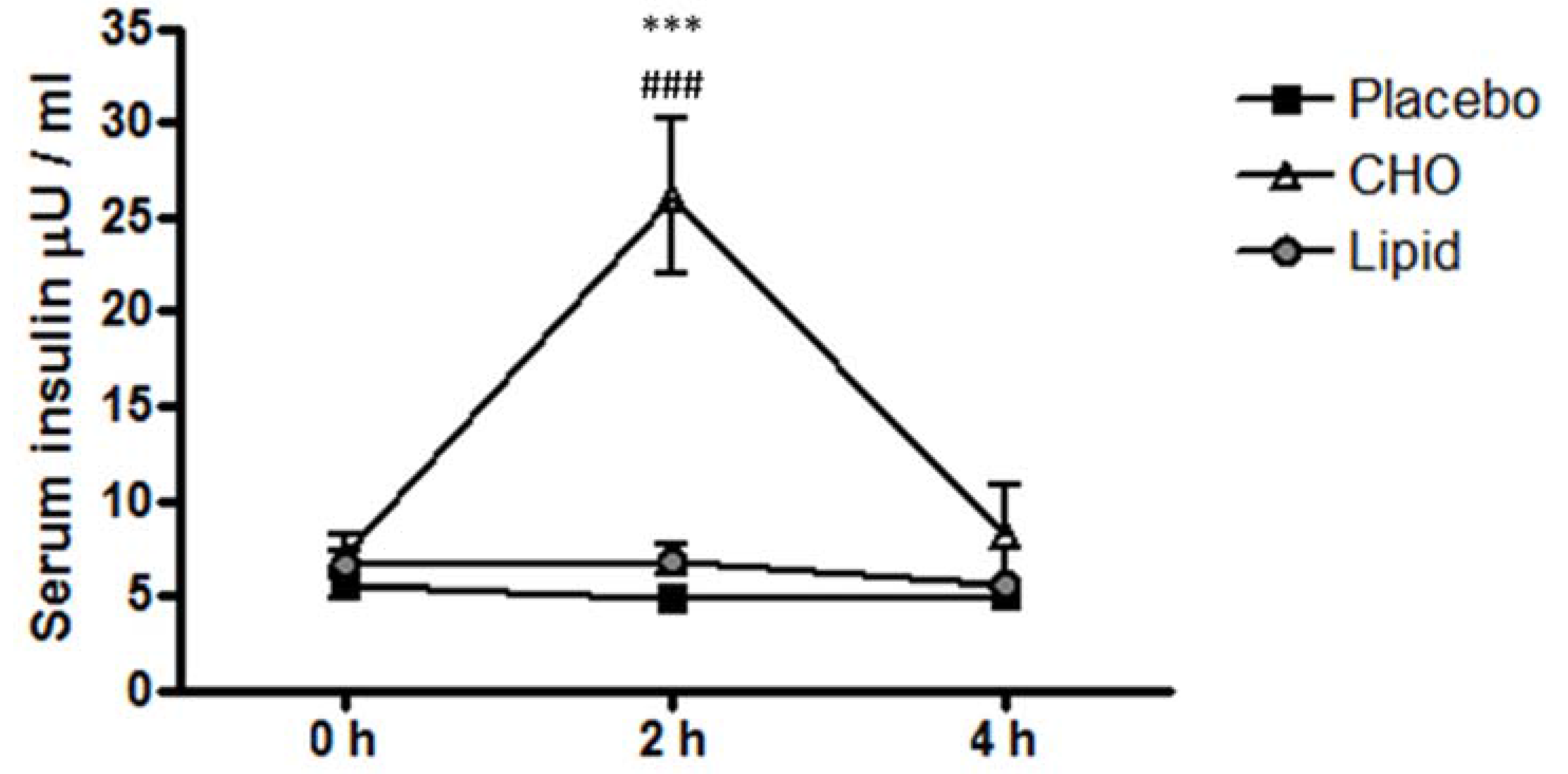
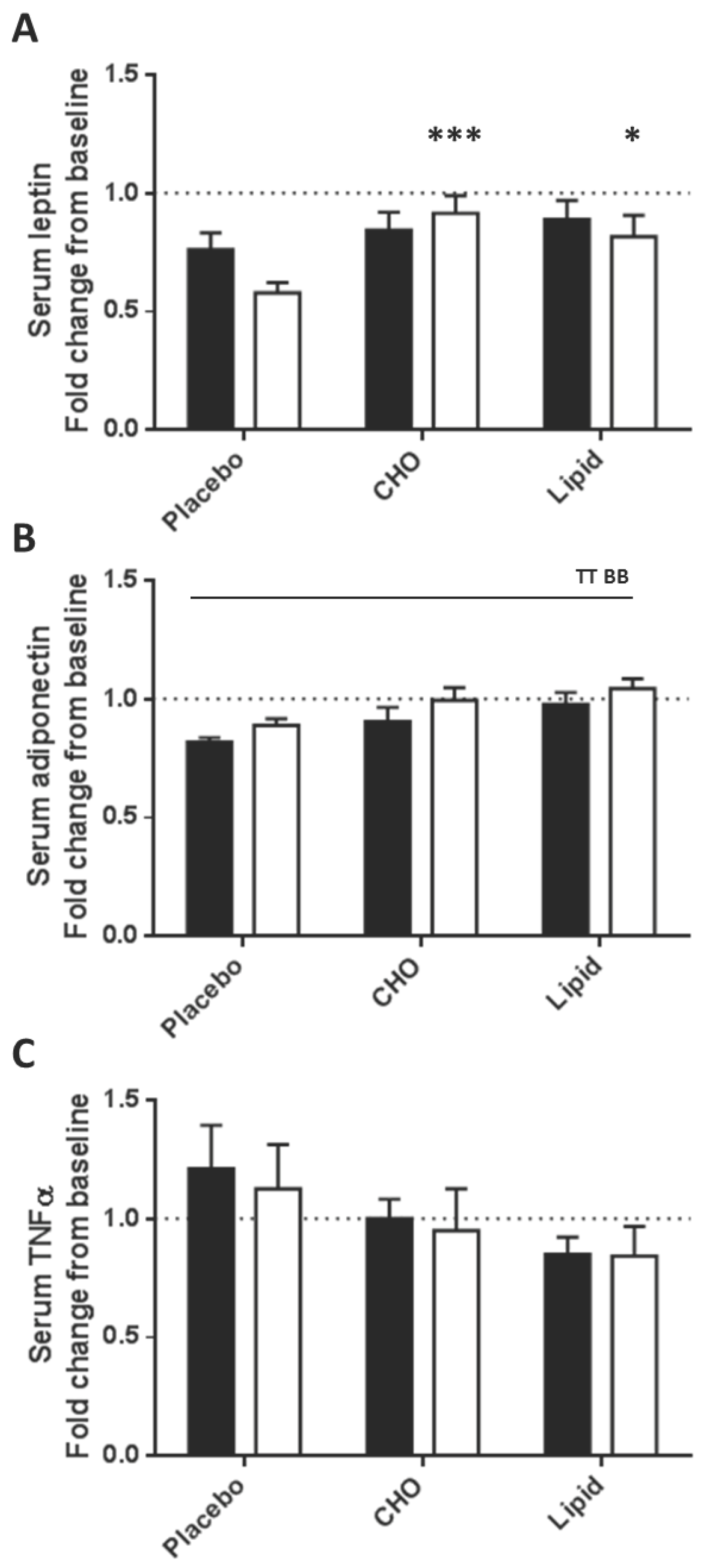

3.3. Subcutaneous Adipose Tissue Inflammatory Gene Expression Levels Were Equally Increased after Placebo, Carbohydrate and Lipid Beverages
| Placebo | Carbohydrate | Lipid | P1 | P2 | P3 | ||
|---|---|---|---|---|---|---|---|
| Adipokines | |||||||
| Leptin | 2 h | 0.69 ± 0.09 | 1.01 ± 0.12 | 1.03 ± 0.13 | 0.129 | 0.001 | 0.027 |
| 4 h | 0.57 ± 0.07 | 0.85 ± 0.10 | 0.86 ± 0.11 | ||||
| Adiponectin | 2 h | 0.86 ± 0.08 | 0.69 ± 0.08 | 0.88 ± 0.17 | 0.658 | 0.007 | 0.389 |
| 4 h | 0.84 ± 0.09 | 0.67 ± 0.07 | 0.90 ± 0.20 | ||||
| Resistin | 2 h | 1.07 ± 0.08 | 1.39 ± 0.20 | 1.17 ± 0.15 | 0.716 | 0.079 | 0.328 |
| 4 h | 0.90 ± 0.08 | 1.13 ± 0.21 | 1.04 ± 0.18 | ||||
| Chemerin | 2 h | 0.90 ± 0.07 | 1.09 ± 0.08 | 0.89 ± 0.09 | 0.424 | 0.105 | 0.291 |
| 4 h | 0.80 ± 0.06 | 0.92 ± 0.10 | 0.91 ± 0.12 | ||||
| Visfatin | 2 h | 3.59 ± 0.73 | 4.19 ± 1.10 | 4.74 ± 1.04 | 0.803 | <0.0001 | 0.718 |
| 4 h | 6.51 ± 1.29 | 5.41 ± 1.43 | 7.10 ± 1.45 | ||||
| PAI-1 | 2 h | 1.16 ± 0.15 | 1.08 ± 0.14 | 1.09 ± 0.20 | 0.830 | 0.064 | 0.978 |
| 4 h | 0.83 ± 0.11 | 0.96 ± 0.11 | 0.97 ± 0.13 | ||||
| Inflammatory Molecules | |||||||
| MCP1 | 2 h | 27.1 ± 6.6 | 34.7 ± 11.8 | 47.3 ± 12.7 | 0.886 | <0.0001 | 0.832 |
| 4 h | 80.8 ± 20.8 | 69.0 ± 21.6 | 78.8 ± 22.6 | ||||
| TNF-α | 2 h | 3.65 ± 1.04 | 3.31 ± 0.74 | 2.89 ± 0.47 | 0.565 | <0.0001 | 0.701 |
| 4 h | 2.05 ± 0.46 | 3.14 ± 0.54 | 2.35 ± 0.37 | ||||
| IL-6 | 2 h | 83.3 ± 28.9 | 241.9 ± 106.4 | 258.0 ± 101.3 | 0.414 | <0.0001 | 0.602 |
| 4 h | 259.7 ± 65.3 | 359.7 ± 138.4 | 241.7 ± 80.5 | ||||
| CD68 | 2 h | 0.87 ± 0.15 | 0.73 ± 0.10 | 0.73 ± 0.10 | 0.243 | 0.042 | 0.280 |
| 4 h | 1.06 ± 0.19 | 0.98 ± 0.18 | 0.63 ± 0.12 | ||||
3.4. Subcutaneous Adipose Tissue Adipokine Gene Expression Levels Differed in Response to Energy/Nutrient-Containing Beverages Compared with the Placebo
4. Discussion
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Frayn, K.N. Adipose tissue as a buffer for daily lipid flux. Diabetologia 2002, 45, 1201–1210. [Google Scholar] [CrossRef] [PubMed]
- Baum, J.I.; Layman, D.K.; Freund, G.G.; Rahn, K.A.; Nakamura, M.T.; Yudell, B.E. A reduced carbohydrate, increased protein diet stabilizes glycemic control and minimizes adipose tissue glucose disposal in rats. J. Nutr. 2006, 136, 1855–1861. [Google Scholar] [PubMed]
- Redinger, R.N. Fat storage and the biology of energy expenditure. Transl. Res. 2009, 154, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Feve, B. Adipogenesis: Cellular and molecular aspects. Best Pract. Res. Clin. Endocrinol. Metab. 2005, 19, 483–499. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.E.; Kehlenbrink, S.; Lee, H.; Hawkins, M.; Yudkin, J.S. Getting the message across: Mechanisms of physiological cross talk by adipose tissue. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E1210–E1229. [Google Scholar] [CrossRef] [PubMed]
- Wrann, C.D.; Laue, T.; Hubner, L.; Kuhlmann, S.; Jacobs, R.; Goudeva, L.; Nave, H. Short-term and long-term leptin exposure differentially affect human natural killer cell immune functions. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E108–E116. [Google Scholar] [CrossRef] [PubMed]
- Romon, M.; Lebel, P.; Fruchart, J.C.; Dallongeville, J. Postprandial leptin response to carbohydrate and fat meals in obese women. J. Am. Coll. Nutr. 2003, 22, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Romon, M.; Lebel, P.; Velly, C.; Marecaux, N.; Fruchart, J.C.; Dallongeville, J. Leptin response to carbohydrate or fat meal and association with subsequent satiety and energy intake. Am. J. Physiol. 1999, 277, E855–E861. [Google Scholar] [PubMed]
- Mitrou, P.; Lambadiari, V.; Maratou, E.; Boutati, E.; Komesidou, V.; Papakonstantinou, A.; Raptis, S.A.; Dimitriadis, G. Skeletal muscle insulin resistance in morbid obesity: The role of interleukin-6 and leptin. Exp. Clin. Endocrinol. Diabetes 2011, 119, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Annuzzi, G.; Bozzetto, L.; Patti, L.; Santangelo, C.; Giacco, R.; di Marino, L.; de Natale, C.; Masella, R.; Riccardi, G.; Rivellese, A.A. Type 2 diabetes mellitus is characterized by reduced postprandial adiponectin response: A possible link with diabetic postprandial dyslipidemia. Metabolism 2010, 59, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Meneses, M.E.; Camargo, A.; Perez-Martinez, P.; Delgado-Lista, J.; Cruz-Teno, C.; Jimenez-Gomez, Y.; Paniagua, J.A.; Gutierrez-Mariscal, F.M.; Tinahones, F.J.; Vidal-Puig, A.; et al. Postprandial inflammatory response in adipose tissue of patients with metabolic syndrome after the intake of different dietary models. Mol. Nutr. Food Res. 2011, 55, 1759–1770. [Google Scholar] [CrossRef] [PubMed]
- Pietraszek, A.; Gregersen, S.; Hermansen, K. Acute effects of dietary fat on inflammatory markers and gene expression in first-degree relatives of type 2 diabetes patients. Rev. Diabet Stud. 2011, 8, 477–489. [Google Scholar] [CrossRef] [PubMed]
- Friedmann-Bette, B.; Schwartz, F.R.; Eckhardt, H.; Billeter, R.; Bonaterra, G.; Kinscherf, R. Similar changes of gene expression in human skeletal muscle after resistance exercise and multiple fine needle biopsies. J. Appl. Physiol. 2012, 112, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Dordevic, A.L.; Bonham, M.P.; Larsen, A.E.; Gran, P.; Sinclair, A.J.; Jowett, J.B.; Konstatopoulos, N.; Cameron-Smith, D. Global Transcriptomic Response in Subcutaneous Abdominal Adipose to Saturated Versus Polyunsaturated Fatty Acid-Rich Meals in Men with Metabolic Syndrome, Unpublished work, 2015.
- Bergström, J. Percutaneous needle biopsy of skeletal muscle in physiological and clinical research. Scand. J. Clin. Lab. Investig. 1975, 35, 609–616. [Google Scholar] [CrossRef]
- Evans, W.; Phinney, S.; Ivy, J. Suction applied to a muscle biopsy maximises sample size. Med. Sci. Sport Exerc. 1982, 14, 101–102. [Google Scholar]
- Smart, K.F.; Aggio, R.B.M.; van Houtte, J.R.; Villas-Bôas, S.G. Analytical platform for metabolome analysis of microbial cells using methyl chloroformate derivatization followed by gas chromatography-mass spectrometry. Nat. Protoc. 2010, 5, 1709–1729. [Google Scholar] [CrossRef] [PubMed]
- Aggio, R.B.M.; Ruggiero, K.; Villas-Bôas, S.G. Pathway activity profiling (PAPi): From the metabolite profile to the metabolic pathway activity. Bioinformatics 2010, 26, 2969–2976. [Google Scholar] [CrossRef] [PubMed]
- Laclaustra, M.; Corella, D.; Ordovas, J.M. Metabolic syndrome pathophysiology: The role of adipose tissue. Nutr. Metab. Cardiovasc. Dis. 2007, 17, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Peairs, A.D.; Rankin, J.W.; Lee, Y.W. Effects of acute ingestion of different fats on oxidative stress and inflammation in overweight and obese adults. Nutr. J. 2011, 10, 122. [Google Scholar] [CrossRef] [PubMed]
- Beisswenger, P.J.; Brown, W.V.; Ceriello, A.; Le, N.A.; Goldberg, R.B.; Cooke, J.P.; Robbins, D.C.; Sarwat, S.; Yuan, H.; Jones, C.A. Meal-induced increases in c-reactive protein, interleukin-6 and tumour necrosis factor alpha are attenuated by prandial + basal insulin in patients with type 2 diabetes. Diabet Med. 2011, 28, 1088–1095. [Google Scholar] [CrossRef] [PubMed]
- Nappo, F.; Esposito, K.; Cioffi, M.; Giugliano, G.; Molinari, A.M.; Paolisso, G.; Marfella, R.; Giugliano, D. Postprandial endothelial activation in healthy subjects and in type 2 diabetic patients: Role of fat and carbohydrate meals. J. Am. Coll Cardiol. 2002, 39, 1145–1150. [Google Scholar] [CrossRef]
- Masson, C.J.; Mensink, R.P. Exchanging saturated fatty acids for (n-6) polyunsaturated fatty acids in a mixed meal may decrease postprandial lipemia and markers of inflammation and endothelial activity in overweight men. J. Nutr. 2011, 141, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Kruse, M.; von Loeffelholz, C.; Hoffmann, D.; Pohlmann, A.; Seltmann, A.C.; Osterhoff, M.; Hornemann, S.; Pivovarova, O.; Rohn, S.; Jahreis, G.; et al. Dietary rapeseed/canola oil supplementation reduces serum lipids and liver enzymes and alters postprandial inflammatory responses in adipose tissue compared to olive oil supplementation in obese men. Mol. Nutr. Food Res. 2015, 59, 507–519. [Google Scholar] [CrossRef] [PubMed]
- Bala, M.; Martin, J.; Kopp, A.; Hanses, F.; Buechler, C.; Schaffler, A. In vivo suppression of visfatin by oral glucose uptake: Evidence for a novel incretin-like effect by glucagon-like peptide-1 (GLP-1). J. Clin. Endocrinol. Metab. 2011, 96, 2493–2501. [Google Scholar] [CrossRef] [PubMed]
- Westerink, J.; Hajer, G.R.; Kranendonk, M.E.; Schipper, H.S.; Monajemi, H.; Kalkhoven, E.; Visseren, F.L. An oral mixed fat load is followed by a modest anti-inflammatory adipocytokine response in overweight patients with metabolic syndrome. Lipids 2014, 49, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Krug, S.; Kastenmuller, G.; Stuckler, F.; Rist, M.J.; Skurk, T.; Sailer, M.; Raffler, J.; Romisch-Margl, W.; Adamski, J.; Prehn, C.; et al. The dynamic range of the human metabolome revealed by challenges. FASEB J. 2012, 26, 2607–2619. [Google Scholar] [CrossRef] [PubMed]
- Boden, G.; Chen, X.; Mozzoli, M.; Ryan, I. Effect of fasting on serum leptin in normal human subjects. J. Clin. Endocrinol. Metab. 1996, 81, 3419–3423. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dordevic, A.L.; Pendergast, F.J.; Morgan, H.; Villas-Boas, S.; Caldow, M.K.; Larsen, A.E.; Sinclair, A.J.; Cameron-Smith, D. Postprandial Responses to Lipid and Carbohydrate Ingestion in Repeated Subcutaneous Adipose Tissue Biopsies in Healthy Adults. Nutrients 2015, 7, 5347-5361. https://doi.org/10.3390/nu7075224
Dordevic AL, Pendergast FJ, Morgan H, Villas-Boas S, Caldow MK, Larsen AE, Sinclair AJ, Cameron-Smith D. Postprandial Responses to Lipid and Carbohydrate Ingestion in Repeated Subcutaneous Adipose Tissue Biopsies in Healthy Adults. Nutrients. 2015; 7(7):5347-5361. https://doi.org/10.3390/nu7075224
Chicago/Turabian StyleDordevic, Aimee L., Felicity J. Pendergast, Han Morgan, Silas Villas-Boas, Marissa K. Caldow, Amy E. Larsen, Andrew J. Sinclair, and David Cameron-Smith. 2015. "Postprandial Responses to Lipid and Carbohydrate Ingestion in Repeated Subcutaneous Adipose Tissue Biopsies in Healthy Adults" Nutrients 7, no. 7: 5347-5361. https://doi.org/10.3390/nu7075224
APA StyleDordevic, A. L., Pendergast, F. J., Morgan, H., Villas-Boas, S., Caldow, M. K., Larsen, A. E., Sinclair, A. J., & Cameron-Smith, D. (2015). Postprandial Responses to Lipid and Carbohydrate Ingestion in Repeated Subcutaneous Adipose Tissue Biopsies in Healthy Adults. Nutrients, 7(7), 5347-5361. https://doi.org/10.3390/nu7075224







