1. Introduction
Protein is one of the main nutrients involved in all aspects of
in vivo iron metabolism including iron absorption, transport, hematopoiesis and storage [
1,
2]. Thus, adequate intake of not only iron, but also protein, is important in the maintenance of normal iron metabolism. Dietary protein differs in the composition of amino acids and the amino acid score by the food origin. Moreover, peptides, which are the digestive form of proteins, are thought to modulate biological functions, and have recently been shown to have various physiological functions. In assessing prevention of iron deficiency anemia (IDA) by diet, it is therefore necessary to confirm the influence and biological use of dietary protein, which coexists with iron, on iron absorption.
Of the three major nutrients (carbohydrate, protein, and lipid), protein has been reported to have the greatest influence on iron absorption [
3,
4,
5,
6]. For example, Cook
et al. reported that soybean protein inhibited iron absorption more than egg white protein by a factor of about 5 [
7]. Although the whole egg is a food that contains a large amount of iron [
8], it is often cited as a food that inhibits iron absorption [
9,
10,
11]. The majority of iron in eggs is found in the yolk. Although it has been reported that the absorptivity of iron from egg origin is low [
12,
13], iron absorption was shown to increase and contribute to a delay in the decrease in hemoglobin concentration in an iron-deficient state where iron demands were high [
14]. The mineral absorption promoting effect of casein phosphopeptide (CPP) of milk origin is well known from the past researches [
15,
16,
17]. Although a number of studies have examined the influence of dietary protein on iron absorption, there are few reports on the contribution of dietary protein in the iron deficient state that is accompanied by abnormal iron metabolism [
3,
4,
5,
6,
7,
8,
9,
10,
11,
12,
13,
14,
15,
16,
17].
We previously examined the effects of various dietary protein sources consumed simultaneously with dietary iron on recovery from IDA. Our previous research showed the possibility that egg white contributes to prompt recovery from IDA compared to soybeans. Furthermore, we showed that the protein form resulted in an increase in serum iron and maturation of red blood cells compared to the peptide form, because the protein form maintained its iron-reducing characteristics in the digestive tract, compared to the peptide form [
18]. In the present study, therefore, we investigated the usefulness of the egg structural protein for recovery from IDA.
2. Experimental Section
2.1. Animal Experimental Protocol
This experimental study was approved by the ethics committee of Kyoto Prefectural University, and performed in accordance with the Guidelines for Animal Experimentation at Kyoto Prefectural University. Thirty-one 4-week-old female Sprague-Dawley rats were used in this study (Japan SLC, Inc., Hamamatsu, Japan). The rats were individually housed in stainless steel cages at a controlled temperature of 22–24 °C, a relative humidity of 40%–60%, and a light cycle of 12 h with free access to distilled water (the iron content of the distilled water was previously measured). Body weight and food intake were recorded at the same time everyday.
The 31 rats were divided into two groups on the basis of body weight. The first group (C group,
n = 6, weighing 106–112 g) was fed a control diet for 42 days. The second group (base LI group,
n = 25, weighing 94–121 g) was fed a low-iron diet for 21 days to induce IDA. IDA rats were then divided into four subgroups based on weight and hemoglobin concentration such that the mean values of these parameters for each subgroup were the same. Each subgroup was fed either an egg white diet (LI-W group,
n = 6, weighing 155–195 g), an ovalbumin diet (LI-A group,
n = 7, weighing 153–177 g), an egg-yolk supplemented diet (LI-Y group,
n = 6, weighing 158–184 g), or the control diet (LI-C group,
n = 6, weighing 163–182 g) for another 21 days. The compositions of the diets used in the experiments are shown in
Table 1. All diets were prepared according to the AIN-76 formulation with one modification (the addition of choline chloride). The low-iron diet contained 0.4 mg Fe/100 g without any ferrous citrate in the mineral mixture. The amount of protein and lipids within all experimental diets was adjusted to be equal to that of the control diet. During the pair- feeding period, the LI-W, LI-A and LI-Y groups were provided with the same amount of diet that was freely provided to the LI-C group, on the following day.
Table 1.
Composition of experimental diet.
Table 1.
Composition of experimental diet.
| | Control Diet | Low Iron Diet | Egg White Diet | Ovalbumin Diet | Egg York Diet |
|---|
| | (g/kg) |
| Casein a | 200 | 200 | - | - | 171 |
| Egg White powder b | - | - | 214 | - | - |
| Ovalbumin c | - | - | - | 211 | - |
| Egg York powder d | - | - | - | - | 81 |
| α-starch | 457 | 457 | 447 | 448 | 455 |
| Sucrose | 228 | 228 | 224 | 225 | 228 |
| Mixed oil e | 50 | 50 | 50 | 50 | |
| Vitamin mixture f | | | 10 | | |
| Mineral mixture g | | | 35 | | |
| Cellose | | | 20 | | |
| | (mg/kg) |
| Iron (III) Citrate | 204 | - | 205 | 196 | 163 |
| Iron content | 39.6 | 4.4 | 40.0 | 39.9 | 40.0 |
Blood was drawn from the tail vein of all of the animals every 4 days during the experimental period. At the end of each study period, the rats were euthanized under ether anesthesia during the early phase of the light cycle in a non-fasted state, and blood samples drawn from the inferior vena cava were collected in tubes with heparin. Samples from the liver and small intestinal mucosa (upper side, 1/4th) were also collected.
2.2. Blood Constituent Analysis
The blood hemoglobin concentration was measured using Hemoglobin B test Wako (Wako Pure Chemical Industries, Osaka, Japan). The hematocrit level was measured after centrifugation of the blood at 12,000 rpm for 5 min at 4 °C. Red blood cell (RBC) counts were determined using a Thoma hemacytometer following a 1:200 dilution with Hayem’s solution in a pipette. Mean cell volume (MCV), mean corpuscular hemoglobin (MCH) and mean cell hemoglobin concentration (MCHC) were calculated as follows:
Serum iron and unsaturated iron binding capacity (UIBC) were measured using Detaminer Fe and UIBC (Kyowa Medix Co., Ltd., Tokyo, Japan) with an automatic biochemical analyzer (CL-8000; Shimadzu Corp., Kyoto, Japan). Total iron binding capacity and serum transferrin saturation were calculated as follows:
2.3. Estimation of Gene Expression
Total RNA was isolated from the homogenized mucosa and liver samples using the Total RNA Isolation mini kit (Agilent Technologies, Inc., Santa Clara, CA, USA), and converted to cDNA using a reverse transcriptase enzyme ReverTra Ace (Toyobo Co., Ltd., Osaka, Japan) according to the manufacturer’s instructions. Each target DNA fragment was amplified using the respective TaqMan gene expression assay kits and a real-time polymerase chain reaction (PCR) system using cDNA as a template. Real-time PCR for gene expression analysis was performed using DNA Engine Opticon and Opticon Monitor software (Bio-Rad Laboratories, Inc., Hercules, CA, USA). TaqMan primer pairs/probes for gene analysis were obtained using a TaqMan Gene Expression Assay (Applied Biosystems, Inc., Carlsbad, CA, USA). Assay IDs were Rn00565927_m1, Rn00591187_m1 and Rn00667869_m1 for DMT1, Ferroportin and β-actin, respectively. Reactions were performed with 10 μL of Premix EX Taq (Takara Bio, Inc., Ohtsu, Japan), 1 μL of the primer pairs/probes sets and 3 μL of cDNA in a final volume of 20 μL. After heating the test sample at 96 °C for 10 s, 50 PCR cycles were performed as follows: 95 °C for 7 s, 60 °C for 30 s, and 72 °C for 20 s. The cycle thresholds of the genes of interest were compared with the housekeeping gene β-actin to determine relative changes in expression.
2.4. Iron Content of Hepatic Tissue
Liver samples were perfused with saline, and treated by the wet ash method using a microwave extraction system (Ethos; Milestone Srl., Sorisole, Italy). The ash was suspended in dilute hydrochloric acid solution after evaporation, and left to dry. Iron concentrations were measured by polarizing Zeeman-effect atomic absorption spectrometry (Z-6100; Hitachi, Ltd., Tokyo, Japan) after suitable dilution. We determined that the coefficient of variation was 0.04. Iron concentrations were expressed on a wet-weight basis.
2.5. Statistical Analysis
Data were presented as means ± standard error (SEM). Before assessing the different variables, we carried out a Bartlett test to check the normal distribution of the variables. Data that fit the normal distribution were compared by 1–way analysis of variance (ANOVA) followed by the Tukey-Kramer test (
Table 2,
Figure 1 and
Figure 2), or Student’s t test (
Table 2). The level of significance was set at
p < 0.05.
Table 2.
Body weight gain, food intake and blood parameters on the day 21 and day 42 after the start of study.
Table 2.
Body weight gain, food intake and blood parameters on the day 21 and day 42 after the start of study.
| A. day 21 | | | |
| | Base LI | C | Student’s t-test
p value |
| Body weight gain (g/day) | 3.0 ± 0.1 | 3.4 ± 0.2 | 0.138 |
| Food intake (g/day) | 11.7 ± 0.1 a | 12.7 ± 0.4 b | 0.034 |
| Hematocrit level (%) | 33.0 ± 0.7 a | 47.1 ± 1.2 b | <0.001 |
| Hemogrobin concentration (g/dL) | 9.7 ± 0.3 a | 15.4 ±0.7 b | <0.001 |
| B. day 42 | | | | | | |
| | LI-W | LI-A | LI-Y | LI-C | C | ANOVA
p value |
| Body weight gain (g/day) | 1.4 ± 0.3 | 1.5 ± 0.1 | 1.3 ± 0.1 | 1.5 ± 0.2 | 1.7 ± 0.1 | 0.196 |
| Food intake (g/day) | 10.9 ± 0.0 a | 10.9 ± 0.0 a | 10.9 ± 0.0 a | 10.9 ± 0.0 a | 11.9 ± 0.5 b | 0.006 |
| Hematocrit level (%) | 49.9 ± 1.5 a | 49.3 ± 1.0 a | 44.4 ± 3.2 b | 48.5 ± 2.8 a | 50.7 ± 1.0 a | 0.002 |
| Hemogrobin concentration (g/dL) | 18.0 ± 0.6 a | 17.8 ± 0.6 a | 15.0 ± 0.6 b | 17.4 ± 1.0 a | 18.2 ± 0.5 a | <0.001 |
| Red blood cell counts (×106/µL) | 14.8 ± 1.1 | 13.0 ± 1.5 | 13.5 ± 1.3 | 14.1 ± 1.8 | 15.8 ± 2.2 | 0.072 |
| Mean cell volume * (fL) | 12.0 ± 0.4 ab | 13.8 ± 1.8 a | 11.2 ± 1.1 b | 12.4 ± 0.7 ab | 11.7 ± 2.0 ab | 0.013 |
| Mean corpuscular hemoglobin ** (pg) | 33.2 ± 1.0 a | 38.4 ± 4.0 b | 33.3 ± 4.5 a | 34.4 ± 2.0 a | 31.6 ± 4.7 a | 0.043 |
| Mean cell hemogrobin concentration *** (%) | 36.0 ± 1.6 ab | 39.0 ± 1.1 ab | 33.7 ± 1.4 a | 31.4 ± 0.3 ab | 36.0 ± 0.6 b | 0.022 |
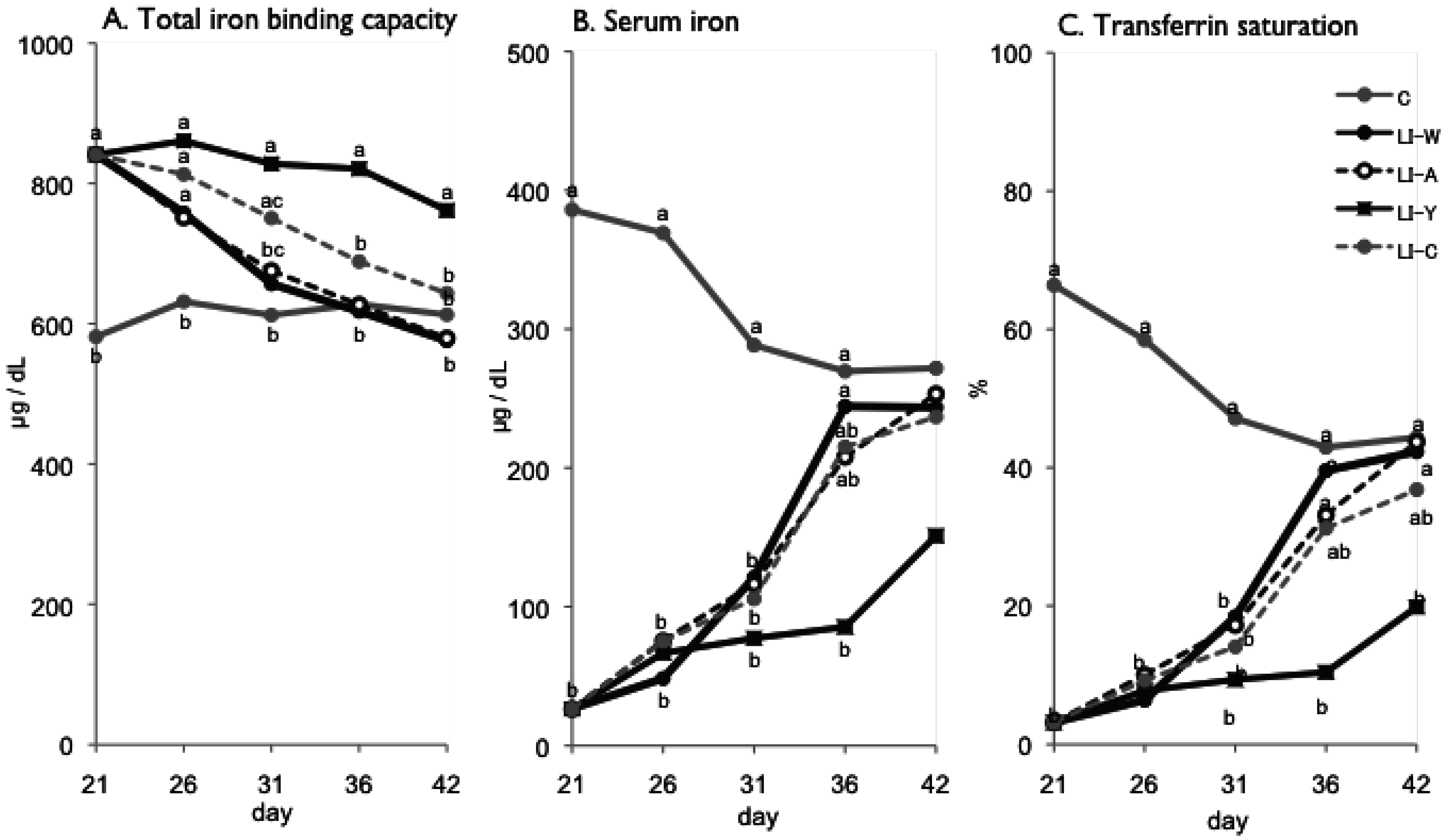
Figure 1.
Total iron binding capacity (A); serum iron (B); transferrin saturation (C) on day 21, 26, 31, 36 and 42 after start of experimental period. Iron deficiency anemia rats, fed low iron diet for 21 days, divided four subgroups, was fed either an egg white diet (LI-W, n = 6), an ovalbumin diet (LI-A, n = 7), an egg york diet (LI-Y, n = 6) or a control diet (LI-C, n = 6) and normal rats fed the control diet (C, n = 6) for 42 days. Values were represented as mean. Values with an unlike letter were significant: p < 0.05.
Figure 1.
Total iron binding capacity (A); serum iron (B); transferrin saturation (C) on day 21, 26, 31, 36 and 42 after start of experimental period. Iron deficiency anemia rats, fed low iron diet for 21 days, divided four subgroups, was fed either an egg white diet (LI-W, n = 6), an ovalbumin diet (LI-A, n = 7), an egg york diet (LI-Y, n = 6) or a control diet (LI-C, n = 6) and normal rats fed the control diet (C, n = 6) for 42 days. Values were represented as mean. Values with an unlike letter were significant: p < 0.05.
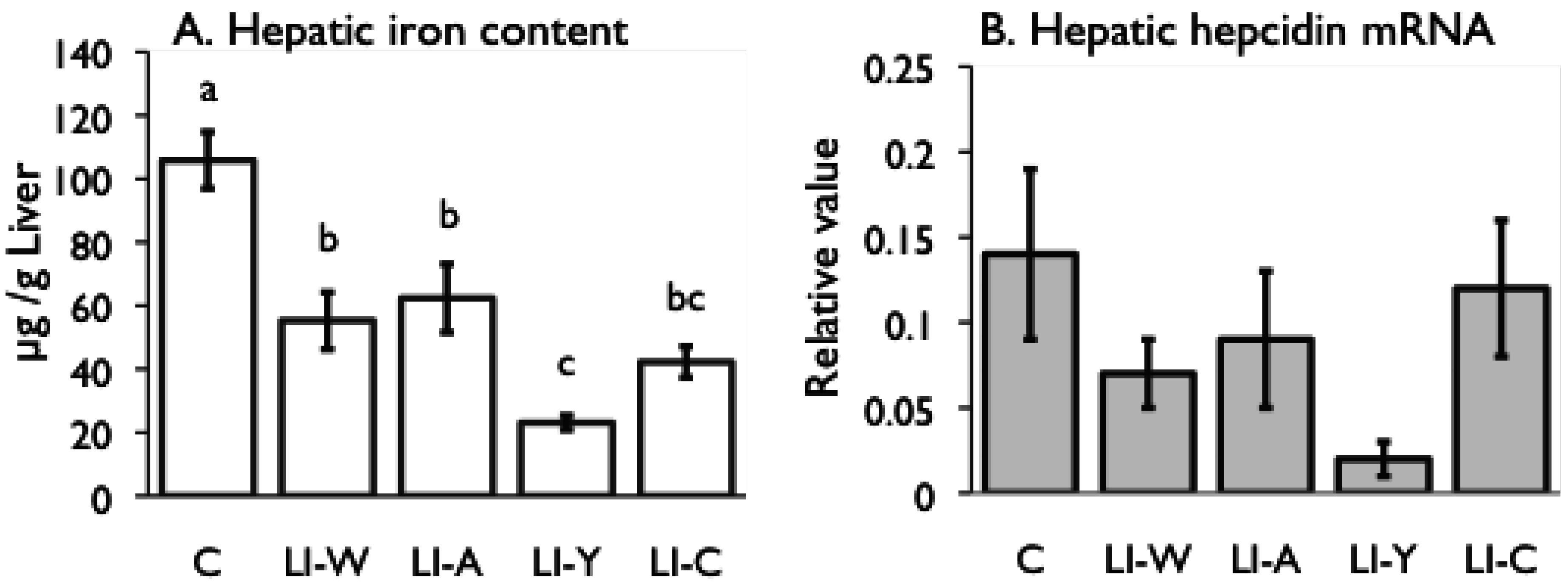
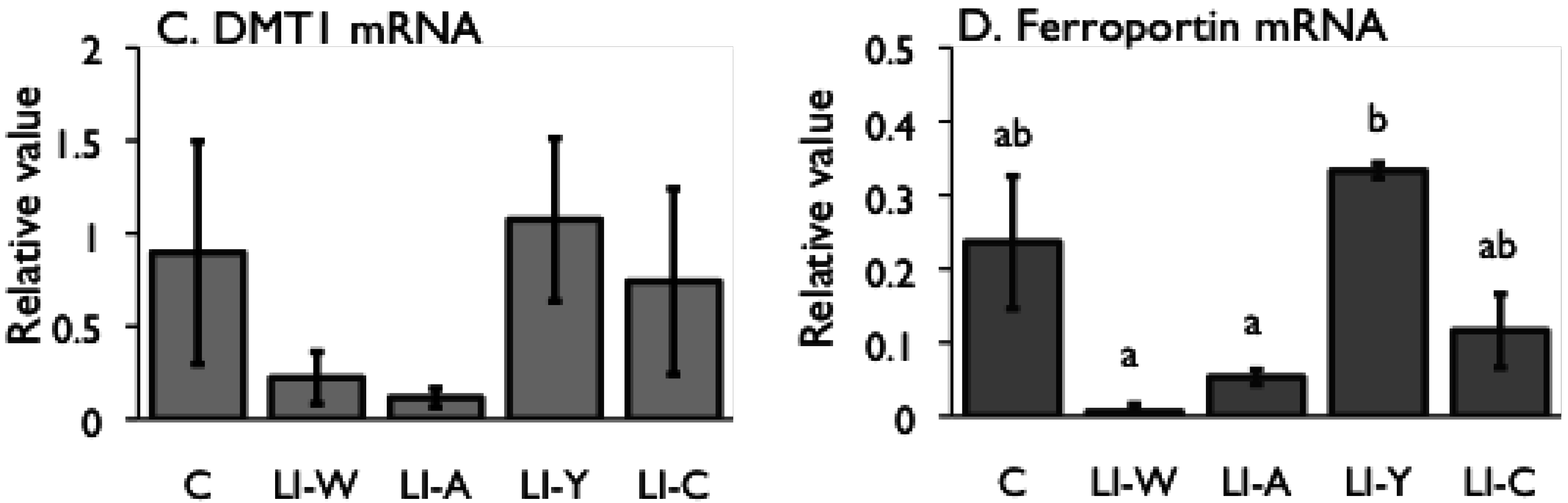
Figure 2.
Iron content (A) and hepcidin mRNA expression (B) in the liver, and iron transporter DMT1 (C) and Ferroportin (D) mRNA expression in small intestines on day 42 after start of study. Iron deficiency anemia rats, fed low iron diet for 21 days, divided four subgroups, was fed either an egg white diet (LI-W, n = 6), an ovalbumin diet (LI-A, n = 7), an egg york diet (LI-Y, n = 6) or a control diet (LI-C, n = 6) and normal rats fed the control diet (C, n = 6) for 42 days. The cycle thresholds of the genes of interest were compared with the housekeeping gene β-actin to determine relative changes in expression. Values were represented as means ± SEM. Values with unlike letter were significant: p < 0.05.
Figure 2.
Iron content (A) and hepcidin mRNA expression (B) in the liver, and iron transporter DMT1 (C) and Ferroportin (D) mRNA expression in small intestines on day 42 after start of study. Iron deficiency anemia rats, fed low iron diet for 21 days, divided four subgroups, was fed either an egg white diet (LI-W, n = 6), an ovalbumin diet (LI-A, n = 7), an egg york diet (LI-Y, n = 6) or a control diet (LI-C, n = 6) and normal rats fed the control diet (C, n = 6) for 42 days. The cycle thresholds of the genes of interest were compared with the housekeeping gene β-actin to determine relative changes in expression. Values were represented as means ± SEM. Values with unlike letter were significant: p < 0.05.
4. Discussion
In the present study, we examined the usefulness of egg constitutive proteins on recovery of IDA. Our results provided new finding that egg yolk protein delayed recovery of IDA while ovalbumin was useful in recovery of IDA, and that the bioavailability of dietary iron varies depending on dietary protein source.
Although the iron content of all diets was equivalent, the transitions of total iron binding capacity, transferring saturation level, and serum iron in the LI-Y group of IDA rats, which were fed the diet containing egg yolk, showed delayed recovery compared with the other groups, and resulted in no recovery at the end of the experimental period. Moreover, the blood properties and hepatic iron content of the LI-Y group were lower than those of the other groups on day 42. These data suggest that egg yolk resulted in delayed recovery from IDA. On the other hand, the mRNA expression of hepatic hepcidin, which regulates iron absorption in the gut [
19,
20], was decreased in the LI-Y group compared to the other diet groups, and it correlated with hepatic iron content. The mRNA expression of ferroportin and DMT1, which are transporters of iron absorption in the small intestine [
21,
22], were up-regulated in the LI-Y group compared to the C group, suggesting that in this group, iron storage was insufficient and iron absorption was promoted by homeostasis. Nevertheless, expression of the iron transporters was up-regulated. One reason for the lack of recovery from IDA in the Li-Y group may be that the absolute quantity of dietary iron transported in the small intestines was low. A previous study reported that intake of egg yolk protein decreased the apparent absorption of iron, calcium and magnesium compared with casein and soy protein in normal rats [
23]. Most of the iron in egg yolk is combined with phosvitin of phosphate protein. Phosvitin is known to have a very high binding capacity for divalent metals, especially iron [
24]. It was observed that the amount of insoluble iron in the small intestines of rats fed the diet containing phosvitin was higher than that in rats fed diets without phosvitin [
25]. In the present study, feeding the diet containing egg yolk may have delayed recovery from IDA because the iron in the egg yolk iron and/or other dietary iron may have strongly combined with phosvitin, and formed an insoluble iron complex in the small intestine. Since phosvitin is a resistant protein [
26], it is possible that the insoluble iron complex was excreted from the body without being used
in vivo. These findings suggest, therefore, that iron from eggs is not readily used
in vivo, and ingredients from egg yolk reduce the bioavailability of dietary iron. In cases of IDA, the choice of egg yolk as the source of protein and iron should be avoided.
Conversely, at the end of the experimental period, the blood parameters of the LI-W group, which was fed egg white, the LI-A group, which was fed ovalbumin, and the LI-C group, which was fed casein, were not significantly different from those of the control group, suggesting that IDA improved in these groups. The hepatic iron content of the LI-W, LI-A, and LI-C groups was significantly lower than the C group, however, and iron storage was not recovered in these groups despite receiving the standard amount of dietary iron given to IDA rats for 3 weeks. The total iron binding capacity of the LI-W and LI-A group was lower than that of the LI-C, but was not significantly different from the C group. In addition, the hepatic iron content of the LI-W and LI-A groups was higher than that of the LI-C group, but there were no significant differences among the three groups. These findings show that IDA of the LI-W and LI-A groups, which were fed egg white protein, was promptly recovered due to inhibition of transferrin production at an early stage compared with the LI-C group, which was fed casein protein. We conclude, therefore, that the egg white protein contributed to an improvement in the iron deficient state compared to casein protein. These results are in agreement with our previous reports [
18]. If iron absorption in the LI-C group, which was fed casein protein, was promoted by CPP, this raises the possibility that egg white protein has the absorption promoting effect on iron that exceeds CPP. On the other hand, a previous study reported that the specific amino acid that accelerated iron absorption exists in animal protein, such as red meat [
27]. These results suggest that the mechanism for promotion of recovery from IDA by egg white protein differs from the rise in iron absorption caused by red meat, since the existence of this amino acid in egg has not been reported even though eggs are from animal sources. Moreover, we assume that the component of egg white protein active in recovery of IDA was ovalbumin, because there were no significant differences in changes in blood parameters and in hepatic iron content between the LI-W and LI-A groups. Albumin possesses a large number of negative carboxylate sites (−CO
2−) on the surface of the molecule, many clustered in groups of three or more [
28]. These sites are suitable for binding iron (III) [
29]. Indeed, albumin has been demonstrated to be a sufficiently powerful ligand for binding iron (III) even when transferrin is not fully saturated [
30]. Therefore, the combination of dietary iron and albumin in the lumen may form a soluble complex that is advantageous for iron absorption.
Despite the fact that the amount of stored iron in the LI-W, LI-A, and LI-C groups was not restored, the ferroportin and DMT1 mRNA levels in the small intestine significantly decreased in these groups compared with the C group. Accordingly, iron absorption by the transporters was suppressed in the LI-W, LI-A, and LI-C groups, even though they were in an iron-deficient state. It is known that the expression of the iron transporter ferroportin is down-regulated by secretion of hepcidin [
31]; however, there was no correlation between hepatic iron content and the level of hepatic hepcidin and ferroportin mRNA expression in this study. Thus, these findings raise the possibility of the presence of another mechanism for regulation of ferroportin expression in the absence of secretion of hepcidin.
One of the limitations of the present study is that we did not directly measure the rate of iron absorption as the trend of dietary iron in the intestine is one of clues to comprehend the usefulness of the dietary iron for the recovery of IDA. In order to elucidate the effects of egg protein on the promotion of iron absorption, further studies should be conducted to investigate iron balance. Moreover, our findings suggest that phosvitin, contained within egg yolk protein, likely reduces the bioavailability of dietary iron. Further research is required to investigate the role of egg yolk phosvitin in the recovery of IDA and its possible usefulness in iron removal therapy. Although the results obtained in this study have some implications for public health, it is necessary to keep in mind that the iron bioavailability varies with different cooking processes, especially of egg yolk.