A Moderate Low-Carbohydrate Low-Calorie Diet Improves Lipid Profile, Insulin Sensitivity and Adiponectin Expression in Rats
Abstract
:1. Introduction
2. Experimental Section
2.1. Animals and Diets
| Components | AL Group | CR1 Group a | CR2 Group b | |||
|---|---|---|---|---|---|---|
| g/kg Diet | % Energy | g/0.8 kg Diet | % Energy | g/0.6 kg Diet | % Energy | |
| Casein | 200.0 | 20.2 | 200.0 | 25.2 | 200.0 | 33.8 |
| Vitamin mix ** | 10.0 | - | 10.0 | - | 10.0 | - |
| Mineral mix ** | 35.0 | - | 35.0 | - | 35.0 | - |
| Sucrose | 50.0 | 5.0 | 50.0 | 6.3 | 50.0 | 8.4 |
| Corn starch | 585.0 | 58.9 | 385.0 | 48.6 | 185.0 | 31.2 |
| Fiber | 50.0 | - | 50.0 | - | 50.0 | - |
| Lipid | 70.0 | 15.9 | 70.0 | 19.9 | 70.0 | 26.6 |
| Total amount (g) | 1000.0 | 100.0 | 800.0 | 100.0 | 600.0 | 100.0 |
| Total Calorie/kg diet (Kcal) | 3970.0 | 3170.0 | 2370.0 | |||
| Energy ratio (%) | ||||||
| Protein/Total | 20 | 25 | 34 | |||
| Carbohydrate/Total | 64 | 55 | 40 | |||
| Lipid/Total | 16 | 20 | 26 | |||
2.2. Biochemical Measurements
2.3. Semiquantitative Reverse Transcriptase Polymerase Chain Reaction
2.4. Statistical Analysis
3. Results
3.1. Calorie Intake and Body Weight

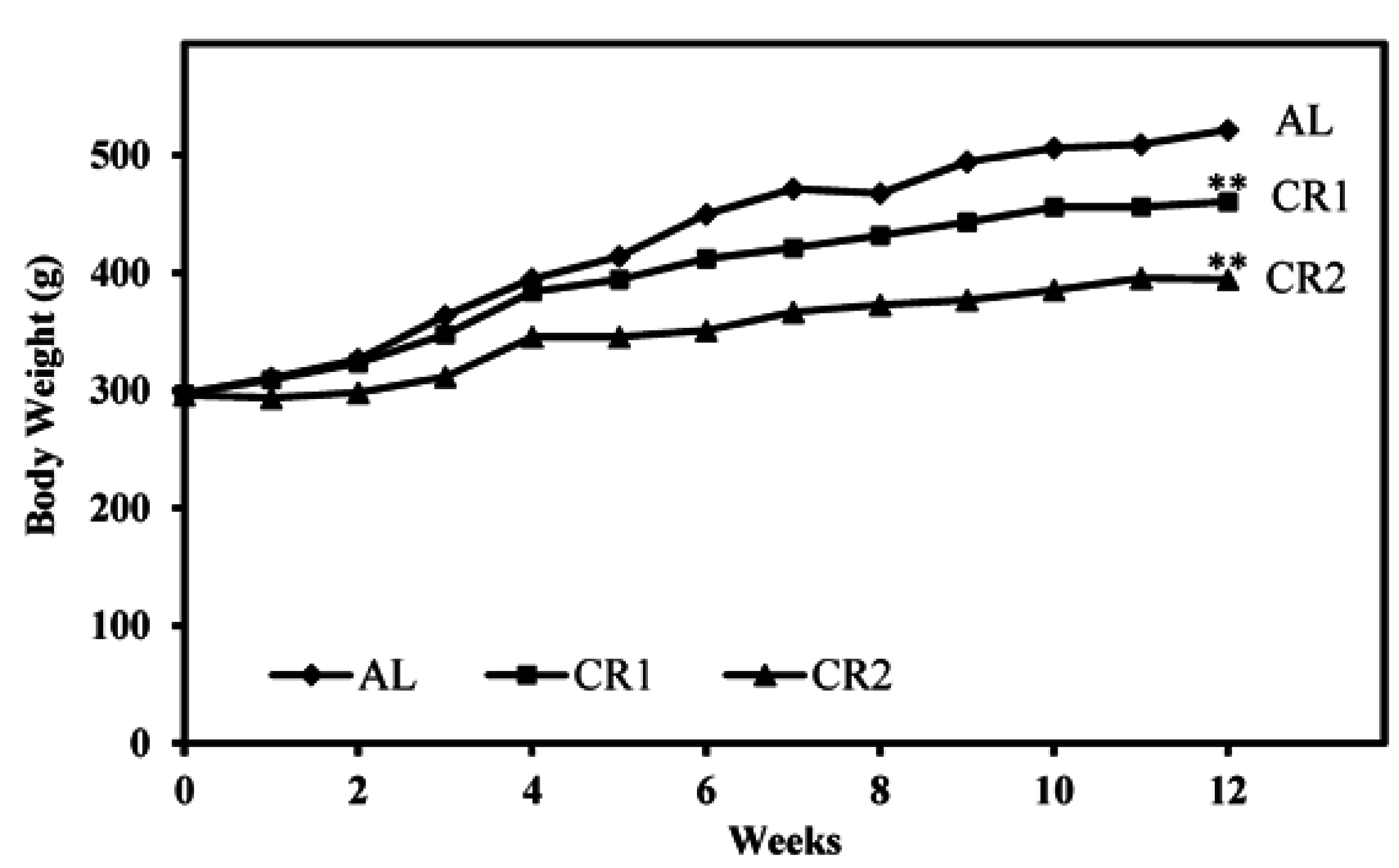
3.2. Changes in Metabolic Parameters
| Group | mmol/L | |||||||
|---|---|---|---|---|---|---|---|---|
| Triglyceride | Total Cholesterol | HDL-C | LDL-C | Total Cholesterol/HDL-C | Glucose | Insulin (IU/mL) | HOMA-IR # | |
| AL | 1.55 ± 0.59 | 1.47 ± 0.40 | 0.69 ± 0.16 | 0.47±0.39 | 2.14 ± 0.53 | 5.54 ± 0.98 | 23.92 ± 8.76 | 5.59 ± 0.41 |
| CR1 | 1.19 ± 0.32 a | 1.58 ± 0.39 | 0.82 ± 0.19 a | 0.52±0.47 | 1.95 ± 0.39 | 5.46 ± 0.67 | 16.02 ± 9.43 | 3.88 ± 0.63 a |
| CR2 | 0.92 ± 0.13 a | 2.09 ± 0.71 a | 0.92 ± 0.22 a | 0.99±0.61 a | 2.25 ± 0.36 | 5.97 ± 0.98 | 10.53 ± 7.59 a | 2.79 ± 0.58 a,b |
| Group | Visceral Fat Mass [g] #,* | Visceral Fat Mass [%] *,Δ |
|---|---|---|
| AL | 16.47 ± 3.76 | 3.29 ± 0.48 |
| CR1 | 12.08 ± 3.71 a | 2.51 ± 0.68 a |
| CR2 | 6.72 ± 2.61 a,b | 1.79 ± 0.69 a,b |
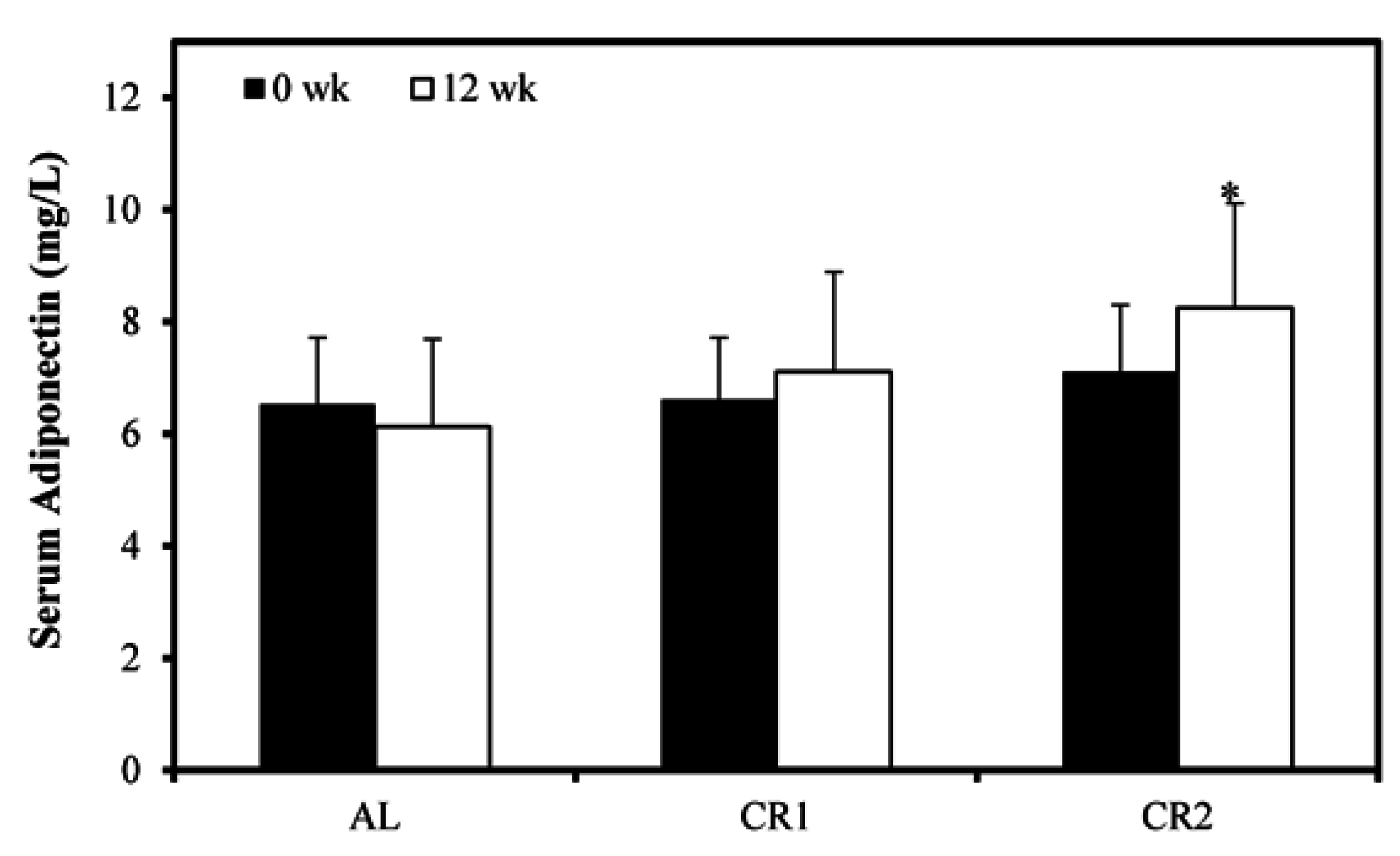
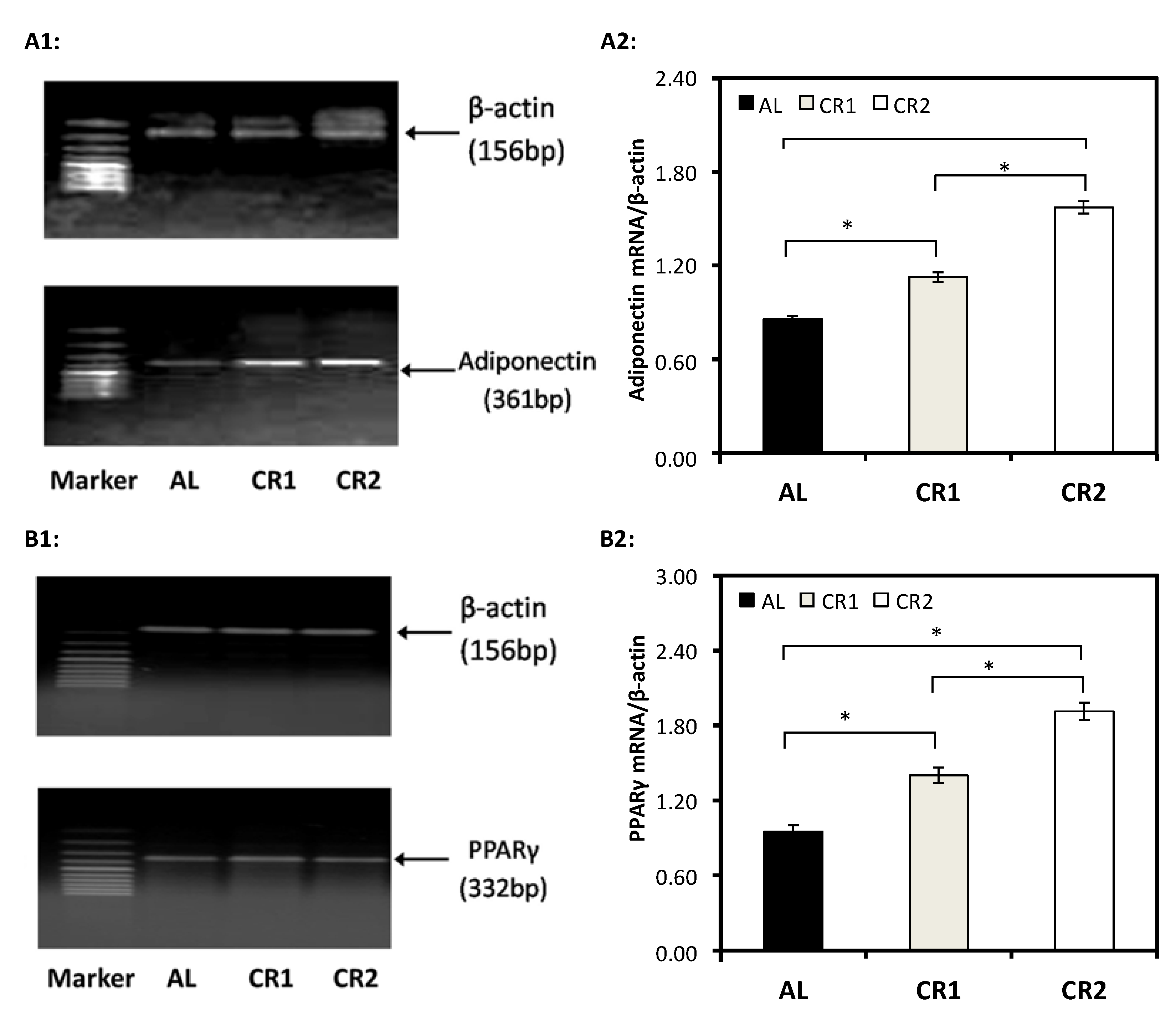
3.3. Gene Expression
3.4. Correlation Analysis
| Independent Variables | Adiponectin | PPARγ | ||
|---|---|---|---|---|
| r | p | r | p | |
| Body weight | −0.389 | 0.001 | −0.425 | 0.005 |
| Triglyceride | −0.345 | 0.042 | −0.532 | 0.032 |
| Total Cholesterol | 0.47 | 0.624 | 0.673 | 0.431 |
| HDL-C † | 0.376 | 0.026 | 0.354 | 0.032 |
| Glucose | 0.530 | 0.231 | 0.492 | 0.485 |
| Insulin | −0.411 | 0.003 | −0.537 | 0.013 |
4. Discussion and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Verdery, R.B.; Walford, R.L. Changes in plasma lipids and lipoproteins in humans during a 2-year period of dietary restriction in biosphere 2. Arch. Intern. Med. 1998, 158, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Walford, R.L.; Mock, D.; Verdery, R.; MacCallum, T. Calorie restriction in biosphere 2: Alterations in physiologic, hematologic, hormonal, and biochemical parameters in humans restricted for a 2-year period. J. Gerontol. A Biol. Sci. Med. Sci. 2002, 57, B211–B224. [Google Scholar] [CrossRef] [PubMed]
- Bordone, L.; Guarente, L. Calorie restriction, sirt1 and metabolism: Understanding longevity. Nat. Rev. Mol. Cell Biol. 2005, 6, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Masuda, J.; Shimagami, H.; Ohta, Y.; Kanda, T.; Saito, K.; Kato, H. Mild caloric restriction up-regulates the expression of prohibitin: A proteome study. Biochem. Biophys. Res. Commun. 2011, 405, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Oana, F.; Takeda, H.; Matsuzawa, A.; Akahane, S.; Isaji, M.; Akahane, M. Adiponectin receptor 2 expression in liver and insulin resistance in db/db mice given a beta3-adrenoceptor agonist. Eur. J. Pharmacol. 2005, 518, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Bjorndal, B.; Burri, L.; Staalesen, V.; Skorve, J.; Berge, R.K. Different adipose depots: Their role in the development of metabolic syndrome and mitochondrial response to hypolipidemic agents. J. Obes. 2011, 2011, 490650. [Google Scholar] [CrossRef] [PubMed]
- Sadashiv; Tiwari, S.; Paul, B.N.; Kumar, S.; Chandra, A.; Dhananjai, S.; Negi, M.P. Adiponectin mrna in adipose tissue and its association with metabolic risk factors in postmenopausal obese women. Hormones (Athens) 2013, 12, 119–127. [Google Scholar] [PubMed]
- Combs, T.P.; Berg, A.H.; Rajala, M.W.; Klebanov, S.; Iyengar, P.; Jimenez-Chillaron, J.C.; Patti, M.E.; Klein, S.L.; Weinstein, R.S.; Scherer, P.E. Sexual differentiation, pregnancy, calorie restriction, and aging affect the adipocyte-specific secretory protein adiponectin. Diabetes 2003, 52, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Vaiopoulos, A.G.; Marinou, K.; Christodoulides, C.; Koutsilieris, M. The role of adiponectin in human vascular physiology. Int. J. Cardiol. 2012, 155, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Iwaki, M.; Matsuda, M.; Maeda, N.; Funahashi, T.; Matsuzawa, Y.; Makishima, M.; Shimomura, I. Induction of adiponectin, a fat-derived antidiabetic and antiatherogenic factor, by nuclear receptors. Diabetes 2003, 52, 1655–1663. [Google Scholar] [CrossRef] [PubMed]
- Combs, T.P.; Wagner, J.A.; Berger, J.; Doebber, T.; Wang, W.J.; Zhang, B.B.; Tanen, M.; Berg, A.H.; O’Rahilly, S.; Savage, D.B.; et al. Induction of adipocyte complement-related protein of 30 kilodaltons by ppargamma agonists: A potential mechanism of insulin sensitization. Endocrinology 2002, 143, 998–1007. [Google Scholar] [PubMed]
- Maeda, N.; Takahashi, M.; Funahashi, T.; Kihara, S.; Nishizawa, H.; Kishida, K.; Nagaretani, H.; Matsuda, M.; Komuro, R.; Ouchi, N.; et al. Ppargamma ligands increase expression and plasma concentrations of adiponectin, an adipose-derived protein. Diabetes 2001, 50, 2094–2099. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; Jeng, C.Y.; Wu, T.J.; Tanaka, S.; Funahashi, T.; Matsuzawa, Y.; Wang, J.P.; Chen, C.L.; Tai, T.Y.; Chuang, L.M. Synthetic peroxisome proliferator-activated receptor-gamma agonist, rosiglitazone, increases plasma levels of adiponectin in type 2 diabetic patients. Diabetes Care 2002, 25, 376–380. [Google Scholar] [CrossRef] [PubMed]
- Sung, B.; Park, S.; Yu, B.P.; Chung, H.Y. Modulation of ppar in aging, inflammation, and calorie restriction. J. Gerontol. A Biol. Sci. Med. Sci. 2004, 59, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Miura, J.; Lu, L.X.; Bernier, M.; DeCabo, R.; Lane, M.A.; Roth, G.S.; Ingram, D.K. Circulating adiponectin levels increase in rats on caloric restriction: The potential for insulin sensitization. Exp. Gerontol. 2004, 39, 1049–1059. [Google Scholar] [CrossRef] [PubMed]
- Volek, J.S.; Fernandez, M.L.; Feinman, R.D.; Phinney, S.D. Dietary carbohydrate restriction induces a unique metabolic state positively affecting atherogenic dyslipidemia, fatty acid partitioning, and metabolic syndrome. Prog. Lipid Res. 2008, 47, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C., Jr. AIN-93 purified diets for laboratory rodents: Final report of the american institute of nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [PubMed]
- Tanno, K.; Okamura, T.; Ohsawa, M.; Onoda, T.; Itai, K.; Sakata, K.; Nakamura, M.; Ogawa, A.; Kawamura, K.; Okayama, A. Comparison of low-density lipoprotein cholesterol concentrations measured by a direct homogeneous assay and by the friedewald formula in a large community population. Clin. Chim. Acta 2010, 411, 1774–1780. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Gerbaix, M.; Metz, L.; Ringot, E.; Courteix, D. Visceral fat mass determination in rodent: Validation of dual-energy X-ray absorptiometry and anthropometric techniques in fat and lean rats. Lipids Health Dis. 2010, 9, 140. [Google Scholar] [CrossRef] [PubMed]
- Fox, C.S.; Massaro, J.M.; Hoffmann, U.; Pou, K.M.; Maurovich-Horvat, P.; Liu, C.Y.; Vasan, R.S.; Murabito, J.M.; Meigs, J.B.; Cupples, L.A.; et al. Abdominal visceral and subcutaneous adipose tissue compartments: Association with metabolic risk factors in the framingham heart study. Circulation 2007, 116, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Freedland, E.S. Role of a critical visceral adipose tissue threshold (CVATT) in metabolic syndrome: Implications for controlling dietary carbohydrates: A review. Nutr. Metab. (Lond.) 2004, 1, 12. [Google Scholar] [CrossRef] [PubMed]
- Wajchenberg, B.L. Subcutaneous and visceral adipose tissue: Their relation to the metabolic syndrome. Endocr. Rev. 2000, 21, 697–738. [Google Scholar] [CrossRef] [PubMed]
- Sasakabe, T.; Haimoto, H.; Umegaki, H.; Wakai, K. Effects of a moderate low-carbohydrate diet on preferential abdominal fat loss and cardiovascular risk factors in patients with type 2 diabetes. Diabetes Metab. Syndr. Obes. 2011, 4, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Sharman, M.J.; Kraemer, W.J.; Love, D.M.; Avery, N.G.; Gomez, A.L.; Scheett, T.P.; Volek, J.S. A ketogenic diet favorably affects serum biomarkers for cardiovascular disease in normal-weight men. J. Nutr. 2002, 132, 1879–1885. [Google Scholar] [PubMed]
- Volek, J.S.; Sharman, M.J.; Forsythe, C.E. Modification of lipoproteins by very low-carbohydrate diets. J. Nutr. 2005, 135, 1339–1342. [Google Scholar] [PubMed]
- Volek, J.S.; Sharman, M.J.; Gomez, A.L.; Scheett, T.P.; Kraemer, W.J. An isoenergetic very low carbohydrate diet improves serum HDL cholesterol and triacylglycerol concentrations, the total cholesterol to HDL cholesterol ratio and postprandial pipemic responses compared with a low fat diet in normal weight, normolipidemic women. J. Nutr. 2003, 133, 2756–2761. [Google Scholar] [PubMed]
- Masoro, E.J.; McCarter, R.J.; Katz, M.S.; McMahan, C.A. Dietary restriction alters characteristics of glucose fuel use. J. Gerontol. 1992, 47, B202–B208. [Google Scholar] [CrossRef] [PubMed]
- Wetter, T.J.; Gazdag, A.C.; Dean, D.J.; Cartee, G.D. Effect of calorie restriction on in vivo glucose metabolism by individual tissues in rats. Am. J. Physiol. 1999, 276, E728–E738. [Google Scholar] [PubMed]
- Yang, X.; Zhang, Y.; Lin, J.; Pen, A.; Ying, C.; Cao, W.; Mao, L. A lower proportion of dietary saturated/monounsaturated/polyunsaturated fatty acids reduces the expression of adiponectin in rats fed a high-fat diet. Nutr. Res. 2012, 32, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Lee, G.D.; Ding, L.; Hu, J.; Qiu, G.; de Cabo, R.; Bernier, M.; Ingram, D.K.; Zou, S. Adipogenic signaling in rat white adipose tissue: Modulation by aging and calorie restriction. Exp. Gerontol. 2007, 42, 733–744. [Google Scholar] [CrossRef] [PubMed]
- Saltiel, A.R.; Kahn, C.R. Insulin signalling and the regulation of glucose and lipid metabolism. Nature 2001, 414, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Tschritter, O.; Fritsche, A.; Thamer, C.; Haap, M.; Shirkavand, F.; Rahe, S.; Staiger, H.; Maerker, E.; Haring, H.; Stumvoll, M. Plasma adiponectin concentrations predict insulin sensitivity of both glucose and lipid metabolism. Diabetes 2003, 52, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; Chen, M.H.; Lee, W.J.; Lee, K.C.; Chao, C.L.; Huang, K.C.; Chen, C.L.; Tai, T.Y.; Chuang, L.M. Adiponectin mRNA levels in the abdominal adipose depots of nondiabetic women. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 896–900. [Google Scholar] [CrossRef] [PubMed]
- Accurso, A.; Bernstein, R.K.; Dahlqvist, A.; Draznin, B.; Feinman, R.D.; Fine, E.J.; Gleed, A.; Jacobs, D.B.; Larson, G.; Lustig, R.H.; et al. Dietary carbohydrate restriction in type 2 diabetes mellitus and metabolic syndrome: Time for a critical appraisal. Nutr. Metab. (Lond.) 2008, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- Krauss, R.M.; Deckelbaum, R.J.; Ernst, N.; Fisher, E.; Howard, B.V.; Knopp, R.H.; Kotchen, T.; Lichtenstein, A.H.; McGill, H.C.; Pearson, T.A.; et al. Dietary guidelines for healthy american adults: A statement for health professionals from the nutrition committee, American heart association. Circulation 1996, 94, 1795–1800. [Google Scholar] [CrossRef] [PubMed]
- Halton, T.L.; Willett, W.C.; Liu, S.; Manson, J.E.; Albert, C.M.; Rexrode, K.; Hu, F.B. Low-carbohydrate-diet score and the risk of coronary heart disease in women. N. Engl. J. Med. 2006, 355, 1991–2002. [Google Scholar] [CrossRef] [PubMed]
- Schneeman, B.O. Carbohydrate: Friend or foe? Summary of research needs. J. Nutr. 2001, 131, 2764S–2765S. [Google Scholar] [PubMed]
- Parks, E.J.; Hellerstein, M.K. Carbohydrate-induced hypertriacylglycerolemia: Historical perspective and review of biological mechanisms. Am. J. Clin. Nutr. 2000, 71, 412–433. [Google Scholar] [PubMed]
- Dreon, D.M.; Fernstrom, H.A.; Miller, B.; Krauss, R.M. Low-density lipoprotein subclass patterns and lipoprotein response to a reduced-fat diet in men. FASEB J. 1994, 8, 121–126. [Google Scholar] [PubMed]
- Dreon, D.M.; Fernstrom, H.A.; Williams, P.T.; Krauss, R.M. A very low-fat diet is not associated with improved lipoprotein profiles in men with a predominance of large, low-density lipoproteins. Am. J. Clin. Nutr. 1999, 69, 411–418. [Google Scholar] [PubMed]
- Retzlaff, B.M.; Walden, C.E.; Dowdy, A.A.; McCann, B.S.; Anderson, K.V.; Knopp, R.H. Changes in plasma triacylglycerol concentrations among free-living hyperlipidemic men adopting different carbohydrate intakes over 2 y: The dietary alternatives study. Am. J. Clin. Nutr. 1995, 62, 988–995. [Google Scholar] [PubMed]
- Yuan, G.; Al-Shali, K.Z.; Hegele, R.A. Hypertriglyceridemia: Its etiology, effects and treatment. CMAJ 2007, 176, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- Fontana, L.; Meyer, T.E.; Klein, S.; Holloszy, J.O. Long-term calorie restriction is highly effective in reducing the risk for atherosclerosis in humans. Proc. Natl. Acad. Sci. USA 2004, 101, 6659–6663. [Google Scholar] [CrossRef] [PubMed]
- Turley, M.L.; Skeaff, C.M.; Mann, J.I.; Cox, B. The effect of a low-fat, high-carbohydrate diet on serum high density lipoprotein cholesterol and triglyceride. Eur. J. Clin. Nutr. 1998, 52, 728–732. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S.; Tjonn, S.L.; Swan, P.D.; White, A.; Hutchins, H.; Sears, B. Ketogenic low-carbohydrate diets have no metabolic advantage over nonketogenic low-carbohydrate diets. Am. J. Clin. Nutr. 2006, 83, 1055–1061. [Google Scholar] [PubMed]
- Daly, M.E.; Paisey, R.; Paisey, R.; Millward, B.A.; Eccles, C.; Williams, K.; Hammersley, S.; MacLeod, K.M.; Gale, T.J. Short-term effects of severe dietary carbohydrate-restriction advice in type 2 diabetes—A randomized controlled trial. Diabet. Med. 2006, 23, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Haimoto, H.; Iwata, M.; Wakai, K.; Umegaki, H. Long-term effects of a diet loosely restricting carbohydrates on HbA1c levels, BMI and tapering of sulfonylureas in type 2 diabetes: A 2-year follow-up study. Diabetes Res. Clin. Pract. 2008, 79, 350–356. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.-H.; Ouyang, C.; Ding, Q.; Song, J.; Cao, W.; Mao, L. A Moderate Low-Carbohydrate Low-Calorie Diet Improves Lipid Profile, Insulin Sensitivity and Adiponectin Expression in Rats. Nutrients 2015, 7, 4724-4738. https://doi.org/10.3390/nu7064724
Chen J-H, Ouyang C, Ding Q, Song J, Cao W, Mao L. A Moderate Low-Carbohydrate Low-Calorie Diet Improves Lipid Profile, Insulin Sensitivity and Adiponectin Expression in Rats. Nutrients. 2015; 7(6):4724-4738. https://doi.org/10.3390/nu7064724
Chicago/Turabian StyleChen, Jie-Hua, Caiqun Ouyang, Qiang Ding, Jia Song, Wenhong Cao, and Limei Mao. 2015. "A Moderate Low-Carbohydrate Low-Calorie Diet Improves Lipid Profile, Insulin Sensitivity and Adiponectin Expression in Rats" Nutrients 7, no. 6: 4724-4738. https://doi.org/10.3390/nu7064724
APA StyleChen, J.-H., Ouyang, C., Ding, Q., Song, J., Cao, W., & Mao, L. (2015). A Moderate Low-Carbohydrate Low-Calorie Diet Improves Lipid Profile, Insulin Sensitivity and Adiponectin Expression in Rats. Nutrients, 7(6), 4724-4738. https://doi.org/10.3390/nu7064724




