Dietary Patterns Derived by Cluster Analysis are Associated with Cognitive Function among Korean Older Adults
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. General Characteristics and Anthropometrics Variables
2.3. Cognitive Function Examination
2.4. Dietary Data
2.5. Statistical Analysis
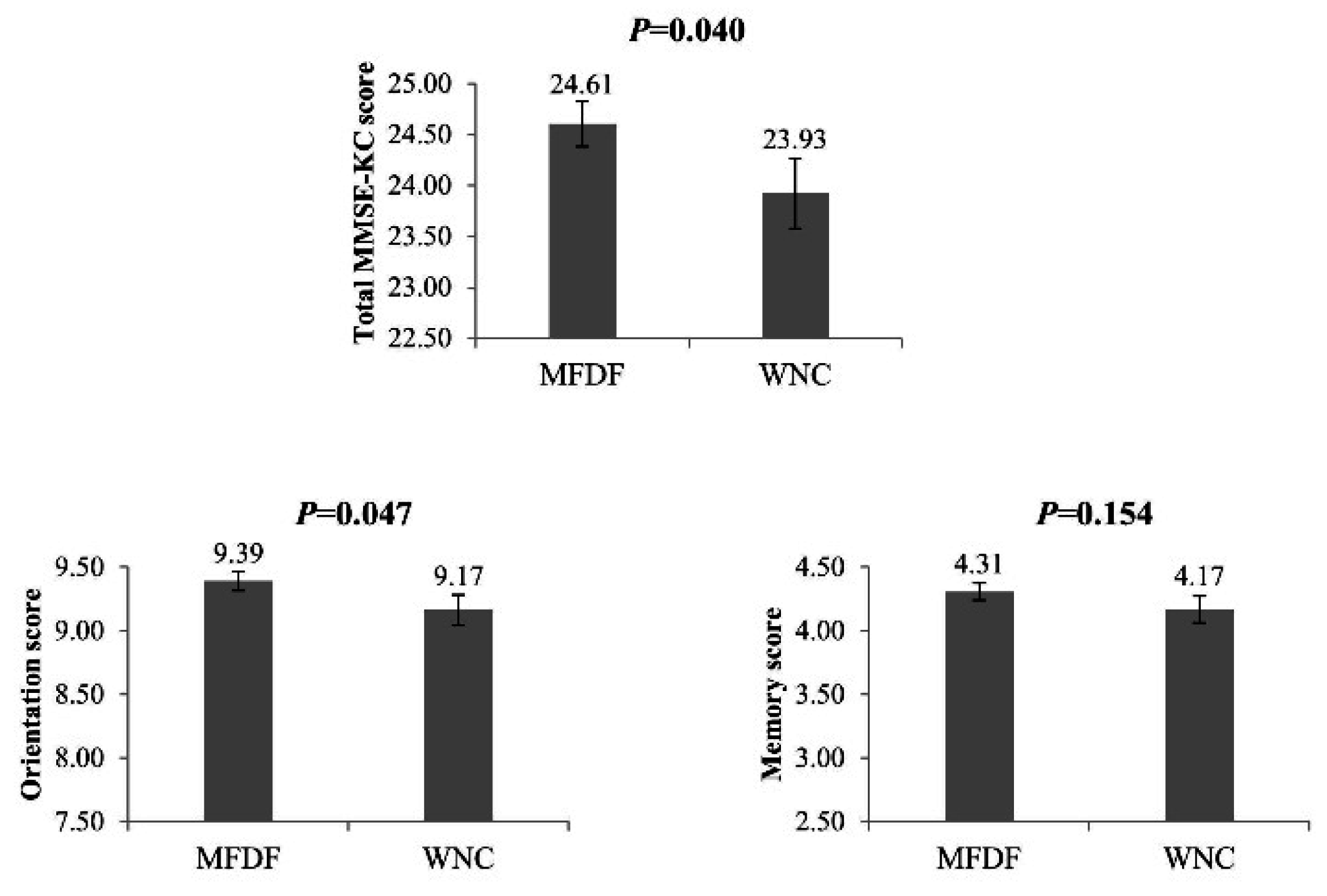
3. Results
| Foods & food groups | MFDF (n = 589) | WNC (n = 176) | p 1 |
|---|---|---|---|
| White rice (g/day) | 0.2 ± 5.6 | 254.4 ± 78.6 | <0.0001 |
| Multigrain rice (g/day) | 251.8 ± 75.1 | 1.0 ± 3.4 | <0.0001 |
| Noodles (g/day) | 19.4 ± 23.1 | 24.9 ± 30.4 | 0.027 |
| Rice cakes (g/day) | 2.6 ± 5.2 | 2.6 ± 8.9 | 0.994 |
| Cereals (g/day) | 0.05 ± 0.5 | 0.1 ± 1.1 | 0.615 |
| Breads (g/day) | 8.1 ± 18.0 | 8.3 ± 20.2 | 0.909 |
| Sweet foods (g/day) | 3.1 ± 8.0 | 3.5 ± 9.4 | 0.564 |
| Nuts (g/day) | 1.0 ± 3.3 | 0.6 ± 1.9 | 0.075 |
| Beans (g/day) | 22.7 ± 28.9 | 23.8 ± 29.8 | 0.664 |
| Eggs (g/day) | 9.3 ± 17.3 | 11.0 ± 19.9 | 0.309 |
| Potatoes (g/day) | 18.8 ± 25.6 | 14.6 ± 27.5 | 0.065 |
| Salty vegetables (g/day) | 154.6 ± 114.3 | 161.8 ± 106.0 | 0.455 |
| Vegetables (g/day) | 63.4 ± 58.2 | 60.1 ± 62.3 | 0.516 |
| Meats (g/day) | 16.2 ± 24.6 | 20.0 ± 24.4 | 0.073 |
| Soups (g/day) | 4.0 ± 10.2 | 3.6 ± 6.6 | 0.554 |
| Fish (g/day) | 43.9 ± 55.0 | 34.8 ± 44.1 | 0.025 |
| Seafood (g/day) | 4.5 ± 8.2 | 6.5 ± 14.5 | 0.090 |
| Dairy products (g/day) | 92.8 ± 127.4 | 67.7 ± 113.1 | 0.019 |
| Soymilk (g/day) | 17.7 ± 45.9 | 27.2 ± 83.8 | 0.149 |
| Coffee (g/day) | 8.2 ± 8.1 | 10.5 ± 9.2 | 0.004 |
| Green tea (g/day) | 22.9 ± 44.6 | 20.9 ± 46.4 | 0.605 |
| Soft drinks (g/day) | 21.1 ± 40.9 | 24.2 ± 45.7 | 0.388 |
| Fruits & fruit juices (g/day) | 172.6 ± 155.8 | 135.4 ± 116.3 | 0.001 |
| Characteristics | MFDF (n = 589) | WNC (n = 176) | p 1 |
|---|---|---|---|
| Age (years) | 67.5 ± 5.0 2 | 67.9 ± 5.7 | 0.311 |
| Height (cm) | 156.3 ± 8.4 | 158.2 ± 8.6 | 0.008 |
| Weight (kg) | 59.8 ± 9.3 | 60.3 ± 9.0 | 0.513 |
| BMI | 24.5 ± 3.2 | 24.1 ± 3.1 | 0.172 |
| Men (%) | 38.2 | 60.2 | <0.0001 |
| Education (%) | 0.355 | ||
| Uneducated | 17.8 | 22.9 | |
| Elementary | 53.1 | 45.7 | |
| Middle school | 12.2 | 12.6 | |
| High school | 11.4 | 14.3 | |
| College or Higher | 5.4 | 4.6 | |
| Occupation | 0.279 | ||
| Office work | 5.3 | 7.4 | |
| Non-office work | 2.0 | 1.7 | |
| Service industry | 4.6 | 4.0 | |
| Farmer | 52.8 | 56.8 | |
| Housework | 13.8 | 8.5 | |
| Unemployed | 19.7 | 17.6 | |
| Others | 1.9 | 4.0 | |
| Marital status | 0.226 | ||
| Currently married, or cohabiting (%) | 78.8 | 83.0 | |
| Alone (%) | 21.2 | 17.1 | |
| Current alcohol drinker (%) | 36.8 | 50.6 | 0.001 |
| Current smoker (%) | 2.2 | 4.0 | 0.197 |
| Regular exerciser (%) | 34.1 | 15.3 | <0.0001 |
| Dietary supplement user (%) | 12.1 | 8.0 | 0.129 |
| Disease | |||
| Cardiovascular disease (%) | 7.1 | 5.7 | 0.503 |
| Hypertension (%) | 36.0 | 28.4 | 0.063 |
| Hyperlipidemia (%) | 3.2 | 2.3 | 0.516 |
| Diabetes (%) | 13.9 | 5.1 | 0.002 |
| Stroke (%) | 2.0 | 3.4 | 0.292 |
| Dietary intakes | MFDF (n = 589) | WNC (n = 176) | p 1 |
|---|---|---|---|
| Total energy (kcal) | 1464.6 ± 18.2 2 | 1422.9 ± 32.7 | 0.270 |
| Percentage of energy | |||
| From carbohydrate (%) | 76.1 ± 0.3 | 78.1 ± 0.5 | 0.0002 |
| From protein (%) | 11.8 ± 0.1 | 11.1 ± 0.2 | <0.0001 |
| From fat (%) | 12.4 ± 0.2 | 10.3 ± 0.4 | 0.0003 |
| Carbohydrate (g) | 275.9 ± 3.2 | 274.4 ± 5.7 | 0.815 |
| Protein (g) | 46.1 ± 0.8 | 40.3 ± 1.4 | 0.0004 |
| Fat (g) | 20.2 ± 0.5 | 17.5 ± 1.0 | 0.015 |
| Total fatty acid (g) | 10.1 ± 0.3 | 9.0 ± 0.6 | 0.102 |
| Saturated fatty acid (g) | 4.3 ± 0.2 | 3.8 ± 0.3 | 0.151 |
| Monounsaturated fatty acid (g) | 4.5 ± 0.2 | 4.1 ± 0.3 | 0.269 |
| Polyunsaturated fatty acid (g) | 2.4 ± 0.1 | 2.1 ± 0.1 | 0.091 |
| n-3 polyunsaturated fatty acid (g) | 0.4 ± 0.02 | 0.3 ± 0.03 | 0.040 |
| β-carotene (μg) | 2567.9 ± 79.9 | 2219.8 ± 143.8 | 0.036 |
| Vitamin C (mg) | 78.9 ± 2.1 | 71.8 ± 3.8 | 0.104 |
| Vitamin E (mg) | 6.6 ± 0.1 | 6.1 ± 0.3 | 0.115 |
| Vitamin D (μg) | 2.0 ± 0.1 | 1.7 ± 0.2 | 0.051 |
| Vitamin B6(mg) | 1.2 ± 0.02 | 1.0 ± 0.03 | 0.001 |
| Vitamin B12(μg) | 2.2 ± 0.1 | 2.2 ± 0.2 | 0.837 |
| Folate (μg) | 417.0 ± 7.1 | 363.2 ± 12.8 | 0.0003 |
| Sodium | 2861.4 ± 68.0 | 2806.4 ± 122.3 | 0.697 |
| Potassium | 2072.6 ± 39.1 | 1754.3 ± 70.3 | <0.0001 |
| Calcium | 368.8 ± 9.7 | 302.8 ± 17.4 | 0.001 |
| Magnesium | 39.7 ± 1.1 | 35.1 ± 2.0 | 0.051 |
| Iron(mg) | 37.4 ± 2.3 | 33.8 ± 4.2 | 0.455 |
| Zinc(mg) | 7.9 ± 0.1 | 7.5 ± 0.2 | 0.197 |
| Selenium(μg) | 64.8 ± 1.0 | 64.0 ± 1.8 | 0.703 |
| MAR | 0.7 ± 0.01 | 0.6 ± 0.01 | <0.0001 |
| Model 1 1 | Model 2 2 | Model 3 3 | |
|---|---|---|---|
| WNC | 1.00(reference) | 1.00 | 1.00 |
| MFDF | 0.67 (0.47, 0.95) | 0.62 (0.43, 0.90) | 0.64 (0.44, 0.94) |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- United Nations. World Population Ageing 2013. Available online: http://www.un.org/en/development/desa/population/publications/ageing/WorldPopulationAgeingReport2013.shtml (accessed on 20 May 2015).
- Ferri, C.P.; Prince, M.; Brayne, C.; Brodaty, H.; Fratiglioni, L.; Ganguli, M.; Hall, K.; Hasegawa, K.; Hendrie, H.; Huang, Y.; et al. Alzheimer’s Disease International Global prevalence of dementia: A Delphi consensus study. Lancet 2005, 366, 2112–2117. [Google Scholar]
- Ministry of Health and Welfare. A Nationwide Survey on the states of the elderly with dementia. Available online: http://www.prism.go.kr/homepage/researchCommon/retrieveResearchDetailPopup.do;jsessionid=96BD6E6308C135F388289CD1B2C7AC32.node02?research_id=1351000-201000126 (accessed on 26 May 2015).
- Petersen, R.C.; Smith, G.E.; Waring, S.C.; Ivnik, R.J.; Tangalos, E.G.; Kokmen, E. Mild cognitive impairment: Clinical characterization and outcome. Arch. Neurol. 1999, 56, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Ganguli, M.; Snitz, B.E.; Saxton, J.A.; Chang, C.H.; Lee, C.; Vander Bilt, J.; Hughes, T.F.; Loewenstein, D.A.; Unverzagt, F.W.; Petersen, R.C. Outcomes of mild cognitive impairment by definition: A population study. Arch. Neurol. 2011, 68, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C.; Doody, R.; Kurz, A.; Mohs, R.C.; Morris, J.C.; Rabins, P.V.; Ritchie, K.; Rossor, M.; Thal, L.; Winblad, B. Current concepts in mild cognitive impairment. Arch. Neurol. 2001, 58, 1985–1992. [Google Scholar] [CrossRef] [PubMed]
- Paleologos, M.; Cumming, R.G.; Lazarus, R. Cohort study of vitamin C intake and cognitive impairment. Am. J. Epidemiol. 1998, 148, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, J.S.; Goodwin, J.M.; Garry, P.J. Association between nutritional status and cognitive functioning in a healthy elderly population. JAMA 1983, 249, 2917–2921. [Google Scholar] [CrossRef] [PubMed]
- Riggs, K.M.; Spiro, A., 3rd; Tucker, K.; Rush, D. Relations of vitamin B-12, vitamin B-6, folate, and homocysteine to cognitive performance in the Normative Aging Study. Am. J. Clin. Nutr. 1996, 63, 306–314. [Google Scholar] [PubMed]
- Eskelinen, M.H.; Ngandu, T.; Helkala, E.L.; Tuomilehto, J.; Nissinen, A.; Soininen, H.; Kivipelto, M. Fat intake at midlife and cognitive impairment later in life: A population-based CAIDE study. Int. J. Geriatr. Psychiatry 2008, 23, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Van Gelder, B.M.; Tijhuis, M.; Kalmijn, S.; Kromhout, D. Fish consumption, n-3 fatty acids, and subsequent 5-y cognitive decline in elderly men: The Zutphen Elderly Study. Am. J. Clin. Nutr. 2007, 85, 1142–1147. [Google Scholar] [PubMed]
- Dangour, A.D.; Whitehouse, P.J.; Rafferty, K.; Mitchell, S.A.; Smith, L.; Hawkesworth, S.; Vellas, B. B-vitamins and fatty acids in the prevention and treatment of Alzheimer’s disease and dementia: A systematic review. J. Alzheimers Dis. 2010, 22, 205–224. [Google Scholar] [PubMed]
- Crichton, G.E.; Bryan, J.; Murphy, K.J. Dietary antioxidants, cognitive function and dementia—A systematic review. Plant Foods Hum. Nutr. 2013, 68, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Akbaraly, T.N.; Singh-Manoux, A.; Marmot, M.G.; Brunner, E.J. Education attenuates the association between dietary patterns and cognition. Dement. Geriatr. Cogn. Disord. 2009, 27, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Andreeva, V.A.; Jeandel, C.; Ferry, M.; Hercberg, S.; Galan, P. A healthy dietary pattern at midlife is associated with subsequent cognitive performance. J. Nutr. 2012, 142, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Samieri, C.; Jutand, M.A.; Féart, C.; Capuron, L.; Letenneur, L.; Barberger-Gateau, P. Dietary patterns derived by hybrid clustering method in older people: Association with cognition, mood, and self-rated health. J. Am. Diet. Assoc. 2008, 108, 1461–1471. [Google Scholar] [CrossRef] [PubMed]
- Corley, J.; Starr, J.M.; McNeill, G.; Deary, I.J. Do dietary patterns influence cognitive function in old age? Int. Psychogeriatr. 2013, 25, 1393–1407. [Google Scholar] [CrossRef] [PubMed]
- Torres, S.J.; Lautenschlager, N.T.; Wattanapenpaiboon, N.; Greenop, K.R.; Beer, C.; Flicker, L.; Alfonso, H.; Nowson, C.A. Dietary Patterns are Associated with Cognition among Older People with Mild Cognitive Impairment. Nutrients 2012, 4, 1542–1551. [Google Scholar] [CrossRef] [PubMed]
- Chan, R.; Chan, D.; Woo, J. A cross sectional study to examine the association between dietary patterns and cognitive impairment in older Chinese people in Hong Kong. J. Nutr. Health Aging 2013, 17, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Kalmijn, S.; van Boxtel, M.P.; Ocké, M.; Verschuren, W.M.; Kromhout, D.; Launer, L.J. Dietary intake of fatty acids and fish in relation to cognitive performance at middle age. Neurology 2004, 62, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Lee, J.H.; Lee, K.U.; Lee, D.Y.; Kim, K.W.; Jhoo, J.H.; Kim, J.H.; Lee, K.H.; Kim, S.Y.; Han, S.H.; Woo, J.I. Development of the Korean version of the Consortium to Establish a Registry for Alzheimer’s Disease Assessment Packet (CERAD-K): Clinical and neuropsychological assessment batteries. J. Gerontol. B Psychol. Sci. Soc. Sci. 2002, 57, P47–P53. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.Y.; Lee, K.U.; Lee, J.H.; Kim, K.W.; Jhoo, J.H.; Kim, S.Y.; Yoon, J.C.; Woo, S.I.; Ha, J.; Woo, J.I. A normative study of the CERAD neuropsychological assessment battery in the Korean elderly. J. Int. Neuropsychol. Soc. 2004, 10, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.; Kim, H.; Kim, K.N.; Son, J.I.; Kim, S.Y.; Tamura, T.; Chang, N. Relationship of cognitive function with B vitamin status, homocysteine, and tissue factor pathway inhibitor in cognitively impaired elderly: A cross-sectional survey. J. Alzheimers Dis. 2013, 33, 853–862. [Google Scholar] [PubMed]
- Kim, Y.N.; Kim, D.H. Decreased serum angiogenin level in Alzheimer’s disease. Prog Neuropsychopharmacol. Biol. Psychiatry 2012, 38, 116–120. [Google Scholar]
- Kang, N.R.; Kim, M.D.; Lee, C.I.; Kwak, Y.S.; Choi, K.M.; Im, H.J.; Park, J.H. The influence of subcortical ischemic lesions on cognitive function and quality of life in late life depression. J. Affect. Disord. 2012, 136, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Ahn, Y.; Kwon, E.; Shim, J.E.; Park, M.K.; Joo, Y.; Kimm, K.; Park, C.; Kim, D.H. Validation and reproducibility of food frequency questionnaire for Korean genome epidemiologic study. Eur. J. Clin. Nutr. 2007, 61, 1435–1441. [Google Scholar] [CrossRef] [PubMed]
- SAS Institute Inc. SAS/STAT® 9.3 User’s Guide; SAS Institute Inc: Cary, NC, USA, 2011; pp. 2241–2266. [Google Scholar]
- The Korean Nutrition Society. Dietary Reference Intakes for Koreans; The Korean Nutrition Society: Seoul, Republic of Korea, 2010. [Google Scholar]
- Botelho, P.B.; Fioratti, C.O.; Abdalla, D.S.; Bertolami, M.C.; Castro, I.A. Classification of Individuals with Dyslipidaemia Controlled by Statins According to Plasma Biomarkers of Oxidative Stress using Cluster Analysis. Br. J. Nutr. 2010, 103, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Villegas, R.; Yang, G.; Gao, Y.T.; Cai, H.; Li, H.; Zheng, W.; Shu, X.O. Dietary patterns are associated with lower incidence of type 2 diabetes in middle-aged women: The Shanghai Women’s Health Study. Int. J. Epidemiol. 2010, 39, 889–899. [Google Scholar]
- Ambrosini, G.L.; Huang, R.C.; Mori, T.A.; Hands, B.P.; O’Sullivan, T.A.; de Klerk, N.H.; Beilin, L.J.; Oddy, W.H. Dietary patterns and markers for the metabolic syndrome in Australian adolescents. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, Y.; Lee, S.Y.; Kim, Y.O.; Chung, Y.S.; Park, S.B. Dietary Patterns and Functional Disability in Older Korean Adults. Maturitas 2013, 76, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.H.; Lee, Y.S.; Chang, H.C.; Moon, M.K.; Song, Y. Association between Dietary Patterns and Blood Lipid Profiles in Korean Adults with Type 2 Diabetes. J. Korean Med. Sci. 2011, 26, 1201–1208. [Google Scholar] [CrossRef] [PubMed]
- Oh, C.; No, J.K.; Kim, H.S. Dietary pattern classifications with nutrient intake and body composition changes in Korean elderly. Nutr. Res. Pract. 2014, 8, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Song, S.J.; Lee, J.E.; Paik, H.Y.; Park, M.S.; Song, Y.J. Dietary patterns based on carbohydrate nutrition are associated with the risk for diabetes and dyslipidemia. Nutr. Res. Pract. 2012, 6, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Lautenschlager, N.T.; Cox, K.L.; Flicker, L.; Foster, J.K.; van Bockxmeer, F.M.; Xiao, J.; Greenop, K.R.; Almeida, O.P. Effect of physical activity on cognitive function in older adults at risk for Alzheimer disease: a randomized trial. JAMA 2008, 300, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Angevaren, M.; Aufdemkampe, G.; Verhaar, H.J.; Aleman, A.; Vanhees, L. Physical activity and enhanced fitness to improve cognitive function in older people without known cognitive impairment. Cochrane Database Syst. Rev. 2008, 16. [Google Scholar] [CrossRef]
- Rockwood, K.; Middleton, L. Physical activity and the maintenance of cognitive function. Alzheimers Dement. 2007, 3, S38–S44. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health and Welfare, Korea Centers for Disease Control and Prevention. Korea Health Statistics 2010: Korea National Health and Nutrition Examination Survey (KNHANES V-1). Available online: https://knhanes.cdc.go.kr/knhanes/sub04/sub04_03.do?classType=7 (accessed on 20 May 2015).
- Rafnsson, S.B.; Dilis, V.; Trichopoulou, A. Antioxidant nutrients and age-related cognitive decline: A systematic review of population-based cohort studies. Eur. J. Nutr. 2013, 52, 1553–1567. [Google Scholar] [CrossRef] [PubMed]
- Sydenham, E.; Dangour, A.D.; Lim, W.S. Omega 3 fatty acid for the prevention of cognitive decline and dementia. Cochrane Database Syst. Rev. 2012, 6, CD005379. [Google Scholar] [PubMed]
- Grodstein, F.; Kang, J.H.; Glynn, R.J.; Cook, N.R.; Gaziano, J.M. A Randomized Trial of Beta Carotene Supplementation and Cognitive Function in Men: The Physicians' Health Study II. Arch. Intern. Med. 2007, 167, 2184–2190. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.G.; Batterham, P.J.; Mackinnon, A.J.; Jorm, A.F.; Hickie, I.; Fenech, M.; Kljakovic, M.; Crisp, D.; Christensen, H. Oral folic acid and vitamin B-12 supplementation to prevent cognitive decline in community-dwelling older adults with depressive symptoms—The Beyond Ageing Project: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 95, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Luchtman, D.W.; Song, C. Cognitive enhancement by omega-3 fatty acids from child-hood to old age: Findings from animal and clinical studies. Neuropharmacology 2013, 64, 550–565. [Google Scholar] [CrossRef] [PubMed]
- Vogel, T.; Dali-Youcef, N.; Kaltenbach, G.; Andrès, E. Homocysteine, vitamin B12, folate and cognitive functions: A systematic and critical review of the literature. Int. J. Clin. Pract. 2009, 63, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, W.; Obeid, R. Homocysteine: A biomarker in neurodegenerative diseases. Clin. Chem. Lab. Med. 2011, 49, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Perrig, W.J.; Perrig, P.; Stähelin, H.B. The relation between antioxidants and memory performance in the old and very old. J. Am. Geriatr. Soc. 1997, 45, 718–724. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, M.; Ninomiya, T.; Ohara, T.; Hirakawa, Y.; Doi, Y.; Hata, J.; Uchida, K.; Shirota, T.; Kitazono, T.; Kiyohara, Y. Self-reported dietary intake of potassium, calcium, and magnesium and risk of dementia in the Japanese: The Hisayama Study. J. Am. Geriatr. Soc. 2012, 60, 1515–1520. [Google Scholar] [CrossRef] [PubMed]
- Cherbuin, N.; Kumar, R.; Sachdev, P.S.; Anstey, K.J. Dietary mineral intake and risk of mild cognitive impairment: The PATH through life project. Front. Aging Neurosci. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Wengreen, H.J.; Neilson, C.; Munger, R.; Corcoran, C. Diet quality is associated with better cognitive test performance among aging men and women. J. Nutr. 2009, 139, 1944–1949. [Google Scholar] [CrossRef] [PubMed]
- Tangney, C.C.; Kwasny, M.J.; Li, H.; Wilson, R.S.; Evans, D.A.; Morris, M.C. Adherence to a Mediterranean-type dietary pattern and cognitive decline in a community population. Am. J. Clin. Nutr. 2011, 93, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Scott, T.; Gao, X.; Maras, J.E.; Bakun, P.J.; Tucker, K.L. Mediterranean diet, healthy eating index 2005, and cognitive function in middle-aged and older Puerto Rican adults. J. Acad. Nutr. Diet. 2013, 113, 276–281.e3. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Lapiscina, E.H.; Clavero, P.; Toledo, E.; Estruch, R.; Salas-Salvadó, J.; San Julian, B.; Sanchez-Tainta, A.; Ros, E.; Valls-Pedret, C.; Martinez-Gonzalez, M.Á. Mediterranean diet improves cognition: The PREDIMED-NAVARRA randomized trial. J. Neurol. Neurosurg. Psychiatry 2013, 84, 1318–1325. [Google Scholar]
- Olsson, E.; Karlström, B.; Kilander, L.; Byberg, L.; Cederholm, T.; Sjögren, P. Dietary patterns and cognitive dysfunction in a 12-year follow-up study of 70 year old men. J. Alzheimers Dis. 2015, 43, 109–119. [Google Scholar] [PubMed]
- Scarmeas, N.; Stern, Y.; Mayeux, R.; Manly, J.J.; Schupf, N.; Luchsinger, J.A. Mediterranean diet and mild cognitive impairment. Arch. Neurol. 2009, 66, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Corrêa Leite, M.L.; Nicolosi, A.; Cristina, S.; Hauser, W.A.; Nappi, G. Nutrition and cognitive deficit in the elderly: A population study. Eur. J. Clin. Nutr. 2001, 55, 1053–1058. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Joung, H. A dairy and fruit dietary pattern is associated with a reduced likelihood of osteoporosis in Korean postmenopausal women. Br. J. Nutr. 2013, 110, 1926–1933. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Jo, I.; Joung, H. A rice-based traditional dietary pattern is associated with obesity in Korean adults. J. Acad. Nutr. Diet. 2012, 112, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Chiu, G.S.; Chatterjee, D.; Darmody, P.T.; Walsh, J.P.; Meling, D.D.; Johnson, R.W.; Freund, G.G. Hypoxia/reoxygenation impairs memory formation via adenosine-dependent activation of caspase 1. J. Neurosci. 2012, 32, 13945–13955. [Google Scholar] [CrossRef] [PubMed]
- Togo, P.; Osler, M.; Sorensen, T.I.; Heitmann, B.L. Food intake patterns and body mass index in observational studies. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 1741–1751. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.; Yu, A.; Choi, B.Y.; Nam, J.H.; Kim, M.K.; Oh, D.H.; Yang, Y.J. Dietary Patterns Derived by Cluster Analysis are Associated with Cognitive Function among Korean Older Adults. Nutrients 2015, 7, 4154-4169. https://doi.org/10.3390/nu7064154
Kim J, Yu A, Choi BY, Nam JH, Kim MK, Oh DH, Yang YJ. Dietary Patterns Derived by Cluster Analysis are Associated with Cognitive Function among Korean Older Adults. Nutrients. 2015; 7(6):4154-4169. https://doi.org/10.3390/nu7064154
Chicago/Turabian StyleKim, Jihye, Areum Yu, Bo Youl Choi, Jung Hyun Nam, Mi Kyung Kim, Dong Hoon Oh, and Yoon Jung Yang. 2015. "Dietary Patterns Derived by Cluster Analysis are Associated with Cognitive Function among Korean Older Adults" Nutrients 7, no. 6: 4154-4169. https://doi.org/10.3390/nu7064154
APA StyleKim, J., Yu, A., Choi, B. Y., Nam, J. H., Kim, M. K., Oh, D. H., & Yang, Y. J. (2015). Dietary Patterns Derived by Cluster Analysis are Associated with Cognitive Function among Korean Older Adults. Nutrients, 7(6), 4154-4169. https://doi.org/10.3390/nu7064154





