Fructose:Glucose Ratios—A Study of Sugar Self-Administration and Associated Neural and Physiological Responses in the Rat
Abstract
:1. Introduction
2. Methods
2.1. Study Overview
2.2. Animals
2.3. Intraoral Surgery
2.4. Apparatus
2.4.1. Operant Self-Administration of Pellets
2.4.2. Operant Self-Administration of Solutions
2.4.3. Home Cage Self-Administration
2.4.4. Taste Reactivity
2.5. Food and Sugars
2.6. Behavioral Testing
2.6.1. Operant Self-Administration of Pellets
2.6.2. Operant Intraoral Self-Administration
2.6.3. Home Cage Self-Administration
2.6.4. Taste Reactivity
2.7. Fos Immunoreactivity
2.8. Solution Hybridization RNase Protection-TCA Precipitation
2.9. Gas Chromatography
2.10. Statistical Analyses
3. Results
3.1. Experiment 1
3.1.1. Taste Reactivity
3.1.2. Operant Self-Administration of Pellets
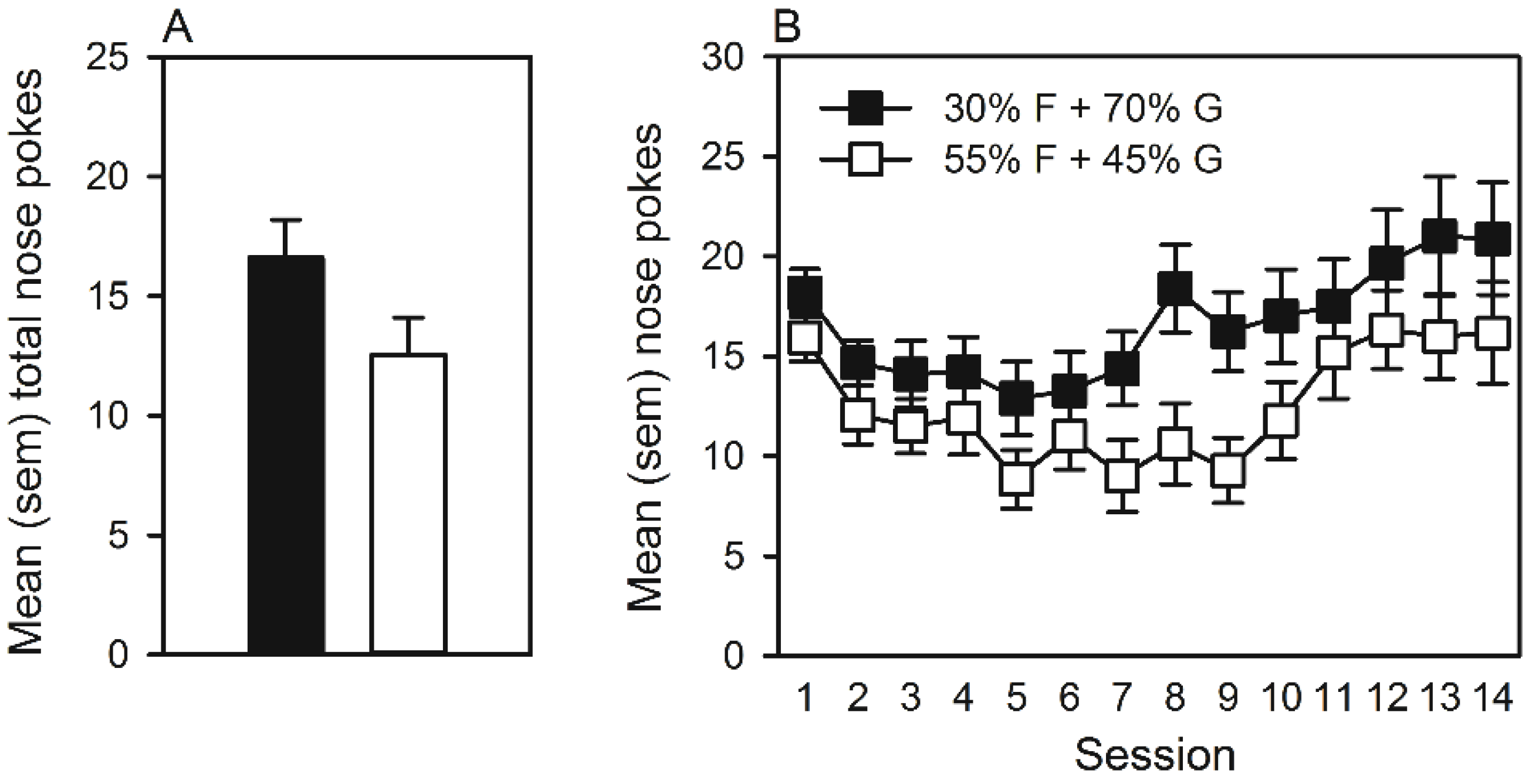
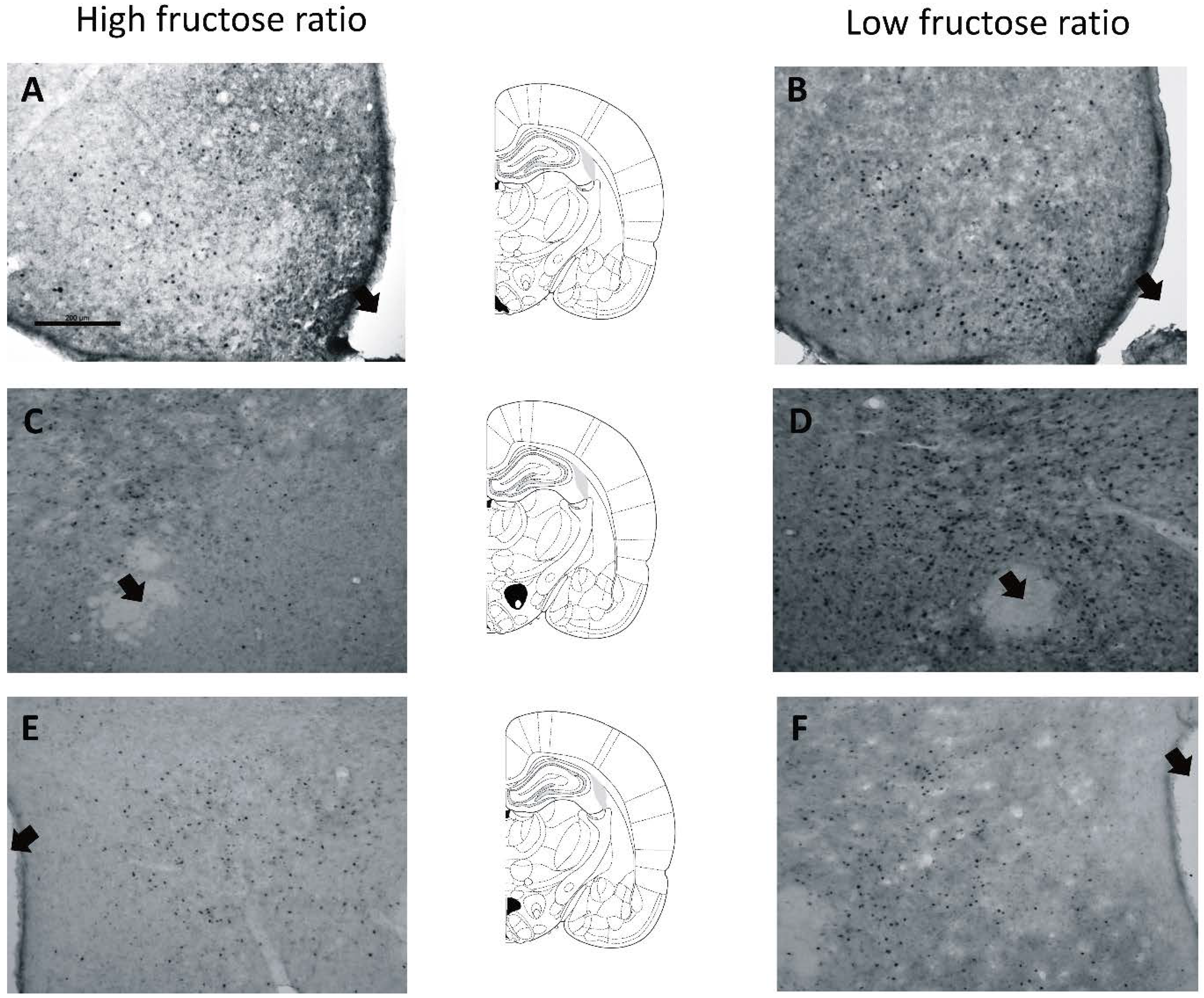
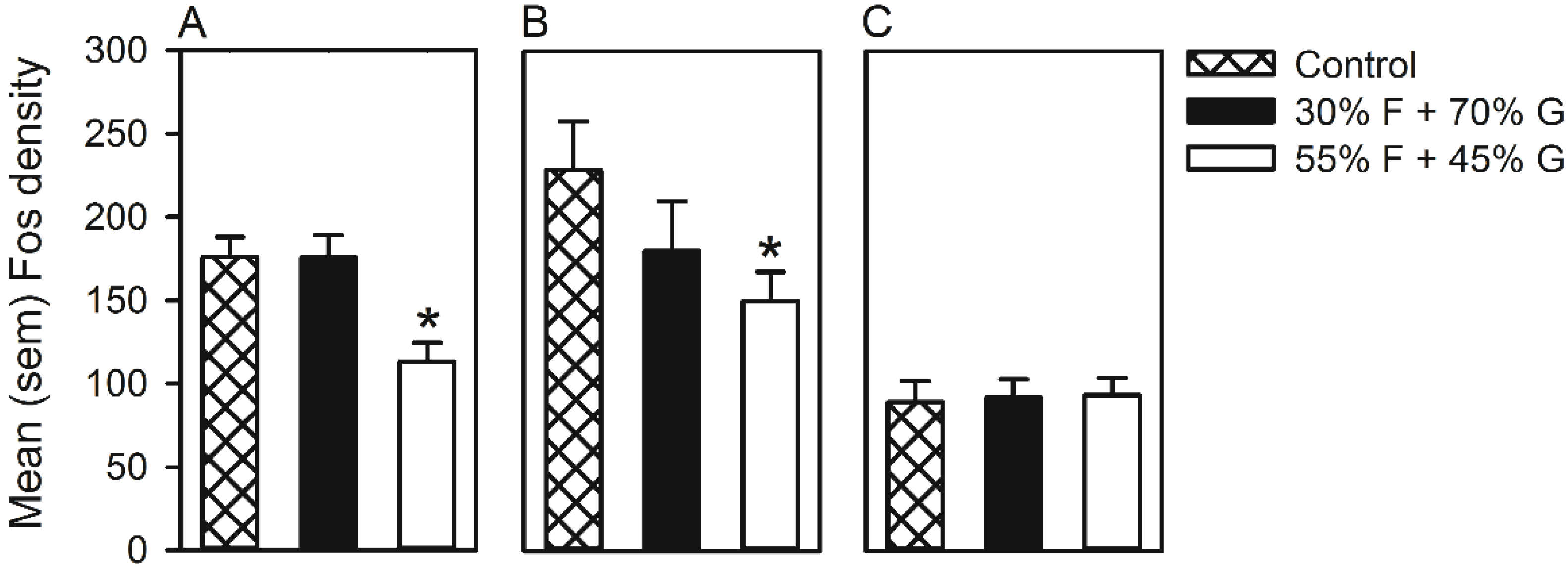
3.2. Experiment 2
3.2.1. Taste Reactivity
3.2.2. Operant Intraoral Self-Administration
| Concentration (%) of saccharin | |||||||
|---|---|---|---|---|---|---|---|
| 0.1% | |||||||
| SA session | 1 | 5 | 10 | 15 | 0.01% | 1% | 10% |
| Infusions | 27.7 (5.5) | 47.4 (11.7) | 39.5 (8.7) | 31.2 (7.5) | 18.9 (7.5) | 16.0 (4.4) | 15.8 (2.2) |
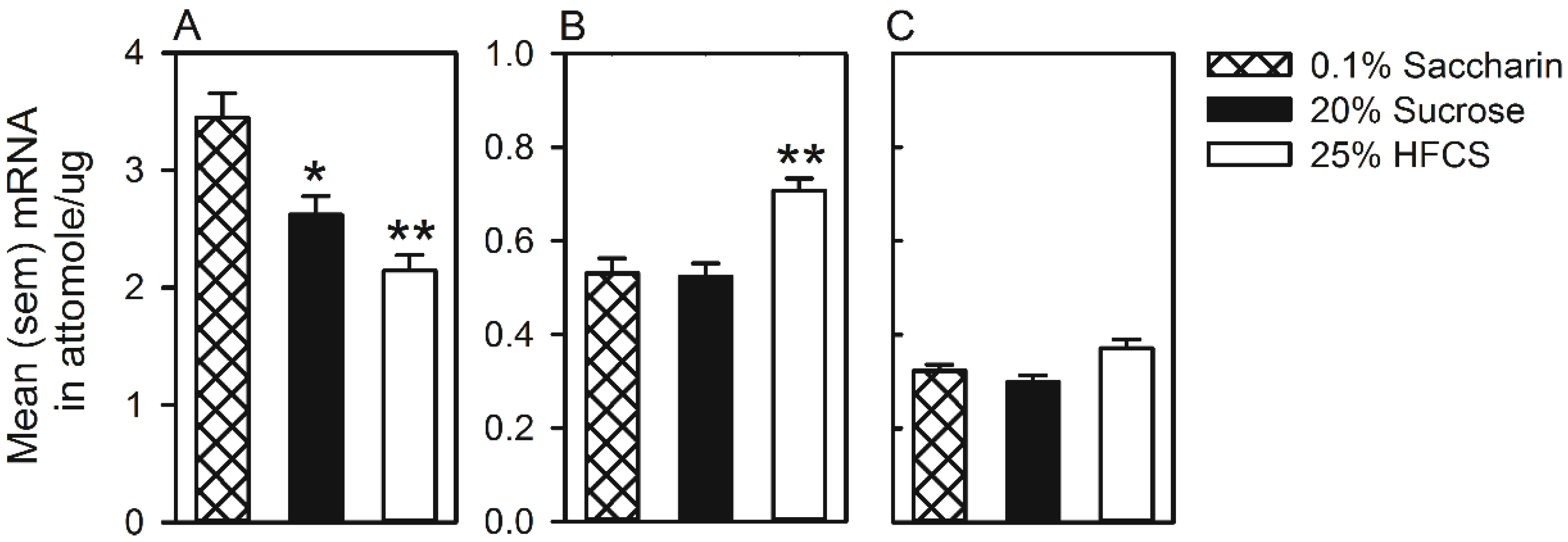
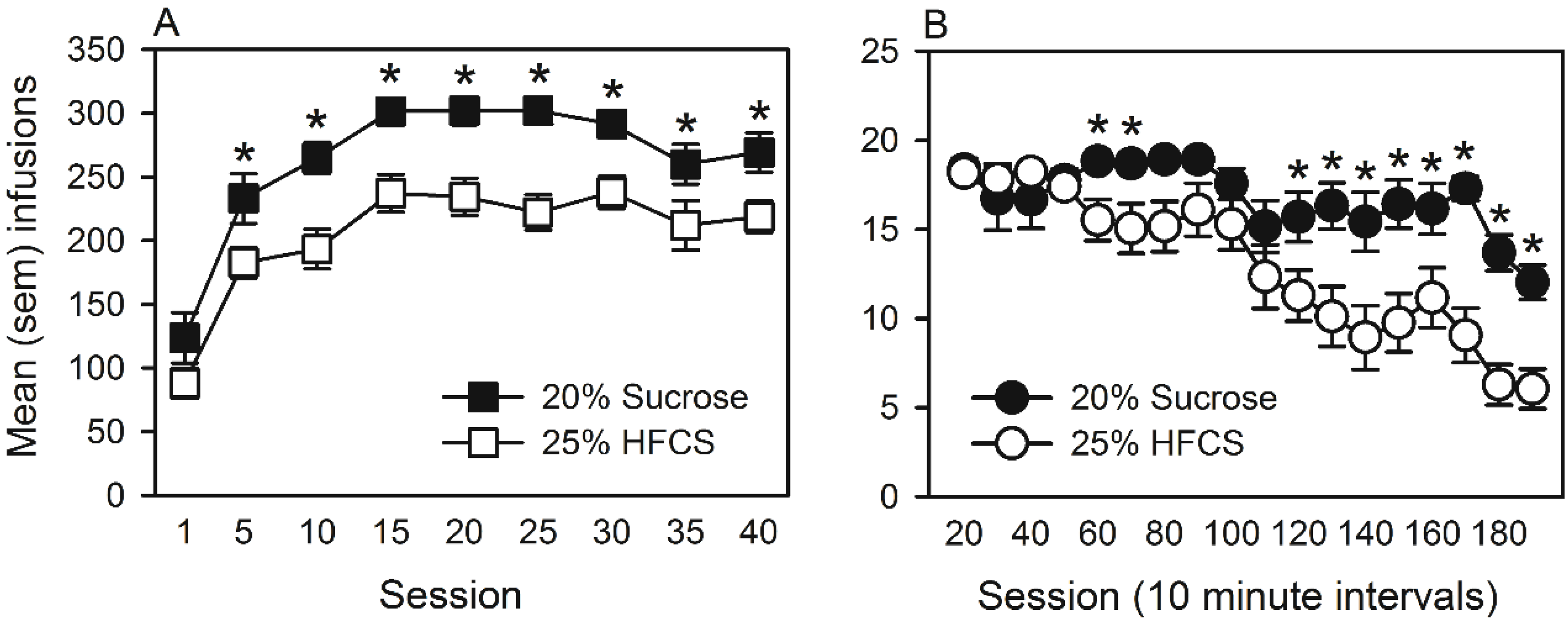
3.3. Experiment 3
Home Cage Self-Administration
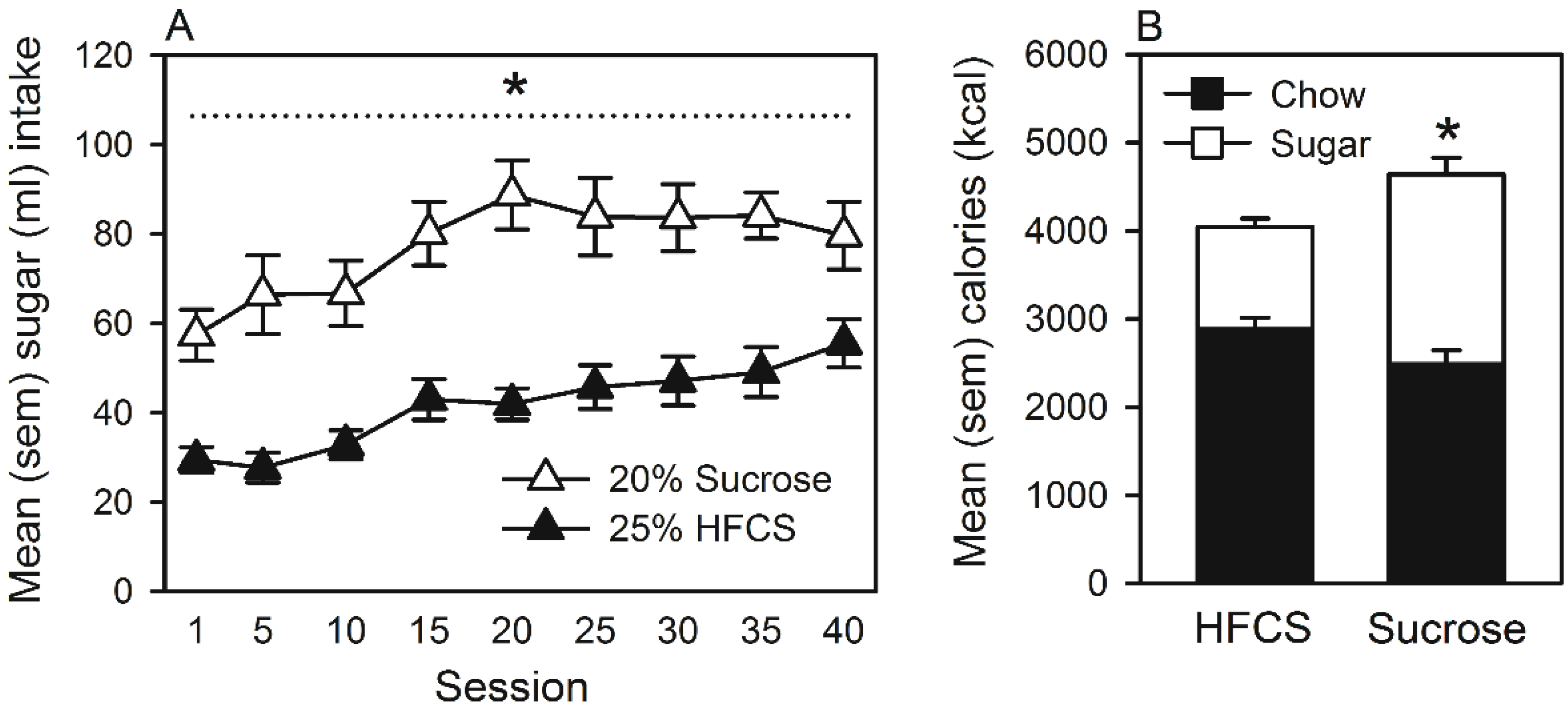
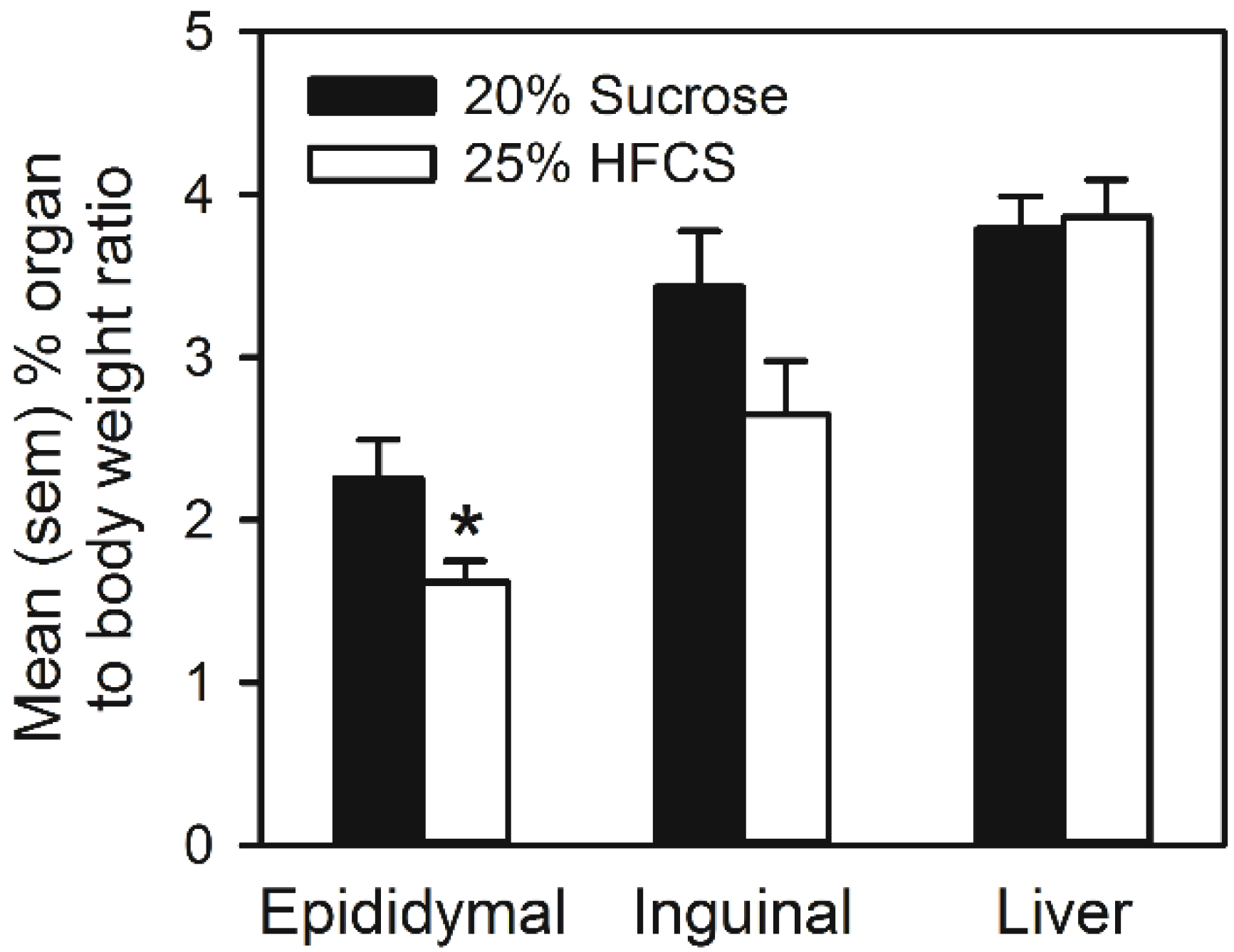
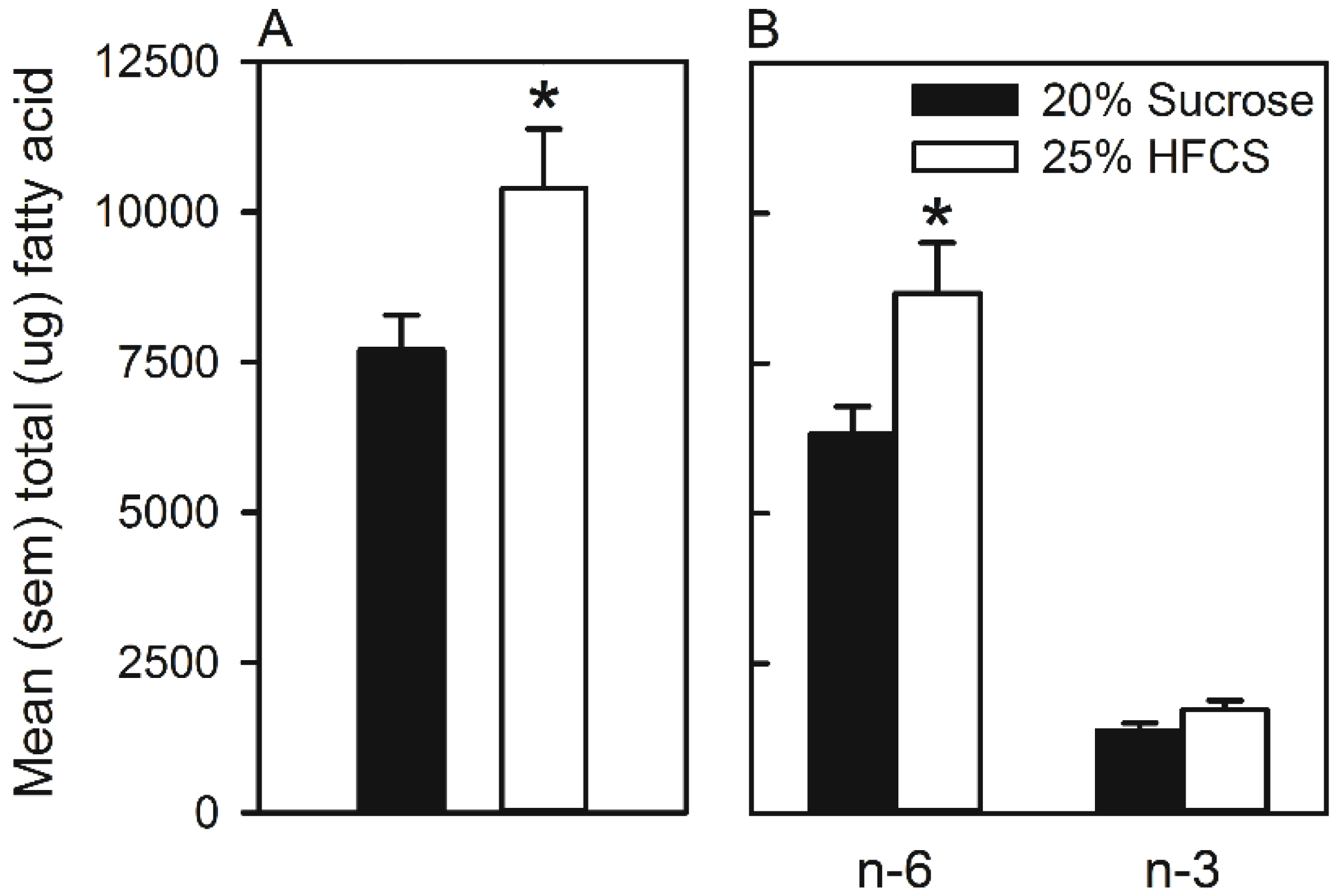
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO. World Health Statistics; World Health Organization: Geneva, Switzerland, 2012; Volume 27. [Google Scholar]
- Twells, L.K.; Gregory, D.M.; Reddigan, J.; Midodzi, W.K. Current and predicted prevalence of obesity in Canada: A trend analysis. Can. Medial Assoc. J. 2014, 2, 18–26. [Google Scholar]
- Van Baak, M.A. Nutrition as a link between obesity and cardiovascular disease: How can we stop the obesity epidemic? Thromb. Haemost. 2013, 110, 689–696. [Google Scholar] [CrossRef] [PubMed]
- National Task Force on the Prevention and Treatment of Obesity. Overweight, Obesity, and Health Risk. Arch. Intern. Med. 2000, 160, 898–904. [Google Scholar]
- Manson, J.E.; Skerrett, P.J.; Greenland, P.; VanItallie, T.B. The escalating pandemics of obesity and sedentary lifestyle. A call to action for clinicians. Arch. Intern. Med. 2004, 164, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Alfredo Martínez, J.; Martínez-Hernández, A.; Enríquez, L.; Moreno-Aliaga, M.J.; Moreno-Moreno, M.J.; Martí, A. Genetics of obesity. Public Health Nutr. 2007, 10, 1138–1144. [Google Scholar] [PubMed]
- Stice, E.; Spoor, S.; Bohon, C.; Small, D.M. Relation between obesity and blunted striatal response to food is moderated by TaqIA A1 allele. Science 2008, 322, 449–452. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, N.S.; Guarnieri, D.J.; DiLeone, R.J. Metabolic hormones, dopamine circuits, and feeding. Front. Neuroendocrinol. 2010, 31, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Wang, G.J.; Fowler, J.S.; Tomasi, D.; Baler, R. Food and drug reward: Overlapping circuits in human obesity and addiction. Curr. Top. Behav. Neurosci. 2012, 11, 1–24. [Google Scholar] [PubMed]
- Christakis, N.A.; Fowler, J.H. The spread of obesity in a large social network over 32 years. N. Engl. J. Med. 2007, 357, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Kirk, S.F.L.; Penney, T.L.; McHugh, T.-L.F. Characterizing the obesogenic environment: The state of the evidence with directions for future research. Obes. Rev. 2010, 11, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Fortuna, J.L. The obesity epidemic and food addiction: Clinical similarities to drug dependence. J. Psychoact. Drugs 2012, 44, 56–63. [Google Scholar] [CrossRef]
- Bray, G.A.; Popkin, B.M. Dietary fat intake does affect obesity! Am. J. Clin. Nutr. 1998, 68, 1157–1173. [Google Scholar] [PubMed]
- Bocarsly, M.E.; Powell, E.S.; Avena, N.M.; Hoebel, B.G. High-fructose corn syrup causes characteristics of obesity in rats: Increased body weight, body fat and triglyceride levels. Pharmacol. Biochem. Behav. 2010, 97, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.J.; Nakagawa, T.; Sanchez-Lozada, L.G.; Shafiu, M.; Sundaram, S.; Le, M.; Ishimoto, T.; Sautin, Y.Y.; Lanaspa, M.A. Sugar, uric acid, and the etiology of diabetes and obesity. Diabetes 2013, 62, 3307–3315. [Google Scholar] [CrossRef] [PubMed]
- Ifland, J.R.; Preuss, H.G.; Marcus, M.T.; Rourke, K.M.; Taylor, W.C.; Burau, K.; Jacobs, W.S.; Kadish, W.; Manso, G. Refined food addiction: A classic substance use disorder. Med. Hypotheses 2009, 72, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Cha, S.H.; Wolfgang, M.; Tokutake, Y.; Chohnan, S.; Lane, M.D. Differential effects of central fructose and glucose on hypothalamic malonyl-CoA and food intake. Proc. Natl. Acad. Sci. USA 2008, 105, 16871–16875. [Google Scholar] [CrossRef] [PubMed]
- Page, K.A.; Chan, O.; Arora, J.; Belfort-Deaguiar, R.; Dzuira, J.; Roehmholdt, B.; Cline, G.W.; Naik, S.; Sinha, R.; Constable, R.T.; et al. Effects of fructose vs. glucose on regional cerebral blood flow in brain regions involved with appetite and reward pathways. JAMA 2013, 309, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Moran, T.H.; McHugh, P.R. Distinctions among three sugars in their effects on gastric emptying and satiety. Am. J. Physiol. 1981, 241, 25–30. [Google Scholar]
- Bray, G.A.; Nielsen, S.J.; Popkin, B.M. Consumption of high-fructose corn syrup in beverages may play a role in the epidemic of obesity. Am. J. Clin. Nutr. 2004, 79, 537–543. [Google Scholar] [PubMed]
- Ferder, L.; Ferder, M.D.; Inserra, F. The role of high-fructose corn syrup in metabolic syndrome and hypertension. Curr. Hypertens. Rep. 2010, 12, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.; Tulloch, A.; Gold, M.S.; Avena, N.M. Hormonal and neural mechanisms of food reward, eating behaviour and obesity. Nat. Rev. Endocrinol. 2014, 10, 540–552. [Google Scholar] [CrossRef] [PubMed]
- Khitan, Z.; Kim, D.H. Fructose: A key factor in the development of metabolic syndrome and hypertension. J. Nutr. Metab. 2013, 2013, 682673. [Google Scholar] [CrossRef] [PubMed]
- White, J.S. Straight talk about high-fructose corn syrup: What it is and what it ain’t. Am. J. Clin. Nutr. 2008, 88, 1716S–1721S. [Google Scholar] [CrossRef] [PubMed]
- White, J.S. Challenging the fructose hypothesis: New perspectives on fructose consumption and metabolism. Adv. Nutr. 2013, 4, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Griffen, S.C.; Bair, B.R.; Swarbrick, M.M.; Keim, N.L.; Havel, P.J. Twenty-four-hour endocrine and metabolic profiles following consumption of high-fructose corn syrup-, sucrose-, fructose-, and glucose-sweetened beverages with meals. Am. J. Clin. Nutr. 2008, 87, 1194–1203. [Google Scholar] [PubMed]
- Soenen, S.; Westerterp-Plantenga, M.S. No differences in satiety or energy intake after high-fructose corn syrup, sucrose, or milk preloads. Am. J. Clin. Nutr. 2007, 86, 1586–1594. [Google Scholar] [PubMed]
- Akhavan, T.; Anderson, G.H. Effects of glucose-to-fructose ratios in solutions on subjective satiety, food intake, and satiety hormones in young men. Am. J. Clin. Nutr. 2007, 86, 1354–1363. [Google Scholar] [PubMed]
- Melanson, K.J.; Zukley, L.; Lowndes, J.; Nguyen, V.; Angelopoulos, T.J.; Rippe, J.M. Effects of high-fructose corn syrup and sucrose consumption on circulating glucose, insulin, leptin, and ghrelin and on appetite in normal-weight women. Nutrition 2007, 23, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Kovács, K.J. c-Fos as a transcription factor: A stressful (re)view from a functional map. Neurochem. Int. 1998, 33, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Kovács, K.J. Measurement of immediate-early gene activation- c-fos and beyond. J. Neuroendocrinol. 2008, 20, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, L.; Hoebel, B.G. Feeding and hypothalamic stimulation increase dopamine turnover in the accumbens. Physiol. Behav. 1988, 44, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Stratford, T.R.; Kelley, A.E. Evidence of a functional relationship between the nucleus accumbens shell and lateral hypothalamus subserving the control of feeding behavior. J. Neurosci. 1999, 19, 11040–11048. [Google Scholar] [PubMed]
- Berthoud, H.-R. Multiple neural systems controlling food intake and body weight. Neurosci. Biobehav. Rev. 2002, 26, 393–428. [Google Scholar] [CrossRef] [PubMed]
- Bellinger, L.L.; Bernardis, L.L. The dorsomedial hypothalamic nucleus and its role in ingestive behavior and body weight regulation: Lessons learned from lesioning studies. Physiol. Behav. 2002, 76, 431–442. [Google Scholar] [CrossRef] [PubMed]
- Grill, H.J.; Norgren, R. The taste reactivity test. I. Mimetic responses to gustatory stimuli in neurologically normal rats. Brain Res. 1978, 143, 263–279. [Google Scholar] [CrossRef] [PubMed]
- Levy, A.; Limebeer, C.L.; Ferdinand, J.; Shillingford, U.; Parker, L.A.; Leri, F. A novel procedure for evaluating the reinforcing properties of tastants in laboratory rats: Operant intraoral self-administration. J. Vis. Exp. 2014, 84, e50956. [Google Scholar] [PubMed]
- Zhou, Y.; Bendor, J.; Hofmann, L.; Randesi, M.; Ho, A.; Kreek, M.J. Mu opioid receptor and orexin/hypocretin mRNA levels in the lateral hypothalamus and striatum are enhanced by morphine withdrawal. J. Endocrinol. 2006, 191, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Georges, F.; Stinus, L.; Bloch, B.; Le Moine, C. Chronic morphine exposure and spontaneous withdrawal are associated with modifications of dopamine receptor and neuropeptide gene expression in the rat striatum. Eur. J. Neurosci. 1999, 11, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Leri, F.; Zhou, Y.; Goddard, B.; Levy, A.; Jacklin, D.; Kreek, M.J. Steady-state methadone blocks cocaine seeking and cocaine-induced gene expression alterations in the rat brain. Eur. Neuropsychopharmacol. 2009, 19, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.A.; Frattali, V.P. Saccharin. Diabetes Care 1989, 12, 74–80. [Google Scholar] [CrossRef] [PubMed]
- López, I.P.; Marti, A.; Milagro, F.I.; de los Angeles Zulet, M.; Moreno-Aliaga, M.J.; Martinez, J.A.; De Miguel, C. DNA microarray analysis of genes differentially expressed in diet-induced (cafeteria) obese rats. Obes. Res. 2003, 11, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Hausman, D.B.; Fine, J.B.; Tagra, K.; Fleming, S.S.; Martin, R.J.; diGirolamo, M. Regional fat pad growth and cellularity in obese zucker rats: Modulation by caloric restriction. Obes. Res. 2003, 11, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Dietary omega-3 fatty acid deficiency and high fructose intake in the development of metabolic syndrome, brain metabolic abnormalities, and non-alcoholic fatty liver disease. Nutrients 2013, 5, 2901–2923. [Google Scholar] [CrossRef] [PubMed]
- Rkieh, N.; Cloke, J.M.; Gallagher, N.; Winters, B.D.; Leri, F. Drugs of abuse as memory modulators: A study of cocaine in rats. Psychopharmacology 2014, 231, 2339–2348. [Google Scholar] [CrossRef] [PubMed]
- Leri, F.; Nahas, E.; Henderson, K.; Limebeer, C.L.; Parker, L.A.; White, N.M. Effects of post-training heroin and d-amphetamine on consolidation of win-stay learning and fear conditioning. J. Psychopharmacol. 2013, 27, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Limebeer, C.L.; Litt, D.E.; Parker, L.A. Effect of 5-HT3 antagonists and a 5-HT(1A) agonist on fluoxetine-induced conditioned gaping reactions in rats. Psychopharmacology 2009, 203, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Neath, K.N.; Limebeer, C.L.; Reilly, S.; Parker, L.A. Increased liking for a solution is not necessary for the attenuation of neophobia in rats. Behav. Neurosci. 2010, 124, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Carroll, M.E.; Morgan, A.D.; Anker, J.J.; Perry, J.L.; Dess, N.K. Selective breeding for differential saccharin intake as an animal model of drug abuse. Behav. Pharmacol. 2008, 19, 435–460. [Google Scholar] [CrossRef] [PubMed]
- Richardson, N.R.; Roberts, D.C. Progressive ratio schedules in drug self-administration studies in rats: A method to evaluate reinforcing efficacy. J. Neurosci. Methods 1996, 66, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Honsberger, M.J.M.; Leri, F. Fos expression in mesocorticolimbic areas during heroin place conditioning. Neuroreport 2008, 19, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Watson, C.; Paxinos, G. The Rat Brain in Stereotaxic Coordinates, 5th ed.; Academic Press: San Diego, CA, USA, 2004. [Google Scholar]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Besson, M.; Pelloux, Y.; Dilleen, R.; Theobald, D.E.; Lyon, A.; Belin-Rauscent, A.; Robbins, T.W.; Dalley, J.W.; Everitt, B.J.; Belin, D. Cocaine modulation of frontostriatal expression of Zif268, D2, and 5-HT2c receptors in high and low impulsive rats. Neuropsychopharmacology 2013, 38, 1963–1973. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.M.; Coscina, D.V.; Fletcher, P.J. The rewarding properties of neuropeptide Y in perifornical hypothalamus vs. nucleus accumbens. Peptides 2000, 21, 1279–1287. [Google Scholar] [CrossRef] [PubMed]
- Pandit, R.; la Fleur, S.E.; Adan, R.A.H. The role of melanocortins and Neuropeptide Y in food reward. Eur. J. Pharmacol. 2013, 719, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Philpot, K.; Smith, Y. CART peptide and the mesolimbic dopamine system. Peptides 2006, 27, 1987–1992. [Google Scholar] [CrossRef] [PubMed]
- Cason, A.M.; Smith, R.J.; Tahsili-Fahadan, P.; Moorman, D.E.; Sartor, G.C.; Aston-Jones, G. Role of orexin/hypocretin in reward-seeking and addiction: Implications for obesity. Physiol. Behav. 2010, 100, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, G.E.; Lyo, D. Anatomical Markers of Activity in Neuroendocrine Systems: Are we all “Fos-ed out ”? J. Neuroendocr. 2002, 14, 259–268. [Google Scholar]
- Robinson, T.; Berridge, K.C. The neural basis of drug craving: An incentive-sensitization theory of addiction. Brain Res. Rev. 1993, 18, 247–291. [Google Scholar] [CrossRef] [PubMed]
- Messier, C.; White, N.M. Contingent and non-contingent actions of sucrose and saccharin reinforcers: Effects on taste preference and memory. Physiol. Behav. 1984, 32, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, J.R.; Phillips, A.G.; Jakubovic, A.; Fibiger, H.C. Increased dopamine metabolism in the nucleus accumbens and striatum following consumption of a nutritive meal but not a palatable non-nutritive saccharin solution. Pharmacol. Biochem. Behav. 1986, 25, 1095–1100. [Google Scholar] [CrossRef] [PubMed]
- Weiss, F.; Lorang, M.T.; Bloom, F.E.; Koob, G.F. Oral alcohol self-administration stimulates dopamine release in the rat nucleus accumbens: Genetic and motivational determinants. J. Pharmacol. Exp. Ther. 1993, 267, 250–258. [Google Scholar] [PubMed]
- Carroll, M.E.; Lac, S.T. Acquisition of i.v. amphetamine and cocaine self-administration in rats as a function of dose. Psychopharmacology 1997, 129, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Deroche-Gamonet, V.; Belin, D.; Piazza, P.V. Evidence for addiction-like behavior in the rat. Science 2004, 305, 1014–1017. [Google Scholar] [CrossRef] [PubMed]
- Sclafani, A.; Ackroff, K. Reinforcement value of sucrose measured by progressive ratio operant licking in the rat. Physiol. Behav. 2003, 79, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.M.; Kenny, P.J. Dopamine D2 receptors in addiction-like reward dysfunction and compulsive eating in obese rats. Nat. Neurosci. 2010, 13, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Spangler, R.; Wittkowski, K.M.; Goddard, N.L.; Avena, N.M.; Hoebel, B.G.; Leibowitz, S.F. Opiate-like effects of sugar on gene expression in reward areas of the rat brain. Brain Res. Mol. Brain Res. 2004, 124, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.; Parkinson, J.A.; Connor, T.M.; Dickinson, A.; Everitt, B.J. Involvement of the central nucleus of the amygdala and nucleus accumbens core in mediating Pavlovian influences on instrumental behaviour. Eur. J. Neurosci. 2001, 13, 1984–1992. [Google Scholar] [CrossRef] [PubMed]
- Bassareo, V.; De Luca, M.A.; Di Chiara, G. Differential impact of pavlovian drug conditioned stimuli on in vivo dopamine transmission in the rat accumbens shell and core and in the prefrontal cortex. Psychopharmacology 2007, 191, 689–703. [Google Scholar] [CrossRef] [PubMed]
- Kelley, A.E. Neural integrative activities of nucleus accumbens subregions in relation to learning and motivation. Psychobiology 1999, 27, 198–213. [Google Scholar]
- Leri, F.; Zhou, Y.; Goddard, B.; Cummins, E.; Kreek, M.J. Effects of high-dose methadone maintenance on cocaine place conditioning, cocaine self-administration, and mu-opioid receptor mRNA expression in the rat brain. Neuropsychopharmacology 2006, 31, 1462–1474. [Google Scholar] [CrossRef] [PubMed]
- Din, J.N.; Newby, D.E.; Flapan, A.D. Omega 3 fatty acids and cardiovascular disease--fishing for a natural treatment. BMJ 2004, 328, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Browning, L.M.; Krebs, J.D.; Moore, C.S.; Mishra, G.D.; O’Connell, M.A.; Jebb, S.A. The impact of long chain n-3 polyunsaturated fatty acid supplementation on inflammation, insulin sensitivity and CVD risk in a group of overweight women with an inflammatory phenotype. Diabetes. Obes. Metab. 2007, 9, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Browning, L.M. n-3 Polyunsaturated fatty acids, inflammation and obesity-related disease. Proc. Nutr. Soc. 2003, 62, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Dangardt, F.; Osika, W.; Chen, Y.; Nilsson, U.; Gan, L.-M.; Gronowitz, E.; Strandvik, B.; Friberg, P. Omega-3 fatty acid supplementation improves vascular function and reduces inflammation in obese adolescents. Atherosclerosis 2010, 212, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Hsu, T.M.; Konanur, V.R.; Taing, L.; Usui, R.; Kayser, B.D.; Goran, M.I.; Kanoski, S.E. Effects of sucrose and high fructose corn syrup consumption on spatial memory function and hippocampal neuroinflammation in adolescent rats. Hippocampus 2015, 25, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Ackroff, K.; Sclafani, A. Rats’ preferences for high fructose corn syrup vs. sucrose and sugar mixtures. Physiol. Behav. 2011, 102, 548–552. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Levy, A.; Marshall, P.; Zhou, Y.; Kreek, M.J.; Kent, K.; Daniels, S.; Shore, A.; Downs, T.; Fernandes, M.F.; Mutch, D.M.; et al. Fructose:Glucose Ratios—A Study of Sugar Self-Administration and Associated Neural and Physiological Responses in the Rat. Nutrients 2015, 7, 3869-3890. https://doi.org/10.3390/nu7053869
Levy A, Marshall P, Zhou Y, Kreek MJ, Kent K, Daniels S, Shore A, Downs T, Fernandes MF, Mutch DM, et al. Fructose:Glucose Ratios—A Study of Sugar Self-Administration and Associated Neural and Physiological Responses in the Rat. Nutrients. 2015; 7(5):3869-3890. https://doi.org/10.3390/nu7053869
Chicago/Turabian StyleLevy, AnneMarie, Paul Marshall, Yan Zhou, Mary Jeanne Kreek, Katrina Kent, Stephen Daniels, Ari Shore, Tiana Downs, Maria Fernanda Fernandes, David M. Mutch, and et al. 2015. "Fructose:Glucose Ratios—A Study of Sugar Self-Administration and Associated Neural and Physiological Responses in the Rat" Nutrients 7, no. 5: 3869-3890. https://doi.org/10.3390/nu7053869
APA StyleLevy, A., Marshall, P., Zhou, Y., Kreek, M. J., Kent, K., Daniels, S., Shore, A., Downs, T., Fernandes, M. F., Mutch, D. M., & Leri, F. (2015). Fructose:Glucose Ratios—A Study of Sugar Self-Administration and Associated Neural and Physiological Responses in the Rat. Nutrients, 7(5), 3869-3890. https://doi.org/10.3390/nu7053869




