No Positive Influence of Ingesting Chia Seed Oil on Human Running Performance
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects
2.2. Research Design
2.3. ALA Bioavailability Study
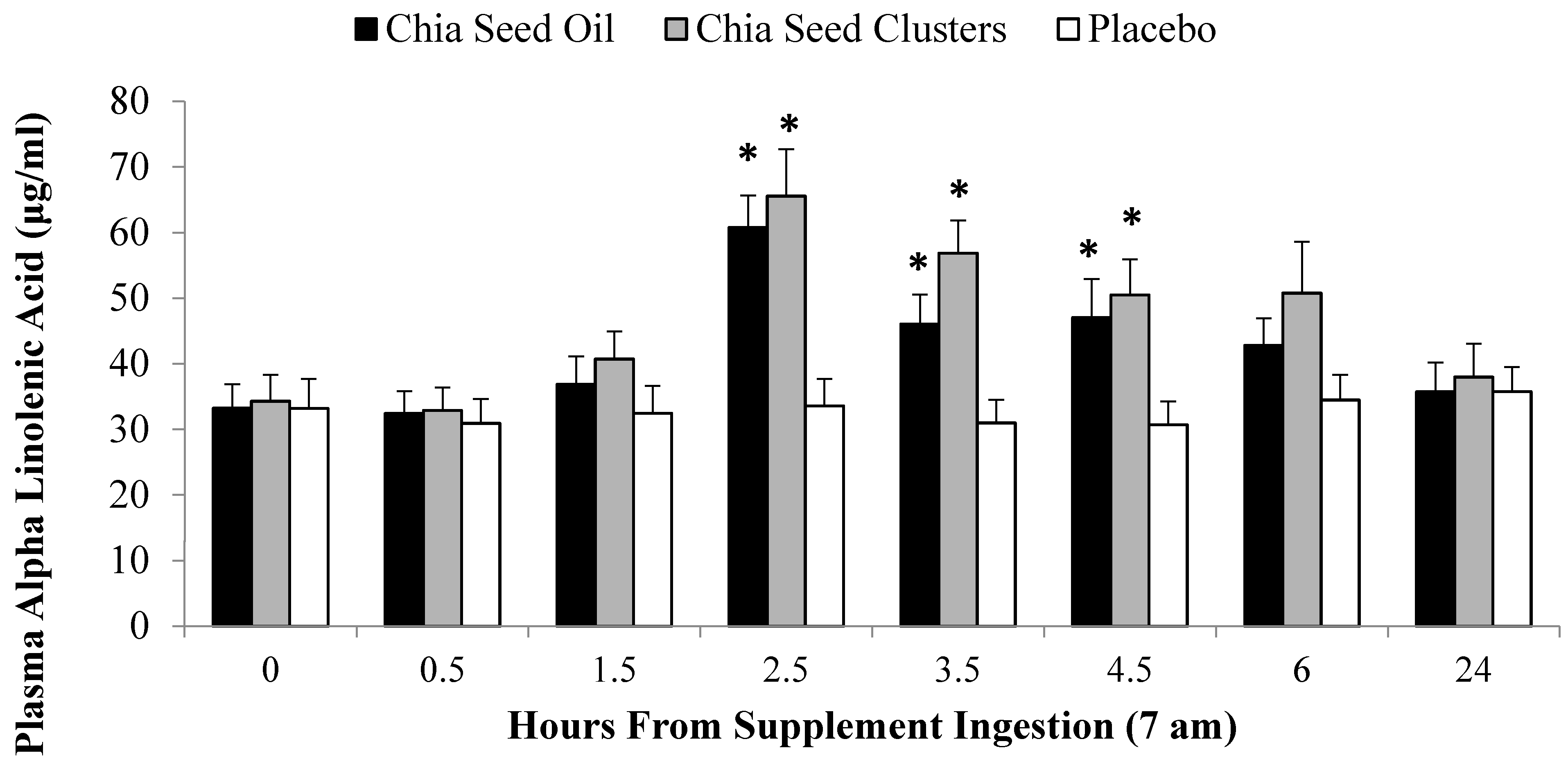
2.4. Exercise Study Protocol
2.5. Complete Blood Count, Lactate, Glucose, Cortisol
2.6. Plasma Cytokines
2.7. Plasma ALA, EPA, DPA, and DHA
2.8. Statistics
3. Results
| Variable | Mean ± SE |
|---|---|
| Age (year) | 38.0 ± 1.7 |
| Height (m) | 1.72 ± 0.02 |
| Weight (kg) | 71.8 ± 3.0 |
| Body fat % | 19.9 ± 1.6 |
| VO2max (mL kg−1 min−1) | 47.9 ± 1.6 |
| Maximal heart rate (beats min−1) | 180 ± 2.5 |
| Water | Chia Seed Oil | p-Value | |
|---|---|---|---|
| Time (h) | 1.86 ± 0.10 | 1.91 ± 0.13 | 0.577 |
| Distance (km) | 19.2 ± 1.1 | 19.8 ± 1.5 | 0.471 |
| VO2 (mL·kg−1 min−1) | 34.4 ± 0.8 | 34.1 ± 0.7 | 0.417 |
| HR (beats min−1) | 154 ± 2.3 | 156 ± 2.2 | 0.015 |
| Ventilation (L min−1) | 69.2 ± 3.4 | 68.0 ± 3.2 | 0.317 |
| RPE (average) | 13.5 ± 0.3 | 13.3 ± 0.3 | 0.438 |
| RER (average) | 0.92 ± 0.01 | 0.92 ± 0.01 | 0.391 |
| Weight change (kg) | 1.4 ± 0.2 | 1.5 ± 0.2 | 0.193 |
| Plasma volume shift (%) | −8.6 ± 1.0 | −10.7 ± 1.5 | 0.194 |
| Lactate, pre-exercise | 0.53 ± 0.04 | 0.55 ± 0.04 | |
| Lactate, post-exercise | 1.11 ± 0.17 | 0.98 ± 0.09 | 0.308 |
| Glucose, pre-exercise | 3.98 ± 0.19 | 4.13 ± 0.12 | |
| Glucose, post-exercise | 4.56 ± 0.22 | 4.47 ± 0.23 | 0.272 |
| Variable | Pre-Run | Post-Run | 1.0-h Post-Run | p-Values: Time; Interaction |
|---|---|---|---|---|
| Cortisol (nmol L−1) | <0.001; 0.055 | |||
| Water | 364 ± 16.6 | 433 ± 33.1 | 359 ± 27.6 | |
| Chia | 375 ± 16.6 | 505 ± 35.9 | 395 ± 27.6 | |
| Leukocytes (109 L−1) | <0.001; 0.208 | |||
| Water | 5.33 ± 0.25 | 16.4 ± 0.9 | 12.7 ± 0.6 | |
| Chia | 5.51 ± 0.28 | 16.4 ± 0.9 | 12.7 ± 0.7 | |
| IL-6 (pg mL−1) | <0.001; 0.368 | |||
| Water | 0.88 ± 0.16 | 9.37 ± 1.67 | 5.79 ± 0.92 | |
| Chia | 1.02 ± 0.28 | 8.77 ± 0.69 | 5.97 ± 0.97 | |
| IL-8 (pg mL−1) | <0.001; 0.116 | |||
| Water | 5.84 ± 0.49 | 13.7 ± 1.5 | 10.7 ± 1.1 | |
| Chia | 5.23 ± 0.42 | 11.8 ± 1.1 | 7.91 ± 0.74 | |
| IL-10 (pg mL−1) | <0.001; 0.680 | |||
| Water | 2.21 ± 0.21 | 38.3 ± 13.7 | 16.7 ± 4.3 | |
| Chia | 2.00 ± 0.19 | 33.7 ± 10.8 | 16.9 ± 4.7 | |
| TNFα (pg mL−1) | <0.001; 0.259 | |||
| Water | 3.86 ± 0.18 | 4.59 ± 0.26 | 4.19 ± 0.23 | |
| Chia | 3.90 ± 0.16 | 4.30 ± 0.20 | 4.10 ± 0.20 |
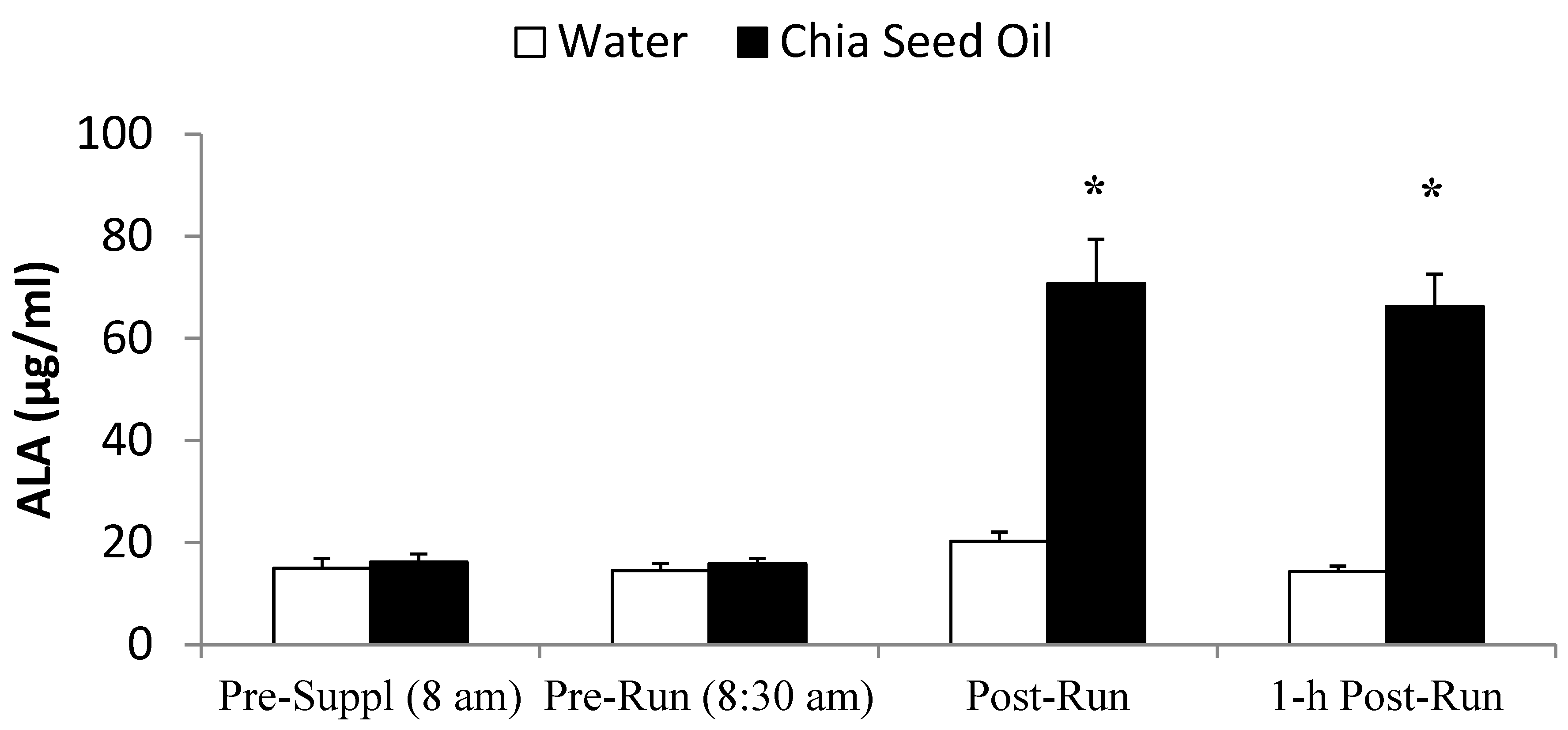
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Burdge, G.C. Metabolism of alpha-linolenic acid in humans. Prostaglandins Leukot Essent. Fat. Acids 2006, 75, 161–168. [Google Scholar] [CrossRef]
- Arterburn, L.M.; Hall, E.B.; Oken, H. Distribution, interconversion, and dose response of n-3 fatty acids in humans. Am. J. Clin. Nutr. 2006, 83, 1467S–1476S. [Google Scholar] [PubMed]
- U.S. Department of Agriculture, Agricultural Research Service. Nutrient Intakes from Food and Beverages: Mean Amounts Consumed per Individual, by Gender and Age. 2014. Available online: http://www.ars.usda.gov/ba/bhnrc/fsrg (accessed on 29 November 2014). [Google Scholar]
- Food and Nutrition Board, Institute of Medicine. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients); Panel on Macronutrients, Panel on Defination of Dietary Fiber, Subcommittees on Upper Reference Levels of Nutrients and Interpretation and Uses of Dietary Reference Intakes, Standing Committee on the Scientific Evaluation of Dietary Reference Intakes, Eds.; National Academy Press: Washington, DC, USA, 2002. [Google Scholar]
- Balk, E.M.; Lichtenstein, A.H.; Chung, M.; Kupelnick, B.; Chew, P.; Lau, J. Effects of omega-3 fatty acids on serum markers of cardiovascular disease risk: A systematic review. Atherosclerosis 2006, 189, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Vedtofte, M.S.; Jakobsen, M.U.; Lauritzen, L.; O’Reilly, E.J.; Virtamo, J.; Knekt, P.; Colditz, G.; Hallmans, G.; Buring, J.; Steffen, L.M.; et al. Association between the intake of α-linolenic acid and the risk of CHD. Br. J. Nutr. 2014, 112, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K. Flaxseed and cardiovascular health. J. Cardiovasc. Pharmacol. 2009, 54, 369–377. [Google Scholar] [CrossRef] [PubMed]
- De Goede, J.; Verschuren, W.M.; Boer, J.M.; Bromhout, D.; Geleijnse, J.M. Alpha-linolenic acid intake and 10-year incidence of coronary heart disease and stroke in 20,000 middle-aged men and women in the Netherlands. PLoS ONE 2011, 6, e17967. [Google Scholar] [CrossRef] [PubMed]
- Egert, S.; Baxheinrich, A.; Lee-Barkey, Y.H.; Tschoepe, D.; Wahrburg, U.; Stratmann, B. Effects of an energy-restricted diet rich in plant-derived α-linolenic acid on systemic inflammation and endothelial function in overweight-to-obese patients with metabolic syndrome traits. Br. J. Nutr. 2014, 112, 1315–1322. [Google Scholar] [CrossRef] [PubMed]
- Geleijnse, J.M.; de Goede, J.; Brouwer, I.A. Alpha-linolenic acid: Is it essential to cardiovascular health? Curr. Atheroscler. Rep. 2010, 12, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Rallidis, L.S.; Paschos, G.; Liakos, G.K.; Velissaridou, A.H.; Anastasiadis, G.; Zampelas, A. Dietary-linolenic acid decreases C-reactive protein, serum amyloid A and interleukin-6 in dyslipidaemic patients. Atherosclerosis 2003, 167, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Bemelmans, W.J.; Lefrandt, J.D.; Feskens, E.J.; van Haelst, P.L.; Broer, J.; Meyboom-de Jong, B.; May, J.F.; Tervaert, J.W.; Smit, A.J. Increased alpha-linolenic acid intake lowers C-reactive protein, but has no effect on markers of atherosclerosis. Eur. J. Clin. Nutr. 2004, 58, 1083–1089. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Etherton, T.D.; Martin, K.R.; West, S.G.; Gillies, P.J.; Kris-Etherton, P.M. Dietary-linolenic acid reduces inflammatory and lipid cardiovascular risk factors in hypercholesterolemic men and women. J. Nutr. 2004, 134, 2991–2997. [Google Scholar] [PubMed]
- Cornish, S.M.; Chilibeck, P.D. Alpha-linolenic acid supplementation and resistance training in older adults. Appl. Phys. Nutr. Metab. 2009, 34, 49–59. [Google Scholar] [CrossRef]
- Nieman, D.C.; Scherr, J.; Luo, B.; Meaney, M.P.; Dréau, D.; Sha, W.; Dew, D.A.; Henson, D.A.; Pappan, K.L. Influence of pistachios on performance and exercise-induced inflammation, oxidative stress, immune dysfunction, and metabolite shifts in cyclists: A randomized, crossover trial. PLoS ONE 2014, 9, e113725. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C.; Cayea, E.J.; Austin, M.D.; Henson, D.A.; McAnulty, S.R.; Jin, F. Chia seed does not promote weight loss or alter disease risk factors in overweight adults. Nutr. Res. 2009, 29, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C.; Gillitt, N.; Jin, F.; Henson, D.A.; Kennerly, K.; Shanely, R.A.; Ore, B.; Su, M.; Schwartz, S. Chia seed supplementation and disease risk factors in overweight women: A metabolomics investigation. J. Altern. Complement. Med. 2012, 18, 700–708. [Google Scholar] [CrossRef] [PubMed]
- Jin, F.; Nieman, D.C.; Sha, W.; Xie, G.; Qiu, Y.; Jia, W. Supplementation of milled chia seeds increases plasma ALA and EPA in postmenopausal women. Plant Foods Hum. Nutr. 2012, 67, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Cruz, O.; Paredes-López, O. Phytochemical profile and nutraceutical potential of chia seeds (Salvia hispanica L.) by ultra high performance liquid chromatography. J. Chromatogr. A 2014, 1346, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Mohd Ali, N.; Yeap, S.K.; Ho, W.Y.; Beh, B.K.; Tan, S.W.; Tan, S.G. The promising future of chia, Salvia hispanica L. J. Biomed. Biotechnol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Illian, T.G.; Casey, J.C.; Bishop, P.A. Omega 3 Chia seed loading as a means of carbohydrate loading. J. Strength Cond. Res. 2011, 25, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Bruce, R.A.; Kusumi, F.; Hosmer, D. Maximal oxygen intake and nomographic assessment of functional aerobic impairment in cardiovascular disease. Am. Heart J. 1973, 85, 546–562. [Google Scholar] [CrossRef] [PubMed]
- Dill, D.B.; Costill, D.L. Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J. Appl. Physiol. 1974, 37, 247–248. [Google Scholar] [PubMed]
- DeLany, J.P.; Windhauser, M.M.; Champagne, C.M.; Bray, G.A. Differential oxidation of individual dietary fatty acids in humans. Am. J. Clin. Nutr. 2000, 72, 905–911. [Google Scholar] [PubMed]
- Clouet, P.; Niot, I.; Bézard, J. Pathway of alpha-linolenic acid through the mitochondrial outer membrane in the rat liver and influence on the rate of oxidation. Comparison with linoleic and oleic acids. Biochem. J. 1989, 263, 867–873. [Google Scholar] [PubMed]
- Dai, J.; Ziegler, T.R.; Bostick, R.M.; Manatunga, A.K.; Jones, D.P.; Goldberg, J.; Miller, A.; Vogt, G.; Wilson, P.W.; Jones, L.; et al. High habitual dietary alpha-linolenic acid intake is associated with decreased plasma soluble interleukin-6 receptor concentrations in male twins. Am. J. Clin. Nutr. 2010, 92, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Etherton, T.D.; Martin, K.R.; Gillies, P.J.; West, S.G.; Kris-Etherton, P.M. Dietary alpha-linolenic acid inhibits proinflammatory cytokine production by peripheral blood mononuclear cells in hypercholesterolemic subjects. Am. J. Clin. Nutr. 2007, 85, 385–391. [Google Scholar] [PubMed]
- Nelson, T.L.; Stevens, J.R.; Hickey, M.S. Inflammatory markers are not altered by an eight week dietary alpha-linolenic acid intervention in healthy abdominally obese adult males and females. Cytokine 2007, 38, 101–106. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nieman, D.C.; Gillitt, N.D.; Meaney, M.P.; Dew, D.A. No Positive Influence of Ingesting Chia Seed Oil on Human Running Performance. Nutrients 2015, 7, 3666-3676. https://doi.org/10.3390/nu7053666
Nieman DC, Gillitt ND, Meaney MP, Dew DA. No Positive Influence of Ingesting Chia Seed Oil on Human Running Performance. Nutrients. 2015; 7(5):3666-3676. https://doi.org/10.3390/nu7053666
Chicago/Turabian StyleNieman, David C., Nicholas D. Gillitt, Mary Pat Meaney, and Dustin A. Dew. 2015. "No Positive Influence of Ingesting Chia Seed Oil on Human Running Performance" Nutrients 7, no. 5: 3666-3676. https://doi.org/10.3390/nu7053666
APA StyleNieman, D. C., Gillitt, N. D., Meaney, M. P., & Dew, D. A. (2015). No Positive Influence of Ingesting Chia Seed Oil on Human Running Performance. Nutrients, 7(5), 3666-3676. https://doi.org/10.3390/nu7053666






