Selenium and Its Supplementation in Cardiovascular Disease—What do We Know?
Abstract
:1. Introduction
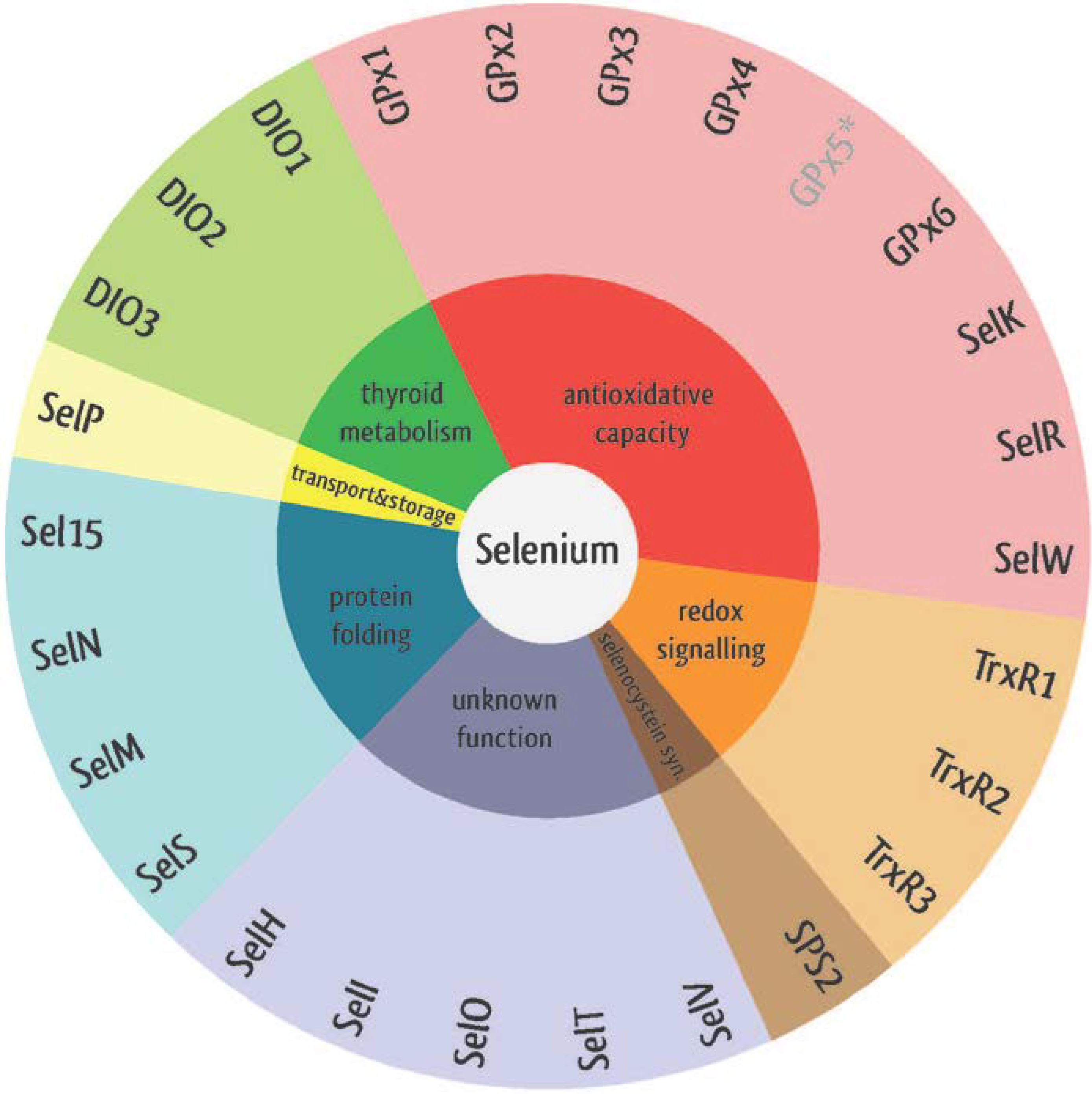
2. The Role of Selenium and Selenoproteins in the Human Body
2.1. Glutathione Peroxidases
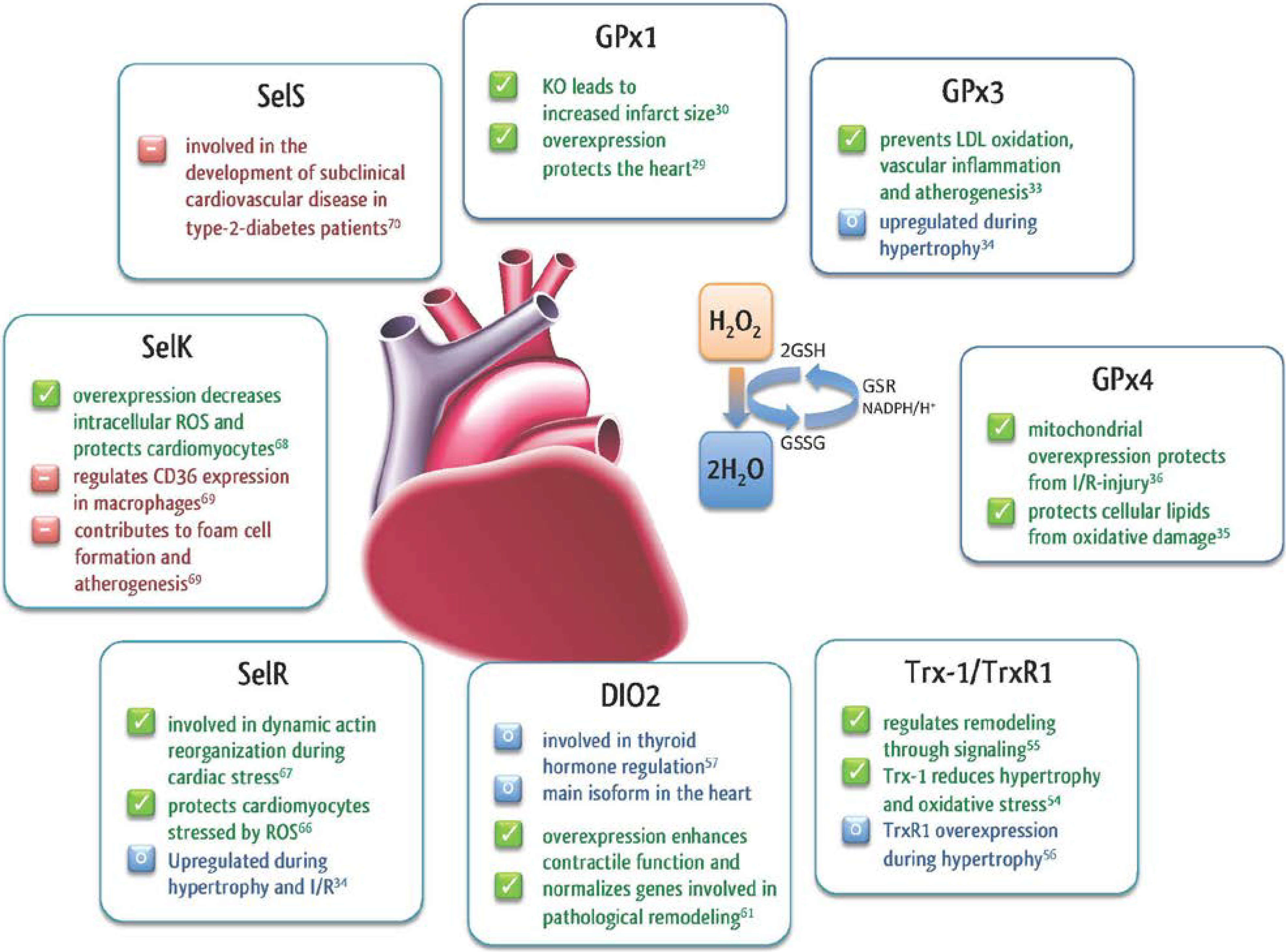
2.2. Thioredoxin Reductase (TrxR)
2.3. Thyroid Hormone Deiodinases (DIO)
2.4. Selenoprotein R (SelR, MsrB1)
2.5. Other Selenoproteins in the Heart
3. The Functional Role of Selenium in the Cardiovascular System
3.1. The Role of Selenium in the Development of Cardiomyopathy—Keshan Disease
3.2. The Role of Selenium and Selenoproteins in Myocardial I/R
3.3. The Role of Selenium in Cardiovascular Disease
3.3.1. Selenium as Biomarker
3.3.2. Selenium in Myocardial Infarction and Cardiovascular Stress
3.3.3. Prophylactic Selenium Supplementation
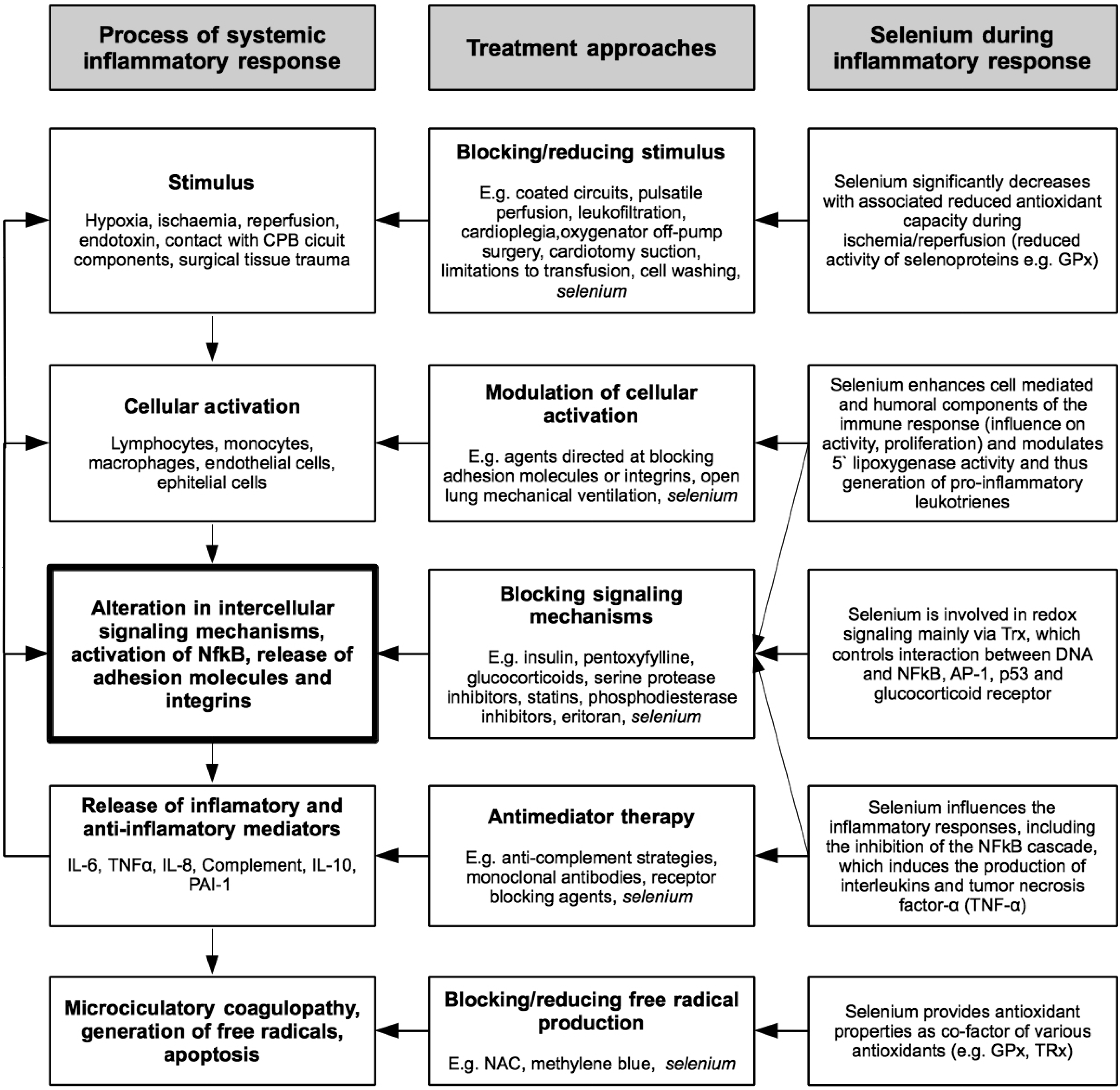
4. The Significance of Selenium in Cardiac Surgery
| Ref. | Study Design | Population | Selenium Salt from and Dosing Regime | Outcomes |
|---|---|---|---|---|
| Leong 2010 [115] | Randomized controlled trial (double-blind) | Patients undergoing elective CABG and/or valve surgery n = 117 | Coenzyme Q10, magnesium orotate, lipoic acid, omega-3 fatty acids and selenium vs. placebo (approximately 2 months before and 1 month after surgery) | Metabolic therapy reduced plasma troponin I, 24 h postoperatively from 1.5 (1.2–1.8) (geometric mean 95% CI) μg L L−1, to 2.1 (1.8–2.6) μg L−1 (p = 0.003) and shortened the mean length of postoperative hospital stay by 1.2 days from 8.1 (7.5–8.7) to 6.9 (6.4–7.4) days (p = 0.004) and reduced hospital costs. Metabolic therapy was inexpensive and had no clinically significant side effects. |
| Stoppe 2011 [113] | Prospective observational study | Patients scheduled for cardiac surgery with CPB n = 60 | - | Fifty patients exhibited a significant selenium deficiency already before surgery. In all patients, blood levels of selenium, copper, and zinc were significantly reduced after end of surgery when compared to preoperative values (selenium: 89.05 ± 12.65 to 70.84 ± 10.46 µg; zinc: 5.15 ± 0.68 to 4.19 ± 0.73 mg L−1; copper: 0.86 ± 0.15 to 0.65 ± 0.14 mg L−1; p < 0.001). Selenium concentrations at end of surgery were independently associated with the postoperative occurrence of multiorgan failure (p = 0.0026, odds ratio 0.8479, 95% confidence interval 0.7617 to 0.9440). |
| Koszta 2012 [114] | Prospective observational study | Patients scheduled for cardiac surgery with CPB n = 197 | - | Selenium levels were significantly lower in non-survivors 102.2 ± 19.5 μg L−1 compared with survivors 111.1 ± 16.9 μg L−1 (p = 0.047), and the mean age, European System for Cardiac Operative Risk Evaluation (EuroSCORE) values, and troponin concentrations were significantly higher in the non-survivors. Lower selenium levels identified as a risk factor for postoperative mortality. |
| Stoppe 2013 [116] | Prospective observational study | Patients scheduled for cardiac surgery with CPB n = 104 | Intravenous bolus of 2.000 μg selenium after induction of anesthesia and 1.000 μg selenium every day further during ICU stay | Preoperative sodium-selenite administration increased selenium blood concentrations to normal values on ICU admission, but failed to prevent a significant decrease of circulating selenium on the first postoperative day. |
| Stevanovic 2014 [110] | Randomized controlled trial (comparison: Off- vs. on-pump CABG) | Patients undergoing elective CABG n = 40 | - | Both groups showed a comparable decrease of circulating selenium concentrations. Likewise, levels of oxidative stress and IL-6 were comparable in both groups. Selenium levels correlated with antioxidant capacity (GPx: r = 0.720; p < 0.001) and showed a negative correlation to myocardial damage (CK-MB: r = −0.571, p < 0.001). Low postoperative selenium levels had a high predictive value for the occurrence of any postoperative complication. |
| Sustain CSX Trial 2014 [117] | Randomized controlled trial (double-blind) | Patients undergoing CABG plus valve surgery, multiple valve replacement surgery, patients with a high perioperative risk profile (≥5% EuroSCORE II). n = 1400 | Intravenous bolus of 2.000 μg selenium after induction of anesthesia and 1.000 μg selenium every day further during ICU stay | On-going, recruiting |
| Haberthuer ClinicalTrials.gov Identifier: NCT01141556 | Randomized controlled trial (double-blind) | Elective all-cause cardiac surgery n = 410 | Loading dose of 4.000 μg, daily dosage of 1.000 μg of selenium vs. placebo | On-going, on analysis |
5. Future Directions and Therapeutic Implications
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Muth, O.H.; Oldfield, J.E.; Remmert, L.F.; Schubert, J.R. Effects of selenium and vitamin E on white muscle disease. Science 1958, 128, 1090. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Alarcon, M.; Cabrera-Vique, C. Selenium in food and the human body: A review. Sci. Total Environ. 2008, 400, 115–141. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.D. Assessment of requirements for selenium and adequacy of selenium status: A review. Eur. J. Clin. Nutr. 2004, 58, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Fairweather-Tait, S.J.; Bao, Y.; Broadley, M.R.; Collings, R.; Ford, D.; Hesketh, J.E.; Hurst, R. Selenium in human health and disease. Antioxid. Redox Signal. 2011, 14, 1337–1383. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Ge, K.; Chen, J.; Chen, X. Selenium-related endemic diseases and the daily selenium requirement of humans. World Rev. Nutr. Diet. 1988, 55, 98–152. [Google Scholar] [PubMed]
- MacFarquhar, J.K.; Broussard, D.L.; Melstrom, P.; Hutchinson, R.; Wolkin, A.; Martin, C.; Burk, R.F.; Dunn, J.R.; Green, A.L.; Hammond, R.; et al. Acute selenium toxicity associated with a dietary supplement. Arch. Intern. Med. 2010, 170, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. Food-chain selenium and human health: Emphasis on intake. Br. J. Nutr. 2008, 100, 254–268. [Google Scholar] [PubMed]
- Lech, T. Suicide by sodium tetraoxoselenate(VI) poisoning. Forensic Sci. Int. 2002, 130, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Arthur, J.R.; McKenzie, R.C.; Beckett, G.J. Selenium in the immune system. J. Nutr. 2003, 133, 1457S–1459S. [Google Scholar] [PubMed]
- McKenzie, R.C.; Rafferty, T.S.; Beckett, G.J. Selenium: An essential element for immune function. Immunol. Today 1998, 19, 342–345. [Google Scholar] [CrossRef] [PubMed]
- Kiremidjian-Schumacher, L.; Roy, M. Selenium and immune function. Z. Ernahrungswiss. 1997, 37, 50–56. [Google Scholar]
- Ahrens, I.; Ellwanger, C.; Smith, B.K.; Bassler, N.; Chen, Y.C.; Neudorfer, I.; Ludwig, A.; Bode, C.; Peter, K. Selenium supplementation induces metalloproteinase-dependent l-selectin shedding from monocytes. J. Leukoc. Biol. 2008, 83, 1388–1395. [Google Scholar] [CrossRef] [PubMed]
- Heyland, D.K.; Dhaliwal, R.; Suchner, U.; Berger, M.M. Antioxidant nutrients: A systematic review of trace elements and vitamins in the critically ill patient. Intensive Care Med. 2004, 31, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Forman, H.J.; Torres, M. Reactive oxygen species and cell signaling: Respiratory burst in macrophage signaling. Am. J. Respir. Crit. Care Med. 2002, 166, S4–S8. [Google Scholar] [CrossRef] [PubMed]
- Arbogast, S.; Ferreiro, A. Selenoproteins and protection against oxidative stress: Selenoprotein N as a novel player at the crossroads of redox signaling and calcium homeostasis. Antioxid. Redox Signal. 2010, 12, 893–904. [Google Scholar] [CrossRef] [PubMed]
- Flores-Mateo, G.; Navas-Acien, A.; Pastor-Barriuso, R.; Guallar, E. Selenium and coronary heart disease: A meta-analysis. Am. J. Clin. Nutr. 2006, 84, 762–773. [Google Scholar] [PubMed]
- Rees, K.; Hartley, L.; Day, C.; Flowers, N.; Clarke, A.; Stranges, S. Selenium supplementation for the primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2013, 1. [Google Scholar] [CrossRef]
- Loscalzo, J. Keshan disease, selenium deficiency, and the selenoproteome. N. Engl. J. Med. 2014, 370, 1756–1760. [Google Scholar] [CrossRef] [PubMed]
- Schrauzer, G.N. Selenomethionine: A review of its nutritional significance, metabolism and toxicity. J. Nutr. 2000, 130, 1653–1656. [Google Scholar] [PubMed]
- Hardy, G.; Hardy, I.; Manzanares, W. Selenium supplementation in the critically ill. Nutr. Clin. Pract. 2012, 27, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Reeves, M.A.; Hoffmann, P.R. The human selenoproteome: Recent insights into functions and regulation. Cell. Mol. Life Sci. 2009, 66, 2457–2478. [Google Scholar] [CrossRef] [PubMed]
- Grumolato, L.; Ghzili, H.; Montero-Hadjadje, M.; Gasman, S.; Lesage, J.; Tanguy, Y.; Galas, L.; Ait-Ali, D.; Leprince, J.; Guerineau, N.C.; et al. Selenoprotein T is a PACAP-regulated gene involved in intracellular Ca2+ mobilization and neuroendocrine secretion. FASEB J. 2008, 22, 1756–1768. [Google Scholar] [CrossRef] [PubMed]
- Prevost, G.; Arabo, A.; Jian, L.; Quelennec, E.; Cartier, D.; Hassan, S.; Falluel-Morel, A.; Tanguy, Y.; Gargani, S.; Lihrmann, I.; et al. The PACAP-regulated gene selenoprotein T is abundantly expressed in mouse and human beta-cells and its targeted inactivation impairs glucose tolerance. Endocrinology 2013, 154, 3796–3806. [Google Scholar] [CrossRef] [PubMed]
- Papp, L.V.; Lu, J.; Holmgren, A.; Khanna, K.K. From selenium to selenoproteins: Synthesis, identity, and their role in human health. Antioxid. Redox Signal. 2007, 9, 775–806. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Rose, A.H.; Hoffmann, P.R. The role of selenium in inflammation and immunity: From molecular mechanisms to therapeutic opportunities. Antioxid. Redox Signal. 2012, 16, 705–743. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohé, R.; Maiorino, M. Glutathione peroxidases. Biochim. Biophys. Acta 2013, 1830, 3289–3303. [Google Scholar] [CrossRef] [PubMed]
- Flohe, L.; Gunzler, W.A.; Schock, H.H. Glutathione peroxidase: A selenoenzyme. FEBS Lett. 1973, 32, 132–134. [Google Scholar] [CrossRef] [PubMed]
- Rotruck, J.T. Selenium: Biochemical role as a component of glutathione peroxidase. Science 1973, 179, 588–590. [Google Scholar]
- Yoshida, T.; Watanabe, M.; Engelman, D.T.; Engelman, R.M.; Schley, J.A.; Maulik, N.; Ho, Y.S.; Oberley, T.D.; Das, D.K. Transgenic mice overexpressing glutathione peroxidase are resistant to myocardial ischemia reperfusion injury. J. Mol. Cell. Cardiol. 1996, 28, 1759–1767. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Maulik, N.; Engelman, R.M.; Ho, Y.S.; Magnenat, J.L.; Rousou, J.A.; Flack, J.E.; Deaton, D.; Das, D.K. Glutathione peroxidase knockout mice are susceptible to myocardial ischemia reperfusion injury. Circulation 1997, 96, 216–220. [Google Scholar]
- Lim, C.C.; Bryan, N.S.; Jain, M.; Garcia-Saura, M.F.; Fernandez, B.O.; Sawyer, D.B.; Handy, D.E.; Loscalzo, J.; Feelisch, M.; Liao, R. Glutathione peroxidase deficiency exacerbates ischemia-reperfusion injury in male but not female myocardium: Insights into antioxidant compensatory mechanisms. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H2144–H2153. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.C.; Mahoney, C.E.; Anderson, L.C.; Ottaviano, F.; Croce, K.; Leopold, J.A.; Zhang, Y.Y.; Tang, S.S.; Handy, D.E.; Loscalzo, J. Glutathione peroxidase-3 deficiency promotes platelet-dependent thrombosis in vivo. Circulation 2011, 123, 1963–1973. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohé, R.; Banning, A.; Schnurr, K. Selenium-dependent enzymes in endothelial cell function. Antioxid. Redox Signal. 2003, 5, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, F.W.; Hashimoto, A.S.; Lee, B.C.; Rose, A.H.; Shohet, R.V.; Hoffmann, P.R. Specific antioxidant selenoproteins are induced in the heart during hypertrophy. Arch. Biochem. Biophys. 2011, 512, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Wortmann, M.; Schneider, M.; Pircher, J.; Hellfritsch, J.; Aichler, M.; Vegi, N.; Kölle, P.; Kuhlencordt, P.; Walch, A.; Pohl, U.; et al. Combined deficiency in glutathione peroxidase 4 and vitamin E causes multiorgan thrombus formation and early death in mice. Circ. Res. 2013, 113, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Hollander, J.M.; Lin, K.M.; Scott, B.T.; Dillmann, W.H. Overexpression of PHGPx and HSP60/10 protects against ischemia/reoxygenation injury. Free Radic. Biol. Med. 2003, 35, 742–751. [Google Scholar] [CrossRef] [PubMed]
- Rose, A.H.; Hoffmann, P.R. Selenoproteins and cardiovascular stress. Thromb. Haemost. 2015, 113, 494–504. [Google Scholar] [CrossRef] [PubMed]
- Arner, E.S.; Holmgren, A. Physiological functions of thioredoxin and thioredoxin reductase. Eur. J. Biochem. FEBS 2000, 267, 6102–6109. [Google Scholar] [CrossRef]
- Maulik, N.; Das, D.K. Emerging potential of thioredoxin and thioredoxin interacting proteins in various disease conditions. Biochim. Biophys. Acta 2008, 1780, 1368–1382. [Google Scholar] [CrossRef] [PubMed]
- Biterova, E.I.; Turanov, A.A.; Gladyshev, V.N.; Barycki, J.J. Crystal structures of oxidized and reduced mitochondrial thioredoxin reductase provide molecular details of the reaction mechanism. Proc. Natl. Acad. Sci. USA 2005, 102, 15018–15023. [Google Scholar] [CrossRef]
- Holmgren, A.; Lu, J. Thioredoxin and thioredoxin reductase: Current research with special reference to human disease. Biochem. Biophys. Res. Commun. 2010, 396, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Tamura, T.; Stadtman, T.C. A new selenoprotein from human lung adenocarcinoma cells: Purification, properties, and thioredoxin reductase activity. Proc. Natl. Acad. Sci. USA 1996, 93, 1006–1011. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.R.; Kim, J.R.; Kwon, K.S.; Yoon, H.W.; Levine, R.L.; Ginsburg, A.; Rhee, S.G. Molecular cloning and characterization of a mitochondrial selenocysteine-containing thioredoxin reductase from rat liver. J. Biol. Chem. 1999, 274, 4722–4734. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.A.; Kirnarsky, L.; Sherman, S.; Gladyshev, V.N. Selenoprotein oxidoreductase with specificity for thioredoxin and glutathione systems. Proc. Natl. Acad. Sci. USA 2001, 98, 3673–3678. [Google Scholar] [CrossRef] [PubMed]
- Lundstrom, J.; Holmgren, A. Protein disulfide-isomerase is a substrate for thioredoxin reductase and has thioredoxin-like activity. J. Biol. Chem. 1990, 265, 9114–9120. [Google Scholar] [PubMed]
- Lundstrom-Ljung, J.; Birnbach, U.; Rupp, K.; Soling, H.D.; Holmgren, A. Two resident ER-proteins, CaBP1 and CaBP2, with thioredoxin domains, are substrates for thioredoxin reductase: Comparison with protein disulfide isomerase. FEBS Lett. 1995, 357, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Bjornstedt, M.; Hamberg, M.; Kumar, S.; Xue, J.; Holmgren, A. Human thioredoxin reductase directly reduces lipid hydroperoxides by NADPH and selenocystine strongly stimulates the reaction via catalytically generated selenols. J. Biol. Chem. 1995, 270, 11761–11764. [Google Scholar] [CrossRef] [PubMed]
- Bjornstedt, M.; Kumar, S.; Holmgren, A. Selenodiglutathione is a highly efficient oxidant of reduced thioredoxin and a substrate for mammalian thioredoxin reductase. J. Biol. Chem. 1992, 267, 8030–8034. [Google Scholar] [PubMed]
- Gromer, S.; Gross, J.H. Methylseleninate is a substrate rather than an inhibitor of mammalian thioredoxin reductase. Implications for the antitumor effects of selenium. J. Biol. Chem. 2002, 277, 9701–9706. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Bjornstedt, M.; Holmgren, A. Selenite is a substrate for calf thymus thioredoxin reductase and thioredoxin and elicits a large non-stoichiometric oxidation of NADPH in the presence of oxygen. Eur. J. Biochem. FEBS 1992, 207, 435–439. [Google Scholar] [CrossRef]
- Ganther, H.E. Selenium metabolism, selenoproteins and mechanisms of cancer prevention: Complexities with thioredoxin reductase. Carcinogenesis 1999, 20, 1657–1666. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Honda, M.; Takabatake, T. Redox regulation of MAPK pathways and cardiac hypertrophy in adult rat cardiac myocyte. J. Am. Coll. Cardiol. 2001, 37, 676–685. [Google Scholar] [CrossRef] [PubMed]
- Berndt, C.; Lillig, C.H.; Holmgren, A. Thiol-based mechanisms of the thioredoxin and glutaredoxin systems: Implications for diseases in the cardiovascular system. Am. J. Physiol. Heart Circ. Physiol. 2007, 292, H1227–H1236. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Yang, G.; Hong, C.; Liu, J.; Holle, E.; Yu, X.; Wagner, T.; Vatner, S.F.; Sadoshima, J. Inhibition of endogenous thioredoxin in the heart increases oxidative stress and cardiac hypertrophy. J. Clin. Investig. 2003, 112, 1395–1406. [Google Scholar] [CrossRef] [PubMed]
- Ago, T.; Sadoshima, J. Thioredoxin and ventricular remodeling. J. Mol. Cell. Cardiol. 2006, 41, 762–773. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Proud, C.G. Ras/Erk signaling is essential for activation of protein synthesis by Gq protein-coupled receptor agonists in adult cardiomyocytes. Circ. Res. 2002, 91, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Schomburg, L.; Köhrle, J. On the importance of selenium and iodine metabolism for thyroid hormone biosynthesis and human health. Mol. Nutr. Food Res. 2008, 52, 1235–1246. [Google Scholar] [CrossRef] [PubMed]
- Mittag, J.; Behrends, T.; Hoefig, C.S.; Vennström, B.; Schomburg, L. Thyroid hormones regulate selenoprotein expression and selenium status in mice. PLoS ONE 2010, 5, e12931. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.C.; Salvatore, D.; Gereben, B.; Berry, M.J.; Larsen, P.R. Biochemistry, cellular and molecular biology, and physiological roles of the iodothyronine selenodeiodinases. Endocr. Rev. 2002, 23, 38–89. [Google Scholar] [CrossRef] [PubMed]
- Klein, I.; Ojamaa, K. Thyroid hormone and the cardiovascular system. N. Engl. J. Med. 2001, 344, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Trivieri, M.G.; Oudit, G.Y.; Sah, R.; Kerfant, B.G.; Sun, H.; Gramolini, A.O.; Pan, Y.; Wickenden, A.D.; Croteau, W.; de Escobar, G.M.; et al. Cardiac-specific elevations in thyroid hormone enhance contractility and prevent pressure overload-induced cardiac dysfunction. Proc. Natl. Acad. Sci. USA 2006, 103, 6043–6048. [Google Scholar] [CrossRef] [PubMed]
- Stadtman, E.R. Protein oxidation and aging. Free Radic. Res. 2006, 40, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Moskovitz, J.; Bar-Noy, S.; Williams, W.M.; Requena, J.; Berlett, B.S.; Stadtman, E.R. Methionine sulfoxide reductase (MsrA) is a regulator of antioxidant defense and lifespan in mammals. Proc. Natl. Acad. Sci. USA 2001, 98, 12920–12925. [Google Scholar] [CrossRef] [PubMed]
- Moskovitz, J.; Flescher, E.; Berlett, B.S.; Azare, J.; Poston, J.M.; Stadtman, E.R. Overexpression of peptide-methionine sulfoxide reductase in Saccharomyces cerevisiae and human T cells provides them with high resistance to oxidative stress. Proc. Natl. Acad. Sci. USA 1998, 95, 14071–14075. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Gladyshev, V.N. Methionine sulfoxide reduction in mammals: Characterization of methionine-R-sulfoxide reductases. Mol. Biol. Cell 2004, 15, 1055–1064. [Google Scholar] [CrossRef] [PubMed]
- Picot, C.R.; Perichon, M.; Lundberg, K.C.; Friguet, B.; Szweda, L.I.; Petropoulos, I. Alterations in mitochondrial and cytosolic methionine sulfoxide reductase activity during cardiac ischemia and reperfusion. Exp. Gerontol. 2006, 41, 663–667. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.C.; Peterfi, Z.; Hoffmann, F.W.; Moore, R.E.; Kaya, A.; Avanesov, A.; Tarrago, L.; Zhou, Y.; Weerapana, E.; Fomenko, D.E.; et al. MsrB1 and MICALs regulate actin assembly and macrophage function via reversible stereoselective methionine oxidation. Mol. Cell 2013, 51, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Qiu, F.; Zhou, H.; Peng, Y.; Hao, W.; Xu, J.; Yuan, J.; Wang, S.; Qiang, B.; Xu, C.; et al. Identification and characterization of selenoprotein K: An antioxidant in cardiomyocytes. FEBS Lett. 2006, 580, 5189–5197. [Google Scholar] [CrossRef] [PubMed]
- Meiler, S.; Baumer, Y.; Huang, Z.; Hoffmann, F.W.; Fredericks, G.J.; Rose, A.H.; Norton, R.L.; Hoffmann, P.R.; Boisvert, W.A. Selenoprotein K is required for palmitoylation of CD36 in macrophages: Implications in foam cell formation and atherogenesis. J. Leukoc. Biol. 2013, 93, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.J.; Lehtinen, A.B.; Xu, J.; Langefeld, C.D.; Freedman, B.I.; Carr, J.J.; Bowden, D.W. Polymorphisms in the Selenoprotein S gene and subclinical cardiovascular disease in the Diabetes Heart Study. Acta Diabetol. 2013, 50, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Feng, H.C.; Walder, K.; Bolton, K.; Sunderland, T.; Bishara, N.; Quick, M.; Kantham, L.; Collier, G.R. Regulation of the selenoprotein SelS by glucose deprivation and endoplasmic reticulum stress—SelS is a novel glucose-regulated protein. FEBS Lett. 2004, 563, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.L.; Wang, S.C.; Gu, B.Q.; Yang, Y.X.; Song, H.B.; Xue, W.L.; Liang, W.S.; Zhang, P.Y. Further investigation on the role of selenium deficiency in the aetiology and pathogenesis of Keshan disease. Biomed. Environ. Sci. 1997, 10, 316–326. [Google Scholar] [PubMed]
- Beck, M.A.; Levander, O.A.; Handy, J. Selenium deficiency and viral infection. J. Nutr. 2003, 133, 1463S–1467S. [Google Scholar] [PubMed]
- Beck, M.A.; Handy, J.; Levander, O.A. Host nutritional status: The neglected virulence factor. Trends Microbiol. 2004, 12, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Jun, E.J.; Ye, J.S.; Hwang, I.S.; Kim, Y.K.; Lee, H. Selenium deficiency contributes to the chronic myocarditis in coxsackievirus-infected mice. Acta Virol. 2011, 55, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Beck, M.A.; Esworthy, R.S.; Ho, Y.S.; Chu, F.F. Glutathione peroxidase protects mice from viral-induced myocarditis. FASEB J. 1998, 12, 1143–1149. [Google Scholar] [PubMed]
- Venardos, K. Effects of dietary selenium on glutathione peroxidase and thioredoxin reductase activity and recovery from cardiac ischemia-reperfusion. J. Trace Elem. Med. Biol. 2004, 18, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Toufektsian, M.C.; Boucher, F.; Pucheu, S.; Tanguy, S.; Ribuot, C.; Sanou, D.; Tresallet, N.; de Leiris, J. Effects of selenium deficiency on the response of cardiac tissue to ischemia and reperfusion. Toxicology 2000, 148, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Tanguy, S.; Toufektsian, M.C.; Besse, S.; Ducros, V.; de Leiris, J.; Boucher, F. Dietary selenium intake affects cardiac susceptibility to ischaemia/reperfusion in male senescent rats. Age Ageing 2003, 32, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Pucheu, S.; Coudray, C.; Tresallet, N.; Favier, A.; de Leiris, J. Effect of dietary antioxidant trace element supply on cardiac tolerance to ischemia-reperfusion in the rat. J. Mol. Cell. Cardiol. 1995, 27, 2303–2314. [Google Scholar] [CrossRef] [PubMed]
- Venardos, K.M.; Perkins, A.; Headrick, J.; Kaye, D.M. Myocardial ischemia-reperfusion injury, antioxidant enzyme systems, and selenium: A review. Curr. Med. Chem. 2007, 14, 1539–1549. [Google Scholar] [CrossRef] [PubMed]
- Tanguy, S.; Boucher, F.; Besse, S.; Ducros, V.; Favier, A.; de Leiris, J. Trace elements and cardioprotection: Increasing endogenous glutathione peroxidase activity by oral selenium supplementation in rats limits reperfusion-induced arrhythmias. J. Trace Elem. Med. Biol. 1998, 12, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Tanguy, S.; Morel, S.; Berthonneche, C.; Toufektsian, M.C.; de Lorgeril, M.; Ducros, V.; Tosaki, A.; de Leiris, J.; Boucher, F. Preischemic selenium status as a major determinant of myocardial infarct size in vivo in rats. Antioxid. Redox Signal. 2004, 6, 792–796. [Google Scholar] [CrossRef] [PubMed]
- Tanguy, S.; Rakotovao, A.; Jouan, M.G.; Ghezzi, C.; de Leiris, J.; Boucher, F. Dietary selenium intake influences Cx43 dephosphorylation, TNF-α expression and cardiac remodeling after reperfused infarction. Mol. Nutr. Food Res. 2011, 55, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Rakotovao, A.; Tanguy, S.; Toufektsian, M.C.; Berthonneche, C.; Ducros, V.; Tosaki, A.; de Leiris, J.; Boucher, F. Selenium status as determinant of connexin-43 dephosphorylation in ex vivo ischemic/reperfused rat myocardium. J. Trace Elem. Med. Biol. 2005, 19, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Rotruck, J.T. Prevention of oxidative damage to rat erythrocytes by dietary selenium. J. Nutr. 1972, 102, 689–696. [Google Scholar] [PubMed]
- Aviram, M.; Fuhrman, B. LDL oxidation by arterial wall macrophages depends on the oxidative status in the lipoprotein and in the cells: Role of prooxidants vs. antioxidants. Mol. Cell. Biochem. 1998, 188, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Navas-Acien, A.; Bleys, J.; Guallar, E. Selenium intake and cardiovascular risk: What is new? Curr. Opin. Lipidol. 2008, 19, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Xun, P.; Liu, K.; Morris, J.S.; Daviglus, M.L.; He, K. Longitudinal association between toenail selenium levels and measures of subclinical atherosclerosis: The CARDIA trace element study. Atherosclerosis 2010, 210, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Loscalzo, J. Effects of nationwide addition of selenium to fertilizers on foods, and animal and human health in Finland: From deficiency to optimal selenium status of the population. J. Trace Elem. Med. Biol. 2014, 370, 1756–1760. [Google Scholar]
- Bleys, J.; Navas-Acien, A.; Guallar, E. Serum selenium levels and all-cause, cancer, and cardiovascular mortality among US adults. Arch. Intern. Med. 2008, 168, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Joseph, J. Selenium and cardiometabolic health: Inconclusive yet intriguing evidence. Am. J. Med. Sci. 2013, 346, 216–220. [Google Scholar] [CrossRef] [PubMed]
- González, J.; Valls, N.; Brito, R.; Rodrigo, R. Essential hypertension and oxidative stress: New insights. World J. Cardiol. 2014, 6, 353–366. [Google Scholar] [CrossRef] [PubMed]
- Nawrot, T.S.; Staessen, J.A.; Roels, H.A.; Den Hond, E.; Thijs, L.; Fagard, R.H.; Dominiczak, A.F.; Struijker-Boudier, H.A. Blood pressure and blood selenium: A cross-sectional and longitudinal population study. Eur. Heart J. 2007, 28, 628–633. [Google Scholar] [CrossRef] [PubMed]
- Berthold, H.K.; Michalke, B.; Krone, W.; Guallar, E.; Gouni-Berthold, I. Influence of serum selenium concentrations on hypertension: The Lipid Analytic Cologne cross-sectional study. J. Hypertens. 2012, 30, 1328–1335. [Google Scholar] [CrossRef] [PubMed]
- Forceville, X.; Mostert, V.; Pierantoni, A.; Vitoux, D.; le Toumelin, P.; Plouvier, E.; Dehoux, M.; Thuillier, F.; Combes, A.; Selenoprotein, P. Rather than Glutathione Peroxidase, as a Potential Marker of Septic Shock and Related Syndromes. Eur. Surg. Res. 2009, 43, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.J.; Hwang, S.Y.; Choi, H.Y.; Yoo, H.J.; Seo, J.A.; Kim, S.G.; Kim, N.H.; Baik, S.H.; Choi, D.S.; Choi, K.M. Serum selenoprotein P levels in patients with type 2 diabetes and prediabetes: Implications for insulin resistance, inflammation, and atherosclerosis. J. Clin. Endocrinol. Metab. 2011, 96, E1325–E1329. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.R.; Zweier, J.L. Cardiac mitochondria and reactive oxygen species generation. Circ. Res. 2014, 114, 524–537. [Google Scholar] [CrossRef] [PubMed]
- Grieve, D.J.; Byrne, J.A.; Cave, A.C.; Shah, A.M. Role of oxidative stress in cardiac remodelling after myocardial infarction. Heart Lung Circ. 2004, 13, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Tsutsui, H.; Kinugawa, S.; Matsushima, S. Mitochondrial oxidative stress and dysfunction in myocardial remodelling. Cardiovasc. Res. 2009, 81, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Kutil, B.; Ostadal, P.; Vejvoda, J.; Kukacka, J.; Cepova, J.; Alan, D.; Krüger, A.; Vondrakova, D. Alterations in serum selenium levels and their relation to troponin I in acute myocardial infarction. Mol. Cell. Biochem. 2010, 345, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Lubos, E.; Sinning, C.R.; Schnabel, R.B.; Wild, P.S.; Zeller, T.; Rupprecht, H.J.; Bickel, C.; Lackner, K.J.; Peetz, D.; Loscalzo, J.; et al. Serum selenium and prognosis in cardiovascular disease: Results from the AtheroGene study. Atherosclerosis 2010, 209, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Eaton, C.B.; Abdul Baki, A.R.; Waring, M.E.; Roberts, M.B.; Lu, B. The association of low selenium and renal insufficiency with coronary heart disease and all-cause mortality: NHANES III follow-up study. Atherosclerosis 2010, 212, 689–694. [Google Scholar] [CrossRef] [PubMed]
- Hercberg, S.; Galan, P.; Preziosi, P.; Bertrais, S.; Mennen, L.; Malvy, D.; Roussel, A.M.; Favier, A.; Briancon, S. The SU.VI.MAX Study: A randomized, placebo-controlled trial of the health effects of antioxidant vitamins and minerals. Arch. Intern. Med. 2004, 164, 2335–2342. [Google Scholar] [CrossRef] [PubMed]
- Hercberg, S.; Bertrais, S.; Czernichow, S.; Noisette, N.; Galan, P.; Jaouen, A.; Tichet, J.; Briancon, S.; Favier, A.; Mennen, L.; et al. Alterations of the lipid profile after 7.5 years of low-dose antioxidant supplementation in the SU.VI.MAX Study. Lipids 2005, 40, 335–342. [Google Scholar] [CrossRef] [PubMed]
- McKeag, N.A.; McKinley, M.C.; Woodside, J.V.; Harbinson, M.T.; McKeown, P.P. The role of micronutrients in heart failure. J. Acad. Nutr. Diet. 2012, 112, 870–886. [Google Scholar] [CrossRef] [PubMed]
- Do Brasil, P.E.A.A.; de Souza, A.P.; Hasslocher-Moreno, A.M.; Xavier, S.S.; Passos, S.R.L.; Moreira, M.D.F.R.; de Oliveira, M.S.; da Silva, G.M.S.; Saraiva, R.M.; de Aguiar, C.C.S.; et al. Selenium Treatment and Chagasic Cardiopathy (STCC): Study protocol for a double-blind randomized controlled trial. Trials 2014, 15, 388. [Google Scholar] [CrossRef] [PubMed]
- Grover, A.; Gorman, K.; Dall, T.M.; Jonas, R.; Lytle, B.; Shemin, R.; Wood, D.; Kron, I. Shortage of cardiothoracic surgeons is likely by 2020. Circulation 2009, 120, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, P.; Schistek, R.; Unger, F. Coronary revascularization in DACH: 1991–2002. Thorac. Cardiovasc. Surg. 2004, 52, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Stevanovic, A.; Coburn, M.; Menon, A.; Rossaint, R.; Heyland, D.; Schalte, G.; Werker, T.; Wonisch, W.; Kiehntopf, M.; Goetzenich, A.; et al. The importance of intraoperative selenium blood levels on organ dysfunction in patients undergoing off-pump cardiac surgery: A randomised controlled trial. PLoS ONE 2014, 9, e104222. [Google Scholar] [CrossRef] [PubMed]
- Hall, R. Identification of inflammatory mediators and their modulation by strategies for the management of the systemic inflammatory response during cardiac surgery. J. Cardiothorac. Vasc. Anesth. 2013, 27, 983–1033. [Google Scholar] [CrossRef] [PubMed]
- Manzanares, W.; Langlois, P.L.; Heyland, D.K. Pharmaconutrition With Selenium in Critically Ill Patients: What Do We Know? Nutr. Clin. Pract. 2015, 30, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Stoppe, C.; Schalte, G.; Rossaint, R.; Coburn, M.; Graf, B.; Spillner, J.; Marx, G.; Rex, S. The intraoperative decrease of selenium is associated with the postoperative development of multiorgan dysfunction in cardiac surgical patients. Crit. Care Med. 2011, 39, 1879–1885. [Google Scholar] [CrossRef] [PubMed]
- Koszta, G.; Kacska, Z.; Szatmari, K.; Szerafin, T.; Fulesdi, B. Lower whole blood selenium level is associated with higher operative risk and mortality following cardiac surgery. J. Anesth. 2012, 26, 812–821. [Google Scholar] [CrossRef] [PubMed]
- Leong, J.Y.; van der Merwe, J.; Pepe, S.; Bailey, M.; Perkins, A.; Lymbury, R.; Esmore, D.; Marasco, S.; Rosenfeldt, F. Perioperative metabolic therapy improves redox status and outcomes in cardiac surgery patients: A randomised trial. Heart Lung Circ. 2010, 19, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Stoppe, C.; Spillner, J.; Rossaint, R.; Coburn, M.; Schalte, G.; Wildenhues, A.; Marx, G.; Rex, S. Selenium blood concentrations in patients undergoing elective cardiac surgery and receiving perioperative sodium selenite. Nutrition 2013, 29, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Stoppe, C.; McDonald, B.; Rex, S.; Manzanares, W.; Whitlock, R.; Fremes, S.; Fowler, R.; Lamarche, Y.; Meybohm, P.; Haberthur, C.; et al. SodiUm SeleniTe Adminstration IN Cardiac Surgery (SUSTAIN CSX-trial): Study design of an international multicenter randomized double-blinded controlled trial of high dose sodium-selenite administration in high-risk cardiac surgical patients. Trials 2014, 15, 339. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.F.; Godfrey, P.J.; Thomson, C.D.; Rea, H.M.; van Rij, A.M. Blood selenium and glutathione peroxidase activity in normal subjects and in surgical patients with and without cancer in New Zealand. Am. J. Clin. Nutr. 1979, 32, 1477–1485. [Google Scholar] [PubMed]
- Laffey, J.G.; Boylan, J.F.; Cheng, D.C.H. The systemic inflammatory response to cardiac surgery: Implications for the anesthesiologist. Anesthesiology 2002, 97, 215–252. [Google Scholar] [CrossRef] [PubMed]
- Méplan, C.; Nicol, F.; Burtle, B.T.; Crosley, L.K.; Arthur, J.R.; Mathers, J.C.; Hesketh, J.E. Relative abundance of selenoprotein P isoforms in human plasma depends on genotype, se intake, and cancer status. Antioxid. Redox Signal. 2009, 11, 2631–2640. [Google Scholar] [CrossRef] [PubMed]
- Méplan, C.; Hughes, D.J.; Pardini, B.; Naccarati, A.; Soucek, P.; Vodickova, L.; Hlavatá, I.; Vrána, D.; Vodicka, P.; Hesketh, J.E. Genetic variants in selenoprotein genes increase risk of colorectal cancer. Carcinogenesis 2010, 31, 1074–1079. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.E.; Xia, Y.; Akesson, B.; Boeglin, M.E.; Burk, R.F. Selenoprotein P concentration in plasma is an index of selenium status in selenium-deficient and selenium-supplemented Chinese subjects. J. Nutr. 1996, 126, 138–145. [Google Scholar] [PubMed]
- Wang, W.Z.; Guo, X.; Duan, C.; Ma, W.J.; Zhang, Y.G.; Xu, P.; Gao, Z.Q.; Wang, Z.F.; Yan, H.; Zhang, Y.F.; et al. Comparative analysis of gene expression profiles between the normal human cartilage and the one with endemic osteoarthritis. Osteoarthr. Cartil. 2009, 17, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Speckmann, B.; Grune, T. Epigenetic effects of selenium and their implications for health. Epigenetics 2015, 10, 179–190. [Google Scholar] [PubMed]
- Treska, V.; Kuntscher, V.; Hasman, D.; Neprasová, P.; Kobr, J.; Racek, J.; Trefil, L.; Hes, O. Importance of selenium for the influence of ischemia-reperfusion syndrome after kidney transplantation from a non-heart beating donor in a pig model. Transplant. Proc. 2002, 34, 3057–3059. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benstoem, C.; Goetzenich, A.; Kraemer, S.; Borosch, S.; Manzanares, W.; Hardy, G.; Stoppe, C. Selenium and Its Supplementation in Cardiovascular Disease—What do We Know? Nutrients 2015, 7, 3094-3118. https://doi.org/10.3390/nu7053094
Benstoem C, Goetzenich A, Kraemer S, Borosch S, Manzanares W, Hardy G, Stoppe C. Selenium and Its Supplementation in Cardiovascular Disease—What do We Know? Nutrients. 2015; 7(5):3094-3118. https://doi.org/10.3390/nu7053094
Chicago/Turabian StyleBenstoem, Carina, Andreas Goetzenich, Sandra Kraemer, Sebastian Borosch, William Manzanares, Gil Hardy, and Christian Stoppe. 2015. "Selenium and Its Supplementation in Cardiovascular Disease—What do We Know?" Nutrients 7, no. 5: 3094-3118. https://doi.org/10.3390/nu7053094
APA StyleBenstoem, C., Goetzenich, A., Kraemer, S., Borosch, S., Manzanares, W., Hardy, G., & Stoppe, C. (2015). Selenium and Its Supplementation in Cardiovascular Disease—What do We Know? Nutrients, 7(5), 3094-3118. https://doi.org/10.3390/nu7053094





