Low Maternal Vitamin B12 Status Is Associated with Lower Cord Blood HDL Cholesterol in White Caucasians Living in the UK
Abstract
:1. Introduction
2. Methods
2.1. Study Population
2.2. Analytical Determinations
2.3. Statistical Analysis
3. Results
3.1. B12, Folate and Homocysteine Status
| Mother | Neonate | |
|---|---|---|
| n = 91 | n = 91 | |
| Age (years) | 32.7 ± 5.9 a | - |
| Weight (Kg) | 77.7 ± 18.1 | 3.57 ± 0.26 |
| Height (m) | 1.62 ± 0.09 | - |
| BMI (early pregnancy) (kg/m2) | 29.4 ± 6.2 | - |
| Glucose (mmol/L) | 4.37 ± 0.42 | 3.88 ± 0.52 |
| Insulin (mIU/L) | 11.6 (12.9, 17.4) b | 8.01 (8.62, 11.9) |
| Triglycerides (mmol/L) | 2.69 (2.62, 3.06) | 0.23 (0.22, 0.26) |
| Cholesterol (mmol/L) | 6.48 (6.31, 6.89) | 1.68 (1.63, 1.84) |
| LDL cholesterol (mmol/L) | 3.53 (3.46, 3.95) | 0.82 (0.79, 0.95) |
| HDL cholesterol (mmol/L) | 1.56 (1.53, 1.72) | 0.74 (0.72, 0.83) |
| HOMA-IR | 1.37 (1.55, 2.09) | 0.99 (1.04, 1.41) |
| Vitamin B12 (ng/L) | 218 (213, 289) | 290 (292, 418) |
| Low B12 status (%) | 36 (40) c | 26 (29) |
| Folate (μg/L) | 10.5 (10.9, 13.2) | 16.8 (16.4, 17.7) |
| Low folate status (%) | 10 (11) | 0 |
| Homocysteine (μmol/L) | 6.23 (6.02, 7.54) | 5.76 (5.64, 6.85) |
| Mothers | Neonate | |||
|---|---|---|---|---|
| Maternal B12 ≥191 (ng/L) | Maternal B12 <191 (ng/L) | Maternal B12 ≥191 (ng/L) | Maternal B12 <191 (ng/L) | |
| n = 55 | n = 36 | n = 55 | n = 36 | |
| Age (years) | 33.0 ± 6.2 a | 32.3 ± 5.6 | - | - |
| Weight (Kg) | 74.3 ± 15.8 | 82.9 ± 20.8 * | 3.58 ± 0.31 | 3.57 ± 0.18 |
| Height (m) | 1.62 ± 0.07 | 1.61 ± 0.11 | - | - |
| BMI (early pregnancy) (kg/m2) | 28.4 ± 6.1 | 30.8 ± 6.4 * | - | - |
| Glucose (mmol/L) | 4.40 ± 0.46 | 4.36 ± 0.34 | 3.85 ± 0.52 | 3.94 ± 0.52 |
| Insulin (mIU/L) | 10.4 (11.7, 18.0) b | 13.7 (12.7, 18.6) | 8.27 (8.33, 12.75) | 7.64 (7.30, 12.40) |
| Triglycerides (mmol/L) | 2.49 (2.37, 2.93) | 3.04 (2.82, 3.48) * | 0.21 (0.20, 0.24) | 0.26 (0.23, 0.32) ** |
| Cholesterol (mmol/L) | 6.23 (5.99, 6.72) | 6.86 (6.51, 7.43) * | 1.72 (1.64, 1.92) | 1.62 (1.51, 1.83) |
| LDL cholesterol (mmol/L) | 3.29 (3.16, 3.82) | 3.91 (3.67, 4.38) * | 0.79 (0.76, 0.90) | 0.87 (0.77, 1.09) |
| HDL cholesterol (mmol/L) | 1.61 (1.54, 1.80) | 1.49 (1.42, 1.70) | 0.79 (0.76, 0.91) | 0.67 (0.62, 0.77) * |
| HOMA-IR | 1.18 (1.37, 2.14) | 1.68 (1.57, 2.27) | 0.94 (0.95, 1.41) | 1.08 (0.98, 1.63) |
| Vitamin B12 (ng/L) | 288 (265, 378) | 146 (139, 155) *** | 367 (354, 544) | 202 (187, 234) *** |
| Low B12 status (%) | 0 | 36 (40) c | 8 (14.5) | 18 (50) |
| Folate (μg/L) | 11.7 (11.5, 14.6) | 9.0 (8.5, 12.4) * | 17.6 (17.0, 18.5) | 15.7 (14.9, 17.2) ** |
| Low folate status (%) | 3 (5.5) | 7 (19.4) | 0 | 0 |
| Homocysteine (μmol/L) | 5.50 (5.26, 6.18) | 7.53 (6.69, 10.1) *** | 4.97 (4.74, 5.59) | 7.09 (6.58, 8.96) *** |
| Smoking | n = 91 | Vitamin B12 (ng/L) | Folate (μg/L) | Homocysteine (μmol/L) | |
|---|---|---|---|---|---|
| Maternal | No (%) | 55 | 245 (227, 357) | 10.7 (10.6, 13.9) | 6.15 (5.61, 8.08) |
| Yes (%) | 45 | 189 (176, 224) ** | 10.1 (9.7, 13.3) | 6.33 (5.90, 7.45) | |
| Neonate | No (%) | 55 | 327 (305, 502) | 16.9 (16.4, 18.2) | 5.42 (5.06, 6.58) |
| Yes (%) | 45 | 252 (232, 364) * | 16.5 (15.8, 17.7) | 6.21 (5.80, 7.68) | |
| Parity | |||||
| Maternal | Para 0 (%) | 18 | 248 (203, 332) | 13.7 (11.8, 17.9) | 6.25 (5.21, 8.03) |
| Para 1 (%) | 48 | 224 (201, 347) | 11.1 (10.8, 14.1) | 6.12 (5.73, 7.43) | |
| Para ≥2 (%) | 34 | 195 (179, 239) * | 8.2 (7.6, 11.6) ** | 6.4 (5.31, 8.97) | |
| Neonate | Para 0 (%) | 18 | 327 (264, 479) | 17.9 (16.6, 19.6) | 5.61 (4.50, 7.59) |
| Para 1 (%) | 48 | 284 (258, 487) | 16.9 (16.3, 18.1) | 5.91 (5.53, 7.30) | |
| Para ≥2 (%) | 34 | 283 (250, 396) | 16.0 (15.2, 17.4) * | 5.67 (5.08, 7.15) | |
| Folate supplement users | |||||
| Maternal | Yes (%) | 85 | 224 (216, 305) | 11.1 (11.3, 14.0) | 6.06 (5.77, 7.44) |
| No (%) | 15 | 187 (154, 245) | 6.8 (5.5, 9.3) *** | 7.42 (6.02, 9.61) * | |
| Neonate | Yes (%) | 85 | 311 (306, 445) | 17.4 (17.0, 18.2) | 5.57 (5.40, 6.53) |
| No (%) | 15 | 213 (134, 391) | 13.5 (12.0, 15.6) *** | 7.01 (5.34, 10.33) * | |
3.2. Maternal B12 and Metabolic Risk of Offspring
| Maternal Variable (SDS) | Neonate’s Metabolic Risk Factors (SDS) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Triglycerides * | Cholesterol * | HDL * | LDL * | Insulin * | Glucose * | Homocysteine * | |||||||||||||||
| Maternal B12 * | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p |
| Model 1 | −0.148 | (−0.38, 0.09) | 0.210 | 0.109 | (−0.11, 0.33) | 0.317 | 0.296 | (0.07, 0.52) | 0.010 | −0.044 | (−0.26, 0.17) | 0.691 | 0.070 | (−0.21, 0.21) | 0.516 | −0.005 | (−0.22, −0.21) | 0.960 | −0.381 | (−0.58, −0.18) | <0.001 |
| Model 2 | −0.086 | (−0.38, 0.21) | 0.562 | 0.178 | (−0.08, 0.44) | 0.173 | 0.294 | (0.05, 0.54) | 0.018 | 0.070 | (−0.26, 0.39) | 0.672 | −0.063 | (−0.17, 0.29) | 0.593 | −0.088 | (−0.14, −0.31) | 0.438 | −0.200 | (−0.35, −0.05) | 0.009 |
| Model 3 | −0.079 | (−0.39, 0.23) | 0.609 | 0.170 | (−0.09, 0.43) | 0.198 | 0.295 | (0.08, 0.51) | 0.009 | 0.056 | (−0.28, 0.39) | 0.736 | −0.062 | (−0.17, 0.29) | 0.602 | −0.093 | (−0.12, 0.30) | 0.378 | |||
| Maternal Folate * | |||||||||||||||||||||
| Model 1 | −0.109 | (−0.33, 0.11) | 0.326 | −0.210 | (−0.42, 0.001) | 0.051 | −0.025 | (−0.25, 0.19) | 0.825 | −0.173 | (−0.38, 0.04) | 0.111 | 0.040 | (−0.17, 0.25) | 0.705 | −0.124 | (−0.33, 0.09) | 0.243 | −0.327 | (−0.53, −0.13) | 0.002 |
| Model 2 | −0.084 | (−0.43, 0.26) | 0.625 | −0.204 | (−0.49, 0.08) | 0.160 | −0.236 | (−0.51, 0.04) | 0.091 | −0.079 | (−0.40, 0.24) | 0.625 | 0.133 | (−0.15, 0.42) | 0.357 | −0.035 | (−0.31, 0.24) | 0.799 | −0.004 | (−0.19, 0.18) | 0.966 |
| Model 3 | −0.093 | (−0.45, 0.26) | 0.600 | −0.200 | (−0.49, 0.09) | 0.169 | −0.209 | (−0.46, 0.04) | 0.099 | −0.082 | (−0.41, 0.24) | 0.616 | 0.136 | (−0.15, 0.43) | 0.353 | −0.039 | (−0.29, 0.22) | 0.763 | |||
| Maternal Homocysteine * | |||||||||||||||||||||
| Model 1 | 0.218 | (−0.00, 0.44) | 0.050 | 0.290 | (−0.07, 0.51) | 0.009 | 0.119 | (−0.13, 0.36) | 0.336 | 0.265 | (0.06, 0.47) | 0.013 | 0.001 | (−0.21, 0.21) | 0.993 | 0.121 | (−0.09, 0.34) | 0.269 | 0.752 | (0.62, 0.89) | <0.001 |
| Model 2 | 0.137 | (−0.19, 0.47) | 0.473 | 0.206 | (−0.08, 0.49) | 0.156 | 0.093 | (−0.23, 0.41) | 0.556 | 0.224 | (−0.09, 0.54) | 0.160 | 0.037 | (−0.23, 0.31) | 0.785 | 0.057 | (−0.21, 0.32) | 0.669 | 0.696 | (0.52, 0.87) | <0.001 |
| Model 3 | 0.126 | (−0.23, 0.48) | 0.474 | 0.230 | (−0.07, 0.53) | 0.129 | 0.158 | (−0.13, 0.45) | 0.278 | 0.248 | (−0.08, 0.57) | 0.130 | 0.043 | (−0.24, 0.32) | 0.759 | 0.089 | (−0.16, 0.34) | 0.469 | |||
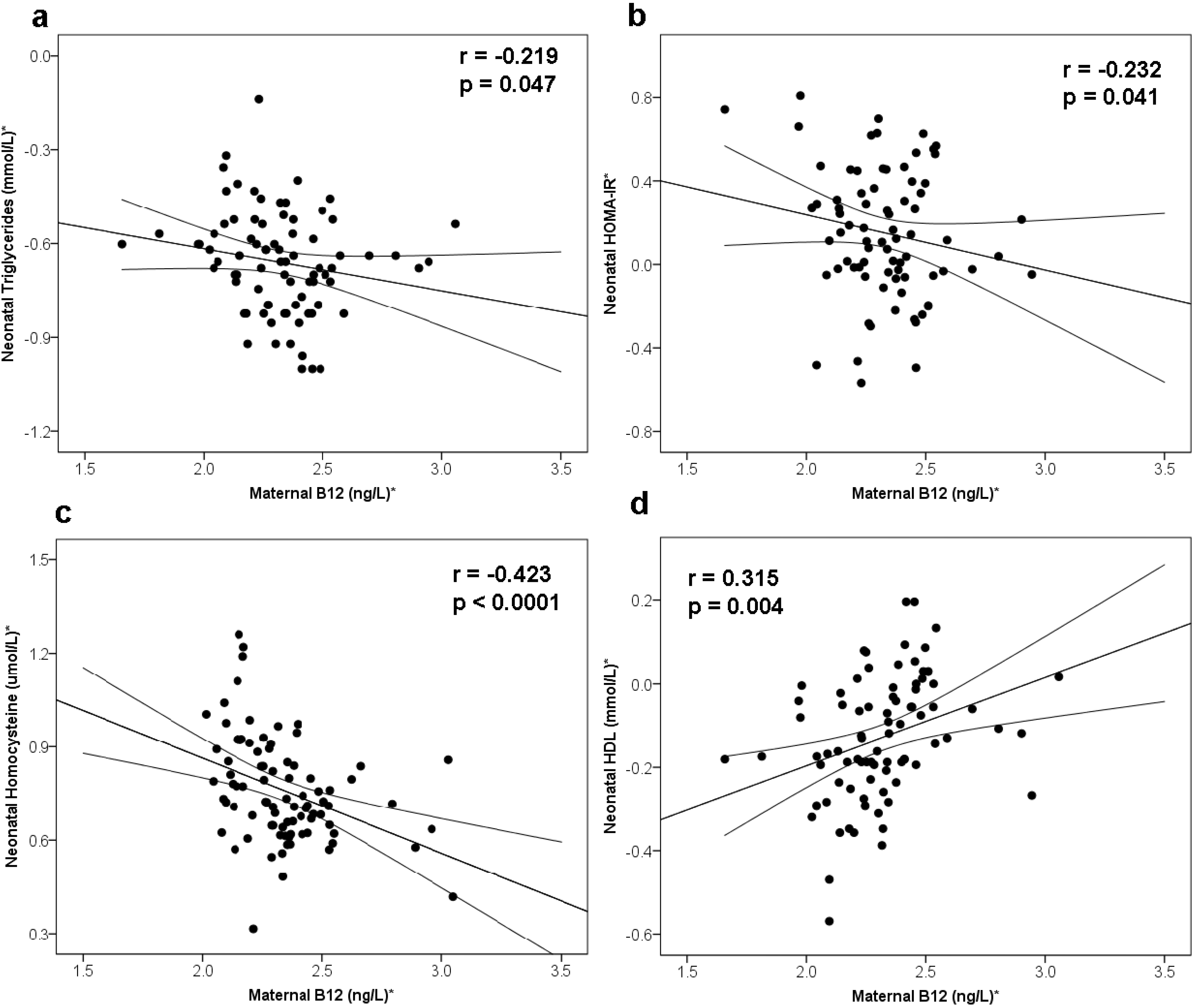
3.3. Maternal Folate and Homocysteine and Metabolic Risk of Offspring
4. Discussion
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cunningham, S.A.; Kramer, M.R.; Narayan, K.M. Incidence of childhood obesity in the united states. N. Engl. J. Med. 2014, 370, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.; Kaufman, F.R. Metabolic complications of childhood obesity: Identifying and mitigating the risk. Diabetes Care 2008, 31, S310–S316. [Google Scholar] [CrossRef] [PubMed]
- Ignell, C.; Claesson, R.; Anderberg, E.; Berntorp, K. Trends in the prevalence of gestational diabetes mellitus in southern sweden, 2003–2012. Acta Obstet. Gynecol. Scand. 2014, 93, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Lawlor, D.A.; Benfield, L.; Logue, J.; Tilling, K.; Howe, L.D.; Fraser, A.; Cherry, L.; Watt, P.; Ness, A.R.; Davey Smith, G.; et al. Association between general and central adiposity in childhood, and change in these, with cardiovascular risk factors in adolescence: Prospective cohort study. BMJ 2010, 341. [Google Scholar] [CrossRef]
- Nayak, C.D.; Agarwal, V.; Nayak, D.M. Correlation of cord blood lipid heterogeneity in neonates with their anthropometry at birth. Indian J. Clin. Biochem. IJCB 2013, 28, 152–157. [Google Scholar] [CrossRef]
- Whincup, P.H.; Kaye, S.J.; Owen, C.G.; Huxley, R.; Cook, D.G.; Anazawa, S.; Barrett-Connor, E.; Bhargava, S.K.; Birgisdottir, B.E.; Carlsson, S.; et al. Birth weight and risk of type 2 diabetes: A systematic review. JAMA 2008, 300, 2886–2897. [Google Scholar] [CrossRef]
- Pettitt, D.J.; Jovanovic, L. Low birth weight as a risk factor for gestational diabetes, diabetes, and impaired glucose tolerance during pregnancy. Diabetes Care 2007, 30 (Suppl. 2), S147–S149. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J.; Osmond, C.; Kajantie, E.; Eriksson, J.G. Growth and chronic disease: Findings in the Helsinki birth cohort. Ann. Hum. Biol. 2009, 36, 445–458. [Google Scholar] [CrossRef] [PubMed]
- Roseboom, T.J.; Painter, R.C.; van Abeelen, A.F.; Veenendaal, M.V.; de Rooij, S.R. Hungry in the womb: What are the consequences? Lessons from the dutch famine. Maturitas 2011, 70, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Yajnik, C.S.; Deshpande, S.S.; Jackson, A.A.; Refsum, H.; Rao, S.; Fisher, D.J.; Bhat, D.S.; Naik, S.S.; Coyaji, K.J.; Joglekar, C.V.; et al. Vitamin B12 and folate concentrations during pregnancy and insulin resistance in the offspring: The pune maternal nutrition study. Diabetologia 2008, 51, 29–38. [Google Scholar] [CrossRef]
- Stewart, C.P.; Christian, P.; Schulze, K.J.; Arguello, M.; LeClerq, S.C.; Khatry, S.K.; West, K.P. Low maternal vitamin B-12 status is associated with offspring insulin resistance regardless of antenatal micronutrient supplementation in rural Nepal. J. Nutr. 2011, 141, 1912–1917. [Google Scholar] [CrossRef] [PubMed]
- Krishnaveni, G.V.; Veena, S.R.; Karat, S.C.; Yajnik, C.S.; Fall, C.H. Association between maternal folate concentrations during pregnancy and insulin resistance in Indian children. Diabetologia 2014, 57, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Dwarkanath, P.; Barzilay, J.R.; Thomas, T.; Thomas, A.; Bhat, S.; Kurpad, A.V. High folate and low vitamin B-12 intakes during pregnancy are associated with small-for-gestational age infants in South Indian women: A prospective observational cohort study. Am. J. Clin. Nutr. 2013, 98, 1450–1458. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, P.; Yajnik, C.S. Role of maternal vitamin B12 on the metabolic health of the offspring: A contributor to the diabetes epidemic? Br. J. Diabetes Vasc. Disease 2010, 10, 109–114. [Google Scholar] [CrossRef]
- Guerra-Shinohara, E.M.; Morita, O.E.; Peres, S.; Pagliusi, R.A.; Sampaio Neto, L.F.; D’Almeida, V.; Irazusta, S.P.; Allen, R.H.; Stabler, S.P. Low ratio of S-adenosylmethionine to S-adenosylhomocysteine is associated with vitamin deficiency in Brazilian pregnant women and newborns. Am. J. Clin. Nutr. 2004, 80, 1312–1321. [Google Scholar] [PubMed]
- Ba, Y.; Yu, H.; Liu, F.; Geng, X.; Zhu, C.; Zhu, Q.; Zheng, T.; Ma, S.; Wang, G.; Li, Z.; et al. Relationship of folate, vitamin B12 and methylation of insulin-like growth factor-II in maternal and cord blood. Eur. J. Clin. Nutr. 2011, 65, 480–485. [Google Scholar] [CrossRef]
- Sinclair, K.D.; Allegrucci, C.; Singh, R.; Gardner, D.S.; Sebastian, S.; Bispham, J.; Thurston, A.; Huntley, J.F.; Rees, W.D.; Maloney, C.A.; et al. DNA methylation, insulin resistance, and blood pressure in offspring determined by maternal periconceptional B vitamin and methionine status. Proc. Natl. Acad. Sci. USA 2007, 104, 19351–19356. [Google Scholar] [CrossRef]
- Garcia, M.M.; Gueant-Rodriguez, R.M.; Pooya, S.; Brachet, P.; Alberto, J.M.; Jeannesson, E.; Maskali, F.; Gueguen, N.; Marie, P.Y.; Lacolley, P.; et al. Methyl donor deficiency induces cardiomyopathy through altered methylation/acetylation of PGC-1alpha by PRMT1 and SIRT1. J. Pathol. 2011, 225, 324–335. [Google Scholar] [CrossRef]
- Kumar, K.A.; Lalitha, A.; Pavithra, D.; Padmavathi, I.J.; Ganeshan, M.; Rao, K.R.; Venu, L.; Balakrishna, N.; Shanker, N.H.; Reddy, S.U.; et al. Maternal dietary folate and/or vitamin B12 restrictions alter body composition (adiposity) and lipid metabolism in Wistar rat offspring. J. Nutr. Biochem. 2013, 24, 25–31. [Google Scholar] [CrossRef]
- Adaikalakoteswari, A.; Jayashri, R.; Sukumar, N.; Venkataraman, H.; Pradeepa, R.; Gokulakrishnan, K.; Anjana, R.; McTernan, P.G.; Tripathi, G.; Patel, V.; et al. Vitamin B12 deficiency is associated with adverse lipid profile in Europeans and Indians with type 2 diabetes. Cardiovasc. Diabetol. 2014, 13. [Google Scholar] [CrossRef]
- Saravanan, P.; Maheshwaran, H.; Stranges, S.; Thorogood, M. Population levels of B12 and folic acid: Do we need to consider B12 fortification to prevent gestational diabetes and cardiovascular risk? Diabet. Med. 2010, 27 (Suppl. 1), 37–188. [Google Scholar] [PubMed]
- Saravanan, P.; Wood, C.; Andersen, N. B12 deficiency is more common than folate deficiency in early pregnancy: Do we need to consider B12 fortification? Diabetologia 2010, 53, S151–S152. [Google Scholar]
- Milman, N.; Byg, K.E.; Bergholt, T.; Eriksen, L.; Hvas, A.M. Cobalamin status during normal pregnancy and postpartum: A longitudinal study comprising 406 Danish women. Eur. J. Haematol. 2006, 76, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Milman, N.; Byg, K.E.; Hvas, A.M.; Bergholt, T.; Eriksen, L. Erythrocyte folate, plasma folate and plasma homocysteine during normal pregnancy and postpartum: A longitudinal study comprising 404 Danish women. Eur. J. Haematol. 2006, 76, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Magera, M.J.; Lacey, J.M.; Casetta, B.; Rinaldo, P. Method for the determination of total homocysteine in plasma and urine by stable isotope dilution and electrospray tandem mass spectrometry. Clin. Chem. 1999, 45, 1517–1522. [Google Scholar] [PubMed]
- Finer, S.; Saravanan, P.; Hitman, G.; Yajnik, C. The role of the one-carbon cycle in the developmental origins of type 2 diabetes and obesity. Diabet. Med. 2014, 31, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Wallace, J.M.; Bonham, M.P.; Strain, J.; Duffy, E.M.; Robson, P.J.; Ward, M.; McNulty, H.; Davidson, P.W.; Myers, G.J.; Shamlaye, C.F.; et al. Homocysteine concentration, related B vitamins, and betaine in pregnant women recruited to the Seychelles child development study. Am. J. Clin. Nutr. 2008, 87, 391–397. [Google Scholar]
- United States Department of Agriculture. USDA Table of Nutrient Retention Factors, Release 5. 2003. Available online: http://www.ars.usda.gov/Main/docs.htm?docid=9448 (accessed on 27 March 2015). [Google Scholar]
- Adaikalakoteswari, A.; Finer, S.; Voyias, P.D.; McCarthy, C.M.; Vatish, M.; Moore, J.; Smart-Halajko, M.; Bawazeer, N.; Al-Daghri, N.M.; McTernan, P.G.; et al. Vitamin B12 insufficiency induces cholesterol biosynthesis by limiting S-adenosylmethionine and modulating the methylation of SREBF1 and LDLR genes. Clin. Epigenetics 2015, 7. [Google Scholar] [CrossRef]
- Schaefer-Graf, U.M.; Graf, K.; Kulbacka, I.; Kjos, S.L.; Dudenhausen, J.; Vetter, K.; Herrera, E. Maternal lipids as strong determinants of fetal environment and growth in pregnancies with gestational diabetes mellitus. Diabetes Care 2008, 31, 1858–1863. [Google Scholar] [CrossRef] [PubMed]
- Schaefer-Graf, U.M.; Meitzner, K.; Ortega-Senovilla, H.; Graf, K.; Vetter, K.; Abou-Dakn, M.; Herrera, E. Differences in the implications of maternal lipids on fetal metabolism and growth between gestational diabetes mellitus and control pregnancies. Diabet. Med. 2011, 28, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Molloy, A.M.; Mills, J.L.; Cox, C.; Daly, S.F.; Conley, M.; Brody, L.C.; Kirke, P.N.; Scott, J.M.; Ueland, P.M. Choline and homocysteine interrelations in umbilical cord and maternal plasma at delivery. Am. J. Clin. Nutr. 2005, 82, 836–842. [Google Scholar] [PubMed]
- Wald, D.S.; Law, M.; Morris, J.K. Homocysteine and cardiovascular disease: Evidence on causality from a meta-analysis. BMJ 2002, 325, 1202. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Salas, P.; Cox, S.E.; Prentice, A.M.; Hennig, B.J.; Moore, S.E. Maternal nutritional status, C(1) metabolism and offspring DNA methylation: A review of current evidence in human subjects. Proc. Nutr. Soc. 2012, 71, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Brindle, N.P.; Zammit, V.A.; Pogson, C.I. Regulation of carnitine palmitoyltransferase activity by malonyl-CoA in mitochondria from sheep liver, a tissue with a low capacity for fatty acid synthesis. Biochem. J. 1985, 232, 177–182. [Google Scholar] [PubMed]
- Cikot, R.J.; Steegers-Theunissen, R.P.; Thomas, C.M.; de Boo, T.M.; Merkus, H.M.; Steegers, E.A. Longitudinal vitamin and homocysteine levels in normal pregnancy. Br. J. Nutr. 2001, 85, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Carmel, R.; Agrawal, Y.P. Failures of cobalamin assays in pernicious anemia. N. Engl. J. Med. 2012, 367, 385–386. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adaikalakoteswari, A.; Vatish, M.; Lawson, A.; Wood, C.; Sivakumar, K.; McTernan, P.G.; Webster, C.; Anderson, N.; Yajnik, C.S.; Tripathi, G.; et al. Low Maternal Vitamin B12 Status Is Associated with Lower Cord Blood HDL Cholesterol in White Caucasians Living in the UK. Nutrients 2015, 7, 2401-2414. https://doi.org/10.3390/nu7042401
Adaikalakoteswari A, Vatish M, Lawson A, Wood C, Sivakumar K, McTernan PG, Webster C, Anderson N, Yajnik CS, Tripathi G, et al. Low Maternal Vitamin B12 Status Is Associated with Lower Cord Blood HDL Cholesterol in White Caucasians Living in the UK. Nutrients. 2015; 7(4):2401-2414. https://doi.org/10.3390/nu7042401
Chicago/Turabian StyleAdaikalakoteswari, Antonysunil, Manu Vatish, Alexander Lawson, Catherine Wood, Kavitha Sivakumar, Philip G. McTernan, Craig Webster, Neil Anderson, Chittaranjan S. Yajnik, Gyanendra Tripathi, and et al. 2015. "Low Maternal Vitamin B12 Status Is Associated with Lower Cord Blood HDL Cholesterol in White Caucasians Living in the UK" Nutrients 7, no. 4: 2401-2414. https://doi.org/10.3390/nu7042401
APA StyleAdaikalakoteswari, A., Vatish, M., Lawson, A., Wood, C., Sivakumar, K., McTernan, P. G., Webster, C., Anderson, N., Yajnik, C. S., Tripathi, G., & Saravanan, P. (2015). Low Maternal Vitamin B12 Status Is Associated with Lower Cord Blood HDL Cholesterol in White Caucasians Living in the UK. Nutrients, 7(4), 2401-2414. https://doi.org/10.3390/nu7042401






