Longitudinal Study of Cytokine Expression, Lipid Profile and Neuronal Growth Factors in Human Breast Milk from Term and Preterm Deliveries
Abstract
:1. Introduction
2. Material & Methods
2.1. Subjects and Design
| Term Gestations (n = 20) | Preterm Gestations (n = 20) | |
|---|---|---|
| Maternal age (year) | 30 (25.0–32.8) | 32.1 (27.2–33.7) |
| Length of pregnancy (week) | 39.2 (38.0–40.0) | 29 (27–315) |
| Mode of delivery | ||
| Vaginal (%) | 10 (50%) | 6 (30%) |
| C-section (%) | 10 (50%) | 14 (70%) |
| Birth weight (g) | 3200 (3000–3650) | 2850 (2500–3000) |
2.2. Breast Milk Samples
2.3. Fatty Acid Analysis
2.4. Protein Array Analysis
2.5. Statistical Analysis and Bioinformatics
3. Results
3.1. Fatty Acids Analysis
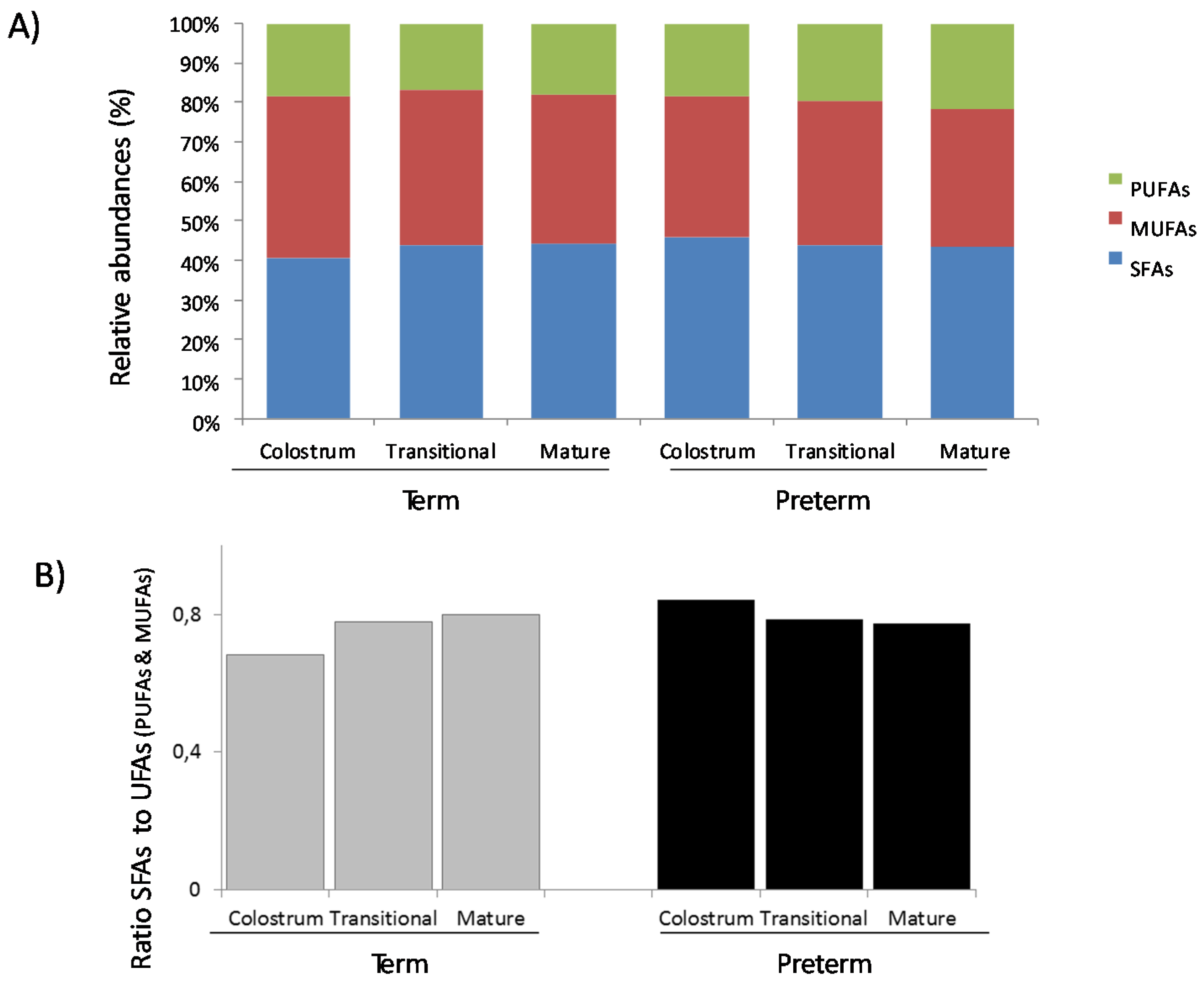
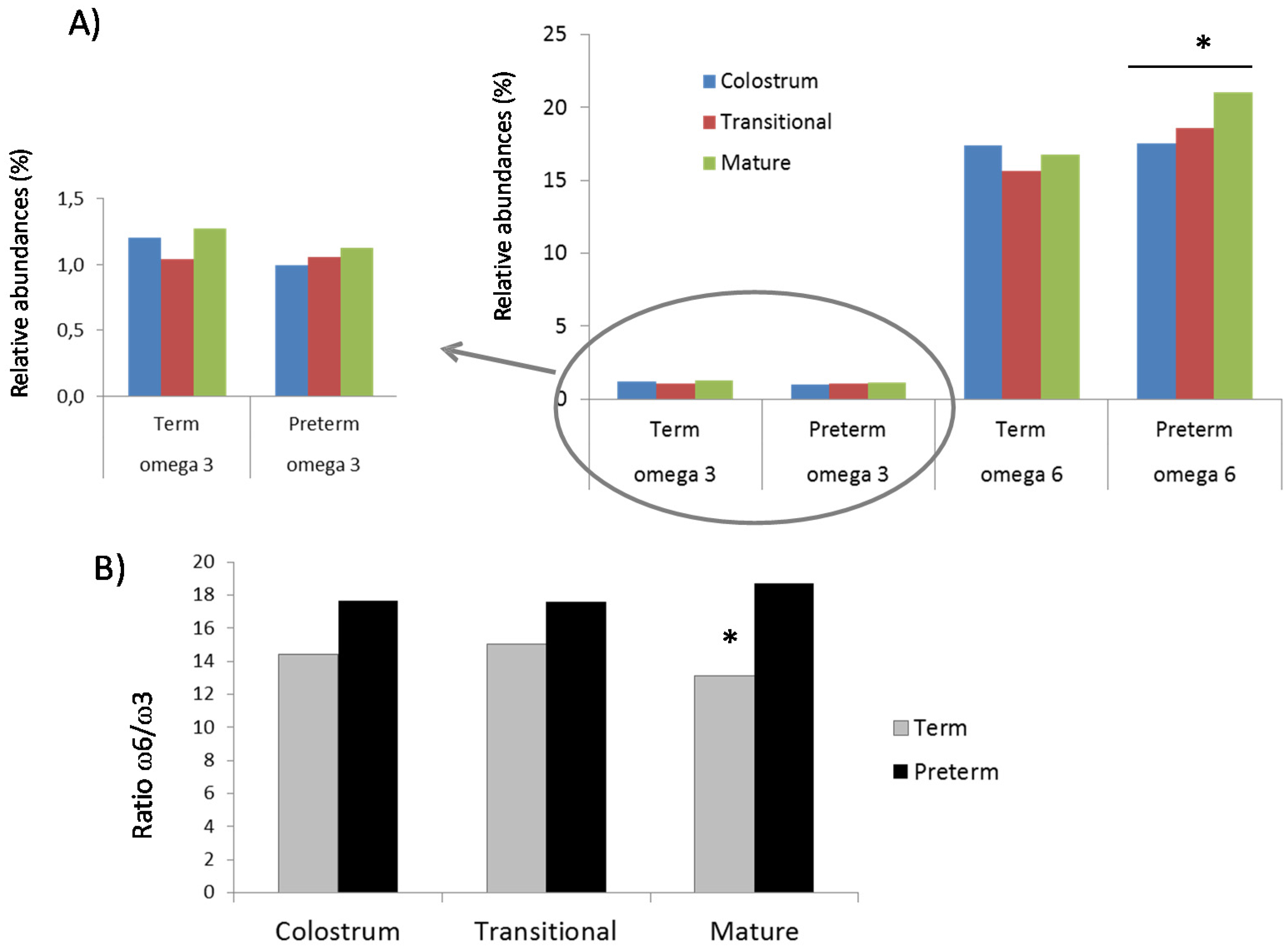
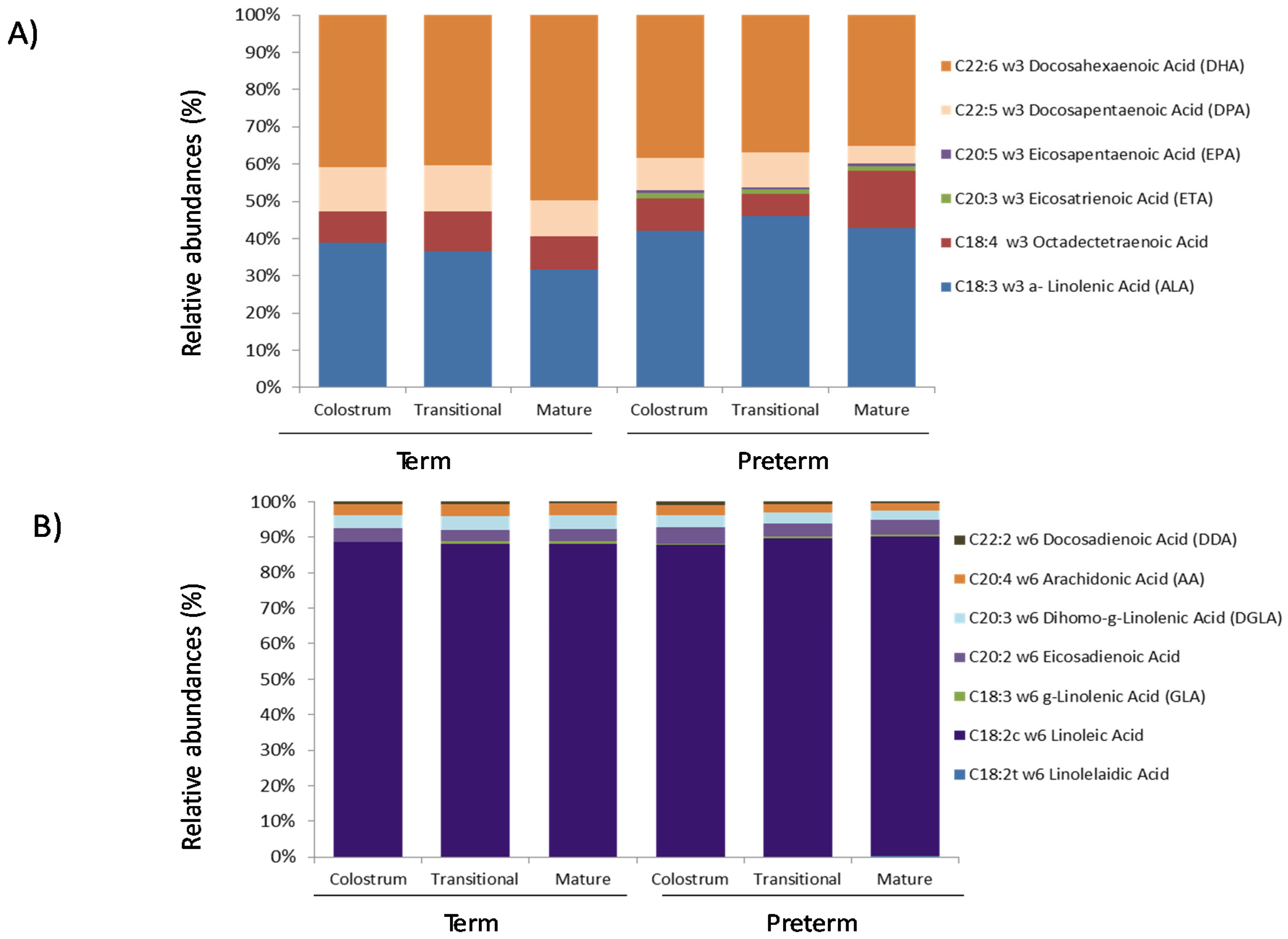
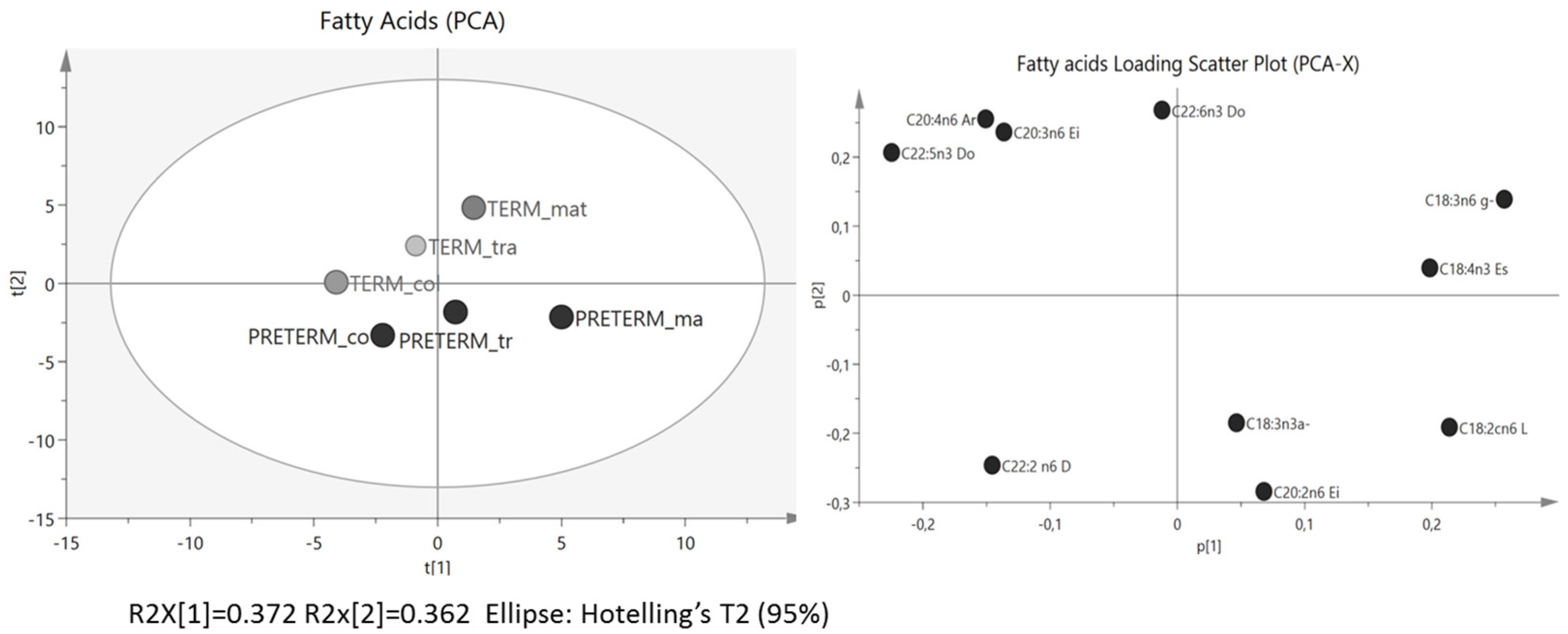
3.2. Protein Array Analysis
| A: Fold change table comparing cytokine profile according to lactation stage | |||
|---|---|---|---|
| Colostrum vs. Transitional Milk | Colostrum vs. Mature Milk | Transitional vs. Mature Milk | |
| Neurtophic factors | |||
| Brain-derived neurotrophic factor (BDNF) | 1.00 | 0.36 * | 0.36 * |
| Ciliary neurotrophic factor (CNTF) | 0.96 | 0.34 * | 0.35 * |
| Glial cell-derived neurotrophic factor (GDNF) | 0.94 | 0.14 * | 0.15 * |
| Neurotrophin-3 | 0.91 | 0.23 * | 0.25 * |
| Other growth factors | |||
| Angiogenin | 0.99 | 0.20 * | 0.21 * |
| Bone morphogenetic protein 4 | 0.85 | 0.07 * | 0.08 * |
| Bone morphogenetic protein 6 | 1.07 | 0.46 * | 0.43 * |
| Epidermal growth factor | 0.84 | 0.13 * | 0.16 * |
| Fibroblast growth factor 6 | 0.65 * | 0.20 * | 0.30 * |
| Fibroblast growth factor 7 | 0.83 | 0.38 * | 0.46 * |
| Fms-related tyrosine kinase-3 ligand | 0.88 | 0.38 * | 0.43 * |
| Insulin-like growth factor-binding protein 1 | 1.44 | 1.05 | 0.73 * |
| Insulin-like growth factor-1 | 1.03 | 0.37 * | 0.35 * |
| Platelet-derived growth factor | 1.08 | 0.58 * | 0.54 * |
| Cytokines | |||
| CK β 8-1 | 1.13 | 0.62 * | 0.55 * |
| IFN-gamma | 0.90 | 1.81 * | 2.01 * |
| IL-1β | 1.06 | 0.88 | 0.83 |
| IL-1α | 0.83 | 0.46 * | 0.55 * |
| IL-2 | 0.91 | 0.44 * | 0.48 * |
| IL-3 | 1.00 | 0.29 * | 0.29 * |
| IL-4 | 1.07 | 0.35 * | 0.33 * |
| IL-5 | 0.89 | 0.33 * | 0.37 * |
| IL-6 | 0.95 | 0.45 * | 0.47 * |
| IL-7 | 0.86 | 0.33 * | 0.38 * |
| IL-10 | 0.76 | 0.35 * | 0.45 * |
| IL-13 | 0.66 | 0.26 * | 0.39 * |
| IL-15 | 0.78 | 0.41 * | 0.53 * |
| Monokine induced by gamma interferon | 0.99 | 0.60 * | 0.61 * |
| Neutrophil-activating protein-2 | 1.01 | 0.50 * | 0.49 * |
| Thymus and activation-regulated chemokine | 0.98 | 0.48 * | 0.49 * |
| Transforming growth factor-β 3 | 0.95 | 0.52 * | 0.55 * |
| Tumor necrosis factor-α | 0.95 | 0.45 * | 0.47 * |
| Tumor necrosis factor-β | 0.95 | 0.20 * | 0.21 * |
| Chemoattractant cytokines | |||
| Eotaxin | 1.14 | 0.30 * | 0.27 * |
| Fractalkine | 0.84 | 0.19 * | 0.23 * |
| Granulocyte chemotactic protein 2 | 0.95 | 0.47 * | 0.49 * |
| Granulocyte-macrophage colony-stimulating factor | 0.88 | 1.41 | 1.61 * |
| I-309 | 0.89 | 0.56 * | 0.63 * |
| Monocyte chemoattractant protein-1 | 1.05 | 0.72 | 0.69 |
| Monocyte chemoattractant protein-2 | 1.03 | 0.34 * | 0.33 * |
| Monocyte chemoattractant protein-3 | 0.91 | 0.40 * | 0.44 * |
| Monocyte chemoattractant protein-4 | 0.99 | 0.38 * | 0.38 * |
| Macrophage colony-stimulating factor | 0.95 | 0.45 * | 0.47 * |
| Human macrophage-derived chemokine | 1.07 | 0.39 * | 0.37 * |
| MIP-1-delta | 0.82 | 0.12 * | 0.15 * |
| MIP-3-α | 1.26 | 0.24 * | 0.19 * |
| Pulmonary and activation-regulated Cytokine (PARC) | 0.83 | 0.18 * | 0.22 * |
| Stromal cell-derived factor-1 | 0.97 | 0.31 * | 0.32 * |
| B: Fold change table comparing cytokine profile according to gestational age | |||
| Preterm vs. Term colostrum | Preterm vs. Term transitional | Preterm vs. Term mature | |
| Neurtophic factors | |||
| Brain-derived neurotrophic factor (BDNF) | 0.89 | 0.90 | 2.14 * |
| Ciliary neurotrophic factor (CNTF) | 0.96 | 0.96 | 2.04 * |
| Glial cell-derived neurotrophic factor (GDNF) | 1.39 | 1.28 | 5.06 * |
| Neurotrophin-3 | 1.10 | 0.98 | 3.60 * |
| Other growth factors | |||
| Angiogenin | 0.74 | 0.89 | 5.23 * |
| Bone morphogenetic protein 4 | 1.00 | 0.48 * | 12.80 * |
| Bone morphogenetic protein 6 | 1.12 | 1.12 | 1.83 * |
| Epidermal growth factor | 0.94 | 6.61 * | 2.78 * |
| Fibroblast growth factor 6 | 1.14 | 1.10 | 1.54 |
| Fibroblast growth factor 7 | 1.01 | 1.85 * | 1.40 |
| Fms-related tyrosine kinase-3 ligand | 1.13 | 1.62 * | 1.34 |
| Insulin-like growth factor-binding protein 1 | 0.70 | 1.27 | 3.80 * |
| Insulin-like growth factor-1 | 0.88 | 0.76 | 2.43 * |
| Platelet-derived growth factor | 1.11 | 0.45 * | 2.68 * |
| Cytokines | |||
| CK β 8-1 | 1.07 | 0.27 * | 3.26 * |
| IFN-gamma | 0.98 | 2.07 * | 0.63 * |
| IL-1β | 1.18 | 0.40 * | 1.70 * |
| IL-1α | 0.95 | 2.54 * | 1.24 |
| IL-2 | 0.87 | 1.44 | 1.59 * |
| IL-3 | 1.13 | 1.46 | 2.15 * |
| IL-4 | 0.90 | 0.86 | 2.39 * |
| IL-5 | 0.84 | 1.95 * | 1.52 * |
| IL-6 | 0.71 | 0.97 | 1.97 * |
| IL-7 | 0.78 | 1.40 | 1.81 * |
| IL-10 | 2.87 * | 4.08 * | 2.09 * |
| IL-13 | 1.99 * | 3.13 * | 2.20 * |
| IL-15 | 1.96 * | 2.57 * | 1.21 |
| Monokine induced by gamma interferon | 0.90 | 1.07 | 1.47 |
| Neutrophil-activating protein-2 | 0.89 | 0.59 * | 3.89 * |
| Thymus and activation-regulated chemokine | 1.03 | 0.90 | 1.67 * |
| Transforming growth factor-β 3 | 0.97 | 1.11 | 1.62 * |
| Tumor necrosis factor-α | 0.88 | 0.85 | 1.79 * |
| Tumor necrosis factor-β | 0.99 | 1.46 | 2.84 * |
| Chemoattractant cytokines | |||
| Eotaxin | 0.97 | 0.26 * | 5.78 * |
| Fractalkine | 1.54 * | 1.27 | 2.71 * |
| Granulocyte chemotactic protein 2 | 1.80 * | 0.80 | 1.97 * |
| Granulocyte-macrophage colony-stimulating factor | 0.99 | 2.53 * | 0.87 |
| I-309 | 1.37 | 0.20 * | 2.21 * |
| Monocyte chemoattractant protein -1 | 1.07 | 0.63 * | 5.97 * |
| Monocyte chemoattractant protein -2 | 0.87 | 0.79 | 2.02 * |
| Monocyte chemoattractant protein -3 | 0.80 | 0.78 | 1.74 * |
| Monocyte chemoattractant protein -4 | 0.97 | 0.86 | 2.79 * |
| Macrophage colony-stimulating factor | 0.56 * | 5.41 * | 5.61 * |
| Human macrophage-derived chemokine | 0.81 | 1.36 | 2.81 * |
| MIP-1-delta | 0.66 | 0.91 | 1.92 * |
| MIP-3-α | 0.67 | 0.40 * | 6.88 * |
| Pulmonary and activation-regulated Cytokine (PARC) | 1.14 | 3.02 * | 3.02 * |
| Stromal cell-derived factor-1 | 0.95 | 1.20 | 1.56 * |
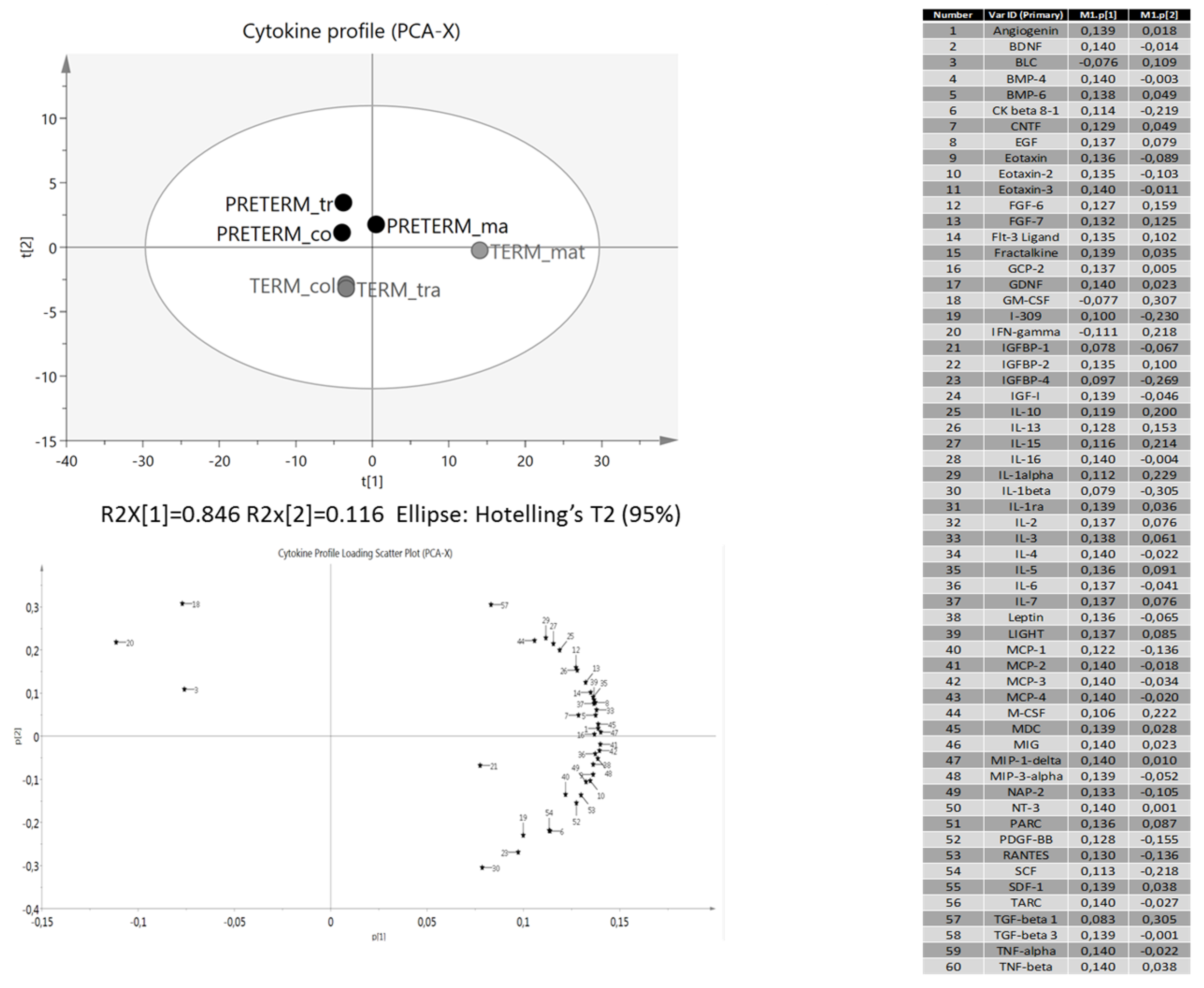
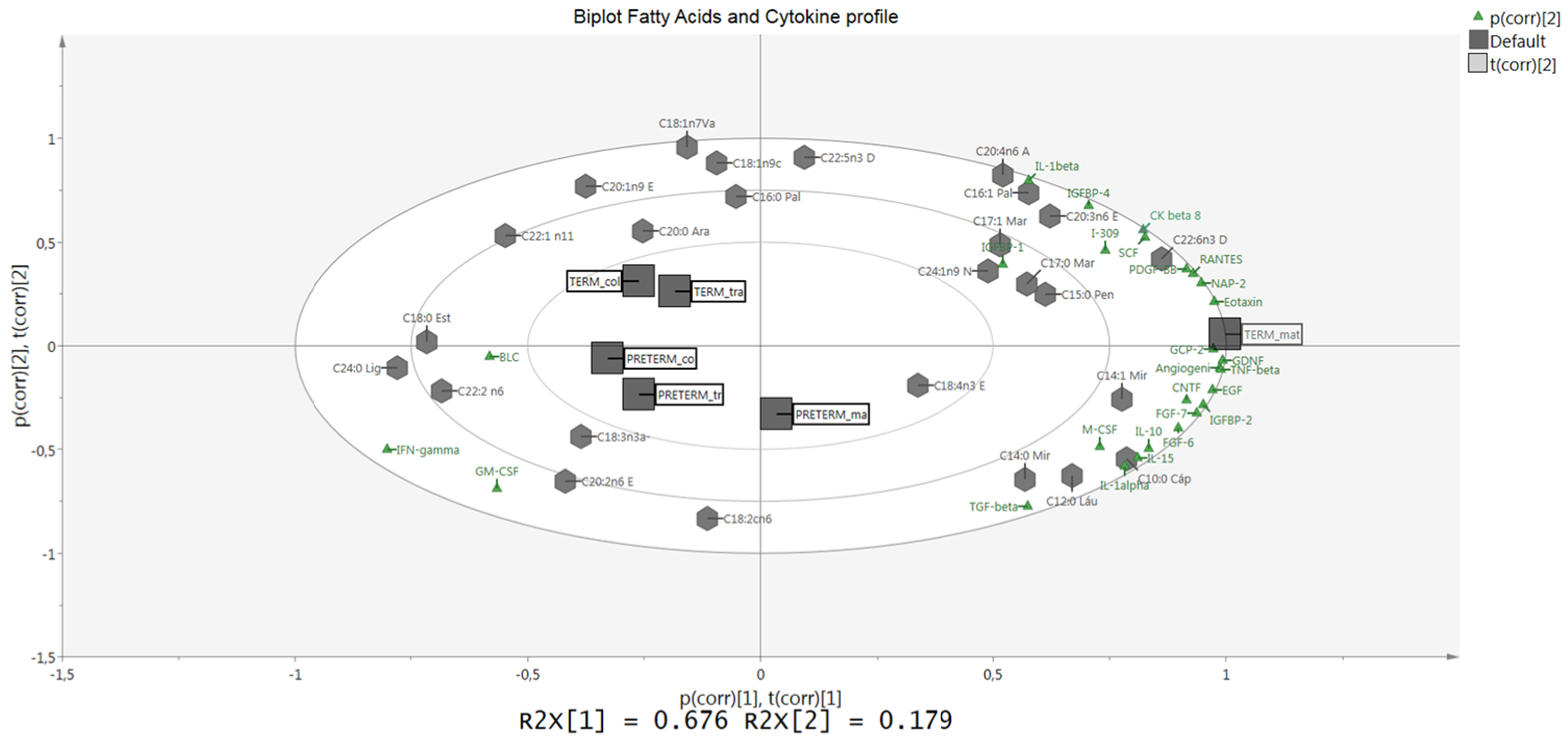
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix
| RayBio® Human Cytokine Antibody G Series VI | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| POS 1 | POS 2 | POS 3 | NEG | NEG | Angiogenin | BDNF | BLC | BMP-4 | BMP-6 | CK β 8-1 | CNTF | EGF | Eotaxin |
| Eotaxin-2 | Eotaxin-3 | FGF-6 | FGF-7 | Fit-3 Ligand | Fractalkine | GCP-2 | GDNF | GM-CSF | I-309 | IFN-gamma | IGFBP-1 | IGFBP-2 | IGFBP-4 |
| IGF-I | IL-10 | IL-13 | IL-15 | IL-16 | IL-1α | IL-1β | IL-1ra | IL-2 | IL-3 | IL-4 | IL-5 | IL-6 | IL-7 |
| Leptin | LIGHT | MCP-1 | MCP-2 | MCP-3 | MCP-4 | M-CSF | MDC | MIG | MIP-1delta | MIP-3α | NAP-2 | NT-3 | PARC |
| PDGF-BB | RANTES | SCF | SDF-1 | TARC | TGF-β1 | TGF-β3 | TNF-α | TNF-β | IC1 | IC2 | IC3 | NEG | NEG |
References
- Walker, A. Breast milk as the gold standard for protective nutrients. J. Pediatr. 2010, 156, S3–S7. [Google Scholar] [CrossRef] [PubMed]
- Lönnerdal, B. Bioactive proteins in human milk: Mechanisms of action. J. Pediatr. 2010, 156, S26–S30. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, R.A.; Lawrence, R.M. Host-resistance factors and immunologic significance of human milk. In Breastfeeding: A Guide for Medical Profession, 7th ed.; Lawrence, R.A., Lawrence, R.M., Eds.; Elsevier: Mosby, MO, USA, 2011; pp. 153–195. [Google Scholar]
- Garofalo, R. Cytokines in human milk. J. Pediatr. 2010, 156, S36–S40. [Google Scholar] [CrossRef] [PubMed]
- Gregory, K.E.; Walker, W.A. Immunologic factors in human milk and disease prevention in the preterm Infant. Curr. Pediatr. Rep. 2013, 1, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Goldman, A.S.; Chheda, S.; Garofalo, R.; Schmalstieg, F.C. Cytokines in human milk: Properties and potential effects upon the mammary gland and the neonate. J. Mammary Gland Biol. Neoplasia 1996, 1, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, M.D.; Srivastava, A.; Brouhard, B.; Saneto, R.; Groh-Warg, S.; Kubit, J. Cytokines in human milk. Res. Commun. Mol. Pathol. Pharmacol. 1996, 93, 263–287. [Google Scholar] [PubMed]
- Kverka, M.; Burianova, J.; Lodinova-Zadnikova, R.; Kocourkova, I.; Cinova, J.; Tuckova, L.; Tlaskalova-Hogenova, H. Cytokine profiling in human colostrum and milk by protein array. Clin. Chem. 2007, 53, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, J.S.; Bryan, D.L.; James, M.J.; Gibson, R.A. Cytokines (IL-1β, IL-6, TNF-α, TGF-β1, and TGF-β2) and prostaglandin E2 in human milk during the first three months postpartum. Pediatr. Res. 1999, 46, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Groer, M.; Duffy, A.; Morse, S.; Kane, B.; Zaritt, J.; Roberts, S.; Ashmeade, T. Cytokines, chemokines, and growth factors in banked human donor milk for preterm infants. J. Hum. Lact. 2014, 24, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Dangat, K.; Kilari, A.; Mehendale, S.; Lalwani, S.; Joshi, S. Higher levels of brain derived neurotrophic factor but similar nerve growth factor in human milk in women with preeclampsia. Int. J. Dev. Neurosci. 2013, 31, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Chalazonitis, A.; Pham, T.D.; Rothman, T.P.; DiStefano, P.S.; Bothwell, M.; Blair-Flynn, J.; Tesarollo, L.; Gershon, M.D. Neurotrophin-3 is required for the survival-differentiation of subsets of developing enteric neurons. J. Neurosci. 2001, 21, 5620–5636. [Google Scholar] [PubMed]
- Brenna, J.T.; Varamini, B.; Jensen, R.G.; Diersen-Schade, D.A.; Boettcher, J.A.; Arterburn, L.M. Docosahexaenoic and arachidonic acid concentrations in human breast milk worldwide. Am. J. Clin. Nutr. 2007, 85, 1457–1464. [Google Scholar] [PubMed]
- Helland, I.B.; Smith, L.; Saarem, K.; Saugstad, O.D.; Drevon, C.A. Maternal supplementation with very-long-chain n-3 fatty acids during pregnancy and lactation augments children’s IQ at 4 years of age. Pediatrics 2003, 111, e39–e44. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.; Morley, R.; Cole, T.J.; Gore, S.M. A randomised multicentre study of human milk versus formula and later development in preterm infants. Arch. Dis. Child. 1994, 70, F141–F146. [Google Scholar] [CrossRef]
- Horwood, L.J.; Fergusson, D.M. Breastfeeding and later cognitive and academic outcomes. Pediatrics 1998, 101, E9. [Google Scholar] [CrossRef] [PubMed]
- Daniels, M.C.; Adair, L.S. Breast-feeding influences cognitive development in Filipino children. J. Nutr. 2005, 135, 2589–2595. [Google Scholar] [PubMed]
- Koletzko, B.; Cetin, I.; Brenna, J.T.; Perinatal Lipid Intake Working Group; Child Health Foundation; Diabetic Pregnancy Study Group; European Association of Perinatal Medicine; European Society for Clinical Nutrition and Metabolism; European Society for Paediatric Gastroenterology, Hepatology and Nutrition, Committee on Nutrition. Dietary fat intakes for pregnant and lactating women. Br. J. Nutr. 2007, 98, 873–875. [Google Scholar] [PubMed]
- Khodayar-Pardo, P.; Mira-Pascual, L.; Collado, M.C.; Martínez-Costa, C. Impact of lactation stage, gestational age and mode of delivery on breast milk microbiota. J. Perinatol. 2014, 34, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.J.; Fallon, E.M.; Kalish, B.T.; Gura, K.M.; Puder, M. The role of the ω-3 fatty acid DHA in the human cycle life. J. Parenter. Enteral Nutr. 2013, 37, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Hadders-Algra, M. Effect of long-chain polyunsaturated fatty acid supplementation on neurodevelopmental outcome in full-term infants. Nutrients 2010, 2, 790–804. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, R.A.; Lawrence, R.M. Biochemistry of human milk. In Breastfeeding: A Guide for Medical Profession, 7th ed.; Lawrence, R.A., Lawrence, R.M., Eds.; Elsevier: Mosby, MO, USA, 2011; pp. 98–152. [Google Scholar]
- López-López, A.; López-Sabater, M.C.; Campoy-Folgoso, C.; Rivero-Urgell, M.; Castellote-Bargalló, A.I. Fatty acid and sn-2 fatty acid composition in human milk from Granada (Spain) and in infant formulas. Eur. J. Clin. Nutr. 2002, 56, 1242–1254. [Google Scholar] [CrossRef] [PubMed]
- Yuhas, R.; Pramuk, K.; Lien, E.L. Human milk fatty acid composition from nine countries varies most in DHA. Lipids 2006, 41, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Mihályi, K.; Györei, E.; Szabó, É.; Marosvölgyi, T.; Lohner, S.; Decsi, T. Contribution of n-3 long-chain polyunsaturated fatty acids to human milk is still low in Hungarian mothers. Eur. J. Peditar. 2015, 174, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Krauss-Etschmann, S.; Shadid, R.; Campoy, C.; Hoster, E.; Demmelmair, H.; Jiménez, M.; Gil, A.; Rivero, M.; Veszprémi, B.; Decsi, T.; et al. Effects of fish-oil and folate supplementation of pregnant women on maternal and fetal plasma concentrations of docosahexaenoic acid and eicosapentaenoic acid: A European randomized multicenter trial. Am. J. Clin. Nutr. 2007, 85, 1392–1400. [Google Scholar] [PubMed]
- Martin, M.A.; Lassek, W.D.; Gaulin, S.J.; Evans, R.W.; Woo, J.G.; Geraghty, S.R.; Davidson, B.S.; Morrow, A.L.; Kaplan, H.S.; Gurven, M.D. Fatty acid composition in the mature milk of Bolivian forager-horticulturalists: Controlled comparisons with a US sample. Matern. Child Nutr. 2012, 8, 404–418. [Google Scholar] [CrossRef] [PubMed]
- Granot, E.; Ishay-Gigi, K.; Malaach, L.; Flidel-Rimon, O. Is there a difference in breast milk fatty acid composition of mothers of preterm and term infants? J. Matern. Fetal Neonatal Med. 2015, 10, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Liu, C.H.; Roberts, A.I.; Das, J.; Xu, G.; Ren, G.; Zhang, Y.; Zhang, L.; Yuan, Z.R.; Tan, H.S.; et al. Granulocyte-macrophage colony-stimulating factor (GM-CSF) and T-cell responses: What we do and don’t know. Cell Res. 2006, 16, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Brandtzaeg, P. Mucosal immunity: Integration between mother and the breast-fed infant. Vaccine 2003, 21, 3382–3388. [Google Scholar] [CrossRef]
- Dvorak, B.; Fituch, C.C.; Williams, C.S.; Hurst, N.M.; Schanler, R.J. Increased epidermal growth factor levels in human milk of mothers with extremely premature infants. Pediatr. Res. 2003, 54, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Erickson, J.E.; Brosenitsch, T.A.; Katz, D.M. Brain-derived neurotrophic factor and glial cell line-derived neurotrophic factor are required simultaneously for survival of dopaminergic primary sensory neurons in vivo. J. Neurosci. 2001, 21, 581–589. [Google Scholar] [PubMed]
- Frost, B.L.; Jilling, T.; Lapin, B.; Maheshwari, A.; Caplan, M.S. Maternal breast milk transforming growth factor-β and feeding intolerance in preterm infants. Pediatr. Res. 2014, 76, 386–389. [Google Scholar] [CrossRef] [PubMed]
- Fichter, M.; Klotz, M.; Hirschberg, D.L.; Waldura, B.; Schofer, O.; Ehnert, S.; Schwarz, L.K.; van Ginneken, C.; Schäfer, K.K. Breast milk contains relevant neurotrophic factors and cytokines for enteric nervous system development. Mol. Nutr. Food Res. 2011, 55, 1592–1596. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Collado, M.C.; Santaella, M.; Mira-Pascual, L.; Martínez-Arias, E.; Khodayar-Pardo, P.; Ros, G.; Martínez-Costa, C. Longitudinal Study of Cytokine Expression, Lipid Profile and Neuronal Growth Factors in Human Breast Milk from Term and Preterm Deliveries. Nutrients 2015, 7, 8577-8591. https://doi.org/10.3390/nu7105415
Collado MC, Santaella M, Mira-Pascual L, Martínez-Arias E, Khodayar-Pardo P, Ros G, Martínez-Costa C. Longitudinal Study of Cytokine Expression, Lipid Profile and Neuronal Growth Factors in Human Breast Milk from Term and Preterm Deliveries. Nutrients. 2015; 7(10):8577-8591. https://doi.org/10.3390/nu7105415
Chicago/Turabian StyleCollado, Maria Carmen, Marina Santaella, Laia Mira-Pascual, Elena Martínez-Arias, Parisá Khodayar-Pardo, Gaspar Ros, and Cecilia Martínez-Costa. 2015. "Longitudinal Study of Cytokine Expression, Lipid Profile and Neuronal Growth Factors in Human Breast Milk from Term and Preterm Deliveries" Nutrients 7, no. 10: 8577-8591. https://doi.org/10.3390/nu7105415
APA StyleCollado, M. C., Santaella, M., Mira-Pascual, L., Martínez-Arias, E., Khodayar-Pardo, P., Ros, G., & Martínez-Costa, C. (2015). Longitudinal Study of Cytokine Expression, Lipid Profile and Neuronal Growth Factors in Human Breast Milk from Term and Preterm Deliveries. Nutrients, 7(10), 8577-8591. https://doi.org/10.3390/nu7105415






