Mediterranean Diet and Cardiodiabesity: A Review
Abstract
:1. Introduction
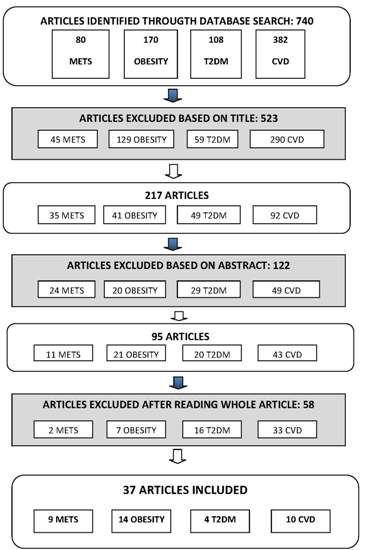
2. Materials and Methods
3. Results
3.1. Characteristics of Study Sample
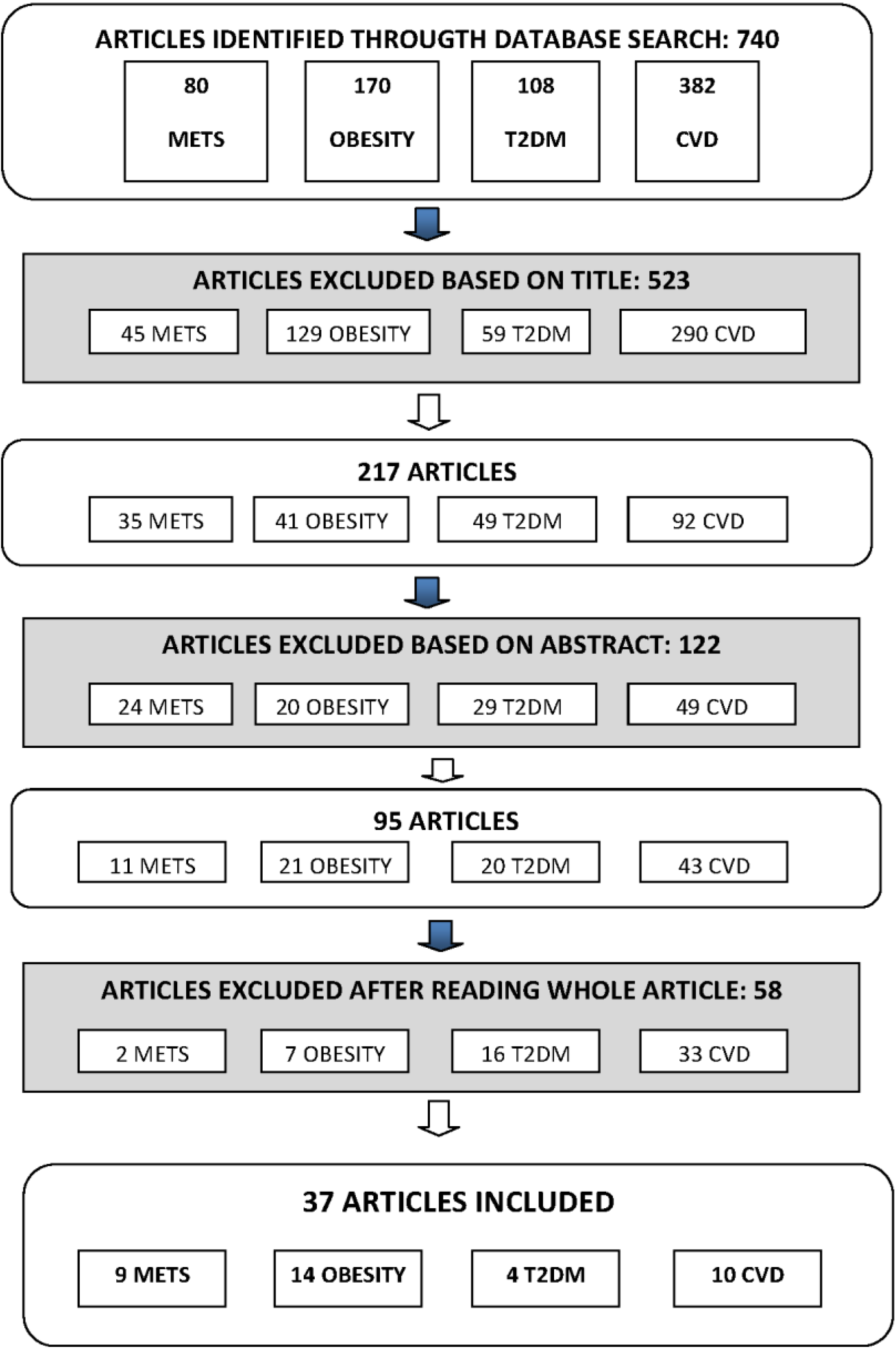
| Author/Year of Publication | Country | Gender | Age (Years) | Follow-Up (Years) | N | Components in the MD Index | Main Outcome | Results | Confounders |
|---|---|---|---|---|---|---|---|---|---|
| Panagiotakos et al. 2005 [47] | Greece | ♂1514 ♀1528 | ♂20–87; ♀20–89 | 1.25 | 3042 | Score based on dietary pyramid [48]: (d) non refined cereals and products, fruits, olive oil, dairy products, wine; (w) fish, poultry, potatoes, olives, legumes, nuts, eggs, sweets; (m) red meat | T2DM | With 10 P increase score: OR 0.79 (95% CI 0.65–0.94), p < 0.05); ARR 1.3% (5.6%–4.3%); RRR 23.2% [(5.6%–4.3%)/5.6%] | Age, sex, BMI, WC, hc, education, financial status, smoking, family history of T2DM |
| Martínez-Glez et al. 2008 [49] | Spain | Men and women | 20–90 | 4.4 | 13,380 | MDS [45]: (+) fatty acid ratio, legumes, grains, fruits, vegetables, fish, wine; (−) dairy products or meat | T2DM | OR 0.41 (95% CI 0.19–0.87) (score 3–6); OR 0.17 (0.04–0.75) (score 7–9). With 2 P increase score RR 0.65, (0.44–0.95), p = 0.04 | Sex, age, years of university education, TEI, BMI, PA, sedentary habits, smoking, family history of T2DM, and personal history of HT |
| Salas-Salvadó et al. 2011 [50] | Spain | Men and women | 55–80 | 4 | 418 | PREDIMED score 14 items [51]: olive oil, vegetables, fruit, fatty acid ratio, legumes, wine, fish, nuts; (−) sweets and carbonated beverages, red meat | T2DM | T2DM incidence MedDiet VOO 10.1% (95% CI 5.1–15.1), MedDiet nuts 11.0% (5.9–16.1), control group 17.9% (11.4–24.4). MedDiet VOO OR 0.49 (0.25–0.97) and nuts groups 0.48 (0.24–0.96). DM incidence was reduced 52% (27–86) | Age, sex, TEI, BMI, WC, PA, smoking status, fasting serum glucose, use of lipid lowering drugs, MedDiet score, and weight change during the study |
| Abiemo et al. 2013 [33] | USA | Men and women | 45–84 | 6 | 5390 | Score was created [45]: vegetables, whole grains, nuts, legumes, fruits, MUFA/SFA, red and processed meat, dairy products, fish, OH | T2DM | MeDiet score: lower baseline mean insulin levels, Q1: 5.8 (95% CI 5.6–6.0) μmol/L; Q5: 4.8 (95% CI 4.6–5.0) μmol/L; p < 0·0001. MedDiet not significantly related risk of T2DM p = 0.64 | Demographic, physiological and behavioral characteristics |
| Panagiotakos et al. 2004 [52] | Greece | Men and women | >18 | / | 2282 | Score created [53]: (d) cereals and products, vegetables, fruits, olive oil, dairy products; (w) fish, poultry, olives, legumes, nuts, potatoes, eggs, sweets; (m) red meat; (+m) wine and high MUFA/SFA ratio | MetS | OD 0.81 (95% CI, 0.68–0.976) | Age, sex, PA, education, smoking, BMI, diet in the past 12 months, TEI |
| Álvarez-León et al. 2006 [54] | Spain | Men and women | >18 | / | 578 | 10-Item score created [55]: cereal, fruits, vegetables, legumes, fish, nuts, alcohol intake from red wine, MUFA/SFA and dairy products | MetS | MD adherence not related to MetS prevalence; T2 OR 1.39 (95% CI, 0.75–2.59); T3 OR 1.37 (95% CI, 0.76–2.46) | Age, sex, PA, education, smoking, BMI, diet in the past 12 months, TEI |
| Tortosa et al. 2007 [56] | Spain | Men and women | 20–90 | 6 | 2563 | MDS [44]: (+) fatty acid ratio, legumes, grains, fruits, vegetables, fish, wine; (−) dairy products or meat. | MetS | MFP (95% CI) Age & sex adjusted score 3–5 OR 0.76 (0.40–1.45); score 6–9 OR 0.18 (0.06–0.56) p = 0.006; Multivariate-adjusted score 3–5 OR 0.80 (0.42–1.54); score 6–9 OR 0.20 (0.06–0.63) | Age, sex, PA, smoking, TEI |
| Salas-Salvadó et al. 2008 [57] | Spain | Men and women | ♂55–80; ♀60–80 | 1 | 1224 | PREDIMED score 14 items [51]: olive oil, vegetables, fruit, fatty acid ratio, legumes, wine, fish, nuts; (−) sweets and carbonated beverages, meat | MetS | MedDiet VOO group OR 1.3 (95% CI 0.8–2.1); MedDiet nuts group OR 1.7 (1.1–2.6) | Sex, age, baseline obesity status, and weight changes |
| Babio et al. 2009 [58] | Spain | Men and women | Mean 67 | / | 808 | 14-P questionnaire [59]: olive oil, vegetables, fruit, fatty acid ratio, legumes, wine, fish, nuts; (−) sweets and carbonated beverages, meat | MetS | ↑MD score OR 0.44 (95% CI, 0.27–0.70) | Age, sex, PA, smoking, TEI |
| Bibiloni et al. 2011 [43] | Spain | Men and women | 12–17 | / | 362 | Score created [45]: high MUFA/SFA ratio; (+m) OH, high legume, cereals and roots, fruit, vegetables, and fish consumption, and low meat and milk consumption | MetS | Q2 MD 6.9%; Q4 5.9%; Glucose levels 0.89 (95% CI 0.40–1.90), triglyceride levels 0.72 (95% CI 0.31–1.77) and HDL-cholesterol levels 0.43 (95% CI 0.19–0.99) | Sex, age, parental educational level, parental socioeconomic status and PA |
| Rumawas et al. 2009 [34] | EEUU | Men and women | Mean 54 | 7 | 2730 | Score created based on MSDPS [60]: whole-grain cereals, fruit, vegetables, dairy, wine, fish, poultry, olives/legumes/nuts, potatoes, eggs, sweets, meat, and olive oil | MetS | Q5 MSDPS ↓ incidence MetS (38.5% vs. 30.1%; p = 0.01) (95% CI) HOMA-IR Q5 3.16 (3.03–3.30) p = 0.02; fasting glucose 97.1 (96.3–98.0) p = 0.03; waist circumference 97.1 (96.7–97.6) p < 0.001; triglycerides 103 (99–107) p < 0.001; HDL-cholesterol 54.0 (53.1–55.0) p = 0.02 | Height, weight, BMI, age, sex, smoking, multivitamin use, ERT, PA, TEI |
| Paletas et al. 2010 [61] | Greece | Men and women | 44.4 ± 13.35 | / | 226 | MDS [45]: (+) fatty acid ratio, legumes, grains, fruits, vegetables, fish, wine; (−) dairy products or meat | MetS | MetS components ↑ non-MD: MetS prevalence 27.3% MedDiet group, 60.2% HF group, and HC group 69.2% p = 0.006 | Sex, BMI, TEI |
| Kesse-Guyot et al. 2013 [62] | France | Men and women | >18 | 6 | 3232 | MDS [45], MSDPS [60] and MED [63] | MetS | ↓ MetS: high MED OR 0.47 (95% CI 0.32–0.69) p = 0.001; high MDS OR 0.50 (95% CI 0.32–0.77) p = 0.03 | Age, gender, education level, smoking status, PA, TEI, antidiabetic, antihypertensive and lipid lowering medications |
| Goulet et al. 2003 [35] | Canada | Women | 30–65 | 0.25 | 77 | MD: (+) cereals, fruit, vegetables, legumes, nuts and seeds, fish, olive oil, wine; (−) dairy products, chicken, eggs, sweets, red meat | OW and OB | ↑ MD, ↓ BMI (mean week 0: 25.8 ± 3.9 kg/m2 to mean week 3 25.6 ± 3.8 kg/m2) p < 0.01; adh less weight (m0: 67.7 ± 11.9 kg to m3: 67.3 ± 11 kg) p < 0.01 | |
| Schröder et al. 2004 [64] | Spain | ♂1403 ♀1468 | 25–74 | 2 | 3162 | MDS [44]: (+) cereals, vegetables, fruit, legumes, nuts, fish, red wine; (−) meat, high fat dairy products | OB | ↑ MD, ↓ OB OR 0.61 p = 0.01; With 5-P MD, less BMI ♂ −0.43 kg/m2 p = 0.030; ♀ −0.68 kg/m2 p = 0.007 | Age, TEI, educational level, smoking, LTPA, smoking and alcohol consumption |
| Trichopoulou et al. 2005 [65] | Greece | ♂9612 ♀13,985 | 20–86 | / | 23,597 | MDS [44]: (+) cereals, vegetables, fruit and nuts, legumes, fish, MUFA/SFA; (+m) OH; (−) meat and dairy products | OB | With 2 P increase score controlling TEI ♂ OR 0.08 (95% CI −0.03–0.20), ♀ OR −0.06 (95% CI −0.16–0.04); With 2 P increase score without controlling TEI ♂ OR 0.21 (95% CI 0.10–0.32), ♀ OR 0.05 (95% CI −0.04–0.15) | Age, years of schooling, smoking, PA, TEI |
| Shubair et al. 2005 [36] | Canada | ♂265 ♀494 | 18–65 | / | 759 | MDP score created: (+) fruit, vegetables, olive oil and garlic, fish and shellfish; (−) meats and poultry, high SFA, foods high in added sugar and low nutrients | OB | ↑ MD, ↓ BMI (β-coefficient −0.186) p = 0.027 | Sex, education, income and marital status |
| Panagiotakos et al. 2006 [66] | Greece | ♂1514 ♀1528 | 18–89 | / | 3042 | MDS [48]: wholegrain cereals, vegetables, fruit, legumes, fish, olive oil, dairy products, chicken, nuts and seeds, olives, potatoes, eggs, sweets, meat, MUFA/SFA | OW and OB | With 5 P increased score OR 0.49 (95% CI 0.42–0.56) (OB & OW); ↑ MD, ↓ BMI (−4 kg) p = 0.001 | Age, sex, PA, metabolism, educational level, smoking status |
| Mendez et al. 2006 [67] | Spain | ♂10,589 ♀17,238 | 29–65 | 3.3 | 27,827 | MDS [65]: (+) cereals, vegetables, fruit, legumes, fish, MUFA/SFA; (+m) OH; (−) meat | OB | S OW subjects ♀ OR 0.73 (95% CI 0.57–0.93), ♂ OR 0.71 (95% CI 0.55–0.93); NS OW subjects ♀ OR 0.99 (95% CI 0.78–1.25), ♂ OR 1.11 (95% CI 0.81–1.52) | Age, special diets related to obesity or related disorders, categorical activity index, education, center, height, parity, smoking status, winter season, follow-up time, health status and changes in lifestyle or health during follow-up |
| Andreoli et al. 2008 [40] | Italy | Women | 25–70 | 0.33 | 47 | MD moderately hypo-caloric: (+) vegetables, fruit, pasta, bread, legumes, fish, olive oil, red wine, meat, dairy products & PA program | Cardiovascular disease risk factors in obese women | ↑ MD, ↓ weight (m0 80.4 ± 15.8 kg to m4 75.2 ± 14.7 kg) p < 0.001; ↑ MD, ↓ BMI (m0 30.7 ± 6.0 kg to m4 28.7 ± 5.6 kg) p < 0.001 | |
| Panagiotakos et al. 2007 [68] | Cyprus | ♂53 ♀97 | 65–100 | / | 150 | MDS: wholegrain cereals, vegetables, fruit, legumes, fish, olive oil, dairy products, chicken, nuts, seeds, olives, potatoes, eggs, sweets, meat | OB | With 10 P increased score OR 0.83 (95% CI −0.25–0.12) p < 0.001 (having one additional risk factor); With 10 P increased score 12% lower OR OB p = 0.001 | Age, sex, smoking habits, PA |
| Romaguera et al. 2009 [37] | 10 European countries | Men and women | 25–70 | 8 | 497,308 | mMDS [44]: (+) vegetables, legumes, fruits, nuts, cereals, fish and seafood, MUFA/SFA; (+m) OH; (−) meat, dairy products | OB | ↑ MD, ↓ WC, for a given BMI ♂ 20.09 (95% CI 20.14–20.04), ♀ 20.06 (95% CI 20.10–20.01); Northern European countries ♂ 20.20 (95% CI 20.23–20.17), ♀ 20.17 (95% CI 20.21–20.13) | Age, educational level (categorical), PA, smoking status, height, menopausal status |
| Beunza et al. 2010 [69] | Spain | Men and women | Mean 38 | 5.7 ± 2.2 | 10,376 | MDS [45] | OW and OB | ↑ MD, ↓ weight 20.059 kg/year 95% CI 20.111–20.008 kg/year, p = 0.02; lowest risk weight gain 4-year 0.76 (95% CI 0.64–0.90) | Age, sex, baseline BMI, PA, sedentary behaviors, smoking, between-meals snacking, TEI |
| Romaguera et al. 2010 [70] | 10 European countries | Men and women | 25–70 | 5 | 373,803 | rMED [71]: (+) vegetables, legumes, fruit, nuts, cereals, fish and seafood, olive oil; (+m) OH; (−) meat, dairy products | OW & OB | ↑ rMED, ↓ weight gain 5-year 20.16 kg (95% CI 20.24, 20.07 kg); less likely to OW or OB 10% (95% CI 4%–18%) | Sex, age, baseline BMI, follow-up, educational level, PA, smoking status, menopausal status, TEI, misreporting of TEI |
| Lazarou et al. 2010 [41] | Cyprus | Men and women | Mean 10.7 ± 0.98 | / | 1140 | KIDMED [72] | OB | ↑ KIDMED, ↓ likely to OW/OB 80% (95% CI 0.041–0.976) | Age, gender, parental obesity status, parental educational level, dietary beliefs and behaviors |
| Farajian et al. 2011 [42] | Greece | Children | 10–12 | / | 4786 | KIDMED [72] | OW & OB | NS NW (score mean 3.70 ± 2.26) & OW/OB (score mean 3.62 ± 2.26), t-test 1.08, p = 0.28 | Age, BMI, waist, body fat mass |
| Martínez-Glez et al. 2012 [73] | Spain | Men and women; 57%♀ | 55–80 | / | 7447 | PREDIMED [51] | OB | With 2 P increase score ♀ WHtR OR −0.65 (95% CI −0.87–0.48), BMI OR −0.37 (95% CI −0.50–0.24), WC OR −0.92 (−1.26–0.58); ♂ WHtR OR −0.58 (95% CI −0.79–0.38), BMI OR −0.25 (95% CI −0.37–0.13), WC OR −0.93 (95% CI −1.29–0.58) | Age, smoking, diabetes status, HT status, PA, educational level, marital status, center, TEI |
| Trichopoulou et al. 2003 [45] | Greece | Men and women | 20–86 | Median 3.7 | 22,043 | 9 Unit MDS [44]: (+) vegetables, legumes, fruits, nuts, cereals, fish; (+m) OH; (−) meat, poultry, dairy products | Fatal CHD events | ↑ MD, ↓ CHD mortality HR 0.67 (95% CI 0.47–0.94) | Sex, age, years of education, smoking status, WtHR, BMI, TEI, energy-expenditure score |
| Knoops et al. 2004 [39] | 11 European Countries | Men and women | 70–90 | 12 | 2339 | mMD [45]: MUFA/SFA, legumes, nuts, seeds, grains, fruit, vegetables and potatoes, meat, dairy products, fish | All-cause and cause specific mortality | ↑ MD score, ↓ mortality: all causes 0.77 (0.68–0.88), CHD 0.61 (0.43–0.88), CVD 0.71 (0.58–0.88) (95% CI) | Other diet and lifestyle factors, sex, age at baseline, BMI, and study population (SENECA vs. FINE). |
| Panagiotakos et al. 2007 [74] | Greece | Men and women | 18–89 | 3042 | Own score [48]: (+) non-refined cereals, fruit, vegetables, legumes, potatoes, fish, olive oil; (−) meat, poultry, full fat dairy products; (+m) OH | CVD risk factors | With 10 P increase score, 10-year follow up 4% lower CHD risk (±0.1%, p < 0.001); ↑ MD, ↓ 10-year follow up CHD risk (rho −0.7, p < 0.001) | No assessment forpotential confounding since all analyses were unadjusted | |
| Buckland et al. 2009 [71] | Spain | Men and women | 29–69 | Mean 10.4 | 41,078 | r-MED [45]: (+) fruits, nuts, seeds, vegetables (no potatoes), legumes, cereals, fresh fish, olive oil; (−) meat, dairy products | CHD events | ↑ MD score, ↓ CHD risk, Z ¼ 0.60 (95% CI 0.47–0.77); with 1 P increase score, 6% reduced risk CHD (95% CI 0.91–0.97) | Age, BMI, educational level, smoking status, PA, TEI, T2DM, hyperlipidemia, HT |
| Fung et al. 2009 [38] | U.K. | Women | 38–63 | 20 | 74,886 | aMED [45]: (+) vegetables (excluding potatoes), fruits, nuts, whole grain, legumes, fish, MUFA/SFA; (+m) OH; (−) meat | Risk of stroke | Q5 ↓, risk CHD RR 0.71 (95% CI 0.62–0.82), p = 0.0001 for CHD; ↓ risk stroke RR 0.87 (95% CI 0.73–1.02), p = 0.03; Q5, ↓ CVD mortality ♀ RR 0.61 (95% CI 0.49–0.76), p = 0.0001 | Age, smoking, BMI, menopausal status, postmenopausal hormone use, TEI, multivitamin intake, OH intake, family history, PA, aspirin |
| Gardener et al. 2011 [32] | EEUU (NY) | Men and women | 69 ± 10 | 9 | 2568 | Own MD Score [45]: dairy, meat, fruit, vegetables (excluding potatoes), legumes, cereals (refined and whole), fish | CHD events | Q5 score (1) HR 0.75 (95% CI 0.56–0.99) p = 0.04; (2) HR 0.80 (0.60–1.06) p = 0.10. With 1P increase score (1) HR 0.94 (0.89–1.00), (2) HR 0.95 (0.90–1.01) | (1) Age, sex, ethnicity, education, PA, TEI, smoking; (2) model 1 and T2DM, HT, hypercholesterolemia, history self-reported cardiac disease |
| Martínez-Glez et al. 2011 [75] | Spain | Men and women | Mean 38 | 4.9 | 13,609 | 9-Point score [45]: (+) MUFA/SFA, legumes, cereals, fruits, nuts, vegetables, fish; (+m) OH; (−) total dairy products, meat | CHD events | ↑ MD, ↓ CVD risk HR 0.41 (95% CI 0.18–0.95); with 2 P increase score, total CVD HR 0.80 (95% CI 0.62–1.02), CHD HR 0.74 (95% CI 0.55–0.99) | Age, sex, TEI, family history CHD, smoking, PA, baseline BMI, HT or use of medication, aspirin, baseline T2DM, baseline dyslipidaemia |
| Menotti et al. 2012 [76] | Italy | Men and women | 45–64 | 5 | 1139 | MAI [77]: (+) cereals, legumes, potatoes, vegetables, fresh dry fruit, fish, wine, virgin olive oil; (−) milk, cheese, meat, eggs, animal fats and margarines, sweet beverages, sugar | CHD events | 20-Year follow-up lnMAI HR 0.74 (95% CI 0.56–0.98) p =0.0338; 40-year follow-up lnMAI HR 0.79 (95% CI 0.65–0.96) p = 0.0168 | Age, cigarette smoking, systolic blood pressure, serum cholesterol, PA, BMI |
| Guallar-Castrillón et al. 2012 [78] | Spain | Men and women | 29–69 | 8–12; median 11 | 40,757 | Own score | CHD events | MD score HR Q2 0.77 (95% CI 0.61–0.98), Q3 0.64 (95% CI 0.50–0.83), Q4 0.56 (95% CI 0.43–0.73), Q5 0.73 (95% CI 0.57–0.94) | Age, sex, BMI, WC, educational level, smoking, PA at work, home and leisure time, T2DM, HT, hypercholesterolemia, cancer, oral contraceptives, menopausal status, hormone replacement therapy, TEI |
| Estruch et al. 2013 [46] | Spain | Men and women | 55–80 | 4.8 | 7447 | PREDIMED [51] | CHD events | MD VOO HR 0.70 (95% CI 0.54–0.92); MD nuts HR 0.72 (95% CI 0.54–0.96) | Sex, age, BMI, cardiovascular-risk-factor status, baseline adherence MedDiet |
3.2. Association between the MedDiet and Cardiodiabesity
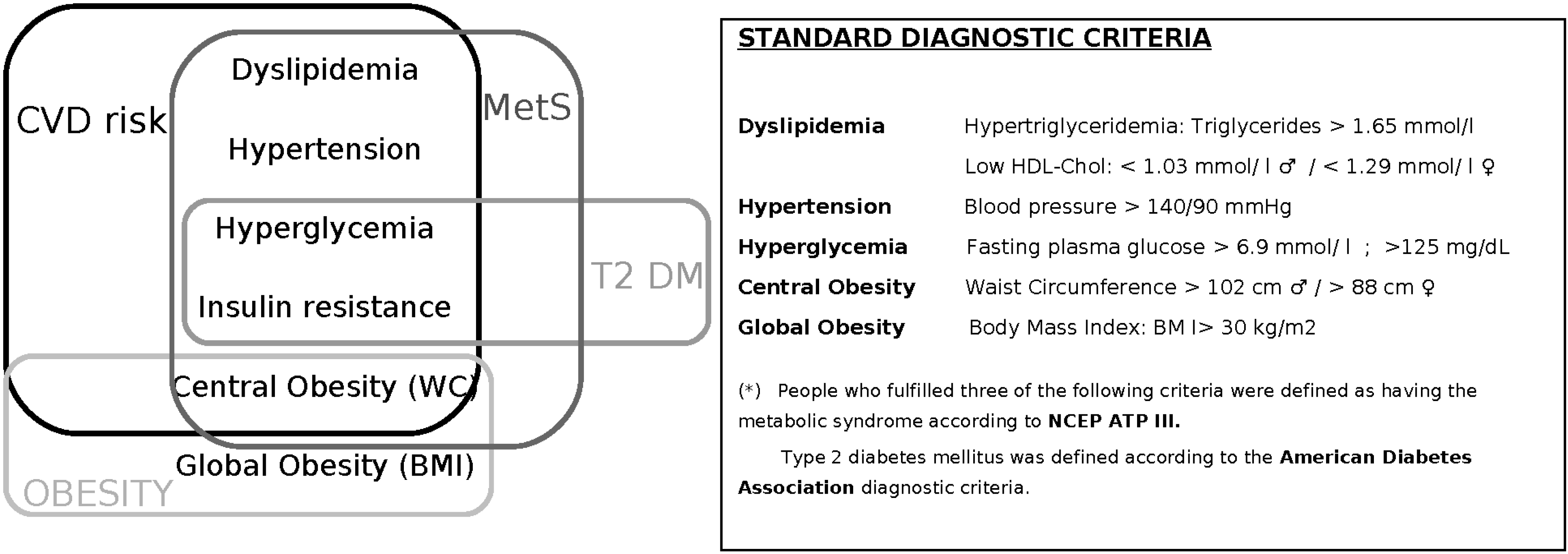
4. Discussion
- (i)
- To develop simple, objective and useful tools to determine the degree of patient adherence to the MedDiet pattern for its application in the clinical setting and general practice.
- (ii)
- To establish consensual recommendations about the MedDiet pattern and psychological approaches to eating habits and lifestyle improvements. Moreover, it is important to foster knowledge of and accessibility to this dietary pattern to health professionals, particularly in primary care where preventive medicine is of greatest, albeit not exclusive, significance. Multidisciplinary approaches and strategies are needed to increase compliance to treatments and even dietary and lifestyle interventions [96].
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Shape up America! Available online: http://www.shapeup.org/ (accessed on 19 June 2014).
- Robertson, D. Cardiodiabetes—Is a joint approach the way forward? Br. J. Cardiol. 2008, 15, S8–S10. [Google Scholar]
- World Health Organization Media Centre (WHO). Diabetes. Available online: http://www.who.int/mediacentre/factsheets/fs312/es/ (accessed on 12 January 2014).
- World Health Organization Media Centre (WHO). 10 Facts on Obesity. Available online: http://www.who.int/features/factfiles/obesity/es/ (accessed on 12 January 2014).
- World Health Organization Media Centre (WHO). Cardiovascular Diseases. Available online: http://www.euro.who.int/en/health-topics/noncommunicable-diseases/cardiovascular-diseases/cardiovascular-diseases2 (accessed on 12 January 2014).
- Rydén, L.; Grant, P.J.; Anker, S.D.; Berne, C.; Cosentino, F.; Danchin, N.; Deaton, C.; Escaned, J.; Hammes, H.P.; Huikuri, H.; et al. ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD: The Task Force on diabetes, pre-diabetes, and cardiovascular diseases of the European Society of Cardiology (ESC) and developed in collaboration with the European Association for the Study of Diabetes (EASD). Eur. Heart J. 2013, 34, 3035–3087. [Google Scholar]
- National Institute for Health and Clinical Excellence (NICE). Clinical Guideline 67. Lipid Modification. Cardiovascular Risk Assessment and the Modification of Blood Lipids for the Primary and Secondary Prevention of Cardiovascular Disease. May 2008. Available online: http://www.nice.org.uk/guidance/cg67/chapter/guidance (accessed on 25 January 2014).
- National Institute for Health and Clinical Excellence (NICE). Clinical Guideline 66. Type 2 Diabetes. May 2008. Available online: http://www.nice.org.uk/guidance/cg87 (accessed on 25 January 2014).
- Grundy, S.M. Hypertriglyceridemia, atherogenic dyslipidemia and the metabolic syndrome. Am. J. Cardiol. 1998, 81, 18B–25B. [Google Scholar]
- Grundy, S.M. Hypertriglyceridemia, insulin resistance and the metabolic syndrome. Am. J. Cardiol. 1999, 83, 25F–29F. [Google Scholar] [CrossRef] [PubMed]
- Hajer, G.H.; van Haeften, T.W.; Visseren, F. Adipose tissue dysfunction in obesity, diabetes, and vascular diseases. Eur. Heart J. 2008, 29, 2959–2971. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Gonzalez, M.A.; Bes-Rastrollo, M.; Serra-Majem, L.; Lairon, D.; Estruch, R.; Trichopoulou, A. Mediterranean food pattern and the primary prevention of chronic disease: Recent developments. Nutr. Rev. 2009, 67, S111–S116. [Google Scholar]
- Serra-Majem, L.; Roman, B.; Estruch, R. Scientific evidence of interventions using the Mediterranean diet: A systematic review. Nutr. Rev. 2006, 64, S27–S47. [Google Scholar]
- Sofi, F.; Cesari, F.; Abbate, R.; Gensini, G.F.; Casini, A. Adherence to Mediterranean diet and health status: Meta-Analysis. BMJ 2008, 337, a1344. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Abbate, R.; Gensini, G.F.; Casini, A. Accruing evidence on benefits of adherence to the Mediterranean diet on health: An updated systematic review and meta-analysis. Am. J. Clin. Nutr. 2010, 92, 1189–1196. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Macchi, C.; Abbate, R.; Gensini, G.F.; Casini, A. Mediterranean diet and health status: An updated meta-analysis and a proposal for a literature-based adherence score. Public Health Nutr. 2013, 29, 1–14. [Google Scholar] [CrossRef]
- Serra-Majem, L.; Bes-Rastrollo, M.; Román, B.; Pfrimer, K.; Sánchez-Villegas, A.; Martínez-González, M.A. Dietary patterns and nutritional adequacy in a Mediterranean country. Br. J. Nutr. 2009, 101, S21–S28. [Google Scholar]
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G.; et al. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar]
- Consenso FESNAD-SEEDO. Recomendaciones nutricionales basadas en la evidencia para la prevención y el tratamiento del sobrepeso y la obesidad en adultos. Rev. Esp. Obes. 2011, 10, 1–78. [Google Scholar]
- Kant, A.K. Dietary patterns and health outcomes. J. Am. Diet. Assoc. 2004, 104, 615–635. [Google Scholar] [CrossRef]
- Bach-Faig, A.; Serra-Majem, L.; Carrasco, J.L.; Roman, B.; Ngo, J.; Bertomeu, I.; Obrador, B. The use of indexes evaluating the adherence to the Mediterranean diet in epidemiological studies: A review. Public Health Nutr. 2006, 9, 132–146. [Google Scholar]
- UNESCO. Representative List of the Intangible Cultural Heritage of Humanity-2010. Available online: http://www.unesco.org/culture/ich/en/RL/00394 (accessed on 28 January 2014).
- Sáez-Almendros, S.; Obrador, B.; Bach-Faig, A.; Serra-Majem, L. Environmental footprints of Mediterranean versus Western dietary patterns: Beyond the health benefits of the Mediterranean diet. Environ. Health 2013, 12, 118. [Google Scholar] [CrossRef]
- Saulle, R.; Semyonov, L.; la Torre, G. Cost and cost-effectiveness of the Mediterranean diet: Results of a systematic review. Nutrients 2013, 5, 4566–4586. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, R.; Bach-Faig, A.; Raido Quintana, B.; Buckland, G.; vaz de Almeida, M.D.; Serra-Majem, L. Worldwide variation of adherence to the Mediterranean diet, in 1961–1965 and 2000–2003. Public Health Nutr. 2009, 12, 1676–1684. [Google Scholar]
- Serra-Majem, L.; Helsing, E. Changing patterns of fat intake in Mediterranean countries. Eur. J. Clin. Nutr. 1993, 47, S13–S20. [Google Scholar]
- Ferro-Luzzi, A.; Sette, S. The Mediterranean diet: An attempt to define its present and past composition. Eur. J. Clin. Nutr. 1989, 43, 13–29. [Google Scholar]
- Panagiotakos, D.B.; Pitsavos, C.; Polychronopoulos, E.; Chrysohoou, C.; Zampelas, A.; Trichopoulou, A. Can a Mediterranean diet moderate the development and clinical progression of coronary heart disease? A systematic review. Med. Sci. Monit. 2004, 10, RA193–RA198. [Google Scholar] [PubMed]
- De Lorgeril, M.; Salen, P. The Mediterranean-style diet for the prevention of cardiovascular diseases. Public Health Nutr. 2006, 9, 118–123. [Google Scholar]
- Buckland, G.; Bach, A.; Serra-Majem, L. Obesity and the Mediterranean diet: A systematic review of observational and intervention studies. Obes. Rev. 2008, 9, 582–593. [Google Scholar] [CrossRef] [PubMed]
- Nordmann, A.J.; Suter-Zimmermann, K.; Bucher, H.C.; Shai, I.; Tuttle, K.H.; Estruch, R.; Briel, M. Meta-Analysis comparing Mediterranean to low-fat diets for modification of cardiovascular risk factors. Am. J. Med. 2011, 124, 841–851. [Google Scholar] [CrossRef] [PubMed]
- Gardener, H.; Wright, C.B.; Gu, Y.; Demmer, R.T.; Boden-Albala, B.; Elkind, M.S.; Sacco, R.L.; Scarmeas, N. Mediterranean-style diet and risk of ischemic stroke, myocardial infarction, and vascular death: The Northern Manhattan Study. Am. J. Clin. Nutr. 2011, 94, 1458–1464. [Google Scholar] [CrossRef] [PubMed]
- Abiemo, E.E.; Alonso, A.; Nettleton, J.A.; Steffen, L.M.; Bertoni, A.G.; Jain, A.; Lutsey, P.L. Relationships of the Mediterranean dietary pattern with insulin resistance and diabetes incidence in the Multi-Ethnic Study of Atherosclerosis (MESA). Br. J. Nutr. 2013, 109, 1490–1497. [Google Scholar] [CrossRef] [PubMed]
- Rumawas, M.E.; Meigs, J.B.; Dwyer, J.T.; McKeown, N.M.; Jacques, P.F. Mediterranean-style dietary pattern, reduced risk of metabolic syndrome traits, and incidence in the Framingham Offspring Cohort. Am. J. Clin. Nutr. 2009, 90, 1608–1614. [Google Scholar] [CrossRef] [PubMed]
- Goulet, J.; Lamarche, B.; Nadeau, G.; Lemieux, S. Effect of a nutritional intervention promoting the Mediterranean food pattern on plasma lipids, lipoproteins and body weight in healthy French-Canadian women. Atherosclerosis 2003, 170, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Shubair, M.M.; McColl, R.S.; Hanning, R.M. Mediterranean dietary components and body mass index in adults: The peel nutrition and heart health survey. Chronic Dis. Can. 2005, 26, 43–51. [Google Scholar] [PubMed]
- Romaguera, D.; Norat, T.; Mouw, T.; May, A.M.; Bamia, C.; Slimani, N.; Travier, N.; Besson, H.; Luan, J.; Wareham, N. Adherence to the Mediterranean diet is associated with lower abdominal adiposity in European men and women. J. Nutr. 2009, 139, 1728–1737. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Rexrode, K.M.; Mantzoros, C.S.; Manson, J.E.; Willett, W.C.; Hu, F.B. Mediterranean diet and incidence of and mortality from coronary heart disease and stroke in women. Circulation 2009, 119, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Knoops, K.T.; de Groot, L.C.; Kromhout, D.; Perrin, A.E.; Moreiras-Varela, O.; Menotti, A.; van Staveren, W.A. Mediterranean diet, lifestyle factors, and 10-year mortality in elderly European men and women: The HALE project. JAMA 2004, 292, 1433–1439. [Google Scholar] [CrossRef] [PubMed]
- Andreoli, A.; Lauro, S.; di Daniele, N.; Sorge, R.; Celi, M.; Volpe, S.L. Effect of a moderately hypoenergetic Mediterranean diet and exercise program on body cell mass and cardiovascular risk factors in obese women. Eur. J. Clin. Nutr. 2008, 62, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Lazarou, C.; Panagiotakos, D.B.; Matalas, A.L. Physical activity mediates the protective effect of the Mediterranean diet on children’s obesity status: The CYKIDS study. Nutrition 2010, 26, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Farajian, P.; Risvas, G.; Karasouli, K.; Pounis, G.D.; Kastorini, C.M.; Panagiotakos, D.B.; Zampelas, A. Very high childhood obesity prevalence and low adherence rates to the Mediterranean diet in Greek children: The GRECO study. Atherosclerosis 2011, 217, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Bibiloni, M.; Martínez, E.; Llull, R.; Maffiotte, E.; Riesco, M.; Llompart, I.; Pons, A.; Tur, J.A. Metabolic syndrome in adolescents in the Balearic Islands, a Mediterranean region. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. Br. Med. J. 1995, 311, 1457–1460. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary prevention of cardiovascular disease with a Mediterranean diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar]
- Panagiotakos, D.B.; Pitsavos, C.; Chrysohoou, C.; Stefanadis, C. The epidemiology of Type 2 diabetes mellitus in Greek adults: The ATTICA study. Diabet. Med. 2005, 22, 1581–1588. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61, 1402S–1406S. [Google Scholar] [PubMed]
- Martínez-González, M.A.; de la Fuente-Arrillaga, C.; Nunez-Cordoba, J.M.; Basterra-Gortari, F.J.; Beunza, J.J.; Vazquez, Z.; Benito, S.; Tortosa, A.; Bes-Rastrollo, M. Adherence to Mediterranean diet and risk of developing diabetes: Prospective cohort study. BMJ 2008, 336, 1348–1351. [Google Scholar]
- Salas-Salvadó, J.; Bulló, M.; Babio, N.; Martínez-González, M.Á.; Ibarrola-Jurado, N.; Basora, J.; Estruch, R.; Covas, M.I.; Corella, D.; Arós, F.; et al. PREDIMED Study Investigators. Reduction in the incidence of type 2 diabetes with the Mediterranean diet: Results of the PREDIMED-Reus nutrition intervention randomized trial. Diabetes Care 2011, 34, 14–19. [Google Scholar]
- Estruch, R.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Ruiz-Gutiérrez, V.; Covas, M.I.; Fiol, M.; Gómez-Gracia, E.; López-Sabater, M.C.; Vinyoles, E.; et al. Effects of a Mediterranean-style diet on cardiovascular risk factors: A randomized trial. Ann. Intern. Med. 2006, 145, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Pitsavos, C.; Chrysohoou, C.; Skoumas, J.; Tousoulis, D.; Toutouza, M.; Toutouzas, P.; Stefanadis, C. Impact of lifestyle habits on the prevalence of the metabolic syndrome among Greek adults from the ATTICA study. Am. Heart J. 2004, 147, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A. From research to education: The Greek experience. Nutrition 2000, 16, 528–531. [Google Scholar] [CrossRef] [PubMed]
- Alvarez León, E.; Henríquez, P.; Serra-Majem, L. Mediterranean diet and metabolic syndrome: A cross-sectional study in the Canary Islands. Public Health Nutr. 2006, 9, 1089–1098. [Google Scholar]
- Henríquez, P.; Doreste, J.; Díaz-Cremades, J.; López-Blanco, F.; Alvarez-León, E.; Serra-Majem, L. Folate status of adults living in the Canary Islands (Spain). Int. J. Vit. Nutr. Res. 2004, 74, 187–192. [Google Scholar]
- Tortosa, A.; Bes-Rastrollo, M.; Sanchez-Villegas, A.; Basterra-Gortari, F.J.; Nuñez-Cordoba, J.M.; Martinez-Gonzalez, M.A. Mediterranean diet inversely associated with the incidence of metabolic syndrome: The SUN prospective cohort. Diabetes Care 2007, 30, 2957–2959. [Google Scholar] [CrossRef] [PubMed]
- Salas-Salvadó, J.; Fernández-Ballart, J.; Ros, E.; Martínez-González, M.A.; Fitó, M.; Estruch, R.; Corella, D.; Fiol, M.; Gómez-Gracia, E.; Arós, F.; et al. Effect of a Mediterranean diet supplemented with nuts on metabolic syndrome status: One-year results of the PREDIMED randomized trial. Arch. Intern. Med. 2008, 168, 2449–2458. [Google Scholar]
- Babio, N.; Bulló, M.; Basora, J.; Martínez-González, M.A.; Fernández-Ballart, J.; Márquez-Sandoval, F.; Molina, C.; Salas-Salvadó, J. Adherence to the Mediterranean diet and risk of metabolic syndrome and its components. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.A.; Fernández-Jarne, E.; Serrano-Martínez, M.; Wright, M.; Gomez-Gracia, E. Development of a short dietary intake questionnaire for the quantitative estimation of adherence to a cardioprotective Mediterranean diet. Eur. J. Clin. Nutr. 2004, 58, 1550–1552. [Google Scholar]
- Rumawas, M.E.; Dwyer, J.Y.; Mckeown, N.M.; Meigs, J.B.; Rogers, G.; Jacques, P.F. The development of the Mediterranean-style dietary pattern score and its application to the American diet in the Framingham Offspring Cohort. J. Nutr. 2009, 139, 1150–1156. [Google Scholar] [CrossRef] [PubMed]
- Paletas, K.; Athanasiadou, E.; Sarigianni, M.; Paschos, P.; Kalogirou, A.; Hassapidou, M.; Tsapas, A. The protective role of the Mediterranean diet on the prevalence of metabolic syndrome in a population of Greek obese subjects. J. Am. Coll. Nutr. 2010, 29, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Ahluwalia, N.; Lassale, C.; Hercberg, S.; Fezeu, L.; Lairon, D. Adherence to Mediterranean diet reduces the risk of metabolic syndrome: A 6-year prospective study. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 677–683. [Google Scholar]
- Issa, C.; Darmon, N.; Salameh, P.; Maillot, M.; Batal, M.; Lairon, D. Mediterranean diet pattern with low consumption of liquid sweets and refined cereals is negatively associated with adiposity in adults from rural Lebanon. Int. J. Obes. (Lond.) 2011, 35, 251–258. [Google Scholar] [CrossRef]
- Schroder, H.; Marrugat, J.; Vila, J.; Covas, M.I.; Elosua, R. Adherence to the traditional Mediterranean diet is inversely associated with body mass index and obesity in a Spanish population. J. Nutr. 2004, 134, 3355–3361. [Google Scholar] [PubMed]
- Trichopoulou, A.; Naska, A.; Orfanos, P.; Trichopoulos, D. Mediterranean diet in relation to body mass index and waist-to-hip ratio: The Greek European Prospective Investigation into Cancer and Nutrition Study. Am. J. Clin. Nutr. 2005, 82, 935–940. [Google Scholar] [PubMed]
- Panagiotakos, D.B.; Chrysohoou, C.; Pitsavos, C.; Stefanadis, C. Association between the prevalence of obesity and adherence to the Mediterranean diet: The ATTICA study. Nutrition 2006, 22, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Mendez, M.A.; Popkin, B.M.; Jakszyn, P.; Berenguer, A.; Tormo, M.J.; Sanchéz, M.J.; Quirós, J.R.; Pera, G.; Navarro, C.; Martinez, C.; et al. Adherence to a Mediterranean diet is associated with reduced 3-year incidence of obesity. J. Nutr. 2006, 136, 2934–2938. [Google Scholar] [PubMed]
- Panagiotakos, D.B.; Polystipioti, A.; Papairakleous, N.; Polychronopoulos, E. Long-Term adoption of a Mediterranean diet is associated with a better health status in elderly people; a cross-sectional survey in Cyprus. Asia Pac. J. Clin. Nutr. 2007, 16, 331–337. [Google Scholar] [PubMed]
- Beunza, J.J.; Toledo, E.; Hu, F.B.; Bes-Rastrollo, M.; Serrano-Martínez, M.; Sánchez-Villegas, A.; Martínez, J.A.; Martínez-González, M.A. Adherence to the Mediterranean diet, long-term weight change, and incident overweight or obesity: The Seguimiento Universidad de Navarra (SUN) cohort. Am. J. Clin. Nutr. 2010, 92, 1484–1493. [Google Scholar] [CrossRef] [PubMed]
- Romaguera, D.; Norat, T.; Vergnaud, A.C.; Mouw, T.; May, A.M.; Agudo, A.; Buckland, G.; Slimani, N.; Rinaldi, S.; Couto, E.; et al. Mediterranean dietary patterns and prospective weight change in participants of the EPIC-PANACEA project. Am. J. Clin. Nutr. 2010, 92, 912–921. [Google Scholar] [CrossRef] [PubMed]
- Buckland, G.; González, C.A.; Agudo, A.; Vilardell, M.; Berenguer, A.; Amiano, P.; Ardanaz, E.; Arriola, L.; Barricarte, A.; Basterretxea, M.; et al. Adherence to the Mediterranean diet and risk of coronary heart disease in the Spanish EPIC Cohort Study. Am. J. Epidemiol. 2009, 170, 1518–1529. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Ribas, L.; Ngo, J.; Ortega, R.M.; García, A.; Pérez-Rodrigo, C.; Aranceta, J. Food, youth and the Mediterranean diet in Spain. Development of KIDMED, Mediterranean Diet Quality Index in children and adolescents. Public Health Nutr. 2004, 7, 931–935. [Google Scholar]
- Martínez-González, M.A.; García-Arellano, A.; Toledo, E.; Salas-Salvadó, J.; Buil-Cosiales, P.; Corella, D.; Covas, M.; Schröder, H.; Arós, F.; Gómez-Gracia, E.; et al. A 14-item Mediterranean diet assessment tool and obesity indexes among high-risk subjects: The PREDIMED trial. PLoS One 2012, 7, e43134. [Google Scholar] [CrossRef]
- Panagiotakos, D.B.; Pitsavos, C.; Arvaniti, F.; Stefanadis, C. Adherence to the Mediterrranean food pattern predicts the prevalence of hypertension, hypercholesterolemia, diabetes and obesity, among healthy adults; the accuracy of the MedDietScore. Prev. Med. 2007, 44, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.A.; García-López, M.; Bes-Rastrollo, M.; Toledo, E.; Martínez-Lapiscina, E.H.; Delgado-Rodriguez, M.; Vazquez, Z.; Benito, S.; Beunza, J.J. Mediterranean diet and the incidence of cardiovascular disease: A Spanish cohort. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 237–244. [Google Scholar]
- Menotti, A.; Alberti-Fidanza, A.; Fidanza, F. The association of the Mediterranean Adequacy Index with fatal coronary events in an Italian middle-aged male population followed for 40 years. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Fidanza, F.; Alberti, A.; Lanti, M.; Menotti, A. Mediterranean adequacy index: Correlation with 25-year mortality from coronary heart disease in the Seven Countries Study. Nutr. Metab. Cardiovasc. Dis. 2004, 14, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Guallar-Castillón, P.; Rodríguez-Artalejo, F.; Tormo, M.J.; Sánchez, M.J.; Rodríguez, L.; Quirós, J.R.; Navarro, C.; Molina, E.; Martínez, C.; Marín, P.; et al. Major dietary patterns and risk of coronary heart disease in middle-aged persons from a Mediterranean country: The EPIC-Spain cohort study. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 192–199. [Google Scholar]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.H.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith, S.C., Jr.; et al. Diagnosis and management of the metabolic syndrome: An american heart association/national heart, lung, and blood institute scientific statement. Circulation 2005, 112, 2735–2752. [Google Scholar] [CrossRef] [PubMed]
- Kastorini, C.M.; Milionis, H.J.; Esposito, K.; Giugliano, D.; Goudevenos, J.A.; Panagiotakos, D.B. The effect of Mediterranean diet on metabolic syndrome and its components. A meta-analysis of 50 studies and 534,906 individuals. J. Am. Coll. Cardiol. 2011, 57, 1299–1313. [Google Scholar]
- Martinez-Gonzalez, M.A.; Bes-Rastrollo, M. Dietary patterns, Mediterranean diet, and cardiovascular disease. Curr. Opin. Lipidol. 2014, 25, 20–26. [Google Scholar]
- Jenkins, D.J.; Kendall, C.W.; Marchie, A.; Faulkner, D.A.; Wong, J.M.; de Souza, R.; Emam, A.; Parker, T.L.; Vidgen, E.; Lapsley, K.G.; et al. Effects of a dietary portfolio of cholesterol-lowering foods vs. lovastatin on serum lipids and C-reactive protein. JAMA 2003, 290, 502–510. [Google Scholar]
- American Public Health Association (APHA). Available online: http://action.apha.org/site/PageServer?pagename=Obesity_Infographic (accessed on 25 January 2014).
- Evert, A.B.; Boucher, J.L.; Cypress, M.; Dunbar, S.A.; Franz, M.J.; Mayer-Davis, E.J.; Neumiller, J.J.; Nwankwo, R.; Verdi, C.L.; Urbanski, P.; et al. Nutrition therapy Recommendations for the management of adults with diabetes. Diabetes Care 2013, 36, 3821–3842. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Willett, W.C. Optimal diets for prevention of coronary heart disease. JAMA 2002, 288, 2569–2578. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Lipids, nutrition and coronary heart disease. In Atherosclerosis and Coronary Artery Disease; Fuster, V., Ross, R., Topol, E.J., Eds.; Lippincott-Raven: Philadelphia, PA, USA, 1996. [Google Scholar]
- Mente, A.; de Koning, L.; Shannon, H.S.; Anand, S.S. A systematic review of the evidence supporting a causal link between dietary factors and coronary heart diseases. Arch. Intern. Med. 2009, 169, 659–669. [Google Scholar] [CrossRef] [PubMed]
- ESC/EAS. Guidelines for the management of dyslipidaemias. Eur. Heart J. 2011, 32, 1769–1818. [Google Scholar]
- Connor, W.E.; Connor, S.L. Should a low-fat, high-carbohydrate diet be recommended for everyone? The case for a low-fat, high-carbohydrate diet. N. Engl. J. Med. 1997, 337, 562–563. [Google Scholar] [CrossRef] [PubMed]
- Jequier, E.; Bray, G.A. Low-Fat diets are preferred? Am. J. Med. 2002, 113, 41S–46S. [Google Scholar] [CrossRef]
- Ferro-Luzzi, A.; Branca, F. Mediterranean Diet, Italian-style: Prototype of a healthy diet. Am. J. Clin. Nutr. 1995, 61, 1338S–1345S. [Google Scholar]
- Chowdhury, R.; Warnakula, S.; Kunutsor, S.; Crowe, F.; Ward, H.A.; Johnson, L.; Franco, O.H.; Butterworth, A.S.; Forouhi, N.G.; Thompson, S.G.; et al. Association of dietary, circulating, and supplement fatty acids with coronary risk: A Systematic review and meta-analysis. Ann. Intern. Med. 2014, 160, 398–406. [Google Scholar] [CrossRef] [PubMed]
- McManus, K.; Antinoro, L.; Sacks, F. A randomized controlled trial of a moderate-fat, low-energy diet compared with a low fat, low-energy diet for weight loss in overweight adults. Int. J. Obes. 2001, 25, 1503–1511. [Google Scholar] [CrossRef]
- Alberti-Fidanza, A.; Fidanza, F.; Chiuchiù, M.P.; Verducci, G.; Fruttini, D. Dietary studies on two rural Italian population groups of the Seven Countries Study. 3. Trend of food and nutrient intake from 1960 to 1991. Eur. J. Clin. Nutr. 1999, 53, 854–860. [Google Scholar]
- Programa de actividades preventivas y promoción de la salud (PAPPS). Guía para ayudar a promover una alimentación saludable. In Grupo de Educación Sanitaria y Promoción de la Salud; SEMFYC: Barcelona, Spain, 2000.
- OMS 2004 Organización Mundial de la Salud. Adherencia a los Tratamientos a Largo Plazo; Pruebas para la Acción; World Health Oranization: Geneva, Switerzland, 2004. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
García-Fernández, E.; Rico-Cabanas, L.; Rosgaard, N.; Estruch, R.; Bach-Faig, A. Mediterranean Diet and Cardiodiabesity: A Review. Nutrients 2014, 6, 3474-3500. https://doi.org/10.3390/nu6093474
García-Fernández E, Rico-Cabanas L, Rosgaard N, Estruch R, Bach-Faig A. Mediterranean Diet and Cardiodiabesity: A Review. Nutrients. 2014; 6(9):3474-3500. https://doi.org/10.3390/nu6093474
Chicago/Turabian StyleGarcía-Fernández, Elena, Laura Rico-Cabanas, Nanna Rosgaard, Ramón Estruch, and Anna Bach-Faig. 2014. "Mediterranean Diet and Cardiodiabesity: A Review" Nutrients 6, no. 9: 3474-3500. https://doi.org/10.3390/nu6093474
APA StyleGarcía-Fernández, E., Rico-Cabanas, L., Rosgaard, N., Estruch, R., & Bach-Faig, A. (2014). Mediterranean Diet and Cardiodiabesity: A Review. Nutrients, 6(9), 3474-3500. https://doi.org/10.3390/nu6093474