Potential Role of Carotenoids as Antioxidants in Human Health and Disease
Abstract
:1. Introduction
2. Carotenoids: Short Overview
2.1. Chemical Structure, Function and Membrane Distribution

2.2. Bioavailability and Fate in Human Body
3. Reactive Oxygen Species
3.1. Major Cellular Sources of ROS
3.2. Types of ROS
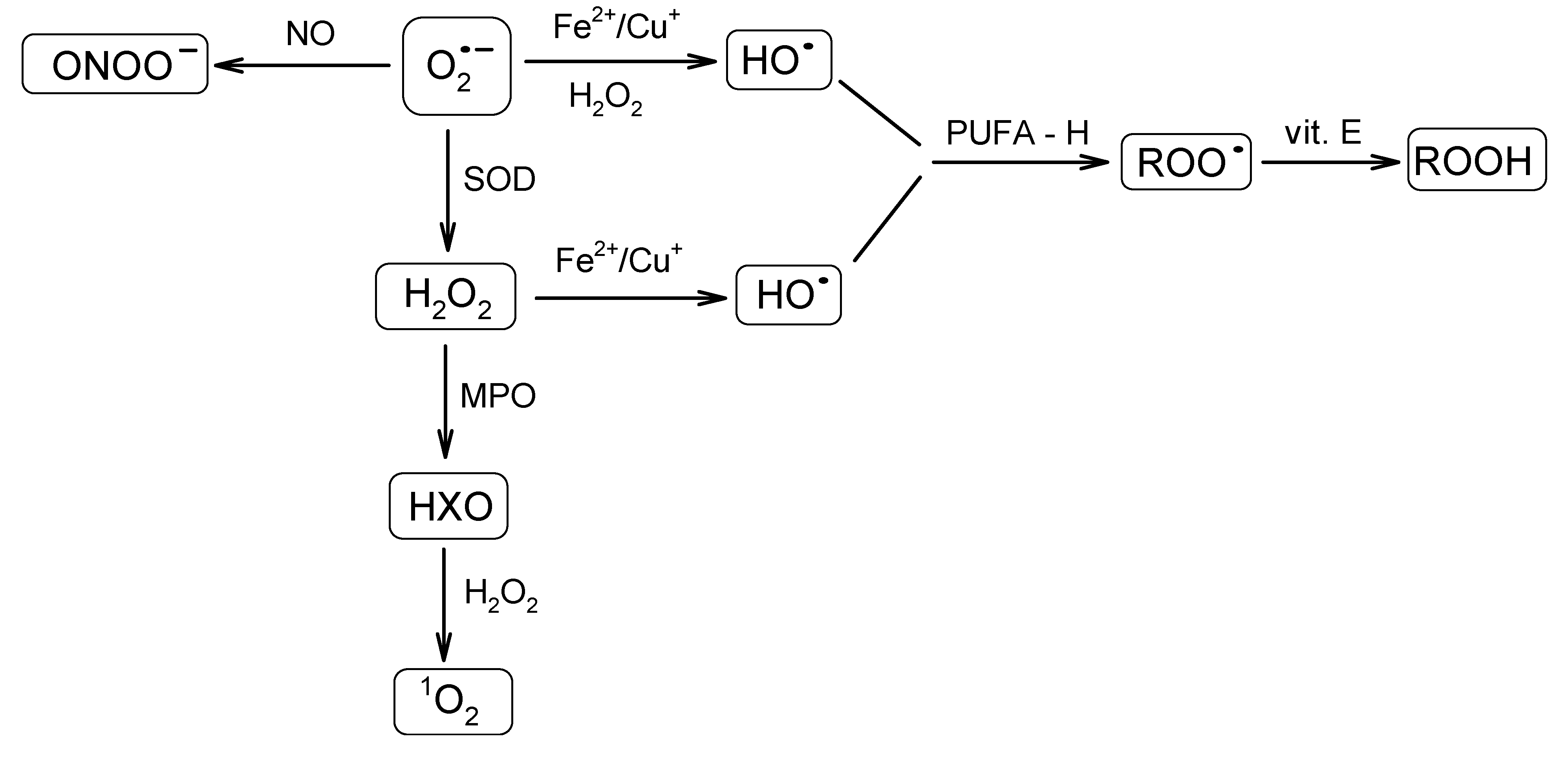
4. ROS: Antioxidants Balance
5. Carotenoids: ROS Interactions
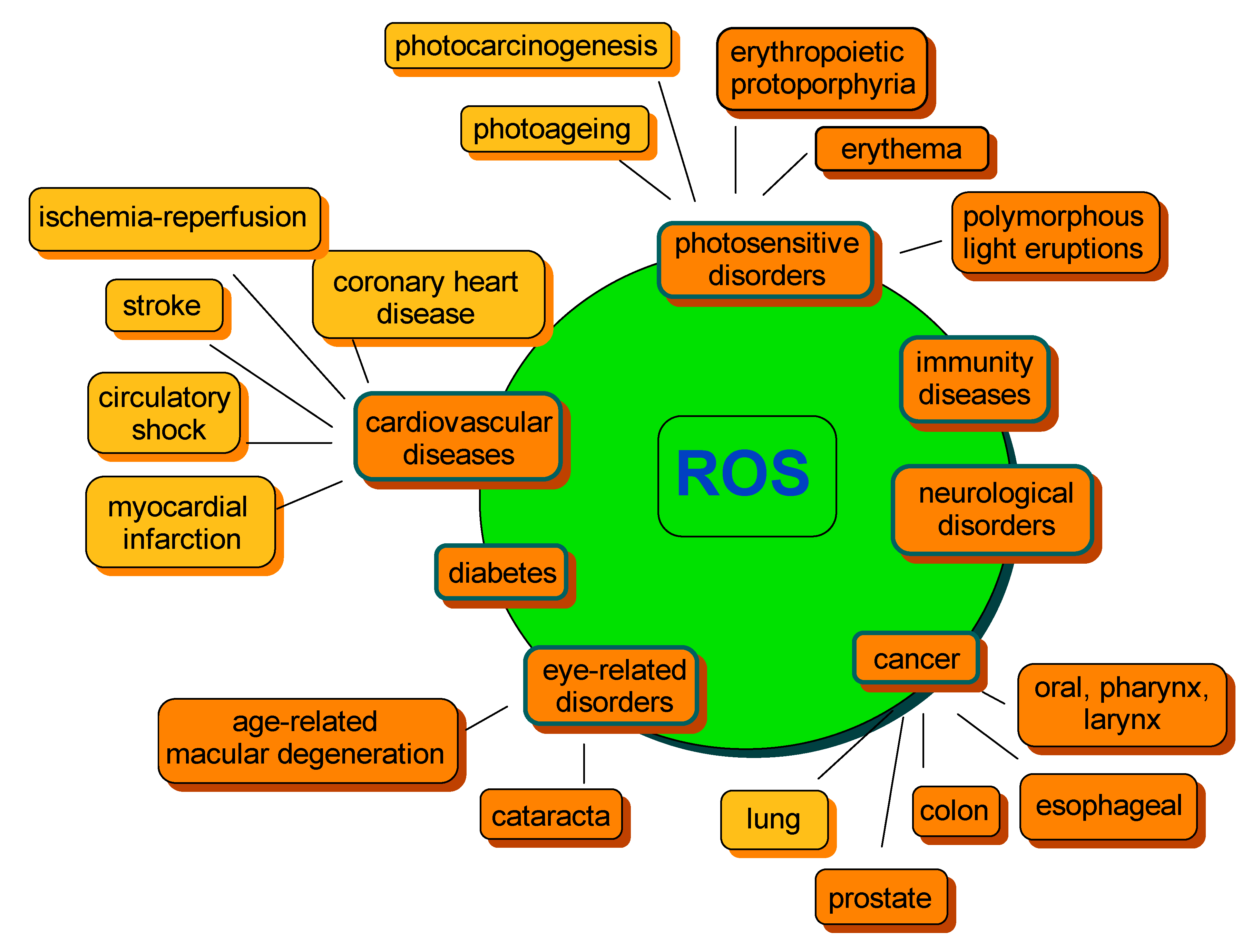
6. Oxidative Stress, Disease and Carotenoids
6.1. Cancer
6.2. Cardiovascular and Related Disorders
6.3. Photosensitivity Disorders
6.4. Other
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Landrum, J.T. Carotenoids: Physical, Chemical, and Biological Functions and Properties; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Scheer, H. The Pigments. In Light-Harvesting Antennas in Photosynthesis; Green, B.R., Parson, W.W., Eds.; Kluwer Academic Publishers: Dordrecht, the Netherlands, 2003; pp. 29–81. [Google Scholar]
- Fiedor, J.; Fiedor, L.; Haessner, R.; Scheer, H. Cyclic ednoperoxides of β-carotene, potential pro-oxidants, as products of chemical quenching of singlet oxygen. Biochim. Biophys. Acta 2005, 1709, 1–4. [Google Scholar] [CrossRef]
- Edge, R.; Truscott, T.G. Properties of Carotenoid Radicals and Excited States and Their Potential Role in Biological Systems. In Carotenoids: Physical, Chemical, and Biological Functions and Properties; Landrum, J.T., Ed.; CRC Press: Boca Raton, FL, USA, 2010; pp. 283–308. [Google Scholar]
- Cvetkovic, D.; Fiedor, L.; Fiedor, J.; Wiśniewska-Becker, A.; Markovic, D. Molecular Base for Carotenoids Antioxidant Activity in Model and Biological Systems: The Health-Related Effects. In Carotenoids: Food Sources, Production and Health Benefits; Yamaguchi, M., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2013; pp. 93–126. [Google Scholar]
- Britton, G.; Liaaen-Jensen, S.; Pfander, H. Carotenoids. Handbook; Birkhauser Verlag: Basel, Switzerland, 2004. [Google Scholar]
- Khachik, F. Distribution and metabolism of dietary carotenoids in humans as a criterion for development of nutritional supplements. Pure Appl. Chem. 2006, 78, 1551–1557. [Google Scholar] [CrossRef]
- Parker, R.S. Carotenoids in human blood and tissues. J. Nutr. 1989, 119, 101–104. [Google Scholar]
- Breecher, G.R.; Khachik, F. Qualitative relationship of dietary and plasma carotenoids in human beings. Ann. N. Y. Acad. Sci. USA 1992, 669, 320–321. [Google Scholar] [CrossRef]
- Bertram, J.S. Cancer prevention by carotenoids: Mechanistic studies in cultured cells. Ann. N. Y. Acad. Sci. USA 1993, 691, 177–191. [Google Scholar] [CrossRef]
- Krinsky, N.I. Micronutrients and their influence on mutagenicity and malignant transformation. Ann. N. Y. Acad. Sci. USA 1993, 686, 229–242. [Google Scholar] [CrossRef]
- Palozza, P.; Serini, S.; Ameruso, M.; Verdecchia, S. Modulation of Intracellular Signaling Pathways by Carotenoids. In Carotenoids: Nutrition and Health; Britton, G., Liaaen-Jensen, S., Pfander, H., Eds.; Birkhauser: Basel, Switzerland, 2009; Volume 5, pp. 211–235. [Google Scholar]
- Wiśniewska, A.; Subczyński, W.K. Effects of polar carotenoids on the shape of the hydrophobic barrier of phospholipid bilayers. Biochim. Biophys. Acta 1998, 1368, 235–246. [Google Scholar]
- Wiśniewska, A.; Subczyński, W.K. Accumulation of macular xanthophylls in unsaturated membrane domains. Free Radic. Biol. Med. 2006, 40, 1820–1826. [Google Scholar] [CrossRef]
- Britton, G. Structure and properties of carotenoids in relation to function. FASEB J. 1995, 9, 1551–1558. [Google Scholar]
- Gabrielska, J.; Gruszecki, W.I. Zeaxanthin (dihydroxy-β-carotene) but not β-carotene rigidifies lipid membranes: A 1H-NMR study of carotenoid-egg phosphatidylcholine liposomes. Biochim. Biophys. Acta 1996, 1285, 167–174. [Google Scholar] [CrossRef]
- Gruszecki, W.I.; Strzałka, K. Carotenoids as moldulators of lipid membrane physical properties. Biochim. Biophys. Acta 2005, 1740, 108–115. [Google Scholar]
- Maiani, G.; Caston, M.J.; Catasta, G.; Toti, E.; Cambrodon, I.G.; Bysted, A.; Granado-Lorencio, F.; Olmedilla-Alonso, B.; Knuthsen, P.; Valoti, M.; et al. Carotenoids: Actual knowledge on food sources, intakes, stability and bioavailability and their protective role in humans. Mol. Nutr. Food Res. 2009, 53, S194–S218. [Google Scholar] [CrossRef]
- Demming-Adams, B.; Adams, R.B. Eye nutrition in context: Mechanisms, implementation, and future directions. Nutrients 2013, 5, 2483–2501. [Google Scholar] [CrossRef]
- Abdel-Aal, E.-S.M.; Akhtar, H.; Zaheer, K.; Ali, R. Dietary sources of lutein and zeaxanthin carotenoids and their role in eye health. Nutrients 2013, 5, 1169–1185. [Google Scholar] [CrossRef]
- Castenmiller, J.J.M.; West, C.E. Bioavailability of carotenoids. Pure Appl. Chem. 1997, 89, 2145–2150. [Google Scholar] [CrossRef]
- Yeum, K.-J.; Russell, R.M. Carotenoid bioavailability and bioconversion. Ann. Rev. Nutr. 2002, 22, 483–504. [Google Scholar] [CrossRef]
- Prince, M.R.; Frisoli, J.K. Beta-carotene accumulation in serum and skin. Am. J. Clin. Nutr. 1993, 57, 175–181. [Google Scholar]
- Olson, J.A. Absorption, transport, and metabolism of carotenoids in humans. Pure Appl. Chem. 1994, 66, 1011–1016. [Google Scholar] [CrossRef]
- Bernhardt, S.; Schlich, E. Impact of different cooking methods on food quality: Retention of lipophilic vitamins in fresh and frozen vegetables. J. Food Eng. 2006, 77, 327–333. [Google Scholar] [CrossRef]
- Fernandez-Garcia, E.; Carvajal-Lerida, I.; Jaren-Galan, M.; Garrido-Fernandez, J.; Perez-Galvez, A.; Hornero-Mendez, D. Carotenoids bioavailability from foods: From plant pigments to efficient biological activities. Food Res. Int. 2012, 46, 438–450. [Google Scholar] [CrossRef]
- Kostic, D.; White, W.S.; Olson, J.A. Intestinal absorption, serum clearance, and interactions between lutein and β-carotene when administrated to human adults in separate or combined oral doses. Am. J. Clin. Nutr. 1995, 62, 602–610. [Google Scholar]
- Deming, D.M.; Erdman, J.W., Jr. Mammalian carotenoid absorption and metabolism. Pure Appl. Chem. 1999, 71, 2213–2223. [Google Scholar] [CrossRef]
- Harrison, E.H. Mechanisms of Intestinal Absorption of Carotenoids: Insights from in Vitro Systems. In Carotenoids: Physical, Chemical, and Biological Functions and Properties; Landrum, J.T., Ed.; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2010; pp. 367–381. [Google Scholar]
- Furr, H.C.; Clark, R.M. Intestinal absorption and tissue distribution of carotenoids. J. Nutr. Biochem. 1997, 8, 364–377. [Google Scholar] [CrossRef]
- Parker, R.S. Absorption, metabolism, and transport of carotenoids. FASEB J. 1996, 10, 542–551. [Google Scholar]
- Stahl, W.; Schwarz, W.; Sundquist, A.R.; Sies, H. cis-trans Isomers of lycopene and β-carotene in human serum and tissues. Arch. Biochem. Biophys. 1992, 294, 173–177. [Google Scholar] [CrossRef]
- Darvin, M.E.; Sterry, W.; Landemann, J.; Vergou, T. The role of carotenoids in human skin. Molecules 2011, 16, 10491–10506. [Google Scholar] [CrossRef]
- Anders, M.W.; Robotham, J.L.; Sheu, S.-S. Mitochondria: New drug targets for oxidative stress-induced diseases. Expert Opin. Drug Metab. Toxicol. 2006, 2, 71–79. [Google Scholar] [CrossRef]
- Inoue, M.; Sato, E.F.; Nishikawa, M.; Park, A.M.; Kira, Y.; Imada, I.; Utsumi, K. Mitochondrial generation of reactive oxygen species and its role in aerobic life. Curr. Med. Chem. 2003, 10, 2495–2505. [Google Scholar] [CrossRef]
- Figueira, T.R.; Barros, M.H.; Camargo, A.A.; Castilho, R.F.; Ferreira, J.C.B.; Kowaltowski, A.J.; Sluse, F.E.; Souza-Pinto, N.C.; Vercesi, A.E. Mitochondria as a source of reactive oxygen and nitrogen species: From molecular mechanisms to human health. Antioxid. Redox Signal. 2013, 18, 2029–2074. [Google Scholar] [CrossRef]
- Starkov, A.A.; Fiskum, G.; Chinopoulos, C.; Lorenzo, B.J.; Browne, S.E.; Patel, M.S.; Beal, M.F. Mitochondrial α-ketoglutarate dehydrogenase complex generates reactive oxygen species. J. Neurosci. 2004, 24, 7779–7788. [Google Scholar] [CrossRef]
- Winterbourn, C.C.; Vissers, M.C.; Kettle, A.J. Myeloperoxidase. Curr. Opin. Hematol. 2000, 7, 53–58. [Google Scholar] [CrossRef]
- Bedard, K.; Krause, K.-H. The NOX family of ROS-generating NADPH oxidases: Physiology and pathophysiology. Physiol. Rev. 2007, 87, 245–313. [Google Scholar] [CrossRef]
- Sheu, S.S.; Nauduri, D.; Anders, M.W. Targeting antioxidants to mitochondria: A new therapeutic direction. Biochim. Biophys. Acta 2006, 1762, 256–265. [Google Scholar] [CrossRef]
- Stadtman, E.R.; Levine, R.L. Protein oxidation. Ann. N. Y. Acad. Sci. USA 2000, 90, 191–208. [Google Scholar]
- Richter, C.; Park, J.W.; Ames, B.N. Normal oxidative damage to mitochondrial and nuclear DNA is extensive. Proc. Natl. Acad. Sci. USA 1988, 85, 6465–6467. [Google Scholar] [CrossRef]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef]
- Babior, B.M. The NADPH oxidase of endothelial cells. IUBMB Life 2000, 50, 267–269. [Google Scholar] [CrossRef]
- Vignais, P.V. The superoxide-generating NADPH oxidase: Structural aspects and activation mechanism. Cell. Mol. Life Sci. 2002, 59, 1428–1459. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Antioxidant defense: Vitamins E and C and carotenoids. Diabetes 1997, 46, S14–S18. [Google Scholar] [CrossRef]
- Fridovich, I. Superoxide anion radical (O2•−), superoxide dismutases, and related matter. J. Biol. Chem. 1997, 272, 18515–18517. [Google Scholar] [CrossRef]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric oxide and peroxynitrite in health and disease. Physiol. Rev. 2007, 87, 315–424. [Google Scholar] [CrossRef]
- Wardman, P. Fluorescent and luminescent probes for measurement of oxidative and nitrosative species in cells and tissues: Progress, pitfalls, and prospects. Free Radic. Biol. Med. 2007, 43, 995–1022. [Google Scholar] [CrossRef]
- Lipinski, B. Hydroxyl radical and its scavengers in health and disease. Oxid. Med. Cell. Longev. 2011, 2011, 809696. [Google Scholar] [CrossRef]
- Chance, B.; Sies, H.; Boveris, A. Hydroperoxide metabolism in mammalian organs. Physiol. Rev. 1979, 59, 527–605. [Google Scholar]
- Boveris, A.; Oshino, N.; Chance, B. The cellular production of hydrogen peroxide. Biochem. J. 1972, 128, 617–630. [Google Scholar]
- Storz, P. Reactive oxygen species in tumor progression. Front. Biosci. 2005, 10, 1881–1896. [Google Scholar] [CrossRef]
- Slauch, J.M. How does the oxidative burst of macrophages kill bacteria? Still an open question. Mol. Microbiol. 2011, 80, 580–583. [Google Scholar] [CrossRef]
- Chen, Y.; Junger, W.G. Measurement of oxidative burst in neutrophils. Meth. Mol. Biol. 2012, 844, 115–124. [Google Scholar] [CrossRef]
- Ogilby, P.R.; Foote, C.S. Chemistry of singlet oxygen. 42. Effect of solvent, solvent isotopic substitution, and temperature on the lifetime of singlet molecular oxygen (1∆g). J. Am. Chem. Soc. 1983, 105, 3423–3430. [Google Scholar] [CrossRef]
- Duthie, S.J.; Collins, A.R.; Duthie, G.G. The Role of Carotenoids in Modulating DNA Stability and Lipid Peroxidation Importance for Human Health. In Fat-Soluble Vitamins; Quinn, P.J., Kagan, V.E., Eds.; Plenum Press: New York, NY, USA, 1998; Volume 30, pp. 181–207. [Google Scholar]
- Girotti, A.W. Mechanisms of lipid peroxidation. J. Free Radic. Biol. Med. 1985, 1, 87–95. [Google Scholar] [CrossRef]
- Ham, A.J.; Liebler, D.C. Vitamin E oxidation in rat liver mitochondria. Biochemistry 1995, 34, 5754–5761. [Google Scholar] [CrossRef]
- El-Agamey, A.; Lowe, G.M.; McGarvey, D.J.; Mortensen, A.; Philip, D.M.; Truscott, T.G.; Young, A.J. Carotenoid radical chemistry and antioxidant/pro-oxidant properties. Arch. Biochem. Biophys. 2004, 430, 37–48. [Google Scholar] [CrossRef]
- Droege, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar]
- Palmer, H.J.; Paulson, K.E. Reactive oxygen species and antioxidants in signal transduction and gene expression. Nutr. Rev. 1997, 55, 353–361. [Google Scholar] [CrossRef]
- Hughes, G.; Murphy, M.P.; Ledgerwood, E.C. Mitochondrial reactive oxygen species regulate the temporal activation of nuclear factor κB to modulate tumor necrosis factor-induced apoptosis: Evidence from mitochondria-targeted antioxidants. Biochem. J. 2005, 389, 83–89. [Google Scholar] [CrossRef]
- Halliwell, B. Phagocyte-derived reactive species: Salvation or suicide? Trends Biochem. Sci. 2006, 31, 509–515. [Google Scholar] [CrossRef]
- Hultqvust, M.; Olsson, L.A.; Gelderman, K.A.; Holmdah, R. The protective role of ROS in autoimmune disease. Trends Immunol. 2009, 30, 201–208. [Google Scholar] [CrossRef]
- Nomura, K.; Imai, H.; Koumura, T.; Kobayashi, T.; Nakagawa, Y. Mitochondrial phospholipid hydroperoxide glutathione peroxidase inhibits the release of cytochrome c from mitochondria by suppressing the peroxidation of cardiolipin in hypoglycaemia-induced apoptosis. Biochem. J. 2000, 351, 183–193. [Google Scholar] [CrossRef]
- Radi, R.; Turrens, J.F.; Chang, L.Y.; Bush, K.M.; Crapo, J.D.; Freeman, B.A. Detection of catalase in rat heart mitochondria. J. Biol. Chem. 1991, 266, 22028–22034. [Google Scholar]
- Ricciarelli, R.; Zingg, J.-M.; Azzi, A. Vitamin E: Protective role of a Janus molecule. FASEB J. 2001, 15, 2314–2325. [Google Scholar] [CrossRef]
- Palozza, P.; Krinsky, N.I. β-Carotene and α-tocopherol are synergistic antioxidants. Arch. Biochem. Biophys. 1992, 297, 184–187. [Google Scholar] [CrossRef]
- Palozza, P.; Moualla, S.; Krinsky, N.I. Effects of β-carotene and α-tocopherol on radical-initiated peroxidation of microsomes. Free Radic. Biol. Med. 1992, 13, 127–136. [Google Scholar] [CrossRef]
- Wrona, M.; Korytowski, W.; Różanowska, M.; Sarna, T.; Truscott, T.G. Cooperation of antioxidants in protection against photosensitized oxidation. Free Radic. Biol. Med. 2003, 35, 1319–1329. [Google Scholar] [CrossRef]
- Boehm, F.; Edge, R.; McGarvey, D.J.; Truscott, T.G. β-Carotene with vitamin E and C offers synergistic cell protection against NOx. FEBS Lett. 1998, 436, 387–389. [Google Scholar] [CrossRef]
- Tinggi, U. Selenium: Its role as antioxidant in human health. Environ. Health Prev. Med. 2008, 13, 102–108. [Google Scholar] [CrossRef]
- Burk, R.F. Selenium, an antioxidant nutrient. Nutr. Clin. Care 2002, 5, 75–79. [Google Scholar] [CrossRef]
- Sautin, Y.Y.; Johnson, R.J. Uric acid: The oxidant-antioxidant paradox. Nucleosides Nucleotides Nucleic Acids 2008, 27, 608–619. [Google Scholar] [CrossRef]
- Grune, T.; Schroeder, P.; Biesalski, H.K. Low Molecular Weight Antioxidants. In The Handbook of Environmental Chemistry; Springer: Berlin, Germany, 2005; Volume 2, pp. 77–90. [Google Scholar]
- Edge, R.; McGarvey, D.J.; Truscott, T.G. The carotenoids as anti-oxidants—A review. J. Photochem. Photobiol. B 1997, 41, 189–200. [Google Scholar]
- Husain, S.R.; Cillard, J.; Cillard, P. Hydroxyl radical scavenging activity of flavonoids. Phytochemistry 1987, 26, 126–133. [Google Scholar]
- Robak, J.; Gryglewski, R.J. Flavonoids are scavengers of superoxide anions. Biochem. Pharmacol. 1988, 37, 837–841. [Google Scholar] [CrossRef]
- Bors, W.; Heller, W.; Michel, C.; Saran, M. Flavonoids as Antioxidants: Determination of Radical-Scavenging Efficiencies. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1990; Volume 186, pp. 343–355. [Google Scholar]
- Ross, J.A.; Kasum, C.M. Dietary flavonoids: Bioavailability, metabolic effects, and safety. Ann. Rev. Nutr. 2002, 22, 19–34. [Google Scholar] [CrossRef]
- Suzuki, K. Anti-oxidants for therapeutic use: Why are only a few drugs in clinical use? Adv. Drug Deliv. Rev. 2009, 61, 287–289. [Google Scholar] [CrossRef]
- Brookes, P.S.; Yoon, Y.; Robotham, J.L.; Anders, M.W.; Sheu, S.S. Calcium, ATP, and ROS: A mitochondrial love-hate triangle. Am. J. Physiol. 2004, 287, C817–C833. [Google Scholar] [CrossRef]
- Wen, X.; Wu, J.; Wang, F.; Liu, B.; Huang, C.; Wei, Y. Deconvoluting the role of reactive oxygen species and autophagy in human diseases. Free Radic. Biol. Med. 2013, 65, 402–410. [Google Scholar] [CrossRef]
- Christensen, R.L. The Electronic States of Carotenoids. In The Photochemistry of Carotenoids; Frank, H.A., Young, A.J., Britton, G., Eds.; Kluwer Academic Publishers: Dordrecht, the Netherlands, 1999; pp. 137–157. [Google Scholar]
- Foote, C.S.; Chang, Y.C.; Denny, R.W. Chemistry of singlet oxygen. X. cis-trans isomerisation of carotenoids by singlet oxygen and probable quenching mechanism. J. Am. Chem. Soc. 1970, 92, 5218–5219. [Google Scholar] [CrossRef]
- Chantrell, S.J.; McAuliffe, C.A.; Munn, R.W. Excited states of protoporphyrin IX dimethyl ester: Reaction of the triplet with carotenoids. J. Chem. Soc. Faraday Trans. I 1977, 73, 858–865. [Google Scholar] [CrossRef]
- Fiedor, J.; Fiedor, L.; Winkler, J.; Scherz, A.; Scheer, H. Photodynamics of the bacteriochlorophyll-carotenoid system. 1. Bacteriochlorophyll-photosensitized oxygenation of β-carotene in acetone. Photochem. Photobiol. 2001, 74, 64–71. [Google Scholar] [CrossRef]
- Fiedor, J.; Fiedor, L.; Kammhuber, N.; Scherz, A.; Scheer, H. Photodynamics of the bacteriochlorophyll-carotenoid system. 2. Influence of central metal, solvent and β-carotene on photobleaching of bacteriochlorophyll derivatives. Photochem. Photobiol. 2002, 76, 145–152. [Google Scholar]
- Stratton, S.P.; Schaefer, W.H.; Liebler, D.C. Isolation and identification of singlet oxygen oxidation products of β-carotene. Chem. Res. Toxicol. 1993, 6, 542–547. [Google Scholar] [CrossRef]
- Martin, H.D.; Ruck, C.; Schmidt, M.; Sell, S.; Beutner, S.; Mayer, B.; Walsh, R. Chemistry of carotenoid oxidation and free radical reactions. Pure Appl. Chem. 1999, 71, 2253–2262. [Google Scholar] [CrossRef]
- Yamauchi, R.; Nobuyuki, H.; Inoue, H.; Kato, K. Products formed by peroxyl radical oxidation of β-carotene. J. Agric. Food Chem. 1993, 41, 708–713. [Google Scholar] [CrossRef]
- Fiedor, J.; Sulikowska, A.; Orzechowska, A.; Fiedor, L.; Burda, K. Antioxidant effects of carotenoids in a model pigment-protein complex. Acta Biochim. Pol. 2012, 59, 61–64. [Google Scholar]
- Galano, A.; Vargas, R.; Martinez, A. Carotenoids can act as antioxidants by oxidizing the superoxide radical anion. Phys. Chem. Chem. Phys. 2010, 12, 193–200. [Google Scholar] [CrossRef]
- Mortensen, A.; Skibsted, L.H.; Sampson, J.; Rice-Evans, C.; Everett, S.A. Comparative mechanisms and rates of free radical scavenging by carotenoid antioxidants. FEBS Lett. 1997, 418, 91–97. [Google Scholar] [CrossRef]
- Kispert, L.D.; Konovalova, T.; Gao, Y. Carotenoid radical cations and dications: EPR, optical, and electrochemical studies. Arch. Biochem. Biophys. 2004, 430, 49–60. [Google Scholar] [CrossRef]
- Chen, C.H.; Han, R.M.; Liang, R.; Fu, L.M.; Wang, P.; Ai, X.C.; Zhang, J.P.; Skibsted, L.H. Direct observation of the β-carotene reaction with hydroxyl radical. J. Phys. Chem. B 2011, 115, 2082–2089. [Google Scholar]
- Burke, M.; Edge, R.; Land, E.J.; McGarvey, D.J.; Truscott, T.G. One-electron reduction potentials of dietary carotenoid radical cations in aqueous micellar environments. FEBS Lett. 2001, 500, 132–136. [Google Scholar] [CrossRef]
- Polyakov, N.E.; Kruppa, A.I.; Leshina, T.V.; Konovalova, T.A.; Kispert, L.D. Carotenoids as antioxidants: Spin trapping EPR and optical study. Free Radic. Biol. Med. 2001, 31, 43–52. [Google Scholar] [CrossRef]
- Polyakov, N.E.; Foscan, A.L.; Bowman, M.K.; Kispert, L.D. Free radical formation in novel carotenoid metal ion complexes with astaxanthin. J. Phys. Chem. B 2010, 114, 16968–16977. [Google Scholar] [CrossRef]
- World Health Organization. Cancer. Available online: http://www.who.int/mediacentre/factsheets/fs297/en/print.html (accessed on 14 October 2013).
- Block, G.; Patterson, B.; Subar, A. Fruit, vegetables, and cancer prevention: A review of the epidemiological evidence. Nutr. Cancer 1992, 18, 1–29. [Google Scholar] [CrossRef]
- Voorrips, L.E.; Goldbohm, A.; Brants, H.A.M.; van Poppel, G.A.F.C.; Sturmans, F.; Hermus, R.J.J.; van den Brandt, P.A. A prospective cohort study on antioxidant and folate intake and male lung cancer risk. Cancer Epidemiol. Biomark. Prev. 2000, 9, 357–365. [Google Scholar]
- Donaldson, M.S. Nutrition and cancer: A review of the evidence for an anti-cancer diet. Nutr. J. 2004, 3, 19. [Google Scholar] [CrossRef]
- Key, T.J. Fruit and vegetables and cancer risk. Br. J. Cancer 2011, 104, 6–11. [Google Scholar] [CrossRef]
- Le Marchand, L.; Hankin, J.H.; Kolonel, L.N.; Beecher, C.R.; Wilkens, L.R.; Zhao, L.P. Intake of specific carotenoids and lung cancer risk. Cancer Epidemiol. Biomarkers Prev. 1993, 2, 183–187. [Google Scholar]
- Mayne, S.T.; Janerich, D.T.; Greenwald, P.; Chorost, S.; Tucci, C.; Zaman, M.B.; Melamed, M.R.; Kiely, M.; McKneally, M.F. Dietary beta carotene and lung cancer risk in U.S. nonsmokers. J. Natl. Cancer Inst. 1994, 86, 33–38. [Google Scholar] [CrossRef]
- Michaud, D.S.; Feskanich, D.; Rimm, E.B.; Colditz, G.A.; Speizer, F.E.; Willett, W.C.; Giovannucci, E. Intake of specific carotenoids and risk of lung cancer in 2 prospective US cohorts. Am. J. Clin. Nutr. 2000, 72, 990–997. [Google Scholar]
- Brennan, P.; Fortes, C.; Butler, J.; Agudo, A.; Benhamou, S.; Darby, S.; Gerken, M.; Jokel, K.H.; Kreuzer, M.; Mallone, S.; et al. A multicenter case-control study of diet and lung cancer among non-smokers. Cancer Causes Control 2000, 11, 49–58. [Google Scholar] [CrossRef]
- The Alpha-Tocopherol; Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta-carotene on the incidence of lung cancer and other cancers in male smokers. N. Engl. J. Med. 1994, 330, 1029–1035. [Google Scholar] [CrossRef]
- Albanes, D.; Heinonen, O.P.; Taylor, P.R.; Virtamo, J.; Edwards, B.K.; Rautalahti, M.; Hartman, A.M.; Palmgren, J.; Freedman, L.S.; Haapakoski, J.; et al. α-ocopherol and β-carotene supplements and lung cancer incidence in the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study: Effects of base-line characteristics and study compliance. J. Natl. Cancer Inst. 1996, 88, 1560–1570. [Google Scholar] [CrossRef]
- Omenn, G.S.; Goodman, G.E.; Thornquist, M.D.; Balmes, J.; Cullen, M.R.; Glass, A.; Keogh, J.P.; Meyskens, F.L., Jr.; Valanis, B.; Williams, J.H., Jr.; et al. Risk factors for lung cancer and for intervention effects in CARET, the Beta-Carotene and Retinol Efficacy Trial. J. Natl. Cancer Inst. 1996, 88, 1550–1559. [Google Scholar] [CrossRef]
- Goodman, G.E.; Thornquist, M.D.; Balmes, J.; Cullen, M.R.; Meysekns, F.L., Jr.; Omenn, G.S.; Valanis, B.; Williams, J.H., Jr. The Beta-Carotene And Retinol Efficacy Trial: Incidence of lung cancer and cardiovascular disease mortality during 6-year follow-up after stopping β-carotene and retinol supplements. J. Natl. Cancer Inst. 2004, 96, 1743–1750. [Google Scholar] [CrossRef]
- Góralczyk, R. β-Carotene and lung cancer in smokers: Review of hypotheses and status of research. Nutr. Cancer 2009, 61, 767–774. [Google Scholar] [CrossRef]
- Mills, P.K.; Beeson, W.L.; Phillips, R.L.; Fraser, G.E. Cohort study of diet, lifestyle, and prostate cancer in Adventist men. Cancer 1989, 64, 598–604. [Google Scholar]
- Chan, J.M.; Gann, P.H.; Giovannucci, E.L. Role of diet in prostate cancer development and progression. J. Clin. Oncol. 2005, 23, 8152–8160. [Google Scholar] [CrossRef]
- Giovannucci, E. A review of epidemiologic studies of tomatoes, lycopene, and prostate cancer. Exp. Biol. Med. 2002, 227, 852–859. [Google Scholar]
- Wertz, K.; Siler, U.; Góralczyk, R. Lycopene: Modes of action to promote prostate health. Arch. Biochem. Biophys. 2004, 430, 127–134. [Google Scholar] [CrossRef]
- Etminan, M.; Takkouche, B.; Caamano-Isorna, F. The role of tomato products and lycopene in the prevention of prostate cancer: A meta-analysis of observational studies. Cancer Epidemiol. Biomark. Prev. 2004, 13, 340–345. [Google Scholar]
- Fraser, M.L.; Lee, A.H.; Binns, C.W. Lycopene and prostate cancer: Emerging evidence. Expert Rev. Anticancer Ther. 2005, 5, 847–854. [Google Scholar] [CrossRef]
- Stacewicz-Sapuntzakis, M.; Bowen, P.E. Role of lycopene and tomato products in prostate health. Biochim. Biophys. Acta 2005, 1740, 202–205. [Google Scholar] [CrossRef]
- Mayne, S.T.; Goodwin, W.J., Jr. Chemoprevention of head and neck cancer. Curr. Opin. Otolaryngol. Head Neck Surg. 1993, 1, 126–132. [Google Scholar]
- Freedman, N.D.; Park, Y.; Subar, A.F.; Hollenbeck, A.R.; Leitzmann, M.F.; Schatzkin, A.; Abnet, C.C. Fruit and vegetable intake and head and neck cancer risk in a large United States prospective cohort study. Int. J. Cancer 2008, 122, 2330–2336. [Google Scholar]
- Cheng, K.K.; Day, N.E. Nutrition and esophageal cancer. Cancer Causes Control 1996, 7, 33–40. [Google Scholar] [CrossRef]
- Bostick, R.M. Nutrition and colon cancer prevention. Nestle Nutr. Workshop Ser. Clin. Perform Programme 2000, 4, 67–85. [Google Scholar] [CrossRef]
- McGarr, S.E.; Ridlon, J.M.; Hylemon, P.B. Diet, anaerobic bacterial metabolism, and colon cancer: A review of the literature. J. Clin. Gastroenterol. 2005, 39, 98–109. [Google Scholar]
- Agarwal, M.; Parameswari, R.P.; Vasanthi, H.R.; Das, D.K. Dynamic action of carotenoids in cardioprotection and maintenance of cardic health. Molecules 2012, 17, 4755–4769. [Google Scholar] [CrossRef]
- Vogiatzi, G.; Tousoulis, D.; Stefanadis, C. The role of oxidative stress in atherosclerosis. Hell. J. Cardiol. 2009, 50, 402–409. [Google Scholar]
- Mayne, S.T. Beta-carotene, carotenoids, and disease prevention in humans. FASEB J. 1996, 10, 690–701. [Google Scholar]
- Voutilainen, S.; Nurmi, T.; Mursu, J.; Rissanen, T.H. Carotenoids and cardiovascular health. Am. J. Clin. Nutr. 2006, 83, 1265–1271. [Google Scholar]
- Tornwall, M.E.; Virtamo, J.; Korhonen, P.A. Effect of α-tocopherol and β-carotene supplementation on coronary heart disease during the 6-year post-trial follow-up in the ATBC study. Eur. Heart J. 2004, 25, 1171–1178. [Google Scholar] [CrossRef]
- Hennekens, C.H.; Buring, J.E.; Manson, J.E.; Stampfer, M.J.; Rosner, B.; Cook, N.R.; Belanger, C.; LaMotte, F.; Gaziano, J.M.; Ridker, P.M.; et al. Lack of effect of long-term supplementation with beta carotene on the incidence of malignant neoplasms and cardiovascular disease. N. Engl. J. Med. 1996, 334, 1145–1149. [Google Scholar] [CrossRef]
- Greenberg, E.R.; Baron, J.A.; Karagas, M.R.; Stukel, T.A.; Nierenberg, D.W.; Stevens, M.M.; Mandel, J.S.; Haile, R.W. Mortality associated with low plasma concentration of beta carotene and the effect or oral supplementation. JAMA 1996, 275, 699–703. [Google Scholar] [CrossRef]
- Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of antioxidant vitamin supplementation in 20,536 high-risk individuals: A randomised placebo-controlled trial. Lancet 2002, 360, 23–33. [Google Scholar] [CrossRef]
- Rapola, J.M.; Virtamo, J.; Ripatti, S.; Huttunen, J.K.; Albanes, D.; Taylor, P.R.; Heinonen, O.P. Randomised trial of α-tocopherol and β-carotene supplements on incidence of major coronary events in men with previous myocardial infarction. Lancet 1997, 349, 1715–1720. [Google Scholar] [CrossRef]
- Polyakov, N.E.; Magyar, A.; Kispert, L.D. Photochemical and optical properties of water-soluble xanthophyll antioxidants: Aggregation vs. complexation. J. Phys. Chem. B 2013, 117, 10173–10182. [Google Scholar] [CrossRef]
- Pashkow, F.J.; Watumull, D.G.; Campbell, C.L. Astaxanthin: A novel potential treatment for oxidative stress and inflammation in cardiovascular disease. Am. J. Cardiol. 2008, 101, 58D–68D. [Google Scholar]
- Johnson, E.J.; Krinsky, N.I. Carotenoids and Coronary Heart Disease. In Carotenoids; Britton, G., Liaaen-Jensen, S., Pfander, H., Eds.; Birkhauser Verlag: Basel, Switzerland, 2009; Volume 5, pp. 287–300. [Google Scholar]
- Willis, I.; Cylus, L. UVA erythema in skin: Is it a sunburn? J. Investig. Dermatol. 1977, 68, 128–129. [Google Scholar]
- Dalle Carbonare, M.; Pathak, M.A. Skin photosensitizing agents and the role of reactive oxygen species in photoageing. J. Photochem. Photobiol. B 1992, 14, 105–124. [Google Scholar]
- Laar von, J.; Stahl, W.; Bolsen, K.; Goerz, G.; Sies, H. β-Carotene serum levels in patients with erythropoietic protoporphyria on treatment with the synthetic all-trans isomer or a natural isomer mixture of β-carotene. J. Photochem. Photobiol. B 1996, 33, 157–162. [Google Scholar]
- Mathews-Roth, M.M. Carotenoids in erythropoietic protoporphyria and other photosensitivity diseases. Ann. N. Y. Acad. Sci. USA 1993, 691, 127–138. [Google Scholar] [CrossRef]
- Mathews-Roth, M.M.; Pathak, M.A.; Parrish, J.A.; Fitzpatrick, T.B.; Kass, E.H.; Toda, K.; Clemens, W. A clinical trial of the effects of oral beta-carotene on the responses of human skin to solar radiation. J. Investig. Dermatol. 1972, 59, 349–353. [Google Scholar]
- Ribaya-Mercado, J.D.; Garmyn, M.; Gilchrest, B.A.; Russell, R.M. Skin lycopene is destroyed preferentially over β-carotene during ultraviolet irradiation in humans. J. Nutr. 1995, 125, 1854–1859. [Google Scholar]
- Garmyn, M.; Ribaya-Mercado, J.D.; Russell, R.M.; Bhawan, J.; Gilchrest, B.A. Effect of beta-carotene supplementation on the human sunburn reaction. Exp. Dermatol. 1995, 4, 104–111. [Google Scholar] [CrossRef]
- Lee, J.; Jiang, S.; Levine, N.; Watson, R.R. Carotenoid supplementation reduces erythema in human skin after simulated solar radiation exposure. Proc. Soc. Exp. Biol. Med. 2000, 223, 170–174. [Google Scholar] [CrossRef]
- Stahl, W.; Heinrich, U.; Jungmann, H.; Sies, H.; Tronnier, H. Carotenoids and carotenoids plus vitamin E protect against ultraviolet light-induced erythema in humans. Am. J. Clin. Nutr. 2000, 71, 795–798. [Google Scholar]
- Stahl, W.; Sies, H. β-Carotene and other carotenoids in protection from sunlight. Am. J. Clin. Nutr. 2012, 96, 1179S–1184S. [Google Scholar] [CrossRef]
- Koepcke, W.; Krutmann, J. Protection from sunburn with β-carotene—A meta-analysis. Photochem. Photobiol. 2008, 84, 284–288. [Google Scholar] [CrossRef]
- Cesarini, J.P.; Michel, L.; Maurette, J.M.; Adhoute, H.; Bejot, M. Immediate effects of UV radiation on the skin: Modification by an antioxidant complex containing carotenoids. Photodermatol. Photoimmunol. Photomed. 2003, 19, 182–189. [Google Scholar] [CrossRef]
- Stahl, W.; Heinrich, U.; Aust, O.; Tronnier, H.; Sies, H. Lycopene-rich products and dietary photoprotection. Photochem. Photobiol. Sci. 2006, 5, 238–242. [Google Scholar]
- Scarmo, S.; Cartmel, B.; Lin, H.; Leffell, D.J.; Welch, E.; Bhosale, P.; Bernstein, P.S.; Mayne, S.T. Significant correlations of dermal total carotenoids and dermal lycopene with their respective plasma levels in healthy adults. Arch. Biochem. Biophys. 2010, 504, 34–39. [Google Scholar] [CrossRef]
- Engelmann, N.; Clinton, S.; Erdman, J. Nutritional aspects of phytoene and phytofluene, carotenoid precursors to lycopene. Adv. Nutr. 2011, 2, 51–61. [Google Scholar] [CrossRef]
- Black, H.S.; deGruijl, F.R.; Forbes, P.D.; Cleaver, J.E.; Ananthaswamy, H.N.; deFabo, E.C.; Ullrich, S.E.; Tyrrell, R.M. Photocarcinogenesis: An overview. J. Photochem. Photobiol. B 1997, 40, 29–47. [Google Scholar]
- Fung, T.T.; Spieglman, D.; Egan, K.M.; Giovannucci, E.; Hunter, D.J.; Willett, W.C. Vitamin and carotenoid intake and risk of squamous cell carcinoma of the skin. Int. J. Cancer 2003, 103, 110–115. [Google Scholar] [CrossRef]
- Dorgan, J.F.; Boakye, N.A.; Fears, T.R.; Schleicher, R.L.; Helsel, W.; Anderson, C.; Robinson, J.; Guin, J.D.; Lessin, S.; Ratnasinghe, L.D.; et al. Serum carotenoids and alpha-tocopherol and risk of nonmelanoma skin cancer. Cancer Epidemiol. Biomark. Prev. 2004, 13, 1276–1282. [Google Scholar]
- Schaumberg, D.A.; Frieling, U.M.; Rifai, N.; Cook, N. No effect of beta-carotene supplementation on risk of nonmelanoma skin cancer among men with low baseline plasma beta-carotene. Cancer Epidemiol. Biomark. Prev. 2004, 13, 1079–1080. [Google Scholar]
- McNaughton, S.A.; Marks, G.C.; Gaffney, P.; Williams, G.; Green, A.C. Antioxidants and basal cell carcinoma of the skin: A nested case-control study. Cancer Causes Control 2005, 16, 609–618. [Google Scholar] [CrossRef]
- Palombo, P.; Fabrizi, G.; Ruocco, V.; Ruocco, E.; Fluhr, J.; Roberts, R.; Morganti, P. Beneficial long-term effects of combined oral/topical antioxidant treatment with the carotenoids lutein and zeaxanthin on human skin: A double-blind, placebo-controlled study. Skin Pharmacol. Physiol. 2007, 20, 199–210. [Google Scholar] [CrossRef]
- Yaar, M.; Gilchrest, B.A. Photoageing: Mechanism, prevention and therapy. Br. J. Dermatol. 2007, 157, 874–887. [Google Scholar] [CrossRef]
- Beatty, S.; Koh, H.H.; Henson, D.; Boulton, M. The role of oxidative stress in the pathogenesis of age-related macular degeneration. Surv. Ophthalmol. 2000, 45, 115–134. [Google Scholar] [CrossRef]
- Koushan, K.; Rusovici, R.; Li, W.; Ferguson, L.R.; Chalam, K.V. The role of lutein in eye-related disease. Nutrients 2013, 5, 1823–1839. [Google Scholar] [CrossRef]
- Loskutova, E.; Nolan, J.; Howard, A.; Beatty, S. Macular pigment and its contribution to vision. Nutrients 2013, 5, 1962–1969. [Google Scholar] [CrossRef]
- Austin, J.; Singhal, N.; Voight, R.; Smaill, F.; Gill, M.J.; Walmsley, S.; Salit, I.; Gimour, J.; Schlech, W.F., III; Choudhri, S.; et al. A community randomized controlled clinical trial of mixed carotenoids and micronutrient supplementation of patients with acquired immunodeficiency syndrome. Eur. J. Clin. Nutr. 2006, 60, 1266–1276. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fiedor, J.; Burda, K. Potential Role of Carotenoids as Antioxidants in Human Health and Disease. Nutrients 2014, 6, 466-488. https://doi.org/10.3390/nu6020466
Fiedor J, Burda K. Potential Role of Carotenoids as Antioxidants in Human Health and Disease. Nutrients. 2014; 6(2):466-488. https://doi.org/10.3390/nu6020466
Chicago/Turabian StyleFiedor, Joanna, and Květoslava Burda. 2014. "Potential Role of Carotenoids as Antioxidants in Human Health and Disease" Nutrients 6, no. 2: 466-488. https://doi.org/10.3390/nu6020466
APA StyleFiedor, J., & Burda, K. (2014). Potential Role of Carotenoids as Antioxidants in Human Health and Disease. Nutrients, 6(2), 466-488. https://doi.org/10.3390/nu6020466



