Anti-Inflammatory Activity of Fruit Fractions in Vitro, Mediated through Toll-Like Receptor 4 and 2 in the Context of Inflammatory Bowel Disease
Abstract
:1. Introduction
2. Experimental Section
2.1. Cell Line and Culture Medium
2.2. Fruits
| Fruits Screened | Selection Criteria | Reference |
|---|---|---|
| Mangosteen | Anti-inflammatory and anti-oxidant properties, high xanthone content | [24,25,26,27] |
| Feijoa | Anti-microbial and anti-oxidant properties | [28] |
| Elderberry | High plant polyphenol content | [29] |
| Cranberry | Chemo-preventative properties including anti-inflammatory effects | [30] |
| Blackcurrant | High plant polyphenol content | [29] |
| Blackberry | High plant polyphenol content | [29] |
| Red raspberry | High plant polyphenol content | [29] |
| Strawberry | High plant polyphenol content | [29] |
| Green Grapes | High plant polyphenol content | [29] |
| Plum | High plant polyphenol content | [29] |
| Pear | High plant polyphenol content | [29] |
| Black Grapes | High plant polyphenol content | [29] |
Fractionation
2.3. HEK-Blue Anti-Inflammatory Screening of Fruit Fractions
Anti-Inflammatory Screening in HEK-Blue Cell Line
3. Results
3.1. HEK Screening Results
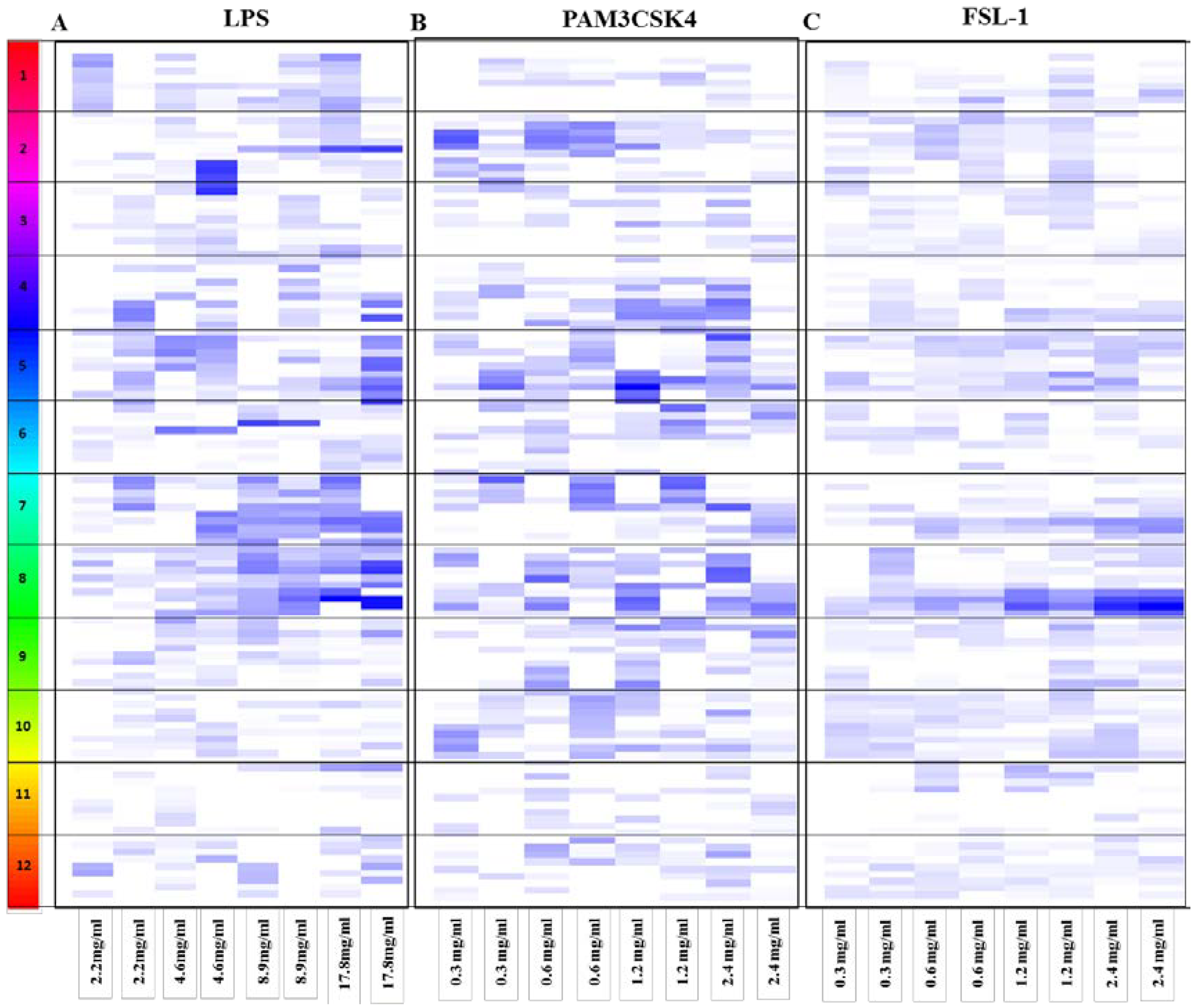
3.2. Dose Response for the Anti-Inflammatory Extracts
| Treated | Fruit & Fraction | Concentration (mg/mL) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 2.2 | 4.4 | 8.9 | 17.8 | ||||||
| A. LPS | Feijoa F4 | −0.03 | 0.00 | 0.03 | 0.02 | 0.05 | 0.09 | 0.16 | 0.15 |
| Feijoa F6 | −0.01 | 0.02 | −0.03 | 0.02 | 0.03 | 0.05 | 0.04 | 0.08 | |
| Black currant F3 | −0.01 | 0.00 | 0.01 | 0.03 | 0.03 | 0.02 | 0.05 | 0.02 | |
| 0.3 | 0.6 | 1.2 | 1.4 | ||||||
| B. PAM3CSK4 | Green grapes F3 | 0.03 | 0.05 | 0.00 | 0.01 | 0.01 | 0.04 | 0.05 | 0.01 |
| Green grapes F4 | 0.03 | 0.02 | 0.03 | 0.02 | 0.02 | 0.04 | 0.05 | 0.01 | |
| Blackberry F4 | 0.03 | 0.04 | 0.01 | 0.04 | 0.04 | 0.09 | 0.05 | 0.04 | |
| Red raspberry F4 | 0.00 | 0.03 | 0.02 | −0.02 | 0.05 | 0.03 | 0.05 | 0.05 | |
| Red raspberry F5 | 0.00 | 0.03 | 0.03 | 0.00 | 0.06 | 0.05 | 0.03 | 0.03 | |
| Strawberry F4 | 0.00 | −0.02 | 0.07 | 0.03 | 0.04 | 0.03 | 0.08 | 0.1 | |
| Strawberry F5 | 0.01 | 0.04 | 0.05 | 0.04 | 0.06 | 0.08 | 0.1 | 0.1 | |
| Feijoa F2 | 0.05 | 0.02 | 0.05 | 0.03 | 0.08 | 0.04 | 0.14 | 0.15 | |
| Feijoa F3 | 0.04 | 0.04 | 0.09 | 0.04 | 0.16 | 0.12 | 0.20 | 0.23 | |
| Feijoa F4 | 0.03 | 0.06 | 0.09 | 0.09 | 0.12 | 0.09 | 0.17 | 0.17 | |
| Feijoa F5 | 0.01 | 0.12 | 0.05 | 0.08 | 0.12 | 0.04 | 0.11 | 0.04 | |
| 0.3 | 0.6 | 1.2 | 1.4 | ||||||
| C. FSL–1 | Blackberry F4 | 0.03 | 0.08 | 0.03 | 0.04 | 0.13 | 0.04 | 0.04 | 0.06 |
| Blackberry F5 | 0.00 | 0.06 | 0.01 | 0.02 | 0.08 | 0.07 | 0.05 | 0.02 | |
| Red raspberry F5 | 0.01 | −0.02 | 0.00 | 0.02 | 0.06 | 0.05 | 0.06 | 0.02 | |
| Green grapes F3 | 0.06 | 0.00 | 0.01 | 0.03 | 0.04 | 0.03 | 0.04 | 0.02 | |
| 2.2 | 4.4 | 8.9 | 17.8 | ||||||
| D. LPS | Feijoa F8 | 0.01 | 0.01 | 0.03 | 0.04 | 0.08 | 0.06 | 0.08 | 0.12 |
| Feijoa F9 | 0.05 | −0.01 | 0.04 | 0.01 | 0.08 | 0.05 | 0.06 | 0.11 | |
| Feijoa F10 | 0.01 | −0.01 | 0.02 | 0.02 | 0.07 | 0.03 | 0.05 | 0.03 | |
| Mangosteen F10 | 0.00 | −0.01 | −0.01 | 0.00 | 0.02 | 0.02 | 0.06 | 0.06 | |
| Elderberry F8 | −0.01 | −0.01 | 0.00 | 0.00 | 0.05 | 0.06 | 0.11 | 0.12 | |
| 0.3 | 0.6 | 1.2 | 1.4 | ||||||
| E. PAM3CSK4 | Elderberry F8 | 0.02 | 0.01 | 0.04 | 0.02 | 0.01 | 0.02 | 0.01 | 0.01 |
| Elderberry F10 | 0.03 | 0.03 | 0.05 | 0.02 | 0.02 | 0.02 | 0.01 | −0.01 | |
| Blackberry F10 | 0.02 | 0.03 | 0.04 | 0.04 | 0.06 | 0.02 | 0.05 | 0.06 | |
| Blackberry F11 | 0.05 | 0.01 | 0.06 | 0.04 | 0.05 | 0.04 | 0.05 | 0.04 | |
| 0.3 | 0.6 | 1.2 | 1.4 | ||||||
| F. FSL–1 | Black grapes F10 | 0.00 | −0.01 | 0.01 | 0.02 | 0.06 | 0.05 | 0.04 | 0.04 |
| Cranberry F9 | 0.00 | −0.01 | 0.02 | 0.00 | 0.02 | 0.06 | 0.03 | 0.02 | |
4. Discussion
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tsianos, E.V.; Katsanos, K. Do we really understand what the immunological disturbances in inflammatory bowel disease mean? World J. Gastroenterol. 2009, 15, 521–525. [Google Scholar] [CrossRef]
- Takeuchi, O.; Akira, S. Pattern recognition receptors and inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Savill, J. Resolution of inflammation: The beginning programs the end. Nat. Immunol. 2005, 6, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. Inflammation 2010: New adventures of an old flame. Cell 2010, 140, 771–776. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Raoof, M.; Chen, Y.; Sumi, Y.; Sursal, T.; Junger, W.; Brohi, K.; Itagaki, K.; Hauser, C.J. Circulating mitochondrial damps cause inflammatory responses to injury. Nature 2010, 464, 104–107. [Google Scholar] [CrossRef]
- Yu, M.; Wang, H.; Ding, A.; Golenbock, D.T.; Latz, E.; Czura, C.J.; Fenton, M.J.; Tracey, K.J.; Yang, H. Hmgb1 signals through Toll-like receptor (TLR) 4 and TLR 2. Shock 2006, 26, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Asea, A.; Rehli, M.; Kabingu, E.; Boch, J.A.; Baré, O.; Auron, P.E.; Stevenson, M.A.; Calderwood, S.K. Novel signal transduction pathway utilized by extracellular HSP70 role of Toll-like receptor (TLR) 2 and TLR 4. J. Biol. Chem. 2002, 277, 15028–15034. [Google Scholar] [PubMed]
- Fiocchi, C. Genes and “in-vironment”: How will our concepts on the pathophysiology of inflammatory bowel disease develop in the future? Dig. Dis. 2013, 30, 2–11. [Google Scholar] [CrossRef]
- Cario, E.; Podolsky, D.K. Differential alteration in intestinal epithelial cell expression of Toll-like receptor 3 (TLR 3) and TLR 4 in inflammatory bowel disease. Infect. Immun. 2000, 68, 7010–7017. [Google Scholar] [CrossRef] [PubMed]
- Abreu, M.T.; Vora, P.; Faure, E.; Thomas, L.S.; Arnold, E.T.; Arditi, M. Decreased expression of Toll-like receptor-4 and MD-2 correlates with intestinal epithelial cell protection against dysregulated proinflammatory gene expression in response to bacterial lipopolysaccharide. J. Immunol. 2001, 167, 1609–1616. [Google Scholar] [CrossRef] [PubMed]
- Otte, J.M.; Cario, E.; Podolsky, D.K. Mechanisms of cross hyporesponsiveness to Toll-like receptor bacterial ligands in intestinal epithelial cells. Gastroenterology 2004, 126, 1054–1070. [Google Scholar] [CrossRef] [PubMed]
- Hommes, D.W. Risks and benefits of biologic therapy for IBD. J. Pediatr. Gastroenterol. Nutr. 2009, 48, 52–53. [Google Scholar] [CrossRef]
- Seow, C.H.; de Silva, S.; Kaplan, G.G.; Devlin, S.M.; Ghosh, S.; Panaccione, R. Managing the risks of IBD therapy. Curr. Gastroenterol. Rep. 2009, 11, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Li, F.X.; Verhoef, M.J.; Best, A.; Otley, A.; Hilsden, R.J. Why patients with inflammatory bowel disease use or do not use complementary and alternative medicine: A canadian national survey. Can. J. Gastroenterol. 2005, 19, 567–573. [Google Scholar] [PubMed]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 2009, 56, 317–333. [Google Scholar] [CrossRef]
- Del Rio, D.; Costa, L.; Lean, M.; Crozier, A. Polyphenols and health: What compounds are involved? Nutr. Metab. Cardiovasc. Dis. 2010, 20, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.; Ballester, I.; Lopez-Posadas, R.; Suarez, M.; Zarzuelo, A.; Martinez-Augustin, O.; de Medina, F.S. Effects of flavonoids and other polyphenols on inflammation. Crit. Rev. Food Sci. Nutr. 2011, 51, 331–362. [Google Scholar] [CrossRef] [PubMed]
- Priego, S.; Feddi, F.; Ferrer, P.; Mena, S.; Benlloch, M.; Ortega, A.; Carretero, J.; Obrador, E.; Asensi, M.; Estrela, J.M.; et al. Natural polyphenols facilitate elimination of HT-29 colorectal cancer xenografts by chemoradiotherapy: A Bcl-2-and superoxide dismutase 2-dependent mechanism. Mol. Cancer Ther. 2008, 7, 3330–3342. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Manach, C.; Morand, C.; Remesy, C.; Jimenez, L. Dietary polyphenols and the prevention of diseases. Crit. Rev. Food Sci. Nutr. 2005, 45, 287–306. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Morand, C.; Manach, C.; Rémésy, C. Absorption and metabolism of polyphenols in the gut and impact on health. Biomed. Pharmacother. 2002, 56, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, H.; Singer, P.; Halpern, Z.; Bruck, R. Polyphenols in the treatment of inflammatory bowel disease and acute pancreatitis. Gut 2007, 56, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Polyphenols and health: Update and perspectives. Arch. Biochem. Biophys. 2010, 501, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Bronstein, I.; Fortin, J.; Voyta, J.; Juo, R.; Edwards, B.; Olesen, C.; Lijam, N.; Kricka, L. Chemiluminescent reporter gene assays: Sensitive detection of the gus and seap gene products. Biotechniques 1994, 17, 172–177. [Google Scholar] [PubMed]
- Bumrungpert, A.; Kalpravidh, R.W.; Chitchumroonchokchai, C.; Chuang, C.C.; West, T.; Kennedy, A.; McIntosh, M. Xanthones from mangosteen prevent lipopolysaccharide-mediated inflammation and insulin resistance in primary cultures of human adipocytes. J. Nutr. 2009, 139, 1185–1191. [Google Scholar] [CrossRef] [PubMed]
- Bumrungpert, A.; Kalpravidh, R.W.; Suksamrarn, S.; Chaivisuthangkura, A.; Chitchumroonchokchai, C.; Failla, M.L. Bioaccessibility, biotransformation, and transport of α-mangostin from Garcinia mangostana (mangosteen) using simulated digestion and Caco-2 human intestinal cells. Mol. Nutr. Food Res. 2009, 53, 54–61. [Google Scholar] [CrossRef]
- Chen, L.-G.; Yang, L.-L.; Wang, C.-C. Anti-inflammatory activity of mangostins from Garcinia mangostana. Food Chem. Toxicol. 2008, 46, 688–693. [Google Scholar] [PubMed]
- Pedraza-Chaverri, J.; Cárdenas-Rodríguez, N.; Orozco-Ibarra, M.; Pérez-Rojas, J.M. Medicinal properties of mangosteen (Garcinia mangostana). Food Chem. Toxicol. 2008, 46, 3227–3239. [Google Scholar] [CrossRef] [PubMed]
- Vuotto, M.L.; Basile, A.; Moscatiello, V.; de Sole, P.; Castaldo-Cobianchi, R.; Laghi, E.; Ielpo, M.T.L. Antimicrobial and antioxidant activities of Feijoa sellowiana fruit. Int. J. Antimicrob. Agents 2000, 13, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Perez-Jimenez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 richest dietary sources of polyphenols: An application of the phenol-explorer database. Eur. J. Clin. Nutr. 2010, 64 (Suppl. 3), 112–120. [Google Scholar] [CrossRef]
- Neto, C.C. Cranberry and its phytochemicals: A review of in vitro anticancer studies. J. Nutr. 2007, 137, 186–193. [Google Scholar]
- Cario, E.; Gerken, G.; Podolsky, D.K. Toll-like receptor 2 controls mucosal inflammation by regulating epithelial barrier function. Gastroenterology 2007, 132, 1359–1374. [Google Scholar] [CrossRef] [PubMed]
- Covert, M.W.; Leung, T.H.; Gaston, J.E.; Baltimore, D. Achieving stability of lipopolysaccharide-induced NF-κb activation. Science 2005, 309, 1854–1857. [Google Scholar] [PubMed]
- Motohashi, N.; Kawase, M.; Shirataki, Y.; Tani, S.; Saito, S.; Sakagami, H.; Kurihara, T.; Nakashima, H.; Wolfard, K.; Mucsi, I.; et al. Biological activity of feijoa peel extracts. Anticancer Res. 2000, 20, 4323–4330. [Google Scholar] [PubMed]
- Giuseppe, R.; Corrado, T. Secondary metabolites from the leaves of feijoa sellowiana berg. Phytochemistry 2004, 65, 2947–2951. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.; Rigano, D.; Pergola, C.; Formisano, C.; Basile, A.; Bramanti, P.; Senatore, F.; Sautebin, L. Inhibition of inducible nitric oxide synthase expression by an acetonic extract from Feijoa sellowiana berg fruits. J. Agric. Food Chem. 2007, 55, 5053–5061. [Google Scholar]
- Cario, E. Barrier-protective function of intestinal epithelial Toll-like receptor 2. Mucosal Immunol. 2008, 1, 62–66. [Google Scholar] [CrossRef]
- Neilson, A.; George, J.; Janle, E.; Mattes, R.; Rudolph, R.; Matusheski, N.; Ferruzzi, M. Influence of chocolate matrix composition on cocoa flavan-3-ol bioaccessibility in vitro and bioavailability in humans. J. Agric. Food Chem. 2009, 57, 9418–9426. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Sang, S.; Lambert, J.D.; Lee, M.J. Bioavailability issues in studying the health effects of plant polyphenolic compounds. Mol. Nutr. Food Res. 2008, 52, 139–151. [Google Scholar]
- Neilson, A.; Ferruzzi, M. Bioavailability and metabolism of bioactive compounds from foods. In Nutrition in the Prevention and Treatment of Disease, 3rd ed.; Coulston, A., Boushey, C., Ferruzzi, M., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2012. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nasef, N.A.; Mehta, S.; Murray, P.; Marlow, G.; Ferguson, L.R. Anti-Inflammatory Activity of Fruit Fractions in Vitro, Mediated through Toll-Like Receptor 4 and 2 in the Context of Inflammatory Bowel Disease. Nutrients 2014, 6, 5265-5279. https://doi.org/10.3390/nu6115265
Nasef NA, Mehta S, Murray P, Marlow G, Ferguson LR. Anti-Inflammatory Activity of Fruit Fractions in Vitro, Mediated through Toll-Like Receptor 4 and 2 in the Context of Inflammatory Bowel Disease. Nutrients. 2014; 6(11):5265-5279. https://doi.org/10.3390/nu6115265
Chicago/Turabian StyleNasef, Noha Ahmed, Sunali Mehta, Pamela Murray, Gareth Marlow, and Lynnette R. Ferguson. 2014. "Anti-Inflammatory Activity of Fruit Fractions in Vitro, Mediated through Toll-Like Receptor 4 and 2 in the Context of Inflammatory Bowel Disease" Nutrients 6, no. 11: 5265-5279. https://doi.org/10.3390/nu6115265
APA StyleNasef, N. A., Mehta, S., Murray, P., Marlow, G., & Ferguson, L. R. (2014). Anti-Inflammatory Activity of Fruit Fractions in Vitro, Mediated through Toll-Like Receptor 4 and 2 in the Context of Inflammatory Bowel Disease. Nutrients, 6(11), 5265-5279. https://doi.org/10.3390/nu6115265






