n-3 Polyunsaturated Fatty Acids and Mechanisms to Mitigate Inflammatory Paracrine Signaling in Obesity-Associated Breast Cancer
Abstract
:1. Introduction
2. Obese Inflammatory Phenotype
3. n-3 Polyunsaturated Fatty Acids and Obesity
4. n-3 Polyunsaturated Fatty Acids and Breast Cancer
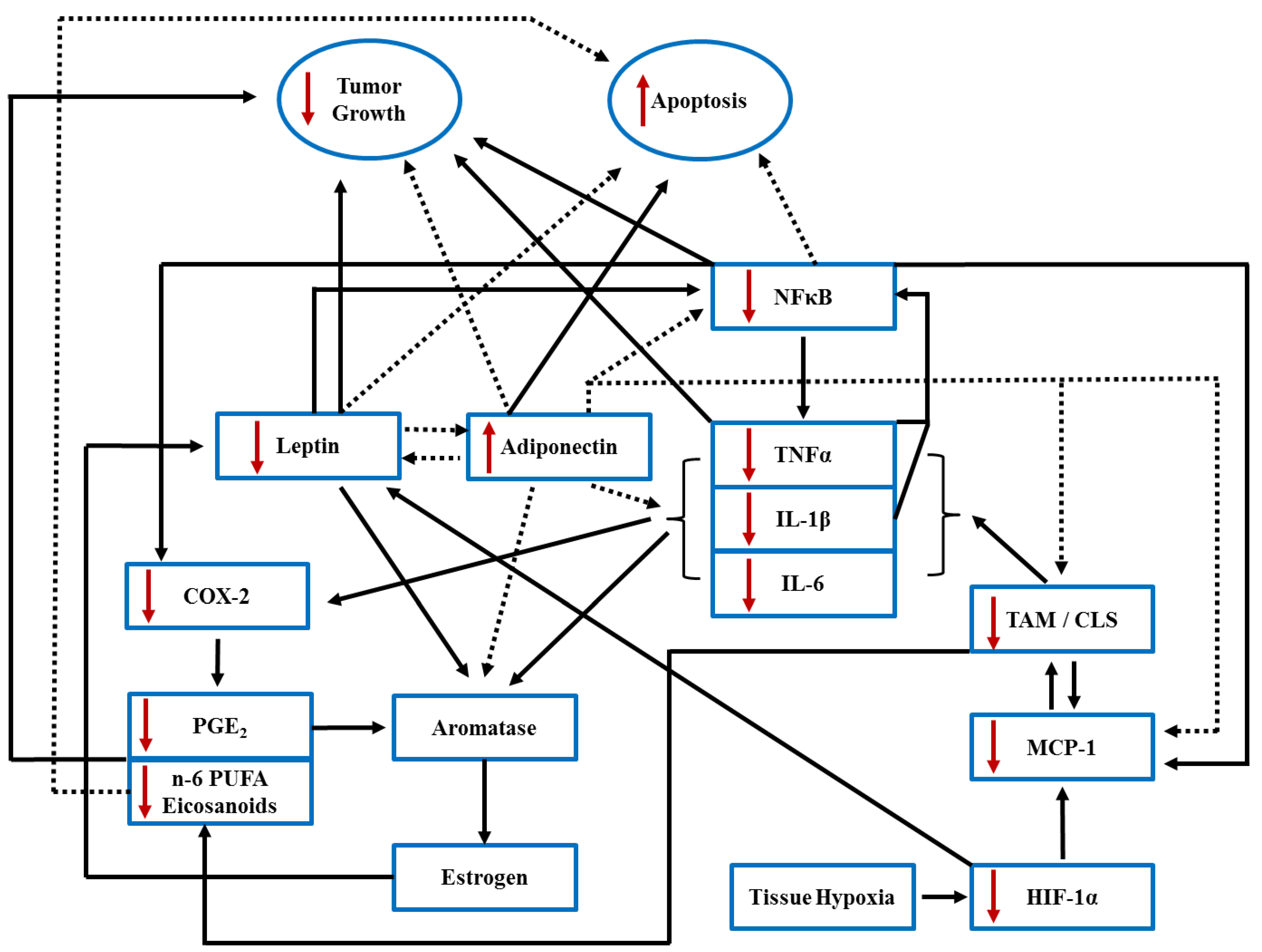
5. n-3 PUFA, Lipid Rafts and Breast Cancer
6. Paracrine Interactions, Inflammatory Mediator Signaling and Breast Cancer
7. The Role of Estrogen and Aromatase Activation in BC
8. Inflammatory and Chemopromotive Fatty Acid Derived Lipid Mediators: Differential Effects of n-6 versus n-3 PUFA
9. Role of Inflammatory Cytokines
10. Role of Leptin
11. Role of Adiponectin
12. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Y.C.; McPherson, K.; Marsh, T.; Gortmaker, S.L.; Brown, M. Health and economic burden of the projected obesity trends in the USA and the UK. Lancet 2011, 378, 815–825. [Google Scholar] [CrossRef] [PubMed]
- Lumeng, C.N.; Saltiel, A.R. Inflammatory links between obesity and metabolic disease. J. Clin. Investig. 2011, 121, 2111–2117. [Google Scholar] [CrossRef] [PubMed]
- Calle, E.E.; Rodriguez, C.; Walker-Thurmond, K.; Thun, M.J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N. Engl. J. Med. 2003, 348, 1625–1638. [Google Scholar] [CrossRef] [PubMed]
- Van Kruijsdijk, R.C.; van der Wall, E.; Visseren, F.L. Obesity and cancer: The role of dysfunctional adipose tissue. Cancer Epidemiol. Biomark. Prev. 2009, 18, 2569–2578. [Google Scholar] [CrossRef]
- Cleary, M.P.; Grossmann, M.E. Minireview: Obesity and breast cancer: The estrogen connection. Endocrinology 2009, 150, 2537–2542. [Google Scholar] [CrossRef] [PubMed]
- Key, T.J.; Appleby, P.N.; Reeves, G.K.; Roddam, A.; Dorgan, J.F.; Longcope, C.; Stanczyk, F.Z.; Stephenson, H.E., Jr.; Falk, R.T.; Miller, R.; et al. Body mass index, serum sex hormones, and breast cancer risk in postmenopausal women. J. Natl. Cancer Inst. 2003, 95, 1218–1226. [Google Scholar] [CrossRef] [PubMed]
- van den Brandt, P.A.; Spiegelman, D.; Yaun, S.S.; Adami, H.O.; Beeson, L.; Folsom, A.R.; Fraser, G.; Goldbohm, R.A.; Graham, S.; Kushi, L.; et al. Pooled analysis of prospective cohort studies on height, weight, and breast cancer risk. Am. J. Epidemiol. 2000, 152, 514–527. [Google Scholar]
- Li, C.I.; Daling, J.R.; Malone, K.E. Incidence of invasive breast cancer by hormone receptor status from 1992 to 1998. J. Clin. Oncol. 2003, 21, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Biglia, N.; Peano, E.; Sgandurra, P.; Moggio, G.; Pecchio, S.; Maggiorotto, F.; Sismondi, P. Body mass index (bmi) and breast cancer: Impact on tumor histopathologic features, cancer subtypes and recurrence rate in pre and postmenopausal women. Gynecol. Endocrinol. 2013, 29, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Garrisi, V.M.; Tufaro, A.; Trerotoli, P.; Bongarzone, I.; Quaranta, M.; Ventrella, V.; Tommasi, S.; Giannelli, G.; Paradiso, A. Body mass index and serum proteomic profile in breast cancer and healthy women: A prospective study. PLoS One 2012, 7, e49631. [Google Scholar] [CrossRef] [PubMed]
- Kamineni, A.; Anderson, M.L.; White, E.; Taplin, S.H.; Porter, P.; Ballard-Barbash, R.; Malone, K.; Buist, D.S. Body mass index, tumor characteristics, and prognosis following diagnosis of early-stage breast cancer in a mammographically screened population. Cancer Causes Control 2013, 24, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, S.; Key, T.J.; Peeters, P.H.; Lahmann, P.H.; Lukanova, A.; Dossus, L.; Biessy, C.; Vineis, P.; Sacerdote, C.; Berrino, F.; et al. Anthropometric measures, endogenous sex steroids and breast cancer risk in postmenopausal women: A study within the epic cohort. Int. J. Cancer 2006, 118, 2832–2839. [Google Scholar] [CrossRef] [PubMed]
- John, E.M.; Phipps, A.I.; Sangaramoorthy, M. Body size, modifying factors, and postmenopausal breast cancer risk in a multiethnic population: The San Francisco bay area breast cancer study. SpringerPlus 2013, 2, 239. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, K.; Bassett, J.K.; MacInnis, R.J.; English, D.R.; Hopper, J.L.; McLean, C.; Giles, G.G.; Baglietto, L. Associations between weight in early adulthood, change in weight, and breast cancer risk in postmenopausal women. Cancer Epidemiol. Biomark. Prev. 2013, 22, 1409–1416. [Google Scholar] [CrossRef]
- Cheraghi, Z.; Poorolajal, J.; Hashem, T.; Esmailnasab, N.; Doosti Irani, A. Effect of body mass index on breast cancer during premenopausal and postmenopausal periods: A meta-analysis. PLoS One 2012, 7, e51446. [Google Scholar] [CrossRef] [PubMed]
- Pierobon, M.; Frankenfeld, C.L. Obesity as a risk factor for triple-negative breast cancers: A systematic review and meta-analysis. Breast Cancer Res. Treat. 2013, 137, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Amadou, A.; Ferrari, P.; Muwonge, R.; Moskal, A.; Biessy, C.; Romieu, I.; Hainaut, P. Overweight, obesity and risk of premenopausal breast cancer according to ethnicity: A systematic review and dose-response meta-analysis. Obes. Rev. 2013, 14, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.A. Impact of obesity on mammary gland inflammation and local estrogen production. J. Mammary Gland Biol. Neoplasia 2014, 19, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, T.; Goel, K.; Correa de Sa, D.; Carter, R.E.; Hodge, D.O.; Kragelund, C.; Kanaya, A.M.; Zeller, M.; Park, J.S.; Kober, L.; et al. Combining body mass index with measures of central obesity in the assessment of mortality in subjects with coronary disease: Role of “Normal weight central obesity”. J. Am. Coll. Cardiol. 2013, 61, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Tanaka, S.; Iwamoto, M.; Fujioka, H.; Takahashi, Y.; Satou, N.; Uchiyama, K. Association between body mass index and breast cancer intrinsic subtypes in Japanese women. Exp. Ther. Med. 2012, 4, 391–396. [Google Scholar] [PubMed]
- Daling, J.R.; Malone, K.E.; Doody, D.R.; Johnson, L.G.; Gralow, J.R.; Porter, P.L. Relation of body mass index to tumor markers and survival among young women with invasive ductal breast carcinoma. Cancer 2001, 92, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Fagherazzi, G.; Chabbert-Buffet, N.; Fabre, A.; Guillas, G.; Boutron-Ruault, M.C.; Mesrine, S.; Clavel-Chapelon, F. Hip circumference is associated with the risk of premenopausal er-/pr- breast cancer. Int. J. Obes. 2012, 36, 431–439. [Google Scholar] [CrossRef]
- Olefsky, J.M.; Glass, C.K. Macrophages, inflammation, and insulin resistance. Annu. Rev. Physiol. 2010, 72, 219–246. [Google Scholar] [CrossRef]
- Pierce, B.L.; Ballard-Barbash, R.; Bernstein, L.; Baumgartner, R.N.; Neuhouser, M.L.; Wener, M.H.; Baumgartner, K.B.; Gilliland, F.D.; Sorensen, B.E.; McTiernan, A.; et al. Elevated biomarkers of inflammation are associated with reduced survival among breast cancer patients. J. Clin. Oncol. 2009, 27, 3437–3444. [Google Scholar] [CrossRef] [PubMed]
- Woodward, W.A.; Cristofanilli, M. Inflammatory breast cancer. Semin. Radiat. Oncol. 2009, 19, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Cariati, M.; Bennett-Britton, T.M.; Pinder, S.E.; Purushotham, A.D. “Inflammatory” Breast cancer. Surg. Oncol. 2005, 14, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Rossmeisl, M.; Jilkova, Z.M.; Kuda, O.; Jelenik, T.; Medrikova, D.; Stankova, B.; Kristinsson, B.; Haraldsson, G.G.; Svensen, H.; Stoknes, I.; et al. Metabolic effects of n-3 pufa as phospholipids are superior to triglycerides in mice fed a high-fat diet: Possible role of endocannabinoids. PLoS One 2012, 7, e38834. [Google Scholar] [CrossRef] [PubMed]
- Rossmeisl, M.; Medrikova, D.; van Schothorst, E.M.; Pavlisova, J.; Kuda, O.; Hensler, M.; Bardova, K.; Flachs, P.; Stankova, B.; Vecka, M.; et al. Omega-3 phospholipids from fish suppress hepatic steatosis by integrated inhibition of biosynthetic pathways in dietary obese mice. Biochim. Biophys. Acta 2014, 1841, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Kalupahana, N.S.; Claycombe, K.; Newman, S.J.; Stewart, T.; Siriwardhana, N.; Matthan, N.; Lichtenstein, A.H.; Moustaid-Moussa, N. Eicosapentaenoic acid prevents and reverses insulin resistance in high-fat diet-induced obese mice via modulation of adipose tissue inflammation. J. Nutr. 2010, 140, 1915–1922. [Google Scholar] [CrossRef] [PubMed]
- Flachs, P.; Mohamed-Ali, V.; Horakova, O.; Rossmeisl, M.; Hosseinzadeh-Attar, M.J.; Hensler, M.; Ruzickova, J.; Kopecky, J. Polyunsaturated fatty acids of marine origin induce adiponectin in mice fed a high-fat diet. Diabetologia 2006, 49, 394–397. [Google Scholar] [CrossRef] [PubMed]
- Neschen, S.; Morino, K.; Rossbacher, J.C.; Pongratz, R.L.; Cline, G.W.; Sono, S.; Gillum, M.; Shulman, G.I. Fish oil regulates adiponectin secretion by a peroxisome proliferator-activated receptor-gamma-dependent mechanism in mice. Diabetes 2006, 55, 924–928. [Google Scholar] [CrossRef] [PubMed]
- Itoh, M.; Suganami, T.; Satoh, N.; Tanimoto-Koyama, K.; Yuan, X.; Tanaka, M.; Kawano, H.; Yano, T.; Aoe, S.; Takeya, M.; et al. Increased adiponectin secretion by highly purified eicosapentaenoic acid in rodent models of obesity and human obese subjects. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1918–1925. [Google Scholar] [CrossRef] [PubMed]
- Nomura, S.; Shouzu, A.; Omoto, S.; Inami, N.; Ueba, T.; Urase, F.; Maeda, Y. Effects of eicosapentaenoic acid on endothelial cell-derived microparticles, angiopoietins and adiponectin in patients with type 2 diabetes. J. Atheroscler. Thromb. 2009, 16, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Gammelmark, A.; Madsen, T.; Varming, K.; Lundbye-Christensen, S.; Schmidt, E.B. Low-dose fish oil supplementation increases serum adiponectin without affecting inflammatory markers in overweight subjects. Nutr. Res. 2012, 32, 15–23. [Google Scholar] [CrossRef]
- Todoric, J.; Loffler, M.; Huber, J.; Bilban, M.; Reimers, M.; Kadl, A.; Zeyda, M.; Waldhausl, W.; Stulnig, T.M. Adipose tissue inflammation induced by high-fat diet in obese diabetic mice is prevented by n-3 polyunsaturated fatty acids. Diabetologia 2006, 49, 2109–2119. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Ide, T. Dietary n-3 fatty acids affect mrna level of brown adipose tissue uncoupling protein 1, and white adipose tissue leptin and glucose transporter 4 in the rat. Br. J. Nutr. 2000, 84, 175–184. [Google Scholar] [PubMed]
- Ruzickova, J.; Rossmeisl, M.; Prazak, T.; Flachs, P.; Sponarova, J.; Veck, M.; Tvrzicka, E.; Bryhn, M.; Kopecky, J. Omega-3 pufa of marine origin limit diet-induced obesity in mice by reducing cellularity of adipose tissue. Lipids 2004, 39, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Winnicki, M.; Somers, V.K.; Accurso, V.; Phillips, B.G.; Puato, M.; Palatini, P.; Pauletto, P. Fish-rich diet, leptin, and body mass. Circulation 2002, 106, 289–291. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.A.; Burke, V.; Puddey, I.B.; Shaw, J.E.; Beilin, L.J. Effect of fish diets and weight loss on serum leptin concentration in overweight, treated-hypertensive subjects. J. Hypertens. 2004, 22, 1983–1990. [Google Scholar] [CrossRef] [PubMed]
- Perez-Echarri, N.; Perez-Matute, P.; Marcos-Gomez, B.; Baena, M.J.; Marti, A.; Martinez, J.A.; Moreno-Aliaga, M.J. Differential inflammatory status in rats susceptible or resistant to diet-induced obesity: Effects of epa ethyl ester treatment. Eur. J. Nutr. 2008, 47, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Puglisi, M.J.; Hasty, A.H.; Saraswathi, V. The role of adipose tissue in mediating the beneficial effects of dietary fish oil. J. Nutr. Biochem. 2011, 22, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Connolly, J.M.; Gilhooly, E.M.; Rose, D.P. Effects of reduced dietary linoleic acid intake, alone or combined with an algal source of docosahexaenoic acid, on mda-mb-231 breast cancer cell growth and apoptosis in nude mice. Nutr. Cancer 1999, 35, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Hardman, W.E.; Munoz, J., Jr.; Cameron, I.L. Role of lipid peroxidation and antioxidant enzymes in omega 3 fatty acids induced suppression of breast cancer xenograft growth in mice. Cancer Cell Int. 2002, 2, 10. [Google Scholar] [CrossRef] [PubMed]
- Hardman, W.E.; Sun, L.; Short, N.; Cameron, I.L. Dietary omega-3 fatty acids and ionizing irradiation on human breast cancer xenograft growth and angiogenesis. Cancer Cell Int. 2005, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Howe, L.R.; Subbaramaiah, K.; Hudis, C.A.; Dannenberg, A.J. Molecular pathways: Adipose inflammation as a mediator of obesity-associated cancer. Clin. Cancer Res. 2013, 19, 6074–6083. [Google Scholar] [CrossRef] [PubMed]
- Leslie, M.A.; Abdelmagid, S.A.; Perez, K.; Muller, W.J.; Ma, D.W. Mammary tumour development is dose-dependently inhibited by n-3 polyunsaturated fatty acids in the mmtv-neu(ndl)-yd5 transgenic mouse model. Lipids Health Dis. 2014, 13, 96. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.W.; Ngo, V.; Huot, P.S.; Kang, J.X. n-3 polyunsaturated fatty acids endogenously synthesized in fat-1 mice are enriched in the mammary gland. Lipids 2006, 41, 35–39. [Google Scholar] [CrossRef] [PubMed]
- MacLennan, M.B.; Clarke, S.E.; Perez, K.; Wood, G.A.; Muller, W.J.; Kang, J.X.; Ma, D.W. Mammary tumor development is directly inhibited by lifelong n-3 polyunsaturated fatty acids. J. Nutr. Biochem. 2013, 24, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Mandal, C.C.; Ghosh-Choudhury, T.; Yoneda, T.; Choudhury, G.G.; Ghosh-Choudhury, N. Fish oil prevents breast cancer cell metastasis to bone. Biochem. Biophys. Res. Commun. 2010, 402, 602–607. [Google Scholar] [CrossRef] [PubMed]
- Manna, S.; Chakraborty, T.; Ghosh, B.; Chatterjee, M.; Panda, A.; Srivastava, S.; Rana, A.; Chatterjee, M. Dietary fish oil associated with increased apoptosis and modulated expression of bax and bcl-2 during 7,12-dimethylbenz(alpha)anthracene-induced mammary carcinogenesis in rats. Prostaglandins Leukot. Essent. Fatty Acids 2008, 79, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Manna, S.; Janarthan, M.; Ghosh, B.; Rana, B.; Rana, A.; Chatterjee, M. Fish oil regulates cell proliferation, protect DNA damages and decrease her-2/neu and c-myc protein expression in rat mammary carcinogenesis. Clin. Nutr. 2010, 29, 531–537. [Google Scholar] [CrossRef] [PubMed]
- Menendez, J.A.; Lupu, R.; Colomer, R. Exogenous supplementation with omega-3 polyunsaturated fatty acid docosahexaenoic acid (dha; 22:6n-3) synergistically enhances taxane cytotoxicity and downregulates her-2/neu (c-erbb-2) oncogene expression in human breast cancer cells. Eur. J. Cancer Prev. 2005, 14, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Menendez, J.A.; Vazquez-Martin, A.; Ropero, S.; Colomer, R.; Lupu, R. Her2 (erbb-2)-targeted effects of the omega-3 polyunsaturated fatty acid, alpha-linolenic acid (ala; 18:3n-3), in breast cancer cells: The “Fat features” Of the “Mediterranean diet” As an “Anti-her2 cocktail”. Clin. Transl. Oncol. 2006, 8, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, N.K.; Rogozina, O.P.; Seppanen, C.M.; Liao, D.J.; Cleary, M.P.; Grossmann, M.E. Combination of intermittent calorie restriction and eicosapentaenoic acid for inhibition of mammary tumors. Cancer Prev. Res. 2013, 6, 540–547. [Google Scholar] [CrossRef]
- Yee, L.D.; Young, D.C.; Rosol, T.J.; Vanbuskirk, A.M.; Clinton, S.K. Dietary (n-3) polyunsaturated fatty acids inhibit her-2/neu-induced breast cancer in mice independently of the ppargamma ligand rosiglitazone. J. Nutr. 2005, 135, 983–988. [Google Scholar] [PubMed]
- Zou, Z.; Bellenger, S.; Massey, K.A.; Nicolaou, A.; Geissler, A.; Bidu, C.; Bonnotte, B.; Pierre, A.S.; Minville-Walz, M.; Rialland, M.; et al. Inhibition of the her2 pathway by n-3 polyunsaturated fatty acids prevents breast cancer in fat-1 transgenic mice. J. Lipid Res. 2013, 54, 3453–3463. [Google Scholar] [CrossRef] [PubMed]
- Kershaw, E.E.; Flier, J.S. Adipose tissue as an endocrine organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556. [Google Scholar] [CrossRef] [PubMed]
- Trayhurn, P.; Wood, I.S. Adipokines: Inflammation and the pleiotropic role of white adipose tissue. Br. J. Nutr. 2004, 92, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Balistreri, C.R.; Caruso, C.; Candore, G. The role of adipose tissue and adipokines in obesity-related inflammatory diseases. Mediat. Inflamm. 2010, 2010, 802078. [Google Scholar] [CrossRef]
- Surmi, B.K.; Hasty, A.H. Macrophage infiltration into adipose tissue: Initiation, propagation and remodeling. Future Lipidol. 2008, 3, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.G.; Javorschi, S.; Hevener, A.L.; Kruszynska, Y.T.; Norman, R.A.; Sinha, M.; Olefsky, J.M. The effect of thiazolidinediones on plasma adiponectin levels in normal, obese, and type 2 diabetic subjects. Diabetes 2002, 51, 2968–2974. [Google Scholar] [CrossRef] [PubMed]
- Peyron-Caso, E.; Fluteau-Nadler, S.; Kabir, M.; Guerre-Millo, M.; Quignard-Boulange, A.; Slama, G.; Rizkalla, S.W. Regulation of glucose transport and transporter 4 (glut-4) in muscle and adipocytes of sucrose-fed rats: Effects of n-3 poly- and monounsaturated fatty acids. Horm. Metab. Res. 2002, 34, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Tishinsky, J.M.; de Boer, A.A.; Dyck, D.J.; Robinson, L.E. Modulation of visceral fat adipokine secretion by dietary fatty acids and ensuing changes in skeletal muscle inflammation. Appl. Physiol. Nutr. Metab. 2014, 39, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Periz, A.; Horrillo, R.; Ferre, N.; Gronert, K.; Dong, B.; Moran-Salvador, E.; Titos, E.; Martinez-Clemente, M.; Lopez-Parra, M.; Arroyo, V.; et al. Obesity-induced insulin resistance and hepatic steatosis are alleviated by omega-3 fatty acids: A role for resolvins and protectins. FASEB J. 2009, 23, 1946–1957. [Google Scholar] [CrossRef] [PubMed]
- Oster, R.T.; Tishinsky, J.M.; Yuan, Z.; Robinson, L.E. Docosahexaenoic acid increases cellular adiponectin mRNA and secreted adiponectin protein, as well as ppargamma mrna, in 3t3-l1 adipocytes. Appl. Physiol. Nutr. Metab. 2010, 35, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Tishinsky, J.M.; Ma, D.W.; Robinson, L.E. Eicosapentaenoic acid and rosiglitazone increase adiponectin in an additive and ppargamma-dependent manner in human adipocytes. Obesity 2011, 19, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Glass, C.K.; Saijo, K. Nuclear receptor transrepression pathways that regulate inflammation in macrophages and t cells. Nat. Rev. Immunol. 2010, 10, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Szanto, A.; Nagy, L. The many faces of ppargamma: Anti-inflammatory by any means? Immunobiology 2008, 213, 789–803. [Google Scholar] [CrossRef]
- Oh, D.Y.; Talukdar, S.; Bae, E.J.; Imamura, T.; Morinaga, H.; Fan, W.; Li, P.; Lu, W.J.; Watkins, S.M.; Olefsky, J.M. Gpr120 is an omega-3 fatty acid receptor mediating potent anti-inflammatory and insulin-sensitizing effects. Cell 2010, 142, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Stulnig, T.M.; Huber, J.; Leitinger, N.; Imre, E.M.; Angelisova, P.; Nowotny, P.; Waldhausl, W. Polyunsaturated eicosapentaenoic acid displaces proteins from membrane rafts by altering raft lipid composition. J. Biol. Chem. 2001, 276, 37335–37340. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Feeding the immune system. Proc. Nutr. Soc. 2013, 72, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. n-3 fatty acids, inflammation and immunity: New mechanisms to explain old actions. Proc. Nutr. Soc. 2013, 72, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ma, D.W.L. The role of n-3 polyunsaturated fatty acids in the prevention and treatment of breast cancer. Nutrients 2014, in press. [Google Scholar]
- Conquer, J.A.; Holub, B.J. Effect of supplementation with different doses of dha on the levels of circulating dha as non-esterified fatty acid in subjects of asian indian background. J. Lipid Res. 1998, 39, 286–292. [Google Scholar] [PubMed]
- Kris-Etherton, P.M.; Taylor, D.S.; Yu-Poth, S.; Huth, P.; Moriarty, K.; Fishell, V.; Hargrove, R.L.; Zhao, G.; Etherton, T.D. Polyunsaturated fatty acids in the food chain in the United States. Am. J. Clin. Nutr. 2000, 71, 179S–188S. [Google Scholar] [PubMed]
- Damsgaard, C.T.; Frokiaer, H.; Lauritzen, L. The effects of fish oil and high or low linoleic acid intake on fatty acid composition of human peripheral blood mononuclear cells. Br. J. Nutr. 2008, 99, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Feskens, E.J.; Kromhout, D. Epidemiologic studies on eskimos and fish intake. Ann. N. Y. Acad. Sci. 1993, 683, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Yee, L.D.; Agarwal, D.; Rosol, T.J.; Lehman, A.; Tian, M.; Hatton, J.; Heestand, J.; Belury, M.A.; Clinton, S.K. The inhibition of early stages of her-2/neu-mediated mammary carcinogenesis by dietary n-3 pufas. Mol. Nutr. Food Res. 2013, 57, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhang, Y.; Jia, C.; Wang, Y.; Lai, P.; Zhou, X.; Wang, Y.; Song, Q.; Lin, J.; Ren, Z.; et al. Mtorc1/2 targeted by n-3 polyunsaturated fatty acids in the prevention of mammary tumorigenesis and tumor progression. Oncogene 2014, 33, 4548–4557. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Lee, Y.S.; Mayoral, R.; Oh, D.Y.; Siu, J.T.; Webster, N.J.; Sears, D.D.; Olefsky, J.M.; Ellies, L.G. Omega-3 fatty acids reduce obesity-induced tumor progression independent of gpr120 in a mouse model of postmenopausal breast cancer. Oncogene 2014. [Google Scholar] [CrossRef]
- Bouwens, M.; van de Rest, O.; Dellschaft, N.; Bromhaar, M.G.; de Groot, L.C.; Geleijnse, J.M.; Muller, M.; Afman, L.A. Fish-oil supplementation induces antiinflammatory gene expression profiles in human blood mononuclear cells. Am. J. Clin. Nutr. 2009, 90, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.S.; Hu, X.J.; Zhao, Y.M.; Yang, J.; Li, D. Intake of fish and marine n-3 polyunsaturated fatty acids and risk of breast cancer: Meta-analysis of data from 21 independent prospective cohort studies. BMJ 2013, 346, f3706. [Google Scholar] [CrossRef] [PubMed]
- Chajes, V.; Torres-Mejia, G.; Biessy, C.; Ortega-Olvera, C.; Angeles-Llerenas, A.; Ferrari, P.; Lazcano-Ponce, E.; Romieu, I. Omega-3 and omega-6 polyunsaturated fatty acid intakes and the risk of breast cancer in mexican women: Impact of obesity status. Cancer Epidemiol. Biomark. Prev. 2012, 21, 319–326. [Google Scholar] [CrossRef]
- Goodstine, S.L.; Zheng, T.; Holford, T.R.; Ward, B.A.; Carter, D.; Owens, P.H.; Mayne, S.T. Dietary (n-3)/(n-6) fatty acid ratio: Possible relationship to premenopausal but not postmenopausal breast cancer risk in U.S. Women. J. Nutr. 2003, 133, 1409–1414. [Google Scholar] [PubMed]
- Maillard, V.; Bougnoux, P.; Ferrari, P.; Jourdan, M.L.; Pinault, M.; Lavillonniere, F.; Body, G.; le Floch, O.; Chajes, V. n-3 and n-6 fatty acids in breast adipose tissue and relative risk of breast cancer in a case-control study in tours, France. Int. J. Cancer 2002, 98, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Bagga, D.; Anders, K.H.; Wang, H.J.; Glaspy, J.A. Long-chain n-3-to-n-6 polyunsaturated fatty acid ratios in breast adipose tissue from women with and without breast cancer. Nutr. Cancer 2002, 42, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Klein, V.; Chajes, V.; Germain, E.; Schulgen, G.; Pinault, M.; Malvy, D.; Lefrancq, T.; Fignon, A.; le Floch, O.; Lhuillery, C.; et al. Low alpha-linolenic acid content of adipose breast tissue is associated with an increased risk of breast cancer. Eur. J. Cancer 2000, 36, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lim, S.Y.; Shin, A.; Sung, M.K.; Ro, J.; Kang, H.S.; Lee, K.S.; Kim, S.W.; Lee, E.S. Fatty fish and fish omega-3 fatty acid intakes decrease the breast cancer risk: A case-control study. BMC Cancer 2009, 9, 216. [Google Scholar] [CrossRef] [PubMed]
- Witt, P.M.; Christensen, J.H.; Schmidt, E.B.; Dethlefsen, C.; Tjonneland, A.; Overvad, K.; Ewertz, M. Marine n-3 polyunsaturated fatty acids in adipose tissue and breast cancer risk: A case-cohort study from Denmark. Cancer Causes Control 2009, 20, 1715–1721. [Google Scholar] [CrossRef] [PubMed]
- Pike, L.J. Lipid rafts: Heterogeneity on the high seas. Biochem. J. 2004, 378, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Staubach, S.; Hanisch, F.G. Lipid rafts: Signaling and sorting platforms of cells and their roles in cancer. Expert Rev. Proteomics 2011, 8, 263–277. [Google Scholar] [CrossRef] [PubMed]
- Hofman, E.G.; Ruonala, M.O.; Bader, A.N.; van den Heuvel, D.; Voortman, J.; Roovers, R.C.; Verkleij, A.J.; Gerritsen, H.C.; van Bergen En Henegouwen, P.M. Egf induces coalescence of different lipid rafts. J. Cell Sci. 2008, 121, 2519–2528. [Google Scholar] [CrossRef] [PubMed]
- Lingwood, D.; Simons, K. Lipid rafts as a membrane-organizing principle. Science 2010, 327, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Patra, S.K. Dissecting lipid raft facilitated cell signaling pathways in cancer. Biochim. Biophys. Acta 2008, 1785, 182–206. [Google Scholar] [PubMed]
- Pike, L.J.; Han, X.; Chung, K.N.; Gross, R.W. Lipid rafts are enriched in arachidonic acid and plasmenylethanolamine and their composition is independent of caveolin-1 expression: A quantitative electrospray ionization/mass spectrometric analysis. Biochemistry 2002, 41, 2075–2088. [Google Scholar] [CrossRef] [PubMed]
- Simons, K.; Sampaio, J.L. Membrane organization and lipid rafts. Cold Spring Harb. Perspect. Biol. 2011, 3, a004697. [Google Scholar] [CrossRef] [PubMed]
- Brzustowicz, M.R.; Cherezov, V.; Caffrey, M.; Stillwell, W.; Wassall, S.R. Molecular organization of cholesterol in polyunsaturated membranes: Microdomain formation. Biophys. J. 2002, 82, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Wassall, S.R.; Brzustowicz, M.R.; Shaikh, S.R.; Cherezov, V.; Caffrey, M.; Stillwell, W. Order from disorder, corralling cholesterol with chaotic lipids. The role of polyunsaturated lipids in membrane raft formation. Chem. Phys. Lipids 2004, 132, 79–88. [Google Scholar] [PubMed]
- Nicolau, D.V., Jr.; Burrage, K.; Parton, R.G.; Hancock, J.F. Identifying optimal lipid raft characteristics required to promote nanoscale protein-protein interactions on the plasma membrane. Mol. Cell. Biol. 2006, 26, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Corsetto, P.A.; Cremona, A.; Montorfano, G.; Jovenitti, I.E.; Orsini, F.; Arosio, P.; Rizzo, A.M. Chemical-physical changes in cell membrane microdomains of breast cancer cells after omega-3 pufa incorporation. Cell Biochem. Biophys. 2012, 64, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Sugimura, K.; Kaji, Y.; Kitamura, Y.; Fujii, M.; Hara, I.; Tachibana, M. Conventional MRI capabilities in the diagnosis of prostate cancer in the transition zone. AJR Am. J. Roentgenol. 2006, 186, 729–742. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Yun, U.J.; Koo, K.H.; Sung, J.Y.; Shim, J.; Ye, S.K.; Hong, K.M.; Kim, Y.N. Down-regulation of lipid raft-associated onco-proteins via cholesterol-dependent lipid raft internalization in docosahexaenoic acid-induced apoptosis. Biochim. Biophys. Acta 2014, 1841, 190–203. [Google Scholar] [CrossRef] [PubMed]
- Schley, P.D.; Brindley, D.N.; Field, C.J. (n-3) pufa alter raft lipid composition and decrease epidermal growth factor receptor levels in lipid rafts of human breast cancer cells. J. Nutr. 2007, 137, 548–553. [Google Scholar] [PubMed]
- Ravacci, G.R.; Brentani, M.M.; Tortelli, T., Jr.; Torrinhas, R.S.; Saldanha, T.; Torres, E.A.; Waitzberg, D.L. Lipid raft disruption by docosahexaenoic acid induces apoptosis in transformed human mammary luminal epithelial cells harboring her-2 overexpression. J. Nutr. Biochem. 2013, 24, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Altenburg, J.D.; Siddiqui, R.A. Omega-3 polyunsaturated fatty acids down-modulate cxcr4 expression and function in mda-mb-231 breast cancer cells. Mol. Cancer Res. 2009, 7, 1013–1020. [Google Scholar] [CrossRef] [PubMed]
- Fulda, S.; Strauss, G.; Meyer, E.; Debatin, K.M. Functional cd95 ligand and cd95 death-inducing signaling complex in activation-induced cell death and doxorubicin-induced apoptosis in leukemic T cells. Blood 2000, 95, 301–308. [Google Scholar] [PubMed]
- Ewaschuk, J.B.; Newell, M.; Field, C.J. Docosahexanoic acid improves chemotherapy efficacy by inducing cd95 translocation to lipid rafts in er(-) breast cancer cells. Lipids 2012, 47, 1019–1030. [Google Scholar] [CrossRef] [PubMed]
- Bollinger, C.R.; Teichgraber, V.; Gulbins, E. Ceramide-enriched membrane domains. Biochim. Biophys. Acta 2005, 1746, 284–294. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Lee, Y.S.; Kim, D.K. Doxorubicin exerts cytotoxic effects through cell cycle arrest and fas-mediated cell death. Pharmacology 2009, 84, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Lorincz, A.M.; Sukumar, S. Molecular links between obesity and breast cancer. Endocr. Relat. Cancer 2006, 13, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Korner, A.; Pazaitou-Panayiotou, K.; Kelesidis, T.; Kelesidis, I.; Williams, C.J.; Kaprara, A.; Bullen, J.; Neuwirth, A.; Tseleni, S.; Mitsiades, N.; et al. Total and high-molecular-weight adiponectin in breast cancer: In vitro and in vivo studies. J. Clin. Endocrinol. Metab. 2007, 92, 1041–1048. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, P.; Combs, T.P.; Shah, S.J.; Gouon-Evans, V.; Pollard, J.W.; Albanese, C.; Flanagan, L.; Tenniswood, M.P.; Guha, C.; Lisanti, M.P.; et al. Adipocyte-secreted factors synergistically promote mammary tumorigenesis through induction of anti-apoptotic transcriptional programs and proto-oncogene stabilization. Oncogene 2003, 22, 6408–6423. [Google Scholar] [CrossRef] [PubMed]
- Grivennikov, S.I.; Karin, M. Inflammatory cytokines in cancer: Tumour necrosis factor and interleukin 6 take the stage. Ann. Rheum. Dis. 2011, 70 (Suppl. 1), i104–i108. [Google Scholar] [CrossRef] [PubMed]
- Horng, T.; Hotamisligil, G.S. Linking the inflammasome to obesity-related disease. Nat. Med. 2011, 17, 164–165. [Google Scholar] [CrossRef] [PubMed]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Dalamaga, M.; Diakopoulos, K.N.; Mantzoros, C.S. The role of adiponectin in cancer: A review of current evidence. Endocr. Rev. 2012, 33, 547–594. [Google Scholar] [CrossRef] [PubMed]
- Fain, J.N. Release of inflammatory mediators by human adipose tissue is enhanced in obesity and primarily by the nonfat cells: A review. Mediat. Inflamm. 2010, 2010, 513948. [Google Scholar] [CrossRef]
- Fain, J.N.; Madan, A.K.; Hiler, M.L.; Cheema, P.; Bahouth, S.W. Comparison of the release of adipokines by adipose tissue, adipose tissue matrix, and adipocytes from visceral and subcutaneous abdominal adipose tissues of obese humans. Endocrinology 2004, 145, 2273–2282. [Google Scholar] [CrossRef] [PubMed]
- Cinti, S.; Mitchell, G.; Barbatelli, G.; Murano, I.; Ceresi, E.; Faloia, E.; Wang, S.; Fortier, M.; Greenberg, A.S.; Obin, M.S. Adipocyte death defines macrophage localization and function in adipose tissue of obese mice and humans. J. Lipid Res. 2005, 46, 2347–2355. [Google Scholar] [CrossRef] [PubMed]
- Subbaramaiah, K.; Howe, L.R.; Bhardwaj, P.; Du, B.; Gravaghi, C.; Yantiss, R.K.; Zhou, X.K.; Blaho, V.A.; Hla, T.; Yang, P.; et al. Obesity is associated with inflammation and elevated aromatase expression in the mouse mammary gland. Cancer Prev. Res. 2011, 4, 329–346. [Google Scholar] [CrossRef]
- Morris, P.G.; Hudis, C.A.; Giri, D.; Morrow, M.; Falcone, D.J.; Zhou, X.K.; Du, B.; Brogi, E.; Crawford, C.B.; Kopelovich, L.; et al. Inflammation and increased aromatase expression occur in the breast tissue of obese women with breast cancer. Cancer Prev. Res. 2011, 4, 1021–1029. [Google Scholar] [CrossRef]
- Bhardwaj, P.; Du, B.; Zhou, X.K.; Sue, E.; Harbus, M.D.; Falcone, D.J.; Giri, D.; Hudis, C.A.; Kopelovich, L.; Subbaramaiah, K.; et al. Caloric restriction reverses obesity-induced mammary gland inflammation in mice. Cancer Prev. Res. 2013, 6, 282–289. [Google Scholar] [CrossRef]
- Ibrahim, M.M. Subcutaneous and visceral adipose tissue: Structural and functional differences. Obes. Rev. 2010, 11, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Dalamaga, M. Obesity, insulin resistance, adipocytokines and breast cancer: New biomarkers and attractive therapeutic targets. World J. Exp. Med. 2013, 3, 34–42. [Google Scholar] [PubMed]
- Ferguson, R.D.; Gallagher, E.J.; Scheinman, E.J.; Damouni, R.; LeRoith, D. The epidemiology and molecular mechanisms linking obesity, diabetes, and cancer. Vitam. Horm. 2013, 93, 51–98. [Google Scholar] [PubMed]
- Rose, D.P.; Vona-Davis, L. Biochemical and molecular mechanisms for the association between obesity, chronic inflammation, and breast cancer. BioFactors 2014, 40, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Baglietto, L.; English, D.R.; Hopper, J.L.; MacInnis, R.J.; Morris, H.A.; Tilley, W.D.; Krishnan, K.; Giles, G.G. Circulating steroid hormone concentrations in postmenopausal women in relation to body size and composition. Breast Cancer Res. Treat. 2009, 115, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Yager, J.D.; Davidson, N.E. Estrogen carcinogenesis in breast cancer. N. Engl. J. Med. 2006, 354, 270–282. [Google Scholar] [CrossRef] [PubMed]
- Key, T.; Appleby, P.; Barnes, I.; Reeves, G.; Endogenous, H.; Breast Cancer Collaborative, G. Endogenous sex hormones and breast cancer in postmenopausal women: Reanalysis of nine prospective studies. J. Natl. Cancer Inst. 2002, 94, 606–616. [Google Scholar] [CrossRef] [PubMed]
- Haffner, S.M.; Katz, M.S.; Dunn, J.F. Increased upper body and overall adiposity is associated with decreased sex hormone binding globulin in postmenopausal women. Int. J. Obes. 1991, 15, 471–478. [Google Scholar] [PubMed]
- Vona-Davis, L.; Rose, D.P. The obesity-inflammation-eicosanoid axis in breast cancer. J. Mammary Gland Biol. Neoplasia 2013, 18, 291–307. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Miki, Y.; Ohuchi, N.; Sasano, H. Intratumoral estrogen production in breast carcinoma: Significance of aromatase. Breast Cancer 2008, 15, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Irahara, N.; Miyoshi, Y.; Taguchi, T.; Tamaki, Y.; Noguchi, S. Quantitative analysis of aromatase mRNA expression derived from various promoters (i.4, i.3, pii and i.7) and its association with expression of TNF-alpha, il-6 and cox-2 mRNAs in human breast cancer. Int. J. Cancer 2006, 118, 1915–1921. [Google Scholar] [CrossRef] [PubMed]
- Brodie, A.; Lu, Q.; Nakamura, J. Aromatase in the normal breast and breast cancer. J. Steroid Biochem. Mol. Biol. 1997, 61, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Macaulay, V.M.; Nicholls, J.E.; Gledhill, J.; Rowlands, M.G.; Dowsett, M.; Ashworth, A. Biological effects of stable overexpression of aromatase in human hormone-dependent breast cancer cells. Br. J. Cancer 1994, 69, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Simpson, E.R.; Mahendroo, M.S.; Nichols, J.E.; Bulun, S.E. Aromatase gene expression in adipose tissue: Relationship to breast cancer. Int. J. Fertil. Menopausal Stud. 1994, 39 (Suppl. 2), S75–S83. [Google Scholar]
- Pasqualini, J.R.; Chetrite, G.S. Recent insight on the control of enzymes involved in estrogen formation and transformation in human breast cancer. J. Steroid Biochem. Mol. Biol. 2005, 93, 221–236. [Google Scholar] [CrossRef] [PubMed]
- Simpson, E.R.; Ackerman, G.E.; Smith, M.E.; Mendelson, C.R. Estrogen formation in stromal cells of adipose tissue of women: Induction by glucocorticosteroids. Proc. Natl. Acad. Sci. USA 1981, 78, 5690–5694. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, J.S.; Elton, R.A.; Miller, W.R. Aromatase activity in adipose tissue from breast quadrants: A link with tumour site. Br. Med. J. 1988, 296, 741–743. [Google Scholar] [CrossRef]
- Bulun, S.E.; Price, T.M.; Aitken, J.; Mahendroo, M.S.; Simpson, E.R. A link between breast cancer and local estrogen biosynthesis suggested by quantification of breast adipose tissue aromatase cytochrome p450 transcripts using competitive polymerase chain reaction after reverse transcription. J. Clin. Endocrinol. Metab. 1993, 77, 1622–1628. [Google Scholar] [PubMed]
- Bulun, S.E.; Chen, D.; Moy, I.; Brooks, D.C.; Zhao, H. Aromatase, breast cancer and obesity: A complex interaction. Trends Endocrinol. Metab. 2012, 23, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E.; Simpson, E.R. Regulation of aromatase expression in human tissues. Breast Cancer Res. Treat. 1994, 30, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Simpson, E.R.; Brown, K.A. Obesity and breast cancer: Role of inflammation and aromatase. J. Mol. Endocrinol. 2013, 51, T51–T59. [Google Scholar] [CrossRef] [PubMed]
- Subbaramaiah, K.; Morris, P.G.; Zhou, X.K.; Morrow, M.; Du, B.; Giri, D.; Kopelovich, L.; Hudis, C.A.; Dannenberg, A.J. Increased levels of cox-2 and prostaglandin e2 contribute to elevated aromatase expression in inflamed breast tissue of obese women. Cancer Discov. 2012, 2, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Pender-Cudlip, M.C.; Krag, K.J.; Martini, D.; Yu, J.; Guidi, A.; Skinner, S.S.; Zhang, Y.; Qu, X.; He, C.; Xu, Y.; et al. Delta-6-desaturase activity and arachidonic acid synthesis are increased in human breast cancer tissue. Cancer Sci. 2013, 104, 760–764. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Reierstad, S.; Lin, Z.; Lu, M.; Brooks, C.; Li, N.; Innes, J.; Bulun, S.E. Prostaglandin e(2) induces breast cancer related aromatase promoters via activation of p38 and c-jun nh(2)-terminal kinase in adipose fibroblasts. Cancer Res. 2007, 67, 8914–8922. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Agarwal, V.R.; Mendelson, C.R.; Simpson, E.R. Estrogen biosynthesis proximal to a breast tumor is stimulated by pge2 via cyclic amp, leading to activation of promoter II of the cyp19 (aromatase) gene. Endocrinology 1996, 137, 5739–5742. [Google Scholar] [PubMed]
- Larsson, S.C.; Kumlin, M.; Ingelman-Sundberg, M.; Wolk, A. Dietary long-chain n-3 fatty acids for the prevention of cancer: A review of potential mechanisms. Am. J. Clin. Nutr. 2004, 79, 935–945. [Google Scholar] [PubMed]
- Shackelford, D.B.; Shaw, R.J. The lkb1-ampk pathway: Metabolism and growth control in tumour suppression. Nat. Rev. Cancer 2009, 9, 563–575. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.A.; McInnes, K.J.; Hunger, N.I.; Oakhill, J.S.; Steinberg, G.R.; Simpson, E.R. Subcellular localization of cyclic amp-responsive element binding protein-regulated transcription coactivator 2 provides a link between obesity and breast cancer in postmenopausal women. Cancer Res. 2009, 69, 5392–5399. [Google Scholar] [CrossRef] [PubMed]
- Purohit, A.; Newman, S.P.; Reed, M.J. The role of cytokines in regulating estrogen synthesis: Implications for the etiology of breast cancer. Breast Cancer Res. 2002, 4, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Nichols, J.E.; Valdez, R.; Mendelson, C.R.; Simpson, E.R. Tumor necrosis factor-alpha stimulates aromatase gene expression in human adipose stromal cells through use of an activating protein-1 binding site upstream of promoter 1.4. Mol. Endocrinol. 1996, 10, 1350–1357. [Google Scholar] [PubMed]
- To, S.Q.; Knower, K.C.; Clyne, C.D. Nfkappab and mapk signalling pathways mediate tnfalpha-induced early growth response gene transcription leading to aromatase expression. Biochem. Biophys. Res. Commun. 2013, 433, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Honma, S.; Shimodaira, K.; Shimizu, Y.; Tsuchiya, N.; Saito, H.; Yanaihara, T.; Okai, T. The influence of inflammatory cytokines on estrogen production and cell proliferation in human breast cancer cells. Endocr. J. 2002, 49, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Panigrahy, D.; Kaipainen, A.; Greene, E.R.; Huang, S. Cytochrome p450-derived eicosanoids: The neglected pathway in cancer. Cancer Metastasis Rev. 2010, 29, 723–735. [Google Scholar] [CrossRef] [PubMed]
- Arnold, C.; Konkel, A.; Fischer, R.; Schunck, W.H. Cytochrome p450-dependent metabolism of omega-6 and omega-3 long-chain polyunsaturated fatty acids. Pharmacol. Rep. 2010, 62, 536–547. [Google Scholar] [CrossRef] [PubMed]
- Bannenberg, G.; Arita, M.; Serhan, C.N. Endogenous receptor agonists: Resolving inflammation. Sci. World J. 2007, 7, 1440–1462. [Google Scholar] [CrossRef]
- Bannenberg, G.L.; Chiang, N.; Ariel, A.; Arita, M.; Tjonahen, E.; Gotlinger, K.H.; Hong, S.; Serhan, C.N. Molecular circuits of resolution: Formation and actions of resolvins and protectins. J. Immunol. 2005, 174, 4345–4355. [Google Scholar] [CrossRef] [PubMed]
- Rose, D.P.; Connolly, J.M. Omega-3 fatty acids as cancer chemopreventive agents. Pharmacol. Ther. 1999, 83, 217–244. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. The role of marine omega-3 (n-3) fatty acids in inflammatory processes, atherosclerosis and plaque stability. Mol. Nutr. Food Res. 2012, 56, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Wei, N.; Wang, B.; Zhang, Q.Y.; Mi, M.T.; Zhu, J.D.; Yu, X.P.; Yuan, J.L.; Chen, K.; Wang, J.; Chang, H. Effects of different dietary fatty acids on the fatty acid compositions and the expression of lipid metabolic-related genes in mammary tumor tissues of rats. Nutr. Cancer 2008, 60, 810–825. [Google Scholar] [CrossRef] [PubMed]
- Kaur, B.; Jorgensen, A.; Duttaroy, A.K. Fatty acid uptake by breast cancer cells (mda-mb-231): Effects of insulin, leptin, adiponectin, and tnfalpha. Prostaglandins Leukot. Essent. Fatty Acids 2009, 80, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.H.; Connolly, J.M.; Rose, D.P. The 12-lipoxygenase gene-transfected mcf-7 human breast cancer cell line exhibits estrogen-independent, but estrogen and omega-6 fatty acid-stimulated proliferation in vitro, and enhanced growth in athymic nude mice. Cancer Lett. 1996, 109, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Connolly, J.M.; Rose, D.P. Enhanced angiogenesis and growth of 12-lipoxygenase gene-transfected mcf-7 human breast cancer cells in athymic nude mice. Cancer Lett. 1998, 132, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Avis, I.; Hong, S.H.; Martinez, A.; Moody, T.; Choi, Y.H.; Trepel, J.; Das, R.; Jett, M.; Mulshine, J.L. Five-lipoxygenase inhibitors can mediate apoptosis in human breast cancer cell lines through complex eicosanoid interactions. FASEB J. 2001, 15, 2007–2009. [Google Scholar] [PubMed]
- Tong, W.G.; Ding, X.Z.; Adrian, T.E. The mechanisms of lipoxygenase inhibitor-induced apoptosis in human breast cancer cells. Biochem. Biophys. Res. Commun. 2002, 296, 942–948. [Google Scholar] [CrossRef] [PubMed]
- Rose, D.P.; Connolly, J.M. Effects of fatty acids and inhibitors of eicosanoid synthesis on the growth of a human breast cancer cell line in culture. Cancer Res. 1990, 50, 7139–7144. [Google Scholar] [PubMed]
- Navarro-Tito, N.; Soto-Guzman, A.; Castro-Sanchez, L.; Martinez-Orozco, R.; Salazar, E.P. Oleic acid promotes migration on mda-mb-231 breast cancer cells through an arachidonic acid-dependent pathway. Int. J. Biochem. Cell Biol. 2010, 42, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Li, Y.; Zhao, Y.; Wang, Q.; You, J.C.; Zhang, X.D.; Ye, L.H. A novel positive feedback loop involving fasn/p-erk1/2/5-lox/ltb4/fasn sustains high growth of breast cancer cells. Acta Pharmacol. Sin. 2011, 32, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Rose, D.P.; Connolly, J.M.; Rayburn, J.; Coleman, M. Influence of diets containing eicosapentaenoic or docosahexaenoic acid on growth and metastasis of breast cancer cells in nude mice. J. Natl. Cancer Inst. 1995, 87, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.L.; Hubbard, N.E. Fatty acids and breast cancer: The role of stem cells. Prostaglandins Leukot. Essent. Fatty Acids 2010, 82, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Sethi, S.; Sarkar, F.H.; Ahmed, Q.; Bandyopadhyay, S.; Nahleh, Z.A.; Semaan, A.; Sakr, W.; Munkarah, A.; Ali-Fehmi, R. Molecular markers of epithelial-to-mesenchymal transition are associated with tumor aggressiveness in breast carcinoma. Transl. Oncol. 2011, 4, 222–226. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yau, C.; Gray, J.W.; Chew, K.; Dairkee, S.H.; Moore, D.H.; Eppenberger, U.; Eppenberger-Castori, S.; Benz, C.C. Enhanced NF kappa b and ap-1 transcriptional activity associated with antiestrogen resistant breast cancer. BMC Cancer 2007, 7, 59. [Google Scholar] [CrossRef] [PubMed]
- Naugler, W.E.; Karin, M. NF-kappab and cancer-identifying targets and mechanisms. Curr. Opin. Genet. Dev. 2008, 18, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Lipids for intravenous nutrition in hospitalised adult patients: A multiple choice of options. Proc. Nutr. Soc. 2013, 72, 263–276. [Google Scholar] [CrossRef] [PubMed]
- Kurtzman, S.H.; Anderson, K.H.; Wang, Y.; Miller, L.J.; Renna, M.; Stankus, M.; Lindquist, R.R.; Barrows, G.; Kreutzer, D.L. Cytokines in human breast cancer: Il-1alpha and il-1beta expression. Oncol. Rep. 1999, 6, 65–70. [Google Scholar] [PubMed]
- Pantschenko, A.G.; Pushkar, I.; Anderson, K.H.; Wang, Y.; Miller, L.J.; Kurtzman, S.H.; Barrows, G.; Kreutzer, D.L. The interleukin-1 family of cytokines and receptors in human breast cancer: Implications for tumor progression. Int. J. Oncol. 2003, 23, 269–284. [Google Scholar] [PubMed]
- Chavey, C.; Bibeau, F.; Gourgou-Bourgade, S.; Burlinchon, S.; Boissiere, F.; Laune, D.; Roques, S.; Lazennec, G. Oestrogen receptor negative breast cancers exhibit high cytokine content. Breast Cancer Res. 2007, 9, R15. [Google Scholar] [CrossRef] [PubMed]
- Candido, J.; Hagemann, T. Cancer-related inflammation. J. Clin. Immunol. 2013, 33 (Suppl. 1), S79–S84. [Google Scholar] [CrossRef]
- Kim, S.; Choi, J.H.; Kim, J.B.; Nam, S.J.; Yang, J.H.; Kim, J.H.; Lee, J.E. Berberine suppresses TNF-alpha-induced mmp-9 and cell invasion through inhibition of ap-1 activity in mda-mb-231 human breast cancer cells. Molecules 2008, 13, 2975–2985. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Deng, J.; Rychahou, P.G.; Qiu, S.; Evers, B.M.; Zhou, B.P. Stabilization of snail by NF-kappab is required for inflammation-induced cell migration and invasion. Cancer Cell 2009, 15, 416–428. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.G.; Li, D.; Stafford, L.J.; Luo, J.; Rodriguez-Villanueva, M.; Wang, Y.; Liu, M. Kiss1 suppresses tnfalpha-induced breast cancer cell invasion via an inhibition of rhoa-mediated NF-kappab activation. J. Cell Biochem. 2009, 107, 1139–1149. [Google Scholar] [CrossRef] [PubMed]
- Kushiro, K.; Nunez, N.P. Ob/ob serum promotes a mesenchymal cell phenotype in b16bl6 melanoma cells. Clin. Exp. Metastasis 2011, 28, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Soria, G.; Ben-Baruch, A. The inflammatory chemokines ccl2 and ccl5 in breast cancer. Cancer Lett. 2008, 267, 271–285. [Google Scholar] [CrossRef] [PubMed]
- Ueno, T.; Toi, M.; Saji, H.; Muta, M.; Bando, H.; Kuroi, K.; Koike, M.; Inadera, H.; Matsushima, K. Significance of macrophage chemoattractant protein-1 in macrophage recruitment, angiogenesis, and survival in human breast cancer. Clin. Cancer Res. 2000, 6, 3282–3289. [Google Scholar] [PubMed]
- Ye, J.; Gao, Z.; Yin, J.; He, Q. Hypoxia is a potential risk factor for chronic inflammation and adiponectin reduction in adipose tissue of ob/ob and dietary obese mice. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E1118–E1128. [Google Scholar] [CrossRef] [PubMed]
- Cancello, R.; Henegar, C.; Viguerie, N.; Taleb, S.; Poitou, C.; Rouault, C.; Coupaye, M.; Pelloux, V.; Hugol, D.; Bouillot, J.L.; et al. Reduction of macrophage infiltration and chemoattractant gene expression changes in white adipose tissue of morbidly obese subjects after surgery-induced weight loss. Diabetes 2005, 54, 2277–2286. [Google Scholar] [CrossRef] [PubMed]
- Itariu, B.K.; Zeyda, M.; Hochbrugger, E.E.; Neuhofer, A.; Prager, G.; Schindler, K.; Bohdjalian, A.; Mascher, D.; Vangala, S.; Schranz, M.; et al. Long-chain n-3 pufas reduce adipose tissue and systemic inflammation in severely obese nondiabetic patients: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 96, 1137–1149. [Google Scholar] [CrossRef] [PubMed]
- Awada, M.; Meynier, A.; Soulage, C.O.; Hadji, L.; Geloen, A.; Viau, M.; Ribourg, L.; Benoit, B.; Debard, C.; Guichardant, M.; et al. n-3 pufa added to high-fat diets affect differently adiposity and inflammation when carried by phospholipids or triacylglycerols in mice. Nutr. Metab. 2013, 10, 23. [Google Scholar] [CrossRef]
- Titos, E.; Rius, B.; Gonzalez-Periz, A.; Lopez-Vicario, C.; Moran-Salvador, E.; Martinez-Clemente, M.; Arroyo, V.; Claria, J. Resolvin d1 and its precursor docosahexaenoic acid promote resolution of adipose tissue inflammation by eliciting macrophage polarization toward an m2-like phenotype. J. Immunol. 2011, 187, 5408–5418. [Google Scholar] [CrossRef] [PubMed]
- Oliver, E.; McGillicuddy, F.C.; Harford, K.A.; Reynolds, C.M.; Phillips, C.M.; Ferguson, J.F.; Roche, H.M. Docosahexaenoic acid attenuates macrophage-induced inflammation and improves insulin sensitivity in adipocytes-specific differential effects between lc n-3 pufa. J. Nutr. Biochem. 2012, 23, 1192–1200. [Google Scholar] [CrossRef] [PubMed]
- De Boer, A.A.; Monk, J.M.; Robinson, L.E. Docosahexaenoic acid decreases pro-inflammatory mediators in an in vitro murine adipocyte macrophage co-culture model. PLoS One 2014, 9, e85037. [Google Scholar] [CrossRef] [PubMed]
- Novak, T.E.; Babcock, T.A.; Jho, D.H.; Helton, W.S.; Espat, N.J. Nf-kappa b inhibition by omega-3 fatty acids modulates lps-stimulated macrophage tnf-alpha transcription. Am. J. Physiol. Lung Cell. Mol. Physiol. 2003, 284, L84–L89. [Google Scholar] [PubMed]
- Weldon, S.M.; Mullen, A.C.; Loscher, C.E.; Hurley, L.A.; Roche, H.M. Docosahexaenoic acid induces an anti-inflammatory profile in lipopolysaccharide-stimulated human thp-1 macrophages more effectively than eicosapentaenoic acid. J. Nutr. Biochem. 2007, 18, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.S.; Dalamaga, M.; Kim, S.Y.; Polyzos, S.A.; Hamnvik, O.P.; Magkos, F.; Paruthi, J.; Mantzoros, C.S. Leptin’s role in lipodystrophic and nonlipodystrophic insulin-resistant and diabetic individuals. Endocr. Rev. 2013, 34, 377–412. [Google Scholar] [CrossRef] [PubMed]
- Widjaja, A.; Stratton, I.M.; Horn, R.; Holman, R.R.; Turner, R.; Brabant, G. Ukpds 20: Plasma leptin, obesity, and plasma insulin in type 2 diabetic subjects. J. Clin. Endocrinol. Metab. 1997, 82, 654–657. [Google Scholar] [CrossRef] [PubMed]
- Jarde, T.; Caldefie-Chezet, F.; Damez, M.; Mishellany, F.; Penault-Llorca, F.; Guillot, J.; Vasson, M.P. Leptin and leptin receptor involvement in cancer development: A study on human primary breast carcinoma. Oncol. Rep. 2008, 19, 905–911. [Google Scholar] [PubMed]
- Revillion, F.; Charlier, M.; Lhotellier, V.; Hornez, L.; Giard, S.; Baranzelli, M.C.; Djiane, J.; Peyrat, J.P. Messenger RNA expression of leptin and leptin receptors and their prognostic value in 322 human primary breast cancers. Clin. Cancer Res. 2006, 12, 2088–2094. [Google Scholar] [CrossRef] [PubMed]
- Neville, M.C.; McFadden, T.B.; Forsyth, I. Hormonal regulation of mammary differentiation and milk secretion. J. Mammary Gland Biol. Neoplasia 2002, 7, 49–66. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, C.; Surmacz, E. Leptin and cancer. J. Cell. Physiol. 2006, 207, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Caldefie-Chezet, F.; Damez, M.; de Latour, M.; Konska, G.; Mishellani, F.; Fusillier, C.; Guerry, M.; Penault-Llorca, F.; Guillot, J.; Vasson, M.P. Leptin: A proliferative factor for breast cancer? Study on human ductal carcinoma. Biochem. Biophys. Res. Commun. 2005, 334, 737–741. [Google Scholar] [CrossRef] [PubMed]
- Dieudonne, M.N.; Machinal-Quelin, F.; Serazin-Leroy, V.; Leneveu, M.C.; Pecquery, R.; Giudicelli, Y. Leptin mediates a proliferative response in human mcf7 breast cancer cells. Biochem. Biophys. Res. Commun. 2002, 293, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, C.; Koda, M.; Cascio, S.; Sulkowska, M.; Kanczuga-Koda, L.; Golaszewska, J.; Russo, A.; Sulkowski, S.; Surmacz, E. Increased expression of leptin and the leptin receptor as a marker of breast cancer progression: Possible role of obesity-related stimuli. Clin. Cancer Res. 2006, 12, 1447–1453. [Google Scholar] [CrossRef]
- Hu, X.; Juneja, S.C.; Maihle, N.J.; Cleary, M.P. Leptin—A growth factor in normal and malignant breast cells and for normal mammary gland development. J. Natl. Cancer Inst. 2002, 94, 1704–1711. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, M.; Kitayama, J.; Nagawa, H. Enhanced expression of leptin and leptin receptor (ob-r) in human breast cancer. Clin. Cancer Res. 2004, 10, 4325–4331. [Google Scholar] [CrossRef] [PubMed]
- Laud, K.; Gourdou, I.; Pessemesse, L.; Peyrat, J.P.; Djiane, J. Identification of leptin receptors in human breast cancer: Functional activity in the t47-d breast cancer cell line. Mol. Cell. Endocrinol. 2002, 188, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Yin, N.; Wang, D.; Zhang, H.; Yi, X.; Sun, X.; Shi, B.; Wu, H.; Wu, G.; Wang, X.; Shang, Y. Molecular mechanisms involved in the growth stimulation of breast cancer cells by leptin. Cancer Res. 2004, 64, 5870–5875. [Google Scholar] [CrossRef] [PubMed]
- Nyante, S.J.; Gammon, M.D.; Kaufman, J.S.; Bensen, J.T.; Lin, D.Y.; Barnholtz-Sloan, J.S.; Hu, Y.; He, Q.; Luo, J.; Millikan, R.C. Common genetic variation in adiponectin, leptin, and leptin receptor and association with breast cancer subtypes. Breast Cancer Res. Treat. 2011, 129, 593–606. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, Y.; Funahashi, T.; Tanaka, S.; Taguchi, T.; Tamaki, Y.; Shimomura, I.; Noguchi, S. High expression of leptin receptor mRNA in breast cancer tissue predicts poor prognosis for patients with high, but not low, serum leptin levels. Int. J. Cancer 2006, 118, 1414–1419. [Google Scholar] [CrossRef] [PubMed]
- Catalano, S.; Mauro, L.; Marsico, S.; Giordano, C.; Rizza, P.; Rago, V.; Montanaro, D.; Maggiolini, M.; Panno, M.L.; Ando, S. Leptin induces, via erk1/erk2 signal, functional activation of estrogen receptor alpha in mcf-7 cells. J. Biol. Chem. 2004, 279, 19908–19915. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, M.E.; Cleary, M.P. The balance between leptin and adiponectin in the control of carcinogenesis—Focus on mammary tumorigenesis. Biochimie 2012, 94, 2164–2171. [Google Scholar] [CrossRef] [PubMed]
- Jarde, T.; Perrier, S.; Vasson, M.P.; Caldefie-Chezet, F. Molecular mechanisms of leptin and adiponectin in breast cancer. Eur. J. Cancer 2011, 47, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Ray, A. Adipokine leptin in obesity-related pathology of breast cancer. J. Biosci. 2012, 37, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Okumura, M.; Yamamoto, M.; Sakuma, H.; Kojima, T.; Maruyama, T.; Jamali, M.; Cooper, D.R.; Yasuda, K. Leptin and high glucose stimulate cell proliferation in mcf-7 human breast cancer cells: Reciprocal involvement of pkc-alpha and ppar expression. Biochim. Biophys. Acta 2002, 1592, 107–116. [Google Scholar] [PubMed]
- Chen, C.; Chang, Y.C.; Liu, C.L.; Chang, K.J.; Guo, I.C. Leptin-induced growth of human zr-75-1 breast cancer cells is associated with up-regulation of cyclin d1 and c-myc and down-regulation of tumor suppressor p53 and p21waf1/cip1. Breast Cancer Res. Treat. 2006, 98, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Seilkop, S.K. The effect of body weight on tumor incidence and carcinogenicity testing in b6c3f1 mice and f344 rats. Fundam. Appl. Toxicol. 1995, 24, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Klurfeld, D.M.; Lloyd, L.M.; Welch, C.B.; Davis, M.J.; Tulp, O.L.; Kritchevsky, D. Reduction of enhanced mammary carcinogenesis in la/n-cp (corpulent) rats by energy restriction. Proc. Soc. Exp. Biol. Med. 1991, 196, 381–384. [Google Scholar] [CrossRef] [PubMed]
- Cleary, M.P.; Juneja, S.C.; Phillips, F.C.; Hu, X.; Grande, J.P.; Maihle, N.J. Leptin receptor-deficient mmtv-tgf-alpha/lepr(db)lepr(db) female mice do not develop oncogene-induced mammary tumors. Exp. Biol. Med. 2004, 229, 182–193. [Google Scholar]
- Cleary, M.P.; Phillips, F.C.; Getzin, S.C.; Jacobson, T.L.; Jacobson, M.K.; Christensen, T.A.; Juneja, S.C.; Grande, J.P.; Maihle, N.J. Genetically obese mmtv-tgf-alpha/lep(ob)lep(ob) female mice do not develop mammary tumors. Breast Cancer Res. Treat. 2003, 77, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Otvos, L., Jr.; Kovalszky, I.; Riolfi, M.; Ferla, R.; Olah, J.; Sztodola, A.; Nama, K.; Molino, A.; Piubello, Q.; Wade, J.D.; et al. Efficacy of a leptin receptor antagonist peptide in a mouse model of triple-negative breast cancer. Eur. J. Cancer 2011, 47, 1578–1584. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.R.; Cherfils, S.; Escobar, M.; Yoo, J.H.; Carino, C.; Styer, A.K.; Sullivan, B.T.; Sakamoto, H.; Olawaiye, A.; Serikawa, T.; et al. Leptin signaling promotes the growth of mammary tumors and increases the expression of vascular endothelial growth factor (vegf) and its receptor type two (vegf-r2). J. Biol. Chem. 2006, 281, 26320–26328. [Google Scholar] [CrossRef] [PubMed]
- Catalano, S.; Marsico, S.; Giordano, C.; Mauro, L.; Rizza, P.; Panno, M.L.; Ando, S. Leptin enhances, via ap-1, expression of aromatase in the mcf-7 cell line. J. Biol. Chem. 2003, 278, 28668–28676. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, C.; Sisci, D.; Surmacz, E. Leptin interferes with the effects of the antiestrogen ici 182,780 in mcf-7 breast cancer cells. Clin. Cancer Res. 2004, 10, 6466–6475. [Google Scholar] [CrossRef] [PubMed]
- Machinal-Quelin, F.; Dieudonne, M.N.; Pecquery, R.; Leneveu, M.C.; Giudicelli, Y. Direct in vitro effects of androgens and estrogens on ob gene expression and leptin secretion in human adipose tissue. Endocrine 2002, 18, 179–184. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, J.S.; Burow, M.E.; Green, A.E.; McLachlan, J.A.; Henson, M.C. Effects of estrogen on leptin gene promoter activation in mcf-7 breast cancer and jeg-3 choriocarcinoma cells: Selective regulation via estrogen receptors alpha and beta. Mol. Cell. Endocrinol. 2001, 176, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wu, Y.M.; Xu, L.; Tang, C.; Zhong, Y.B. [Influence of hypoxia on leptin and leptin receptor gene expression of c57bl/6j mice]. Zhonghua Jie He He Hu Xi Za Zhi 2005, 28, 173–175. [Google Scholar] [PubMed]
- Ambrosini, G.; Nath, A.K.; Sierra-Honigmann, M.R.; Flores-Riveros, J. Transcriptional activation of the human leptin gene in response to hypoxia. Involvement of hypoxia-inducible factor 1. J. Biol. Chem. 2002, 277, 34601–34609. [Google Scholar] [CrossRef] [PubMed]
- Grosfeld, A.; Andre, J.; Hauguel-De Mouzon, S.; Berra, E.; Pouyssegur, J.; Guerre-Millo, M. Hypoxia-inducible factor 1 transactivates the human leptin gene promoter. J. Biol. Chem. 2002, 277, 42953–42957. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.; Liu, X.; Shen, W.; Deckelbaum, R.J.; Qi, K. The regulation of leptin, leptin receptor and pro-opiomelanocortin expression by n-3 pufas in diet-induced obese mice is not related to the methylation of their promoters. Nutr. Metab. 2011, 8, 31. [Google Scholar] [CrossRef]
- Zeidan, A.; Javadov, S.; Chakrabarti, S.; Karmazyn, M. Leptin-induced cardiomyocyte hypertrophy involves selective caveolae and rhoa/rock-dependent p38 mapk translocation to nuclei. Cardiovasc. Res. 2008, 77, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, T.; Yamauchi, T. Adiponectin and adiponectin receptors. Endocr. Rev. 2005, 26, 439–451. [Google Scholar] [CrossRef] [PubMed]
- Waki, H.; Yamauchi, T.; Kamon, J.; Kita, S.; Ito, Y.; Hada, Y.; Uchida, S.; Tsuchida, A.; Takekawa, S.; Kadowaki, T. Generation of globular fragment of adiponectin by leukocyte elastase secreted by monocytic cell line thp-1. Endocrinology 2005, 146, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, T.; Kamon, J.; Ito, Y.; Tsuchida, A.; Yokomizo, T.; Kita, S.; Sugiyama, T.; Miyagishi, M.; Hara, K.; Tsunoda, M.; et al. Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 2003, 423, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Ryan, A.S.; Berman, D.M.; Nicklas, B.J.; Sinha, M.; Gingerich, R.L.; Meneilly, G.S.; Egan, J.M.; Elahi, D. Plasma adiponectin and leptin levels, body composition, and glucose utilization in adult women with wide ranges of age and obesity. Diabetes Care 2003, 26, 2383–2388. [Google Scholar] [CrossRef] [PubMed]
- Vona-Davis, L.; Rose, D.P. Adipokines as endocrine, paracrine, and autocrine factors in breast cancer risk and progression. Endocr. Relat. Cancer 2007, 14, 189–206. [Google Scholar] [CrossRef] [PubMed]
- Kern, P.A.; di Gregorio, G.B.; Lu, T.; Rassouli, N.; Ranganathan, G. Adiponectin expression from human adipose tissue: Relation to obesity, insulin resistance, and tumor necrosis factor-alpha expression. Diabetes 2003, 52, 1779–1785. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, Y.; Funahashi, T.; Kihara, S.; Taguchi, T.; Tamaki, Y.; Matsuzawa, Y.; Noguchi, S. Association of serum adiponectin levels with breast cancer risk. Clin. Cancer Res. 2003, 9, 5699–5704. [Google Scholar] [PubMed]
- Mantzoros, C.; Petridou, E.; Dessypris, N.; Chavelas, C.; Dalamaga, M.; Alexe, D.M.; Papadiamantis, Y.; Markopoulos, C.; Spanos, E.; Chrousos, G.; et al. Adiponectin and breast cancer risk. J. Clin. Endocrinol. Metab. 2004, 89, 1102–1107. [Google Scholar] [CrossRef] [PubMed]
- Macis, D.; Guerrieri-Gonzaga, A.; Gandini, S. Circulating adiponectin and breast cancer risk: A systematic review and meta-analysis. Int. J. Epidemiol. 2014, 43, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Jia, J.; Dong, S.; Zhang, C.; Yu, S.; Li, L.; Mao, C.; Wang, D.; Chen, J.; Yuan, G. Circulating adiponectin levels and the risk of breast cancer: A meta-analysis. Eur. J. Cancer Prev. 2014, 23, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.Y.; Wang, M.; Ma, Z.B.; Yu, L.X.; Zhang, Q.; Gao, D.Z.; Wang, F.; Yu, Z.G. The role of adiponectin in breast cancer: A meta-analysis. PLoS One 2013, 8, e73183. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, M.E.; Ray, A.; Nkhata, K.J.; Malakhov, D.A.; Rogozina, O.P.; Dogan, S.; Cleary, M.P. Obesity and breast cancer: Status of leptin and adiponectin in pathological processes. Cancer Metastasis Rev. 2010, 29, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Takahata, C.; Miyoshi, Y.; Irahara, N.; Taguchi, T.; Tamaki, Y.; Noguchi, S. Demonstration of adiponectin receptors 1 and 2 mRNA expression in human breast cancer cells. Cancer Lett. 2007, 250, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Jarde, T.; Caldefie-Chezet, F.; Damez, M.; Mishellany, F.; Perrone, D.; Penault-Llorca, F.; Guillot, J.; Vasson, M.P. Adiponectin and leptin expression in primary ductal breast cancer and in adjacent healthy epithelial and myoepithelial tissue. Histopathology 2008, 53, 484–487. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.B.; Chow, K.H.; Xu, A.; Lam, K.S.; Liu, J.; Wong, N.S.; Moon, R.T.; Shepherd, P.R.; Cooper, G.J.; Wang, Y. Adiponectin haploinsufficiency promotes mammary tumor development in mmtv-pyvt mice by modulation of phosphatase and tensin homolog activities. PLoS One 2009, 4, e4968. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lam, J.B.; Lam, K.S.; Liu, J.; Lam, M.C.; Hoo, R.L.; Wu, D.; Cooper, G.J.; Xu, A. Adiponectin modulates the glycogen synthase kinase-3beta/beta-catenin signaling pathway and attenuates mammary tumorigenesis of mda-mb-231 cells in nude mice. Cancer Res. 2006, 66, 11462–11470. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, S.; Miyoshi, Y.; Ishihara, H.; Noguchi, S. Growth-inhibitory effect of adiponectin via adiponectin receptor 1 on human breast cancer cells through inhibition of s-phase entry without inducing apoptosis. Breast Cancer Res. Treat. 2008, 112, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, E.; Benaitreau, D.; Dieudonne, M.N.; Leneveu, M.C.; Serazin, V.; Giudicelli, Y.; Pecquery, R. Adiponectin mediates an antiproliferative response in human mda-mb 231 breast cancer cells. Oncol. Rep. 2008, 20, 971–977. [Google Scholar] [PubMed]
- Pfeiler, G.; Hudelist, G.; Wulfing, P.; Mattsson, B.; Konigsberg, R.; Kubista, E.; Singer, C.F. Impact of adipor1 expression on breast cancer development. Gynecol. Oncol. 2010, 117, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Jarde, T.; Caldefie-Chezet, F.; Goncalves-Mendes, N.; Mishellany, F.; Buechler, C.; Penault-Llorca, F.; Vasson, M.P. Involvement of adiponectin and leptin in breast cancer: Clinical and in vitro studies. Endocr. Relat. Cancer 2009, 16, 1197–1210. [Google Scholar] [CrossRef] [PubMed]
- Dieudonne, M.N.; Bussiere, M.; dos Santos, E.; Leneveu, M.C.; Giudicelli, Y.; Pecquery, R. Adiponectin mediates antiproliferative and apoptotic responses in human mcf7 breast cancer cells. Biochem. Biophys. Res. Commun. 2006, 345, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Taliaferro-Smith, L.; Nagalingam, A.; Zhong, D.; Zhou, W.; Saxena, N.K.; Sharma, D. Lkb1 is required for adiponectin-mediated modulation of ampk-s6k axis and inhibition of migration and invasion of breast cancer cells. Oncogene 2009, 28, 2621–2633. [Google Scholar] [CrossRef] [PubMed]
- Nkhata, K.J.; Ray, A.; Schuster, T.F.; Grossmann, M.E.; Cleary, M.P. Effects of adiponectin and leptin co-treatment on human breast cancer cell growth. Oncol. Rep. 2009, 21, 1611–1619. [Google Scholar] [PubMed]
- Villarreal-Molina, M.T.; Antuna-Puente, B. Adiponectin: Anti-inflammatory and cardioprotective effects. Biochimie 2012, 94, 2143–2149. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, N.; Walsh, K. Adiponectin as an anti-inflammatory factor. Clin. Chim. Acta 2007, 380, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Folco, E.J.; Rocha, V.Z.; Lopez-Ilasaca, M.; Libby, P. Adiponectin inhibits pro-inflammatory signaling in human macrophages independent of interleukin-10. J. Biol. Chem. 2009, 284, 25569–25575. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monk, J.M.; Turk, H.F.; Liddle, D.M.; De Boer, A.A.; Power, K.A.; Ma, D.W.L.; Robinson, L.E. n-3 Polyunsaturated Fatty Acids and Mechanisms to Mitigate Inflammatory Paracrine Signaling in Obesity-Associated Breast Cancer. Nutrients 2014, 6, 4760-4793. https://doi.org/10.3390/nu6114760
Monk JM, Turk HF, Liddle DM, De Boer AA, Power KA, Ma DWL, Robinson LE. n-3 Polyunsaturated Fatty Acids and Mechanisms to Mitigate Inflammatory Paracrine Signaling in Obesity-Associated Breast Cancer. Nutrients. 2014; 6(11):4760-4793. https://doi.org/10.3390/nu6114760
Chicago/Turabian StyleMonk, Jennifer M., Harmony F. Turk, Danyelle M. Liddle, Anna A. De Boer, Krista A. Power, David W.L. Ma, and Lindsay E. Robinson. 2014. "n-3 Polyunsaturated Fatty Acids and Mechanisms to Mitigate Inflammatory Paracrine Signaling in Obesity-Associated Breast Cancer" Nutrients 6, no. 11: 4760-4793. https://doi.org/10.3390/nu6114760
APA StyleMonk, J. M., Turk, H. F., Liddle, D. M., De Boer, A. A., Power, K. A., Ma, D. W. L., & Robinson, L. E. (2014). n-3 Polyunsaturated Fatty Acids and Mechanisms to Mitigate Inflammatory Paracrine Signaling in Obesity-Associated Breast Cancer. Nutrients, 6(11), 4760-4793. https://doi.org/10.3390/nu6114760




