A Psycho-Genetic Study of Hedonic Responsiveness in Relation to “Food Addiction”
Abstract
:1. Introduction
1.1. Hedonic Responsiveness and Capacity for Reward
1.2. The Biological Basis of Food Addiction
Brain Opioid Pathways and Food Reward
1.3. The Current Study
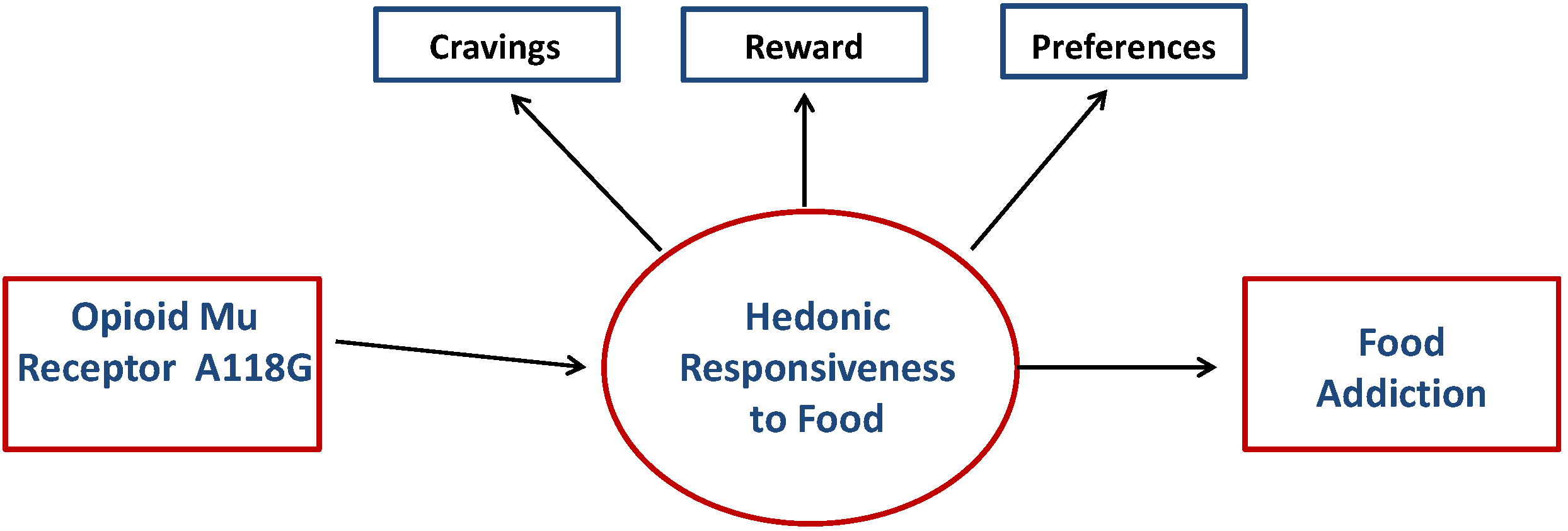
2. Methods
2.1. Participants
2.2. Measures
2.2.1. Genotyping
2.2.2. Self-Report Questionnaires
2.3. Procedures
2.4. Statistical Analyses
3. Results
3.1. Descriptive Statistics
| Group | Allele | Genotype | |||
|---|---|---|---|---|---|
| G | A | GG | GA | AA | |
| Food Addiction | 9 | 41 | 2 (8%) | 5 (20%) | 18 (72%) |
| Non-Food Addiction | 44 | 184 | 5 (4.4%) | 34 (29.8%) | 75 (65.8) |
| Variable | GG | GA | AA | F |
|---|---|---|---|---|
| Age | 31.9 (6.5:26–44) | 33.2 (6.2: 25–45) | 32.6 (6.6:25–47) | 0.22 |
| BMI | 31.1 (8.0:19.5–40.9) | 32.2 (8.6: 19.6–51.4) | 33.9 (8.4:19.0–60.1) | 0.82 |
| Hedonic Responsiveness | 0.5 (0.9:−0.6–1.6) a | −0.4 (0.8:−2.4–1.4) | 0.1 (1.0:−2.4–2.5) a | 5.31 ** |
| YFAS Symptom Score | 3.1 (2.1:1–7) | 2.2 (1.7:0–6) | 2.9 (2.0:0–7) | 1.95 |
3.2. Indirect Effects

| Polymorphism | Bootstrap Estimate | SE | BC 95% CI Lower | BC 95% CI Upper |
|---|---|---|---|---|
| AA vs. GA | 0.71 * | 0.23 | 0.30 | 1.19 |
| GG vs. GA | 1.16 * | 0.50 | 0.26 | 2.21 |
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association Press: Washington, WA, USA, 2013. [Google Scholar]
- Potenza, M.N. Non-substance addictive behaviors in the context of DSM-5. Addict. Behav. 2014, 39, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.; Carter, J.C. If certain foods are addictive, how might this change the treatment of compulsive overeating and obesity? Curr. Addict. Rep. 2014, 1, 89–95. [Google Scholar] [CrossRef]
- Gearhardt, A.N.; Davis, C.; Kushner, R.; Brownell, K. The addiction potential of hyperpalatable foods. Curr. Drug Abuse Rev. 2011, 4, 140–145. [Google Scholar] [CrossRef] [PubMed]
- De Pierre, J.A.; Puhl, R.M.; Luedicke, J. Public perceptions of food addiction: A comparison with alcohol and tobacco. J. Subst. Use 2014, 19, 1–6. [Google Scholar] [CrossRef]
- Latner, J.D.; Puhl, R.M.; Murakami, J.M.; O’Brien, K.S. Food addiction as a causal model of obesity. Effects on stigma, blame, and perceived psychopathology. Appetite 2014, 77C, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Meinzer, M.C.; Pettit, J.W.; Leventhal, A.M.; Hill, R.M. Explaining the covariance between attention-deficit hyperactivity disorder and depressive symptoms: The role of hedonic responsivity. J. Clin. Psychol. 2012, 68, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Leventhal, A.M.; Chasson, G.S.; Tapia, E.; Miller, E.K.; Pettit, J.W. Measuring hedonic capacity in depression: A psychometric analysis of three anhedonia scales. J. Clin. Psychol. 2006, 62, 1545–1558. [Google Scholar] [CrossRef] [PubMed]
- Davis, C. From passive overeating to “food addiction”: A spectrum of compulsion and severity. ISRN Obes. 2013, 2013. [Google Scholar] [CrossRef]
- George, O.; Koob, G.F. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci. Biobehav. Rev. 2010, 2, 232–247. [Google Scholar] [CrossRef]
- Davis, C.; Levitan, R.D.; Kaplan, A.S.; Carter, J.C.; Reid, C.; Curtis, C.; Patte, K.; Hwang, R.; Kennedy, J.L. Reward sensitivity and the D2 dopamine receptor gene: A case-control study of binge eating disorder. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2008, 32, 620–628. [Google Scholar] [CrossRef]
- Davis, C.; Levitan, R.D.; Yilmaz, Z.; Kaplan, A.S.; Carter, J.C.; Kennedy, J.L. Binge eating disorder and the dopamine D2 receptor: Genotypes and sub-phenotypes. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2012, 38, 328–335. [Google Scholar] [CrossRef]
- Schienle, A.; Schafer, A.; Hermann, A.; Vaitl, D. Binge-eating disorder: Reward sensitivity and brain activation to images of food. Biol. Psychiatry 2009, 65, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Curtis, C.; Davis, C. A qualitative study of binge eating disorder and obesity from an addiction perspective. Eat. Disord. 2014, 22, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Lowe, M.R.; Butryn, M.L.; Didie, E.R.; Annunziato, R.A.; Thomas, J.G.; Crerand, C.E.; Ochner, C.N.; Coletta, M.C.; Bellace, D.; Wallaert, M.; et al. The Power of Food Scale: A new measure of the psychological influence of the food environment. Appetite 2009, 53, 114–118. [Google Scholar] [CrossRef]
- Davis, C.; Loxton, N.J.; Levitan, R.D.; Kaplan, A.S.; Carter, J.C.; Kennedy, J.L. “Food addiction” and its associations with a dopaminergic multilocus genetic profile. Physiol. Behav. 2013, 118, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Gearhardt, A.N.; Yokum, S.; Orr, P.T.; Stice, E.; Corbin, W.R.; Brownell, K.D. Neural correlates of food addiction. Arch. Gen. Psychiatry 2011, 32, E1–E9. [Google Scholar]
- Davis, C.; Curtis, C.; Levitan, R.D.; Carter, J.C.; Kaplan, A.S.; Kennedy, J.L. Evidence that “food addiction” is a valid phenotype of obesity. Appetite 2011, 57, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Davis, C. Compulsive overeating as an addictive behaviour: Overlap between food addiction and binge eating disorder. Curr. Obes. Rep. 2013, 2, 171–178. [Google Scholar] [CrossRef]
- Gearhardt, A.N.; Boswell, R.G.; White, M.A. The association of “food addiction” with disordered eating and body mass index. Eat. Behav. 2014, 15, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Gearhardt, A.N.; White, M.A.; Masheb, R.M.; Morgan, P.T.; Crosby, R.D.; Grilo, C.M. An examination of the food addiction construct in patients with binge eating disorder. Int. J. Eat. Disord. 2012, 45, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Berridge, K.C. “Liking” and “wanting” food rewards: Brain substrates and roles in eating disorders. Physiol. Behav. 2009, 97, 537–550. [Google Scholar] [CrossRef] [PubMed]
- Kelley, A.E.; Bakshi, V.P.; Haber, S.N.; Steininger, T.L.; Will, M.J.; Zhang, M. Opioid modulation of taste hedonics within the ventral striatum. Physiol. Behav. 2002, 76, 365–377. [Google Scholar] [CrossRef] [PubMed]
- Katsuura, Y.; Taha, S.A. Mu opioid receptor antagonism in the nucleus accumbens shell blocks consumption of a preferred sucrose solution in an anticipatory contrast paradigm. Neuroscience 2014, 261, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Cambridge, V.C.; Ziauddeen, H.; Nathan, P.J.; Subramaniam, N.; Dodds, C.; Chamberlain, S.R.; Koch, A.; Maltby, K.; Skeggs, A.L.; Napolitano, A.; et al. Neural and behavioral effects of a novel mu opioid receptor antagonist in binge-eating obese people. Biol. Psychiatry 2013, 73, 887–894. [Google Scholar] [CrossRef]
- Kelley, A.E.; Will, M.J.; Steininger, T.L.; Zhang, M.; Haber, S.N. Restricted daily consumption of a highly palatable food (chocolate Ensure®) alters striatal encephalin gene expression. Eur. J. Neurosci. 2003, 18, 2592–2598. [Google Scholar] [CrossRef] [PubMed]
- Daubenmier, J.; Lustig, R.H.; Hecht, F.M.; Kristeller, J.; Woolley, J.; Adam, T.; Dallman, M.; Epel, E. A new biomarker of hedonic eating? Appetite 2014, 92–100. [Google Scholar] [CrossRef]
- Bond, C.; LaForge, K.S.; Tian, M.; Melia, D.; Zhang, S.; Borg, L.; Gong, J.; Schluger, J.; Strong, J.A.; Leal, S.M.; et al. Single-nucleotide polymorphism in the human mu opioid receptor gene alters beta-endorphin binding and activity: Possible implications for opioid addiction. Proc. Natl. Acad. Sci. USA 1998, 95, 9608–9613. [Google Scholar] [CrossRef]
- Barr, C.S.; Schwandt, M.L.; Lindell, S.G.; Higley, J.D.; Maestropien, D.; Goldman, D.; Suomi, S.J.; Heilig, M. Variation at the mu-opioid receptor gene (OPRM1) influences attachment behavior in infant primates. Proc. Natl. Acad. Sci. USA 2008, 105, 5277–5281. [Google Scholar] [CrossRef] [PubMed]
- Deb, I.; Chakraborty, J.; Gangopadhyay, P.K.; Choudhury, S.R.; Das, S. Single-nucleotide polymorphism (A118G) in exon 1 of OPRM1 gene causes alteration in downstream signalling by mu-opioid receptor and may contribute to the genetic risk for addiction. J. Neurochem. 2010, 112, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Bart, G.; Kreek, M.J.; Ott, J.; LaForge, K.S.; Proudnikov, D.; Pollak, L.; Heilig, M. Increased attributable risk related to a functional mu-opioid receptor gene polymorphism in association with alcohol dependence in central Sweden. Neuropsychopharmacology 2005, 30, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Ray, L.A.; Bujarski, S.; MacKillop, J.; Courtney, K.E.; Monti, P.M.; Miotto, K. Subjective response to alcohol among alcohol-dependent individuals: Effects of the mu-opioid receptor (OPRM1) gene and alcoholism severity. Alcohol. Clin. Exp. Res. 2013, 37, E116–E124. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.A.; Kim, J.W.; Song, J.Y.; Park, S.; Lee, H.J.; Chung, J.H. Association of polymorphisms in nicotinic acetylcholine receptor α4 subunit gene (CHRNA4), μ-opioid receptor gene (OPRM1), and ethanol-matabolizing enzyme genes with alcoholism in Korean patients. Alcohol 2004, 34, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Luo, X.; Kranzler, H.R.; Lappalainen, J.; Yang, B.-Z.; Krupitsky, E.; Zvartau, E.; Gelernter, J. Association between two μ-opioid receptor gene (OPRM1) haplotype blocks and drug or alcohol dependence. Hum. Mol. Genet. 2006, 15, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Miranda, R.; Ray, L.; Justus, A.; Meyerson, L.A.; Knopik, V.S.; McGeary, J.; Monti, P.M. Initial evidence of an association between OPRM1 and adolescent alcohol misuse. Alcohol. Clin. Exp. Res. 2010, 34, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Ray, L.A.; Hutchinson, K.E. A polymorphism of the mu opioid receptor gene and sensitivity to the effects of alcohol in humans. Alcohol. Clin. Exp. Res. 2004, 28, 1789–1795. [Google Scholar] [CrossRef] [PubMed]
- Filbey, F.M.; Ray, L.; Smolen, A.; Claus, E.D.; Audette, A.; Hutchison, K.E. Differential neural response to alcohol priming and alcohol taste cues is associated with DRD4 VNTR and OPRM1 genotypes. Alcohol. Clin. Exp. Res. 2008, 32, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Troisi, A.; Frazzetto, G.; Carola, V.; di Lorenzo, G.; Coviello, M.; D’Amato, F.R.; Moles, A.; Siracusano, A.; Gross, C. Social hedonic capacity is associated with the A118G polymorphism of the μ-opioid receptor gene (OPRM1) in adult healthy volunteers and psychiatric patients. Soc. Neurosci. 2011, 6, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Gearhardt, A.N.; Corbin, W.R.; Brownell, K.D. Preliminary Validation of the Yale Food Addiction Scale. Appetite 2009, 52, 430–436. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association Press: Washington, WA, USA, 1994. [Google Scholar]
- Lahiri, D.K.; Nurnburger, J.I., Jr. A rapid non-enzymatic method for the preparation of HMV DNA from blood for RFLP analysis. Nucleic Acids Res. 1991, 19, 5444. [Google Scholar] [CrossRef] [PubMed]
- Lotsch, J.; Geisslinger, G. Are μ-opioid receptor polymorphisms important for clinical opioid therapy? Trends Mol. Med. 2005, 11, 82–89. [Google Scholar]
- Barrett, J.C.; Fry, B.; Maller, J.; Daly, M.J. Haploview: Analysis and visualization of LD and haplotype maps. Bioinformatics 2005, 21, 263–265. [Google Scholar] [CrossRef] [PubMed]
- Geiselman, P.J.; Anderson, A.M.; Dowdy, M.L.; West, D.B.; Redmann, S.M.; Smith, S.R. Reliability and validity of a macronutrient self-selection paradigm and a food preference questionnaire. Physiol. Behav. 1998, 63, 919–928. [Google Scholar] [CrossRef] [PubMed]
- Cappelleri, J.C.; Bushmakin, A.G.; Gerber, R.A.; Leidy, N.K.; Sexton, C.C.; Karlsson, J.; Lowe, M.R. Evaluating the Power of Food Scale in obese subjects and a general sample of individuals: Development and measurement properties. Int. J. Obes. 2009, 33, 913–922. [Google Scholar] [CrossRef]
- Cepeda-Benito, A.; Gleaves, D.H.; Williams, T.L.; Erath, S.A. The development and validation of the state and trait food-cravings questionnaires. Behav. Ther. 2000, 31, 151–173. [Google Scholar] [CrossRef]
- Hayes, A.F.; Preacher, K.J. Statistical mediation analysis with a multicategorical independent variable. Br. J. Math. Stat. Psychol. 2014, 67, 451–470. [Google Scholar] [CrossRef] [PubMed]
- Hair, J.F.; Black, B.; Babin, B.; Anderson, R.E.; Tatham, R.L. Multivariate Data Analysis; Pearson Education Inc.: Saddle River, NJ, USA, 2009. [Google Scholar]
- Shrout, P.E.; Bolger, N. Mediation in experimental and nonexperimental studies: New procedures and recommendations. Psychol. Meth. 2002, 7, 422–445. [Google Scholar] [CrossRef]
- Hayes, A.F. Introduction to mediation, moderation, and conditional process analysis. In A Regression-Based Approach; Guilford Press: New York, NY, USA, 2013. [Google Scholar]
- Hansson, B.; Westerberg, L. On the correlation between heterozygosity and fitness in natural populations. Mol. Ecol. 2002, 11, 2467–2474. [Google Scholar] [CrossRef] [PubMed]
- Ray, L.A.; Courtney, K.E.; Hutchison, K.E.; MacKillop, J.; Galvan, A.; Ghahremari, D.G. Initial evidence that OPRM1 genotype moderates ventral and dorsal striatum functional connectivity during alcohol cues. Am. Clin. Exp. Res. 2014, 38, 78–89. [Google Scholar] [CrossRef] [Green Version]
- Domino, E.F.; Evans, C.L.; Ni, L.S.; Guthrie, S.K.; Koeppe, R.A. Tobacco smoking produces greater striatal dopamine release in G-allele carriers with mu opioid receptor A118G polymorphism. Prog. Neuropsychopharmacol. Biol. Psychiatry 2012, 38, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Haerian, B.S.; Haerian, M.S. OPRM1 rs1799971 polymorphism and opioid dependence evidence from a meta-analysis. Pharmacogenomics 2013, 14, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Berthoud, H.R.; Lenard, N.R.; Shin, A.C. Food reward, hyperphagia, and obesity. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, 1266–1277. [Google Scholar] [CrossRef]
- Johnson, P.M.; Kenny, P.J. Dopamine D2 receptors in addiction-like reward dysfunction and compulsive eating in obese rats. Nat. Neurosci. 2010, 13, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Martine, S.I.; Maniam, J.; South, T.; Holmes, N.; Westbrook, R.F.; Morris, M.J. Extended exposure to a palatable cafeteria diet alters gene expression in brain regions implicated in reward, and withdrawal from this diet alters gene expression in brain regions associated with stress. Behav. Brain Res. 2014, 265, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Blum, K.; Chen, A.L.C.; Giordano, J.; Borsten, J.; Chen, T.J.H.; Hauser, M.; Simpatico, T.; Femino, J.; Braverman, E.R.; Debmayla, B. The addictive brain: All roads lead to dopamine. J. Psychoact. Drugs 2012, 44, 134–143. [Google Scholar] [CrossRef]
- Heber, D.; Carpenter, C.L. Addictive genes and the relationship to obesity and inflammation. Mol. Neurobiol. 2011, 44, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Lavi-Avnon, Y.; Yadid, G.; Overstreet, D.H.; Weller, A. Abnormal patterns of maternal behavior in a genetic animal model of depression. Physiol. Behav. 2005, 84, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Padrao, G.; Mallorqui, A.; Cucurell, D.; Marco-Pallares, J.; Rodriguez-Fornellis, A. Neurophysiological differences in reward processing in anhedonics. Cog. Affect. Behav. Neurosci. 2013, 13, 102–115. [Google Scholar] [CrossRef]
- Davis, C.; Fox, J. Sensitivity to reward and body mass index (BMI): Evidence for a non-linear relationship. Appetite 2008, 50, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Hommer, D.W.; Bjork, J.M.; Gilman, J.M. Imaging brain response to reward in addictive behaviors. Ann. N. Y. Acad. Sci. 2011, 1216, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, Z.; Davis, C.; Loxton, N.J.; Kaplan, A.S.; Levitan, R.D.; Carter, J.C.; Kennedy, J.L. Association between MC4R rs17882313 polymorphism and overeating behaviors. Int. J. Obes. 2014. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Davis, C.; Loxton, N.J. A Psycho-Genetic Study of Hedonic Responsiveness in Relation to “Food Addiction”. Nutrients 2014, 6, 4338-4353. https://doi.org/10.3390/nu6104338
Davis C, Loxton NJ. A Psycho-Genetic Study of Hedonic Responsiveness in Relation to “Food Addiction”. Nutrients. 2014; 6(10):4338-4353. https://doi.org/10.3390/nu6104338
Chicago/Turabian StyleDavis, Caroline, and Natalie J. Loxton. 2014. "A Psycho-Genetic Study of Hedonic Responsiveness in Relation to “Food Addiction”" Nutrients 6, no. 10: 4338-4353. https://doi.org/10.3390/nu6104338
APA StyleDavis, C., & Loxton, N. J. (2014). A Psycho-Genetic Study of Hedonic Responsiveness in Relation to “Food Addiction”. Nutrients, 6(10), 4338-4353. https://doi.org/10.3390/nu6104338




